WO2014118272A1 - Antimir-122 oligonucleotide carbohydrate conjugates - Google Patents
Antimir-122 oligonucleotide carbohydrate conjugates Download PDFInfo
- Publication number
- WO2014118272A1 WO2014118272A1 PCT/EP2014/051793 EP2014051793W WO2014118272A1 WO 2014118272 A1 WO2014118272 A1 WO 2014118272A1 EP 2014051793 W EP2014051793 W EP 2014051793W WO 2014118272 A1 WO2014118272 A1 WO 2014118272A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- region
- lna
- oligomer
- antisense oligonucleotide
- nucleotides
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/549—Sugars, nucleosides, nucleotides or nucleic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
- C12N2310/113—Antisense targeting other non-coding nucleic acids, e.g. antagomirs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/323—Chemical structure of the sugar modified ring structure
- C12N2310/3231—Chemical structure of the sugar modified ring structure having an additional ring, e.g. LNA, ENA
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
- C12N2320/32—Special delivery means, e.g. tissue-specific
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2330/00—Production
- C12N2330/30—Production chemically synthesised
Definitions
- the invention relates conjugated oligonucleotides which target microRNA-122 which comprise an antisense oligomer which is complementary to a microRNA-122 target sequence wherein the antisense oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
- Oligonucleotide conjugates have been extensively evaluated for use in siRNAs, where they are considered essential in order to obtain sufficient in vivo potency.
- WO2004/044141 refers to modified oligomeric compounds that modulate gene expression via an RNA interference pathway.
- the oligomeric compounds include one or more conjugate moieties that can modify or enhance the pharmacokinetic and pharmacodynamic properties of the attached oligomeric compound.
- WO2012/083046 reports on a galactose cluster-pharmacokinetic modulator targeting moiety for siRNAs.
- single stranded antisense oligonucleotides are typically administered therapeutically without conjugation or formulation.
- the main target tissues for antisense oligonucleotides are the liver and the kidney, although a wide range of other tissues are also accessible by the antisense modality, including lymph node, spleen, bone marrow.
- Cholesterol conjugates have been used to target siRNAs and antagomirs to the liver - see for example, Soutscheck et al., Nature 2004 vol. 432 173 - 178 and Krijtzfeldt et al., Nature 2005 vol 438, 685 - 689.
- the use of cholesterol as a liver targeting entity was found to be essential for in vivo activity.
- HCV Hepatitis C virus
- miravirsen which targets microRNA- 122, an essential host factor for HCV infection in the liver, is being developed for treatment of HCV and has shown efficacy for the treatment of hepatitis C (HCV) in both chimpanzees and human patients.
- Miravirsen is a microRNA-122 inhibitor.
- microRNA-122 (miR-122) is a liver specific microRNA. miR-122 which is involved in lipid and cholesterol metabolism, and inhibition of miR-122 in vivo in rodents results in a reduction in serum cholesterol levels. The molecular mechanisms whereby miR-122 regulated cholesterol metabolism are apparently, at present unknown.
- the present invention is based upon the discovery that the potency, bio-distribution and therapeutic index of single stranded antisense oligonucleotides targeting microRNA-122 can be vastly improved by the conjugation of the oligonucleotide to a carbohydrate conjugate, such as a GalNAc conjugate. The invention therefore provides novel therapeutic agents for treatment of HCV or cholesterol associated metabolic disease.
- PCT/EP2014/051781 which refers to LNA oligomer conjugates is also hereby incorporated by reference in its entirity.
- the invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
- HCV hepatitis C infection
- the invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to a conjugate group which comprises at least one N-acetylgalactosamine (GalNac) group.
- the invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to a conjugate group which comprises a galactose cluster.
- the invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to a conjugate group which comprises three N-acetylgalactosamine (GalNAc) groups (a trivalent GalNAc cluster).
- HCV hepatitis C infection
- GalNAc N-acetylgalactosamine
- the antisense oligonucleotide comprises a contiguous nucleotide sequence which is complementary to at least 8 contiguous nucleotides present in a hsa-miR-122 sequence.
- the contiguous nucleotide sequence of the antisense oligonucleotide is fully complementary to a corresponding region of hsa-miR-122.
- Such compounds are are refered to as targeting a microR-122 nucleic acid, such as hsa-miR-122.
- the contiguous nucleotide sequence of the antisense oligonucleotide comprises a region which is fully complementary to the hsa-miR-122 seed region.
- the contiguous nucleotide sequence of the antisense oligonucleotide comprises the sequence 5'-CACACTCC-3' or 5'-ACACTCC-3'.
- the invention also provides for a poly oligomeric oligomer targeting miR-122.
- the antisense oligonucleotide according to the invention may therefore comprise a first region which is complementary to at least 7 consecutive nucleotides of hsa-miR-122, and a second region which is complementary to at least 7 consecutive nucleotides of hsa-miR-122, wherein the first and the second regions are linked via a region physiologically labile nucleotides, such as 1 - 5 phosphodiester linked DNA nucleotides.
- the first and second regions may each be as according to the oligonucleotide region of the (mono-oligomeric) other compounds of the invention, and may in some embodiments be the same and in some be different oligomer sequences.
- the first and the second regions are 7 - 10 LNA nucleotides in length which each target the microRNA seed region.
- the nucleosides of the first and the second regions may be linked by phosphorothioate linkages.
- the invention provides for an antisense oligonucleotide which targets microRNA-122, for the treatment of high cholesterol, wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
- the antisense oligonucleotide of the invention comprises LNA nucleotides.
- the invention provides for an antisense oligonucleotide according to the invention for the treatment of a metabolic disease, such as a cholesterol associated metabolic disease, such as elevated cholesterol.
- the invention provides for an antisense oligonucleotide according to the invention for the treatment of hepatitis C infection.
- the invention provides for an antisense oligonucleotide which targets microRNA-122- wherein said antisense oligonucleotide is conjugated to an asialoglycoprotein receptor targeting moiety, for use in medicine.
- the invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the treatment of a metabolic disease or viral disease.
- the invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the treatment of HCV infection.
- the invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the improvement of hepatic function in a subject.
- the invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the reduction in necroinflammation in a subject.
- the invention provides for a method of treatment of HCV infection in a subject suffering from hepatitis, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention, to the subject.
- the invention provides for a method of treatment of HCV infection in a subject infected with HCV, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention, to the subject.
- the invention provides for a method of treatment of a metabolic disease a subject in need of treatment, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the imvention, to the subject.
- the uinvention provides for a method of improving the haptic function a subject in need of improved hepatic function, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention to the subject.
- the invention provides for a method of reducing necroinflammation a subject in need of reduced necroinflammation, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention, to the subject.
- the invention provides for an LNA antisense oligomer which targets microRNA-122 (which may be referred to as region A herein) comprising an antisense oligomer and an asialoglycoprotein receptor targeting conjugate moiety, such as a GalNAc moiety, which may form part of a further region (referred to as region C).
- the invention provides for an LNA antisense oligomer which targets microRNA-122
- region A (which may be referred to as region A herein) comprising an antisense oligomer and a GalNAc moiety, such as a trivalent GalNAc moiety which may form part of a further region (referred to as region C).
- the invention provides for an LNA antisense oligomer which targets microRNA-122 which is covalently joined to (e.g. linked to) a (non-nucleoside) carbohydrate moiety, such as a carbohydrate conjugate moiety.
- a carbohydrate moiety is not a linear carbohydrate polymer.
- the carbohydrate moiety may however be multi-valent, such as, for example 2, 3, 4 or 4 identical or non-identical carbohydrate moieties may be covalently joined to the oligomer, optionally via a linker or linkers.
- the invention provides for an LNA antisense oligomer (conjugate) comprising an LNA antisense oligomer and a conjugate moiety which comprises a carbohydrate, such as a carbohydrate conjugate moiety.
- the invention provides for the oligomeric compound according to the invention for use in the inhibition of miroRNA-122 target in a cell.
- the use is in vitro. In some embodiments the use is in vivo.
- the invention also provides for an oligomer which targets microRNA-122, such as an LNA oligomer which targets microRNA-122, comprising a contiguous region of 7 - 23 phosphorothioate linked nucleosides, and further comprising between 1 and 6 DNA nucleosides which are contiguous with the LNA oligomer, wherein the internucleoside linkages between the DNA, and/or adjacent to the DNA nucleoside(s), is physiologically labile, such as is / are phosphodiester linkages.
- an LNA oligomer may be in the form of a conjugate, as described herein. When conjugated, the conjugate may, for example a carbohydrate, such as a GalNac conjugate, such as a GalNac cluster, e.g. triGalNac, or another conjugate as described herein.
- FIG. 1 Examples of tri-GalNac conjugates which may be used.
- Conjuagtes 1 - 4 illustrate 4 suitable GalNac conjugate moieties, and conjugates 1 a - 4a refer to the same conjugates with an additional linker moiety (Y) which is used to link the conjugate to the oligomer (region A or to a biocleavable linker, such as region B).
- the wavy line represents the covalent link to the oligomer.
- FIG. 2 Silencing of miR-122 in the mouse liver by seed-targeting tiny LNA.
- (a) RNA blot analysis of liver RNAs from mice after treatment with three intravenous doses of 20 mg/kg tiny antimiR-122, 15-mer antimiR-122 or LNA scramble control or with saline.
- FIG. 1 Total Cholesterol analysis at pre-dose, day 4 and day7. Cholesterol is upregulated due to decreased miR122.
- FIG. 4 Expression of Aldo A and Bckdk was measured by standard TaqMan Q-PCR assays. The mRNA levels of these genes are upregulated due to decreased miR122.
- the invention provides for an antisense oligonucleotide which targets microRNA-122 (miR- 122), such as for use in the treatment of hepatitis C infection (HCV) or a metabolic disorder, wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
- the invention relates to LNA oligomeric compounds, such as LNA antisense oligonucleotides, which are covalently linked to a non-nucleotide carbohydrate conjugate group.
- the compound of the invention does not comprise RNA (units).
- the oligomer has a single contiguous sequence which is a linear molecule or is synthesized as a linear molecule. The oligomer may therefore be single stranded molecule.
- the oligomer does not comprise short regions of, for example, at least 3, 4 or 5 contiguous nucleotides, which are complementary to equivalent regions within the same oligomer (i.e. duplexes).
- the oligomer in some embodiments, may be not (essentially) double stranded.
- the oligomer is essentially not double stranded, such as is not a siRNA.
- the oligomeric compound is not in the form of a duplex with a (substantially) complementary oligonucleotide - e.g. is not an siRNA.
- oligomer in the context of the present invention, refers to a molecule formed by covalent linkage of two or more nucleotides (i.e. an oligonucleotide).
- a single nucleotide (unit) may also be referred to as a monomer or unit.
- nucleoside refers to a sequence of nucleotides or monomers, what is referred to is the sequence of bases, such as A, T, G, C or U.
- the oligomer may consists or comprises of a contiguous nucleotide sequence of from 7 - 30, such as 7 - 26 or 8 - 25, such as 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24, 25 nucleotides in length, such as 10 - 20 nucleotides in length.
- the length of the LNA oligomer is 10 - 16 nucleotides, such as 12, 13 or 14 nucleosides. In some embodiments, the LNA oligomer is 7, 8, 9 nucleosides in length, such as a "Tiny" LNA.
- the oligomers comprise or consist of a contiguous nucleotide sequence of a total of from 10 - 22, such as 12 - 18, such as 13 - 17 or 12 - 16, such as 13, 14, 15, 16 contiguous nucleotides in length.
- the oligomers comprise or consist of a contiguous nucleotide sequence of a total of 10, 1 1 , 12, 13, or 14 contiguous nucleotides in length.
- the oligomer according to the invention consists of no more than 22 nucleotides, such as no more than 20 nucleotides, such as no more than 18 nucleotides, such as 15, 16 or 17 nucleotides. In some embodiments the oligomer of the invention comprises less than 20 nucleotides. It should be understood that when a range is given for an oligomer, or contiguous nucleotide sequence length it includes the lower an upper lengths provided in the range, for example from (or between) 10 - 30, includes both 10 and 30.
- the use of the carbohydrate conjugates according to the invention have been found to be particularly suitable for short LNA oligomers, including short gapmers, mixmers or totalmers (tinys) such as LNA oligomers of less than 20, such as less than 18, such as 16nts or less or 15 or 14nts or less.
- the invention relates to LNA oligomeric compounds, such as LNA antisense oligonucleotides, which are covalently linked to a non-nucleotide carbohydrate conjugate group.
- LNA oligomer is an oligomer which comprises at least one LNA nucleoside, and may typically comprise at least 20% LNA nucleosides, such as at least 30% LNA nucleosides, such as at least 40% such as at least 50% LNA (not including region B).
- short LNA oligomers may be 7 - 12 nucleotides wherein at least 70% of the nucleotides are LNA (excluding region B), such as at least 80% LNA, such as at least 90% LNA, or all nucleotides are LNA nucleotides.
- Such short LNAs may be joined together via region B to form multiple oligomer conjugates (poly oligomer conjugates).
- the invention provides LNA antisense oligomer conjugate, comprising a LNA antisense oligomer and a conjugate moiety which comprises a
- carbohydrate such as a carbohydrate conjugate moiety, covalently bound to the LNA antisense oligomer.
- LNA oligomeric compounds also referred herein as LNA oligomers or LNA oligonucleotides
- An LNA oligomer comprises at least one "Locked Nucleic Acid” (LNA) nucleoside, such as a nucleoside which comprises a covalent bridge (also referred to a radical) between the 2' and 4' position (a 2' - 4' bridge).
- LNA nucleosides are also referred to as "bicyclic nucleosides”.
- the LNA oligomer is typically a single stranded antisense oligonucleotide.
- the LNA oligomer comprises or is a gapmer. In some embodiments the LNA oligomer comprises or is a mixmer. In some embodiments the LNA oligomer comprises or is a totalmer.
- the nucleoside analogues present in the oligomer are all LNA, and the oligomer may, optionally further comprise RNA or DNA, such as DNA nucleosides (e.g. in a gapmer or mixmer).
- the internucleoside linkages of the oligomer comprise at least one internucleoside linkage other than phosphodiester, such as at least one, such as at least 50%, such as at least 75%, such as at least 90%, such as 100% of the internucleoside linkages in the oligomer (or oligomer region or region A) are other than phosphodiester.
- the internucleoside linkages other than phosphodiester are sulphur containing internucleoside linkages, such as phosphorothioate, phosphorodithioate such as phosphorothioate.
- the oligomer may comprise at least one phosphorothioate internucleoside linkage, such as at least two, three or four phosphorothioate linkages, and in some embodiments at least 50% of the internucleoside linkages may be phosphorothioate, such as at least 75%, at least 90% or all internucleoside linkages (other than those, optionally present in a cleavable linker) may be phosphorothioate.
- the internucleoside linkages between the two terminal nucleosides at the 5' end, the 3' end or both the 5' and 3' end (other than in region B, when present), are sulphur containing internucleoside linkages, such as phosphorothioate.
- the oligomer comprises at least one region of consecutive DNA nucleosides, such as a region of 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12
- the internucleoside linkage between the DNA nucleosides is a sulphur containing internucleoside linkage such as phosphorothiote.
- the central region of a gapmer ( ⁇ '), which typically comprises a region of consecutive DNA nucleosides has are sulphur containing internucleoside linkages, such as phosphorothioate, between the nucleosides, such as between consecutive DNA nucleosides and/or between DNA nucleosides and nucleic acid analogue nucleosides, such as the sugar modified nucleosides referred to herein, such as LNA.
- LNA Locked Nucleic Acid Nucleosides
- Bicyclic nucleoside analogues include nucleoside analogues typically which comprise a bridge (or biradical) linking the second and forth carbon of the ribose ring, (C4 * -C2 * bridge or biradical).
- the presence of the biradical between the 2 nd and 4 th carbon locks the ribose into a 3' endo- (north) confirmation, and as such bicyclic nucleoside analogues with a C2 * -C4 * biradical are often referred to as Locked nucleic acid (LNA), or bicyclic nucleic acids (BNA).
- LNA Locked nucleic acid
- BNA bicyclic nucleic acids
- nucleosides of the LNA oligomer may be modified nucleosides, also referred to as nucleoside analogues herein, such as sugar modified nucleoside analogues, for example bicyclic nucleoside analogues (e.g. LNA) and/or 2'substituted nucleoside analogues.
- nucleoside analogues such as sugar modified nucleoside analogues, for example bicyclic nucleoside analogues (e.g. LNA) and/or 2'substituted nucleoside analogues.
- the nucleoside analogues present in the oligomer all comprise the same sugar modification, for example are all bicyclic nucleoside analogues, such as they may be (optionally independently) selected from the group consisting of beta-D-X-LNA or alpha-L-X-LNA (wherein X is oxy, amino or thio), or other LNAs disclosed herein including, but not limited to,(R/S) cET, cMOE or 5'-Me-LNA.
- the oligomer may comprise at least one bicyclic nucleoside (LNA) and at least one further nucleoside analogue, such as one or more 2'substituted nucleoside.
- LNA bicyclic nucleoside
- nucleoside analogue such as one or more 2'substituted nucleoside.
- some or all of the nucleosides of the oligomer may be modified nucleosides, also referred to as nucleoside analogues herein.
- the oligomer or oligomer region (region A) comprises at least one, such as at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21 , at least 22, at least 23, at least 24 or 25 nucleoside analogues.
- nucleoside analogues are (optionally independently) selected from the group consisting of bicyclic nucleoside analogues (such as LNA), and/or 2' substituted nucleoside analogues, such as (optionally independently) selected from the group consisting of 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino- DNA units, 2'-AP, 2'-FANA, 2'-(3-hydroxy)propyl, and 2'-fluoro-DNA units, and/or other (optionally) sugar modified nucleoside analogues such as morpholino, peptide nucleic acid (PNA), CeNA, unlinked nucleic acid (UNA), hexitol nucleoic acid (HNA).
- PNA peptide nucleic acid
- CeNA unlinked nucleic acid
- HNA hexitol nucleoic acid
- the further nucleoside analogues increase the affinity of the first region for its target nucleic acid (or a complementary DNA or RNA sequence).
- Various nucleoside analogues are disclosed in Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, hereby incorporated by reference.
- the LNA oligomer comprises at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, for example all nucleoside analogues (or in a totalmer all nucleosides) bicyclic nucleoside analogues, such as LNA, e.g. beta-D-X-LNA or alpha-L- X-LNA (wherein X is oxy, amino or thio, such as oxy), or other LNAs disclosed herein including, but not limited to,(R/S) cET, cMOE or 5'-Me-LNA.
- the LNA oligomer comprises of DNA and sugar modified nucleoside analogues, such as bicyclic nucleoside analogues and/or 2'substituted nucleoside analogues. In some embodiments, the oligomer comprises of DNA and LNA nucleoside units.
- WO05/013901 WO07/027775, WO07027894 refers to fully 2'substituted oligomers, such as fully 2'-0-MOE.
- the first region of the oligomer may comprise of 2' substituted nucleosides.
- WO07/027775 also refers to MOE, LNA, DNA mixmers for use in targeting microRNAs.
- the oligomer of the invention targets the liver expressed microRNA-122, such as hsa- miR-122. Oligomer's targeting miR-122 are disclosed in WO2007/1 12754,
- WO2007/1 12753, WO2009/043353 may be mixmers, such as SPC3649, also referred to as miravirsen (which has the sequence 5'-CcAttGTcaCaCtCC-3' (SEQ ID NO 1 ) , where capital letters are beta-D-oxy LNA, small letters are DNA, fully phosphorothioate and LNA C are 5-methyl cyctosine), or a tiny LNA, such as those disclosed in WO2009/043353 (e.g.
- the miR-122 targeting oligomers have a length of 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17 or 18 nucleotides in length.
- the miR- 122 targeting oligomers a sequence which is fully complementary to miR-122 as measured accross the length of the oligomer, and preferably include the sequence 5'-CACACTCC-3'. According to miRBase, the mature microRNA-122 sequence is 5'
- the oligomer targeting a microRNA such as miR-122 is complementary to a corresponding region of the microRNA accorss the length of the oligomer and in some embodiments the 3' nucleoside of the oligomer is compelmentary to (i.e. aligns to) the first, second, third or fourth 5' nucleotides of the microRNA, such as miR-122, such as the second 5' nucleotide of the microRNA, such as miR-122.
- the disease may be a viral disease, such as hepatitis, including hepatitis B and hepatitis C, or a metabolic disease related to elevated cholesterol, such as atherosclosis and hyperlipidaemia and related disorders. Oligomers targeting miR-122 may also be used for the improvement of hepatic function (see e.g.
- the liver disease may be a disease or disorder selected from the group consisting of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis; or from the group consisting of a disease or disorder selected from the group consisting of cytomegalovirus infection, schistosomiasis infection and Leptospirosis infection.
- the invention provides a method of down-regulating or inhibiting the expression of the target in a cell which is expressing the target, said method comprising administering the oligomer or conjugate according to the invention to said cell to down-regulating or inhibiting the expression of the target in said cell.
- the cell is a mammalian cell such as a human cell.
- the administration may occur, in some embodiments, in vitro.
- the first region (or first and second region) form a single contiguous nucleobase sequence which is complementary, to a corresponding region of a microRNA- 122 such as miR-122a (i.e. targets), such as the hsa-miR-122 sequences (miRBase release 20: MI0000442), such as:
- miR-122 has been indicated in HCV infection, where it is an essential host factor required for maintenance of the infection. Inhibitors of miR-122 may therefore be used in the treatment of hepatitis C infection.
- the invention therefore provides for the oligomer according to the invention which targets miR-122 for use in the treatment of HCV infection.
- the invention further provides for a method of treatment of HCV infection, wherein said method comprises the administration of the oligomer of the invention to a subject in need to said treatment.
- the invention provides for the use of an oligomer of the invention which targets miR- 122, for the manufacture of a medicament for the treatment of HCV infection.
- the invention provides for a method of treating HCV infection, said method
- the invention provides for an in vivo or in vitro method for the inhibition of miR-122 in a cell which is expressing miR-122, such as an HCV infected cell or a HCV replicon expressing cell, said method comprising administering an oligomer or conjugate or pharmaceutical composition according to the invention to said cell so as to inhibit miR-122 in said cell.
- miR-122 has also been indicated in cholesterol metabolism, and it has been suggested that inhibition of miR-122 may be used for a treatment to reduce plasma cholesterol levels (Esau, Cell Metab. 2006 Feb;3(2):87-98.)
- Inhibitors of miR-122 may therefore be used in a treatment to reduce plasma cholesterol levels, or in the treatment of a metabolic disease associated with elevated levels of cholesterol (related disorders), such as indications selected from the group consisting of liver steatosis,_atherosclerosis, hyperlipidaemia, hypercholesterolemia, familiar hypercholesterolemia, dyslipidemias, coronary artery disease (CAD), and coronary heart disease (CHD)
- a metabolic disease associated with elevated levels of cholesterol related disorders
- indications selected from the group consisting of liver steatosis,_atherosclerosis, hyperlipidaemia, hypercholesterolemia, familiar hypercholesterolemia, dyslipidemias, coronary artery disease (CAD), and coronary heart disease (CHD)
- Compounds of the invention which target miR-122 may be used in the treatment of elevated cholesterol levels or related disorders.
- the invention therefore provides for the oligomer according to the invention which targets miR-122 for use in the treatment of elevated cholesterol levels or related disorders.
- the invention further provides for a method of treatment of elevated cholesterol levels or related disorders, wherein said method comprises the administration of the oligomer of the invention to a subject in need to said treatment.
- the invention provides for the use of an oligomer of the invention which targets miR- 122, for the manufacture of a medicament for the treatment of elevated cholesterol levels or related disorders.
- the invention provides for a method of treating elevated cholesterol levels or related disorders, said method comprising administering an effective amount of an oligomer according to the invention which targets miR-122, to a patient suffering from said disorder.
- the invention provides for an in vivo or in vitro method for the inhibition of miR-122 in a cell which is expressing miR-122, such as an HCV infected cell or a HCV replicon expressing cell, said method comprising administering an oligomer or conjugate or pharmaceutical composition according to the invention to said cell so as to inhibit miR-122 in said cell.
- Oligomer's targeting miR-122 are disclosed in WO2007/1 12754, WO2007/1 12753,
- WO2009/043353 may be mixmers, such as SPC3649, also referred to as miravirsen see below, or a tiny LNA, such as those disclosed in WO2009/043353 (e.g. 5'-ACACTCC- 3', 5'-CACACTCC-3', 5'-TCACACTCC-3', (SEQ ID NOs 3 - 5) where capital letters are beta- D_oxy LNA, fully phosphorothioate and LNA C are 5-methyl cyctosine).
- SPC3649 also referred to as miravirsen see below
- a tiny LNA such as those disclosed in WO2009/043353 (e.g. 5'-ACACTCC- 3', 5'-CACACTCC-3', 5'-TCACACTCC-3', (SEQ ID NOs 3 - 5) where capital letters are beta- D_oxy LNA, fully phosphorothioate and LNA C are 5-methyl cyctosine).
- the miR-122 targeting oligomers have a length of 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17 or 18 (or 19, 20, 21 , 22 or 23 nucleotides) in length.
- the miR- 122 targeting oligomers a sequence which is fully complementary to miR-122 as measured accross the length of the oligomer, and preferably include the sequence 5'-CACACTCC-3' (SEQ ID NO 3).
- the oligomer targeting a microRNA such as miR- 122 is complementary to a corresponding region of the microRNA accorss the length of the oligomer and in some embodiments the 3' nucleoside of the oligomer is compelmentary to (i.e. aligns to) the first, second, third or fourth 5' nucleotides of the microRNA, such as miR- 122, such as the second 5' nucleotide of the microRNA, such as miR-122.
- the following is an oligomers which targets the hsa-miR-122 (human miR-122), and may be used as region A in the compounds of the invention.
- Miravirsen 5'- m CsCsA s t s t s GsT s c s as m Csas m Csts m Cs m C -3' (SEQ ID NO 1 )
- miR-122 targeting compounds which may be used in the context of the present invention (region A) are disclosed in WO2007/027894, WO2007/027775.
- the contiguous nucleotide sequence comprises no more than a single mismatch when hybridizing to the target sequence.
- Region B may however be non- complementary and may therefore be disregarded when determining the degree of complementarity.
- the degree of “complementarity” is expressed as the percentage identity (or percentage homology) between the sequence of the oligomer (or region thereof) and the sequence of the target region (or the reverse complement of the target region) that best aligns therewith. The percentage is calculated by counting the number of aligned bases that are identical between the 2 sequences, dividing by the total number of contiguous monomers in the oligomer, and multiplying by 100. In such a comparison, if gaps exist, it is preferable that such gaps are merely mismatches rather than areas where the number of monomers within the gap differs between the oligomer of the invention and the target region.
- corresponding to and “corresponds to” refer to the comparison between the nucleotide sequence of the oligomer (i.e. the nucleobase or base sequence) or contiguous nucleotide sequence (a first region) and the equivalent contiguous nucleotide sequence of a further sequence selected from either i) a sub-sequence of the reverse complement of the nucleic acid target. Nucleotide analogues are compared directly to their equivalent or corresponding nucleotides.
- a first sequence which corresponds to a further sequence under i) or ii) typically is identical to that sequence over the length of the first sequence (such as the contiguous nucleotide sequence) or, as described herein may, in some embodiments, is at least 80% homologous to a corresponding sequence, such as at least 85%, at least 90%, at least 91 %, at least 92%at least 93%, at least 94%, at least 95%, at least 96% homologous, such as 100% homologous (identical).
- nucleotide analogue and “corresponding nucleotide” are intended to indicate that the nucleotide in the nucleotide analogue and the naturally occurring nucleotide are identical.
- the "corresponding nucleotide analogue” contains a pentose unit (different from 2-deoxyribose) linked to an adenine.
- the contiguous nucleobase sequence of the oligomer may therefore be
- nucleotide refers to a glycoside comprising a sugar moiety
- nucleotide analogue a base moiety and a covalently linked group (linkage group), such as a phosphate or phosphorothioate internucleotide linkage group, and covers both naturally occurring nucleotides, such as DNA or RNA, and non-naturally occurring nucleotides comprising modified sugar and/or base moieties, which are also referred to as "nucleotide analogues" herein.
- a single nucleotide (unit) may also be referred to as a monomer or nucleic acid unit.
- nucleoside and nucleotide are used to refer to both naturally occurring nucleotides/sides, such as DNA and RNA, as well as nucleotide/side analogues.
- nucleoside is commonly used to refer to a glycoside comprising a sugar moiety and a base moiety, and may therefore be used when referring to the nucleotide units, which are covalently linked by the internucleoside linkages between the nucleotides of the oligomer.
- nucleotide is often used to refer to a nucleic acid monomer or unit, and as such in the context of an oligonucleotide may refer to the base - such as the "nucleotide sequence”, typically refer to the nucleobase sequence (i.e. the presence of the sugar backbone and internucleoside linkages are implicit).
- nucleotide may refer to a nucleic acid monomer or unit, and as such in the context of an oligonucleotide may refer to the base - such as the "nucleotide sequence", typically refer to the nucleobase sequence (i.e. the presence of the sugar backbone and internucleoside linkages are implicit).
- nucleoside for example the term “nucleotide” may be used, even when specifying the presence or nature of the linkages between the nucleosides.
- the 5' terminal nucleotide of an oligonucleotide does not comprise a 5' internucleoside linkage group, although may or may not comprise a 5' terminal group.
- Non-naturally occurring nucleotides include nucleotides which have modified sugar moieties, such as bicyclic nucleotides or 2' modified nucleotides, such as 2' substituted nucleotides.
- Nucleotide analogues are variants of natural nucleotides, such as DNA or RNA nucleotides, by virtue of modifications in the sugar and/or base moieties. Analogues could in principle be merely “silent” or “equivalent” to the natural nucleotides in the context of the oligonucleotide, i.e. have no functional effect on the way the oligonucleotide works to inhibit target gene expression. Such "equivalent” analogues may nevertheless be useful if, for example, they are easier or cheaper to manufacture, or are more stable to storage or manufacturing conditions, or represent a tag or label.
- the analogues will have a functional effect on the way in which the oligomer works to inhibit expression; for example by producing increased binding affinity to the target and/or increased resistance to intracellular nucleases and/or increased ease of transport into the cell.
- nucleoside analogues are described by e.g. Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, and in Scheme 1 :
- the oligomer may thus comprise or consist of a simple sequence of natural occurring nucleotides - preferably 2'-deoxynucleotides (referred here generally as "DNA”), but also possibly ribonucleotides (referred here generally as "RNA”), or a combination of such naturally occurring nucleotides and one or more non-naturally occurring nucleotides, i.e. nucleotide analogues.
- DNA 2'-deoxynucleotides
- RNA ribonucleotides
- nucleotide analogues may suitably enhance the affinity of the oligomer for the target sequence.
- nucleotide analogues examples include tricyclic nucleic acids, for example please see WO2013154798 and WO2013154798 which are hereby incorporated by reference.
- Oligomeric compounds such as antisense oligonucleotides, such as the compounds referred to herein, including region A, and in some optional embodiments, region B, may contain one or more nucleosides wherein the sugar group has been modified.
- Such sugar modified nucleosides may impart enhanced nuclease stability, increased binding affinity, or some other beneficial biological property to the antisense compounds.
- nucleosides comprise a chemically modified
- the oligomer, or first region thereof comprises at least one , such as at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21 , at least 22, at least 23, at least 24 or 25 nucleoside analogues, such as sugar modified nucleoside analogues.
- nucleoside analogues are (optionally independently selected from the group consisting of bicyclic nucleoside analogues (such as LNA), and/or 2' substituted nucleoside analogues, such as (optionally independently) selected from the group consisting of 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino-DNA units, 2'-AP, 2'- FANA, 2'-(3-hydroxy)propyl, and 2'-fluoro-DNA units, and/or other (optionally) sugar modified nucleoside analogues such as morpholino, peptide nucleic acid (PNA), CeNA, unlinked nucleic acid (UNA), hexitol nucleoic acid (HNA). bicyclo-HNA (see e.g.
- the nucleoside analogues increase the affinity of the first region for its target nucleic acid (or a complementary DNA or RNA sequence).
- the oligomer comprises at least one bicyclic nucleotide analogue, such as LNA.
- the first region comprises of at least one bicyclic nucleoside analogues (e.g. LNA) and/or 2'substituted nucleoside analogues.
- the nucleoside analogues present in the oligomer all comprise the same sugar modification.
- At least one nucleoside analogue present in the first region is a bicyclic nucleoside analogue, such as at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, for example all nucleoside analogues (except the DNA and or RNA nucleosides of region B) are sugar modified nucleoside analogues, such as such as bicyclic nucleoside analogues, such as LNA, e.g.
- beta-D-X-LNA or alpha-L- X-LNA (wherein X is oxy, amino or thio), or other LNAs disclosed herein including, but not limited to,(R/S) cET, cMOE or 5'-Me-LNA.
- substituent groups including 5' and 2' substituent groups
- BNA bicyclic nucleic acids
- Examples of chemically modified sugars include, 2'-F-5'-methyl substituted nucleoside (see, PCT International Application WO 2008/101 157, published on 8/21/08 for other disclosed 5', 2'-bis substituted nucleosides), replacement of the ribosyl ring oxygen atom with S with further substitution at the 2'-position (see, published U.S. Patent Application
- nucleosides having modified sugar moieties include, without limitation, nucleosides comprising 5'-vinyl, 5'-methyl (R or S), 4'-S, 2'-F, 2'-OCH 3 , and 2'-0(CH 2 )2 O CH 3 substituent groups.
- bicyclic nucleosides refer to modified nucleosides comprising a bicyclic sugar moiety.
- examples of bicyclic nucleosides include, without limitation, nucleosides comprising a bridge between the 4' and the 2' ribosyl ring atoms.
- compounds provided herein include one or more bicyclic nucleosides wherein the bridge comprises a 4' to 2' bicyclic nucleoside.
- 4' to 2' bicyclic nucleosides include, but are not limited to, one of the formulae: 4'-(CH 2 )- O -2' (LNA); 4'- (CH 2 )-S-2'; 4 -(CH 2 ) 2 - O -2' (ENA); 4'-CH(CH 3 )- O -2' and 4'-CH(CH 2 OCH 3 )-0-2 * , and analogs thereof (see, U.S.
- PCT/US2008/064591 PCT/US2008/066154, and PCT/US2008/068922.
- Each of the foregoing bicyclic nucleosides can be prepared having one or more stereochemical sugar configurations including for example a-L-ribofuranose and beta -D-ribofuranose (see PCT international application PCT DK98/00393, published on March 25, 1999 as WO 99/14226).
- the bridge of a bicyclic sugar moiety is, -[C(R a )(Rb)] n -, - [C(Ra)( b)] n - O -, -C(R a R b )-N(R)- O - or, -C(R a R b )- O -N(R)-.
- the bridge is 4'-CH 2 -2', 4'-(CH 2 ) 2 -2', 4'- (CH 2 ) 3 -2', 4'-CH 2 - O -2', 4 * -(CH 2 )2- O -2', 4'-CH 2 - O - N(R)-2', and 4'-CH 2 -N(R)- O -2'-, wherein each R is, independently, H, a protecting group, or Ci-Ci 2 alkyl.
- bicyclic nucleosides are further defined by isomeric
- a nucleoside comprising a 4'-2' methylene-oxy bridge
- a nucleoside may be in the a-L configuration or in the beta - D configuration.
- a-L-methyleneoxy (4'-CH 2 - 0-2') BNA's have been incorporated into antisense oligonucleotides that showed antisense activity (Frieden et al, Nucleic Acids Research, 2003, 21 , 6365- 6372).
- bicyclic nucleosides include, but are not limited to, (A) a-L- Methyleneoxy (4'-CH 2 -0-2') BNA, (B) beta -D-Methyleneoxy (4'-CH 2 -0-2') BNA, (C)
- Bx is the base moiety and R is, independently, H, a protecting group or Ci-C 2 alkyl.
- R is, independently, H, a protecting group or Ci-C 2 alkyl.
- Bx is a heterocyclic base moiety
- R c is Ci-Ci 2 alkyl or an amino protecting group
- T a and T b are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium.
- bicyclic nucleoside having Formula II having Formula II:
- Bx is a heterocyclic base moiety
- T a and T b are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium;
- Z a is CrC 6 alkyi, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, substituted CrC 6 alkyi, substituted C 2 -C 6 alkenyl, substituted C 2 -C 6 alkynyl, acyl, substituted acyl, substituted amide, thiol, or substituted thio.
- Bx is a heterocyclic base moiety
- T a and T b are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium;
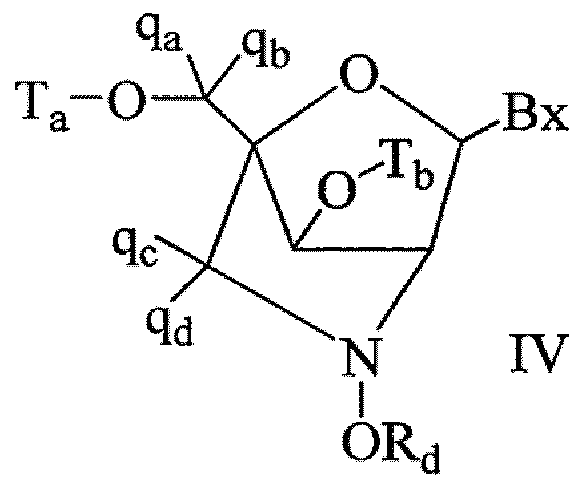
- bicyclic nucleoside having Formula IV having Formula IV:
- Bx is a heterocyclic base moiety
- T a and T b are each, independently H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium;
- R d is Ci-C 6 alkyl, substituted Ci-C 6 alkyl, C 2 -C 6 alkenyl, substituted C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, substituted C 2 -C 6 alkynyl; each q b , q c and q d is, independently, H, halogen, Ci-C 6 alkyl, substituted Ci-C 6 alkyl, C 2 -Ce alkenyl, substituted C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, or substituted C 2 -C6 alkynyl, CrC 6 alkoxyl, substituted Q- C 6 alkoxyl, acyl, substituted acyl, d- C 6 aminoalkyl, or substituted Ci-C 6 aminoalkyl;
- Bx is a heterocyclic base moiety
- T a and T b are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium;
- the bichang nucleoside has Formula VI:
- Bx is a heterocyclic base moiety
- 4'-2' bicyclic nucleoside or “4' to 2' bicyclic nucleoside” refers to a bicyclic nucleoside comprising a furanose ring comprising a bridge connecting the 2' carbon atom and the 4' carbon atom.
- nucleosides refer to nucleosides comprising modified sugar moieties that are not bicyclic sugar moieties.
- sugar moiety, or sugar moiety analogue, of a nucleoside may be modified or substituted at any position.
- 2'-modified sugar means a furanosyl sugar modified at the 2' position.
- such modifications include substituents selected from: a halide, including, but not limited to substituted and unsubstituted alkoxy, substituted and
- 2' modifications are selected from substituents including, but not limited to: 0[(CH 2 ) n O] m CH 3 , 0(CH 2 ) threadNH 2 , 0(CH 2 ) administratCH 3 , 0(CH 2 ) threadONH 2 ,
- modified nucleosides comprise a 2'-MOE side chain ⁇ see, e.g., Baker et al., J. Biol. Chem., 1997, 272, 1 1944-12000).
- a "modified tetrahydropyran nucleoside” or “modified THP nucleoside” means a nucleoside having a six-membered tetrahydropyran "sugar” substituted in for the pentofuranosyl residue in normal nucleosides (a sugar surrogate). Modified ?THP
- nucleosides include, but are not limited to, what is referred to in the art as hexitol nucleic acid (HNA), anitol nucleic acid (ANA), manitol nucleic acid (MNA) ⁇ see Leumann, CJ. Bioorg. and Med. Chem. (2002) 10:841 -854), fluoro HNA (F-HNA), or those compounds having Formula X:
- HNA hexitol nucleic acid
- ANA anitol nucleic acid
- MNA manitol nucleic acid
- F-HNA fluoro HNA
- Bx is a heterocyclic base moiety
- T 3 and T 4 are each, independently, an internucleoside linking group linking the
- tetrahydropyran nucleoside analog to the antisense compound or one of T 3 and T 4 is an internucleoside linking group linking the tetrahydropyran nucleoside analog to the antisense compound and the other of T 3 and T4 is H, a hydroxyl protecting group, a linked conjugate group, or a 5' or 3'-terminal group;
- the modified THP nucleosides of Formula X are provided wherein q m , q n , q P , q r , q s , qt, and q u are each H. In some embodiments, at least one of q m , q n , q , q r , q s , qt and q u is other than H. In some embodiments, at least one of q m , q n , q , q r , q s , qtand q u is methyl. In some embodiments, THP nucleosides of Formula X are provided wherein one of Ri and R 2 is F. In some embodiments, Riis fluoro and R 2 is H, R-i is methoxy and R 2 is H, and Ri is methoxyethoxy and R 2 is H.
- 2'-modified or “2'-substituted” refers to a nucleoside comprising a sugar comprising a substituent at the 2' position other than H or OH. 2'-modified
- 2'-modifed nucleosides may further comprise other modifications, for example, at other positions of the sugar and/or at the nucleobase.
- 2'-F refers to a sugar comprising a fluoro group at the 2' position.
- 2'-OMe or “2'-OCH 3 " or “2'-0-methyl” each refers to a nucleoside comprising a sugar comprising an -OCH 3 group at the 2' position of the sugar ring.
- oligonucleotide refers to a compound comprising a plurality of linked nucleosides.
- an oligonucleotide comprises one or more ribonucleosides (RNA) and/or deoxyribonucleosides (DNA).
- nucleosides for incorporation into antisense compounds ⁇ see, e.g., review article: Leumann, J. C, Bioorganic and Medicinal Chemistry, 2002, 10, 841 -854). Such ring systems can undergo various additional substitutions to enhance activity. Methods for the preparations of modified sugars are well known to those skilled in the art. In nucleotides having modified sugar moieties, the nucleobase moieties (natural, modified, or a combination thereof) are maintained for hybridization with an appropriate nucleic acid target.
- antisense compounds comprise one or more nucleotides having modified sugar moieties.
- the modified sugar moiety is 2'- MOE.
- the 2'-MOE modified nucleotides are arranged in a gapmer motif.
- the modified sugar moiety is a cEt.
- the cEt modified nucleotides are arranged throughout the wings of a gapmer motif.
- R 4* and R 2* together designate the biradical -0-CH(CH 2 OCH 3 )- (2'0-methoxyethyl bicyclic nucleic acid - Seth at al., 2010, J. Org. Chem) - in either the R- or S- configuration.
- R 4* and R 2* together designate the biradical -0-CH(CH 2 CH 3 )- (2'0-ethyl bicyclic nucleic acid - Seth at al., 2010, J. Org. Chem). - in either the R- or S- configuration.
- R 4* and R 2* together designate the biradical -0-CH(CH 3 )-. - in either the R- or S- configuration. In some embodiments, R 4* and R 2* together designate the biradical -0-CH 2 -0-CH 2 - - (Seth at al., 2010, J. Org. Chem).
- R 4* and R 2* together designate the biradical -O-NR-CH 3 - (Seth at al., 2010, J. Org. Chem) .
- the LNA units have a structure selected from the following group:
- conjugates of the present invention such as trivalent GalNAc conjugates are therefore highly useful in reducing the nephrotoxicity of LNA compounds, such as cET compounds.
- the oligomer may thus comprise or consist of a simple sequence of natural occurring nucleotides - preferably 2'-deoxynucleotides (referred to here generally as "DNA”), but also possibly ribonucleotides (referred to here generally as "RNA”), or a combination of such naturally occurring nucleotides and one or more non-naturally occurring nucleotides, i.e. nucleotide analogues.
- nucleotide analogues may suitably enhance the affinity of the oligomer for the target sequence.
- affinity-enhancing nucleotide analogues in the oligomer such as BNA, (e.g.) LNA or 2'-substituted sugars, can allow the size of the specifically binding oligomer to be reduced, and may also reduce the upper limit to the size of the oligomer before nonspecific or aberrant binding takes place.
- the oligomer comprises at least 1 nucleoside analogue. In some embodiments the oligomer comprises at least 2 nucleotide analogues. In some embodiments, the oligomer comprises from 3-8 nucleotide analogues, e.g. 6 or 7 nucleotide analogues. In the by far most preferred embodiments, at least one of said nucleotide analogues is a BNA, such as locked nucleic acid (LNA); for example at least 3 or at least 4, or at least 5, or at least 6, or at least 7, or 8, of the nucleotide analogues may be BNA, such as LNA. In some embodiments all the nucleotides analogues may be BNA, such as LNA.
- LNA locked nucleic acid
- the oligomers of the invention which are defined by that sequence may comprise a corresponding nucleotide analogue in place of one or more of the nucleotides present in said sequence, such as BNA units or other nucleotide analogues, which raise the duplex stability/T m of the oligomer/target duplex (i.e. affinity enhancing nucleotide analogues).
- a preferred nucleotide analogue is LNA, such as oxy-LNA (such as beta-D-oxy-LNA, and alpha-L-oxy-LNA), and/or amino-LNA (such as beta-D-amino-LNA and alpha-L-amino- LNA) and/or thio-LNA (such as beta-D-thio-LNA and alpha-L-thio-LNA) and/or ENA (such as beta-D-ENA and alpha-L-ENA).
- LNA such as oxy-LNA (such as beta-D-oxy-LNA, and alpha-L-oxy-LNA), and/or amino-LNA (such as beta-D-amino-LNA and alpha-L-amino- LNA) and/or thio-LNA (such as beta-D-thio-LNA and alpha-L-thio-LNA) and/or ENA (such as beta-D-ENA and alpha-L-EN
- the further nucleotide analogues present within the oligomer of the invention are independently selected from, for example: 2'-0-alkyl-RNA units, 2'-amino- DNA units, 2'-fluoro-DNA units, BNA units, e.g. LNA units, arabino nucleic acid (ANA) units, 2'-fluoro-ANA units, HNA units, INA (intercalating nucleic acid -Christensen, 2002. Nucl.
- nucleotide analogues present in the oligomer of the invention, such as the first region, or contiguous nucleotide sequence thereof.
- the further nucleotide analogues are 2'-0-methoxyethyl-RNA
- oligonucleotide of the invention may comprise nucleotide analogues which are
- At least one of said nucleotide analogues is 2'-MOE-RNA, such as 2, 3, 4, 5, 6, 7, 8, 9 or 10 2'-MOE-RNA nucleotide units.
- at least one of said nucleotide analogues is 2'-fluoro DNA, such as 2, 3, 4, 5, 6, 7, 8, 9 or 10 2'-fluoro-DNA nucleotide units.
- the oligomer according to the invention comprises at least one BNA, e.g. Locked Nucleic Acid (LNA) unit, such as 1 , 2, 3, 4, 5, 6, 7, or 8 BNA LNA units, such as from 3 - 7 or 4 to 8 BNA LNA units, or 3, 4, 5, 6 or 7 BNA/LNA units.
- BNA Locked Nucleic Acid
- all the nucleotide analogues are BNA, such as LNA.
- the oligomer may comprise both beta-D-oxy-LNA, and one or more of the following LNA units: thio-LNA, amino-LNA, oxy-LNA, and/or ENA in either the beta-D or alpha-L configurations or combinations thereof.
- the oligomer (such as the first and optionally second regions) may comprise both BNA and LNA and DNA units.
- the combined total of LNA and DNA units is 10-25, such as 10 - 24, preferably 10-20, such as 10 - 18, such as 12-16.
- the nucleotide sequence of the oligomer, of first region thereof, such as the contiguous nucleotide sequence consists of at least one BNA, e.g. LNA and the remaining nucleotide units are DNA units.
- the oligomer, or first region thereof comprises only BNA, e.g. LNA, nucleotide analogues and naturally occurring nucleotides (such as RNA or DNA, most preferably DNA nucleotides), optionally with modified internucleotide linkages such as phosphorothioate.
- BNA e.g. LNA
- nucleotide analogues such as RNA or DNA, most preferably DNA nucleotides
- naturally occurring nucleotides such as RNA or DNA, most preferably DNA nucleotides
- modified internucleotide linkages such as phosphorothioate.
- nucleobase refers to the base moiety of a nucleotide and covers both naturally occurring a well as non-naturally occurring variants. Thus, “nucleobase” covers not only the known purine and pyrimidine heterocycles but also heterocyclic analogues and tautomeres thereof. It will be recognised that the DNA or RNA nucleosides of region B may have a naturally occurring and/or non-naturally occurring nucleobase(s), such as DNA nucleobases independently selected from the group A, C, T and G, or the group C, T and G.
- nucleobases include, but are not limited to adenine, guanine, cytosine, thymidine, uracil, xanthine, hypoxanthine, 5-methylcytosine, isocytosine, pseudoisocytosine, 5-bromouracil, 5-propynyluracil, 6-aminopurine, 2-aminopurine, inosine, diaminopurine, and 2-chloro-6-aminopurine.
- the nucleobases may be independently selected from the group consisting of adenine, guanine, cytosine, thymidine, uracil, 5- methylcytosine.
- the nucleobases may be independently selected from the group consisting of adenine, guanine, cytosine, thymidine, and 5-methylcytosine.
- at least one of the nucleobases present in the oligomer is a modified nucleobase selected from the group consisting of 5-methylcytosine, isocytosine, pseudoisocytosine, 5-bromouracil, 5-propynyluracil, 6-aminopurine, 2-aminopurine, inosine, diaminopurine, and 2-chloro-6-aminopurine.
- LNA refers to a bicyclic nucleoside analogue which comprises a C2 * - C4 * biradical (a bridge), and is known as "Locked Nucleic Acid”. It may refer to an LNA monomer, or, when used in the context of an "LNA oligonucleotide", LNA refers to an oligonucleotide containing one or more such bicyclic nucleotide analogues.
- bicyclic nucleoside analogues are LNA nucleotides, and these terms may therefore be used interchangeably, and is such embodiments, both are be characterised by the presence of a linker group (such as a bridge) between C2' and C4' of the ribose sugar ring.
- a linker group such as a bridge
- Y is selected from the group consisting of -0-, -CH 2 0-, -S-, -NH-, N(R e ) and/or - CH 2 -;
- Z and Z * are independently selected among an internucleotide linkage, R H , a terminal group or a protecting group;
- B constitutes a natural or non-natural nucleotide base moiety (nucleobase), and
- R H is selected from hydrogen and Ci -4 -alkyl;
- R a , R b R c , R d and R e are, optionally independently, selected from the group consisting of hydrogen, optionally substituted Ci-i 2 -alkyl, optionally substituted C 2- i2-alkenyl, optionally substituted C 2- i2-alkynyl, hydroxy, Ci-i 2 -alkoxy, C 2- i 2 -alkoxyalkyl, C 2- i 2 -alkenyloxy, carboxy, Ci-i
- R a , R b R c , R d and R e are, optionally independently, selected from the group consisting of hydrogen and Ci -6 alkyl, such as methyl.
- asymmetric groups may be found in either R or S orientation, for example, two exemplary
- stereochemical isomers include the beta-D and alpha-L isoforms, which may be illustrated as follows:
- thio-LNA comprises a locked nucleotide in which Y in the general formula above is selected from S or -CH 2 -S-.
- Thio-LNA can be in both beta-D and alpha-L- configuration.
- amino-LNA comprises a locked nucleotide in which Y in the general formula above is selected from -N(H)-, N(R)-, CH 2 -N(H)-, and -CH 2 -N(R)- where R is selected from hydrogen and Ci -4 -alkyl.
- Amino-LNA can be in both beta-D and alpha-L- configuration.
- Oxy-LNA comprises a locked nucleotide in which Y in the general formula above represents -0-. Oxy-LNA can be in both beta-D and alpha-L-configuration.
- ENA comprises a locked nucleotide in which Y in the general formula above is -CH 2 -0- (where the oxygen atom of -CH 2 -0- is attached to the 2'-position relative to the base B).
- R e is hydrogen or methyl.
- LNA is selected from beta-D-oxy-LNA, alpha-L-oxy-LNA, beta-D-amino-LNA and beta-D-thio-LNA, in particular beta-D-oxy-LNA.
- an oligomeric compound may function via non RNase mediated degradation of target mRNA, such as by steric hindrance of translation, or other methods,
- the oligomers of the invention are capable of recruiting an
- RNase endoribonuclease
- an oligomer is deemed essentially incapable of recruiting
- the RNaseH initial rate is less than 1 %, such as less than 5%, such as less than 10% or less than 20% of the initial rate determined using the equivalent DNA only oligonucleotide, with no 2' substitutions, with phosphorothioate linkage groups between all nucleotides in the oligonucleotide, using the methodology provided by Example 91 - 95 of EP 1 222 309.
- an oligomer is deemed capable of recruiting RNaseH if, when provided with the complementary RNA target, and RNaseH, the RNaseH initial rate, as measured in pmol/l/min, is at least 20%, such as at least 40 %, such as at least 60 %, such as at least 80 % of the initial rate determined using the equivalent DNA only oligonucleotide, with no 2' substitutions, with phosphorothioate linkage groups between all nucleotides in the oligonucleotide, using the methodology provided by Example 91 - 95 of EP 1 222 309.
- Most antisense oligonucleotides are compounds which are designed to recruit RNase enzymes (such as RNaseH) to degrade their intended target.
- RNase enzymes such as RNaseH
- Such compounds include DNA phosphorothioate oligonucleotides and gapmer, headmers and tailmers. These compounds typically comprise a region of at least 5 or 6 DNA nucleotides, and in the case of gapmers are flanked on either side by affinity enhancing nucleotide analogues.
- the oligomers of the present invention may operate via an RNase (such as RNaseH) independent mechanism.
- RNase such as RNaseH
- examples of oligomers which operate via a non-RNaseH (or non- RNase) mechanism are mixmers and totalmers.
- 'mixmer' refers to oligomers which comprise both naturally and non-naturally occurring nucleotides, where, as opposed to gapmers, tailmers, and headmers there is no contiguous sequence of more than 5, and in some embodiments no more than 4
- the mixmer does not comprise more than 5 consecutive nucleoside analogues, such as BNA (LNA), and in some embodiments no more than 4 consecutive, such as no more than three consecutive, consecutive nucleoside analogues, such as BNA (LNA).
- the remaining nucleosides may, for example be DNA nucleosides, and/or in non-bicyclic nucleoside analogues, such as those referred to herein, for example, 2' substituted nucleoside analogues, such as 2'-0-MOE and or 2'fluoro.
- the oligomer according to the invention maybe mixmers - indeed various mixmer designs are highly effective as oligomer or first region thereof, particularly when targeting microRNA (antimiRs), microRNA binding sites on mRNAs (Blockmirs) or as splice switching oligomers (SSOs). See for example WO2007/1 12754 (LNA-AntimiRsTM), WO2008/131807 (LNA splice switching oligos),
- the oligomer or mixmer may comprise of BNA and 2' substituted nucleoside analogues, optionally with DNA nucleosides - see for example see WO07027894 and WO2007/1 12754 which are hereby incorporated by reference.
- Specific examples include oligomers or first regions which comprise LNA, 2'-0-MOE and DNA, LNA, 2'fluoro and 2 -O-MOE, 2'-0-MOE and 2'fluoro, 2'-0-MOE and 2'fluoro and LNA, or LNA and 2'-0-MOE and LNA and DNA.
- the oligomer or mixmer comprises or consists of a contiguous nucleotide sequence of repeating pattern of nucleotide analogue and naturally occurring nucleotides, or one type of nucleotide analogue and a second type of nucleotide analogues.
- the repeating pattern may, for instance be every second or every third nucleotide is a nucleotide analogue, such as BNA (LNA), and the remaining nucleotides are naturally occurring nucleotides, such as DNA, or are a 2'substituted nucleotide analogue such as 2'MOE of 2'fluoro analogues as referred to herein, or, in some embodiments selected form the groups of nucleotide analogues referred to herein. It is recognised that the repeating pattern of nucleotide analogues, such as LNA units, may be combined with nucleotide analogues at fixed positions - e.g. at the 5' or 3' termini.
- LNA BNA
- the first nucleotide of oligomer or mixmer, counting from the 3' end is a nucleotide analogue, such as an LNA nucleotide.
- the second nucleotide of the oligomer or mixmer, counting from the 3' end is a nucleotide analogue, such as an LNA nucleotide.
- the seventh and/or eighth nucleotide of the oligomer or mixmer are nucleotide analogues, such as LNA nucleotides.
- the 5' terminal of olifgmer or mixmer is a nucleotide analogue, such as an LNA nucleotide.
- the above design features may, in some embodiments be incorporated into the mixmer design, such as antimiR mixmers.
- the oligomer or mixmer does not comprise a region of more than 4 consecutive DNA nucleotide units or 3 consecutive DNA nucleotide units. In some embodiments, the mixmer does not comprise a region of more than 2 consecutive DNA nucleotide units.
- the oligomer or mixmer comprises at least a region consisting of at least two consecutive nucleotide analogue units, such as at least two consecutive LNA units.
- the oligomer or mixmer comprises at least a region consisting of at least three consecutive nucleotide analogue units, such as at least three consecutive LNA units.
- the oligomer or mixmer of the invention does not comprise a region of more than 7 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 6 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 5 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 4 consecutive nucleotide analogue units, such as LNA units.
- the oligomer or mixmer of the invention does not comprise a region of more than 3 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 2 consecutive nucleotide analogue units, such as LNA units.
- the following embodiments may apply to mixmers or totalmer oligomers (e.g. as region A):
- the oligomer (e.g. region A) of the invention may, in some embodiments, comprise of at least two alternating regions of LNA and non-LNA nucleotides (such as DNA or 2' substituted nucleotide analogues).
- the oligomer of the invention may, in some embodiments, comprise a contiguous sequence of formula: 5' ([LNA nucleotides]i -5 and [non-LNA nucleotides]i- 4 ) 2 - i2- 3'.
- the 5' nucleotide of the contiguous nucleotide sequence (or the oligomer) is an LNA nucleotide.
- the 3' nucleotide of the contiguous nucleotide sequence is a nucleotide analogue, such as LNA, or the 2, 3, 4, 5 3' nucleotides are nucleotide analogues, such as LNA nucleotides, or other nucleotide analogues which confer enhanced serum stability to the oligomer.
- a nucleotide analogue such as LNA
- 2, 3, 4, 5 3' nucleotides are nucleotide analogues, such as LNA nucleotides, or other nucleotide analogues which confer enhanced serum stability to the oligomer.
- the contiguous nucleotide sequence of the oligomer has a formula 5' ([LNA nucleotides]i -5 - [non-LNA nucleotides]i -4 ) 2 - n - [LNA nucleotides]i -5 3'.
- the contiguous nucleotide sequence of the oligomer has 2, 3 or 4 contiguous regions of LNA and non-LNA nucleotides - e.g. comprises formula 5' ([LNA nucleotides]i- 5 and [non-LNA nucleotides]i- 4 ) 2 - 3, optionally with a further 3' LNA region [LNA nucleotides]i -5 .
- the contiguous nucleotide sequence of the oligomer comprises 5' ([LNA nucleotides]i-3 and [non-LNA nucleotides]i -3 )2 - 5, optionally with a further 3' LNA region [LNA nucleotides]i -3 .
- the contiguous nucleotide sequence of the oligomer comprises 5' ([LNA nucleotides]i- 3 and [non-LNA nucleotides]i- 3 ) 3 , optionally with a further 3' LNA region [LNA nucleotides]i -3 .
- non-LNA nucleotides are all DNA nucleotides.
- the non-LNA nucleotides are independently or dependency selected from the group consisting of DNA units, RNA units, 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino-DNA units, and 2'-fluoro-DNA units. In some embodiments the non-LNA nucleotides are (optionally independently selected from the group consisting of 2' substituted nucleoside analogues, such as (optionally
- sugar modified nucleoside analogues such as morpholino, peptide nucleic acid (PNA), CeNA, unlinked nucleic acid (UNA), hexitol nucleoic acid (HNA). bicyclo- HNA (see e.g.
- the nucleoside analogues increase the affinity of the first region for its target nucleic acid (or a complementary DNA or RNA sequence).
- Various nucleoside analogues are disclosed in Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, hereby incorporated by reference.
- the non-LNA nucleotides are DNA nucleotides. In some embodiments, the non-LNA nucleotides are DNA nucleotides. In some embodiments, the non-LNA nucleotides are DNA nucleotides. In some embodiments, the non-LNA nucleotides are DNA nucleotides. In some embodiments, the non-LNA nucleotides are DNA nucleotides. In some embodiments, the non-LNA nucleotides are DNA nucleotides.
- the oligomer or contiguous nucleotide sequence comprises of LNA nucleotides and optionally other nucleotide analogues (such as the nucleotide analogues listed under non-LNA nucleotides) which may be affinity enhancing nucleotide analogues and/or nucleotide analogues which enhance serum stability.
- the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of said nucleotide analogues.
- the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of LNA nucleotides.
- the oligomer or contiguous nucleotide sequence is 8 - 12, such as 8 - 10, or 10 - 20, such as 12 - 18 or 14 - 16 nts in length.
- the oligomer or contiguous nucleotide sequence is capable of forming a duplex with a complementary single stranded RNA nucleic acid molecule with phosphodiester internucleoside linkages, wherein the duplex has a T m of at least about 60°C, such as at least 65°C.
- T m Assay The oligonucleotide: Oligonucleotide and RNA target (PO) duplexes are diluted to 3 mM in 500 ml RNase-free water and mixed with 500 ml 2x T m - buffer (200mM NaCI, 0.2mM EDTA, 20mM Naphosphate, pH 7.0). The solution is heated to 95°C for 3 min and then allowed to anneal in room temperature for 30 min.
- the duplex melting temperatures (T m ) is measured on a Lambda 40 UVA IS Spectrophotometer equipped with a Peltier temperature programmer PTP6 using PE Templab software (Perkin Elmer). The temperature is ramped up from 20°C to 95°C and then down to 25°C, recording absorption at 260 nm. First derivative and the local maximums of both the melting and annealing are used to assess the duplex T m .
- a totalmer is a single stranded oligomer which only comprises non-naturally occurring nucleosides, such as sugar-modified nucleoside analogues.
- the first region according to the invention maybe totalmers - indeed various totalmer designs are highly effective as oligomers or first region thereofs, e.g. particularly when targeting microRNA (antimiRs).
- the totalmer comprises or consists of at least one XYX or YXY sequence motif, such as a repeated sequence XYX or YXY, wherein X is LNA and Y is an alternative (i.e. non LNA) nucleotide analogue, such as a 2'-0- MOE RNA unit and 2'-fluoro DNA unit.
- the above sequence motif may, in some
- embodiments be XXY, XYX, YXY or YYX for example.
- the totalmer may comprise or consist of a contiguous nucleotide sequence of between 7 and 16 nucleotides, such as 9, 10, 1 1 , 12, 13, 14, or 15 nucleotides, such as between 7 and 12 nucleotides.
- the contiguous nucleotide sequence of the totolmer comprises of at least 30%, such as at least 40%, such as at least 50%, such as at least 60%, such as at least 70%, such as at least 80%, such as at least 90%, such as 95%, such as 100% BNA (LNA) units.
- LNA BNA
- the remaining units may be selected from the non-LNA nucleotide analogues referred to herein in, such those selected from the group consisting of 2'-0_alkyl-RNA unit, 2'-OMe-RNA unit, 2'-amino-DNA unit, 2'-fluoro-DNA unit, LNA unit, PNA unit, HNA unit, INA unit, and a 2'MOE RNA unit, or the group 2'-OMe RNA unit and 2'-fluoro DNA unit.
- the totalmer consist or comprises of a contiguous nucleotide sequence which consists only of LNA units. In some embodiments, the totalmer, such as the LNA totalmer, is between 7 - 12 nucleoside units in length. In some embodiments, the totalmer (as the oligomer or first region thereof) may be targeted against a microRNA (i.e. be antimiRs) - as referred to WO2009/043353, which are hereby incorporated by reference.
- a microRNA i.e. be antimiRs
- the oligomer or contiguous nucleotide sequence comprises of LNA nucleotides and optionally other nucleotide analogues which may be affinity enhancing nucleotide analogues and/or nucleotide analogues which enhance serum stability.
- the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of said nucleotide analogues.
- the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of LNA nucleotides. MicroRNA modulation via the oligomer of the invention
- the oligomer or first region thereof is an oligomer, such as an LNA-antimiR®, which comprises or consists of a contiguous nucleotide sequence which is corresponds to or is fully complementary to a mature microRNA or part thereof.
- the oligomer may in some embodiments, target (i.e. comprises or consists of a contiguous nucleotide sequence which is fully complementary to (a corresponding region of) a microRNA.
- the microRNA may be a liver expressed microRNA, such as miR-122.
- the oligomer according to the invention consists or comprises of a contiguous nucleotide sequence which corresponds to or is fully complementary to hsa- miR122 (NR_029667.1 Gl:262205241 ), such as the mature hsa-miR-122. In some embodiments the oligomer according to the invention, consists or comprises of a contiguous nucleotide sequence which corresponds to or is fully complementary to hsa-miR122
- the oligomer when the oligomer or first region thereof targets miR-122, the oligomer is for the use in the treatment of hepatitis C infection.
- oligomer or first region thereof 'antimiR' designs and oligomers are disclosed in WO2007/1 12754, WO2007/1 12753, PCT/DK2008/000344 and US provisional applications 60/979217 and 61/028062, all of which are hereby incorporated by reference.
- the oligomer or first region thereof is an antimiR which is a mixmer or a totalmer.
- the term AntimiR may therefore be replaces with the term oligomer.
- AntimiR oligomers are oligomers which consist or comprise of a contiguous nucleotide sequence which is fully complementary to, or essentially complementary to (i.e. may comprise one or two mismatches), to a microRNA sequence, or a corresponding subsequence thereof.
- the antimiR may be comprise a contiguous nucleotide sequence which is complementary or essentially complementary to the entire mature microRNA, or the antimiR may be comprise a contiguous nucleotide sequence which is complementary or essentially complementary to a sub-sequence of the mature microRNA or pre-microRNA - such a sub-sequence (and therefore the
- corresponding contiguous nucleotide sequence is typically at least 8 nucleotides in length, such as between 8 and 25 nucleotides, such as 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24 nucleotides in length, such as between 10-17 or 10-16 nucleotides, such as between 12 - 15 nucleotides.
- Numerous designs of AnitmiRs have been suggested, and typically antimiRs for therapeutic use, such as the contiguous nucleotide sequence thereof comprise one or more nucleotide analogues units.
- the antimiR may have a gapmer structure as herein described.
- other designs may be preferable, such as mixmers, or totalmers.
- WO2007/1 12754 and WO2007/1 12753 both hereby incorporated by reference, provide antimiR oligomers and antimiR oligomer designs where the oligomers which are complementary to mature microRNA
- a subsequence of the antimiR corresponds to the miRNA seed region.
- the first or second 3' nucleobase of the oligomer corresponds to the second 5' nucleotide of the microRNA sequence.
- nucleobase units 1 to 6 (inclusive) of the oligomer as measured from the 3' end the region of the oligomer are complementary to the microRNA seed region sequence.
- nucleobase units 1 to 7 (inclusive) of the oligomer as measured from the 3' end the region of the oligomer are complementary to the microRNA seed region sequence.
- nucleobase units 2 to 7 (inclusive) of the oligomer as measured from the 3' end the region of the oligomer are complementary to the microRNA seed region sequence.
- the antimiR oligomer comprises at least one nucleotide analogue unit, such as at least one LNA unit, in a position which is within the region complementary to the miRNA seed region.
- the antimiR oligomer may, in some embodiments
- embodiments comprise at between one and 6 or between 1 and 7 nucleotide analogue units, such as between 1 and 6 and 1 and 7 LNA units, in a position which is within the region complementary to the miRNA seed region.
- the antimiR of the invention is 7, 8 or 9 nucleotides long, and comprises a contiguous nucleotide sequence which is complementary to a seed region of a human or viral microRNA, and wherein at least 80 %, such as 85%, such as 90%, such as 95%, such as 100% of the nucleotides are LNA.
- the antimiR of the invention is 7, 8 or 9 nucleotides long, and comprises a contiguous nucleotide sequence which is complementary to a seed region of a human or viral microRNA, and wherein at least 80 % of the nucleotides are LNA, and wherein at least 80%, such as 85%, such as 90%, such as 95%, such as 100% of the internucleotide bonds are phosphorothioate bonds.
- the antimiR comprises one or two LNA units in positions three to eight, counting from the 3' end. This is considered advantageous for the stability of the A- helix formed by the oligo:microRNA duplex, a duplex resembling an RNA:RNA duplex in structure.
- nucleoside monomers of the oligomers e.g. first and second regions
- oligomers e.g. first and second regions
- linkage groups e.g. first and second linkage groups
- each monomer is linked to the 3' adjacent monomer via a linkage group.
- the 5' monomer at the end of an oligomer does not comprise a 5' linkage group, although it may or may not comprise a 5' terminal group.
- linkage group or "internucleotide linkage” are intended to mean a group capable of covalently coupling together two nucleotides. Specific and preferred examples include phosphate groups and phosphorothioate groups.
- nucleotides of the oligomer of the invention or contiguous nucleotides sequence thereof are coupled together via linkage groups.
- each nucleotide is linked to the 3' adjacent nucleotide via a linkage group.
- Suitable internucleotide linkages include those listed within WO2007/031091 , for example the internucleotide linkages listed on the first paragraph of page 34 of
- Suitable sulphur (S) containing internucleotide linkages as provided herein may be preferred, such as phosphorothioate or phosphodithioate.
- Phosphorothioate internucleotide linkages are also preferred, particularly for the first region, such as in gapmers, mixmers, antimirs splice switching oligomers, and totalmers.
- the internucleotide linkages in the oligomer may, for example be phosphorothioate or boranophosphate so as to allow RNase H cleavage of targeted RNA.
- Phosphorothioate is preferred, for improved nuclease resistance and other reasons, such as ease of manufacture.
- the nucleotides and/or nucleotide analogues are linked to each other by means of phosphorothioate groups.
- at least 50%, such as at least 70%, such as at least 80%, such as at least 90% such as all the internucleoside linkages between nucleosides in the first region are other than phosphodiester (phosphate), such as are selected from the group consisting of phosphorothioate phosphorodithioate, or boranophosphate.
- nucleosides in the first region are phosphorothioate.
- WO09124238 refers to oligomeric compounds having at least one bicyclic nucleoside attached to the 3' or 5' termini by a neutral internucleoside linkage.
- the oligomers of the invention may therefore have at least one bicyclic nucleoside attached to the 3' or 5' termini by a neutral internucleoside linkage, such as one or more phosphotriester,
- methylphosphonate MMI, amide-3, formacetal or thioformacetal.
- the remaining linkages may be phosphorothioate.
- conjugate is intended to indicate a heterogenous molecule formed by the covalent attachment (“conjugation") of the oligomer as described herein has one or more non-nucleotide, or non-polynucleotide moieties conjugated thereto.
- the conjugate group is a carbohydrate moiety.
- the conjugate is or may comprise a carbohydrate or comprises a carbohydrate group.
- the carbohydrate is selected from the group consisting of galactose, lactose, n-acetylgalactosamine, mannose, and mannose-6- phosphate.
- the conjugate group is or may comprise mannose or mannose-6-phosphate.
- Carbohydrate conjugates may be used to enhance delivery or activity in a range of tissues, such as liver and/or muscle. See, for example, EP1495769, W099/65925, Yang et al., Bioconjug Chem (2009) 20(2): 213-21. Zatsepin & Oretskaya Chem Biodivers. (2004) 1 (10): 1401 -17.
- the oligomer may further comprise one or more additional conjugate moieties, of which lipophilic or hydrophobic moieties are particularly interesting. These may for example, act as pharmacokinetic modulators, and may be covalently linked to either the carbohydrate conjugate, a linker linking the carbohydrate conjugate to the oligomer or a linker linking multiple carbohydrate conjugates (multi-valent) conjugates, or to the oligomer, optionally via a linker, such as a bio cleavable linker.
- additional conjugate moieties of which lipophilic or hydrophobic moieties are particularly interesting. These may for example, act as pharmacokinetic modulators, and may be covalently linked to either the carbohydrate conjugate, a linker linking the carbohydrate conjugate to the oligomer or a linker linking multiple carbohydrate conjugates (multi-valent) conjugates, or to the oligomer, optionally via a linker, such as a bio cleavable linker.
- the compound of the invention may further comprise one or more additional conjugate moieties, of which lipophilic or hydrophobic moieties are particularly interesting, such as when the conjugate group is a carbohydrate moiety.
- Such lipophilic or hydrophobic moieties may act as pharmacokinetic modulators, and may be covalently linked to either the carbohydrate conjugate, a linker linking the carbohydrate conjugate to the oligomer or a linker linking multiple carbohydrate conjugates (multi-valent) conjugates, or to the oligomer, optionally via a linker, such as a bio cleavable linker.
- the oligomer or conjugate moiety may therefore comprise a pharmacokinetic modulator, such as a lipophilic or hydrophobic moieties. Such moieties are disclosed within the context of siRNA conjugates in WO2012/082046.
- the hydrophobic moiety may comprise a C8 - C36 fatty acid, which may be saturated or un-saturated. In some embodiments, C10, C12, C14, C16, C18, C20, C22, C24, C26, C28, C30, C32 and C34 fatty acids may be used.
- the hydrophobic group may have 16 or more carbon atoms.
- hydrophobic groups may be selected from the group comprising:sterol, cholesterol, palmitoyl, hexadec-8-enoyl, oleyl, (9E, 12E)-octadeca-9,12-dienoyl, dioctanoyl, and C16- C20 acyl.
- hydrophobic groups having fewer than 16 carbon atoms are less effective in enhancing polynucleotide targeting, but they may be used in multiple copies (e.g. 2x, such as 2x C8 or C10, C12 or C14) to enhance efficacy.
- Pharmacokinetic modulators useful as polynucleotide targeting moieties may be selected from the group consisting of: cholesterol, alkyl group, alkenyl group, alkynyl group, aryl group, aralkyi group, aralkenyl group, and aralkynyl group, each of which may be linear, branched, or cyclic.
- Pharmacokinetic modulators are preferably hydrocarbons, containing only carbon and hydrogen atoms. However, substitutions or heteroatoms which maintain hydrophobicity, for example fluorine, may be permitted.
- GalNac conjugates for use with LNA oligomers do not require a pharmacokinetic modulator, and as such, in some embodiments, the GalNac conjugate is not covalently linked to a lipophilic or hydrophobic moiety, such as those described here in, e.g. do not comprise a C8 - C36 fatty acid or a sterol.
- the invention therefore also provides for LNA oligomer GalNac conjugates which do not comprise a lipophilic or hydrophobic pharmacokinetic modulator or conjugate moiety/group.
- the invention provides LNA antisense oligonucleotides which are conjugated to an asialoglycoprotein receptor targeting moiety.
- the conjugate moiety (such as the third region or region C) comprises an asialoglycoprotein receptor targeting moiety, such as galactose, galactosamine, N-formyl-galactosamine, Nacetylgalactosamine, N-propionyl-galactosamine, N-n-butanoyl-galactosamine, and N-isobutanoylgalactos-amine.
- the conjugate comprises a galactose cluster, such as N- acetylgalactosamine trimer.
- the conjugate moiety comprises an GalNAc (N-acetylgalactosamine), such as a mono-valent, di-valent , tri-valent of tetra-valent GalNAc.
- Trivalent GalNAc conjugates may be used to target the compound to the liver.
- GalNAc conjugates have been used with methylphosphonate and PNA antisense oligonucleotides (e.g. US 5,994517 and Hangeland et al., Bioconjug Chem. 1995 Nov- Dec;6(6):695-701 ) and siRNAs (e.g. WO2009/126933, WO2012/089352 &
- GalNAc conjugate moieties can include, for example, oligosaccharides and carbohydrate clusters such as Tyr-Glu-Glu-(aminohexyl GalNAc)3 (YEE(ahGalNAc)3; a glycotripeptide that binds to Gal/Gal N Ac receptors on hepatocytes, see, e.g., Duff, et al., Methods Enzymol, 2000, 313, 297); lysine-based galactose clusters (e.g., L3G4; Biessen, et al., Cardovasc.
- oligosaccharides and carbohydrate clusters such as Tyr-Glu-Glu-(aminohexyl GalNAc)3 (YEE(ahGalNAc)3; a glycotripeptide that binds to Gal/Gal N Ac receptors on hepatocytes, see, e.g., Duff, et al., Methods Enzymol, 2000
- conjugates can include oligosaccharides that can bind to carbohydrate recognition domains (CRD) found on the asiologlycoprotein-receptor (ASGP- R).
- CCD carbohydrate recognition domains
- ASGP- R asiologlycoprotein-receptor
- WO2012/083046 discloses siRNAs withGalNAc conjugate moieties which comprise cleavable pharmacokinetic modulators, which are suitable for use in the present invention, the preferred pharmacokinetic modulators are C16 hydrophobic groups such as palmitoyl, hexadec-8-enoyl, oleyl, (9E, 12E)-octadeca-9,12-dienoyl, dioctanoyi, and C16-C20 acyl.
- the ⁇ 46 cleavable pharmacokinetic modulators may also be cholesterol.
- the 'targeting moieties may be selected from the group consisting of: galactose, galactosamine, N-formyl-galactosamine, N-acetylgalactosamine, Npropionyl- galactosamine, N-n-butanoyl-galactosamine, N-iso-butanoylgalactos-amine, galactose cluster, and N-acetylgalactosamine trimer and may have a pharmacokinetic modulator selected from the group consisting of: hydrophobic group having 16 or more carbon atoms, hydrophobic group having 16-20 carbon atoms, palmitoyl, hexadec-8-enoyl, oleyl, (9E,12E)-octadeca-9,12dienoyl, dioctanoyi, and C16-C20 acyl, and cholesterol.
- a pharmacokinetic modulator selected from the group consisting of: hydrophobic group
- GalNac clusters disclosed in ⁇ 46 include: (E)-hexadec-8-enoyl (C16), oleyl (C18), (9,E,12E)-octadeca-9,12-dienoyl (C18), octanoyl (C8), dodececanoyl (C12), C-20 acyl, C24 acyl, dioctanoyi (2xC8).
- the targeting moiety-pharmacokinetic modulator targeting moiety may be linked to the polynucleotide via a physiologically labile bond or, e.g. a disulfide bond, or a PEG linker.
- the invention also relates to the use of phospodiester linkers, such as DNA phosphodiester linkers, between the oligomer and the conjugate group (these are referred to as region B herein, and suitably are positioned between the LNA oligomer and the carbohydrate conjugate group).
- phospodiester linkers such as DNA phosphodiester linkers
- a preferred targeting ligand is a galactose cluster.
- a galactose cluster comprises a molecule having e.g. comprising two to four terminal galactose derivatives.
- the term galactose derivative includes both galactose and derivatives of galactose having affinity for the asialoglycoprotein receptor equal to or greater than that of galactose.
- a terminal galactose derivative is attached to a molecule through its C-l carbon.
- the asialoglycoprotein receptor (ASGPr) is unique to hepatocytes and binds branched galactose-terminal glycoproteins.
- a preferred galactose cluster has three terminal galactosamines or galactosamine derivatives each having affinity for the asialoglycoprotein receptor.
- a more preferred galactose cluster has three terminal N-acetyl- galactosamines.
- Other terms common in the art include tri-antennary galactose, tri-valent galactose and galactose trimer. It is known that tri-antennary galactose derivative clusters are bound to the ASGPr with greater affinity than bi-antennary or mono-antennary galactose derivative structures (Baenziger and Fiete, 1980, Cell, 22, 61 1 -620; Connolly et al., 1982,1. Biol.
- a galactose cluster may comprise two or preferably three galactose derivatives each linked to a central branch point.
- the galactose derivatives are attached to the central branch point through the C-l carbons of the saccharides.
- the galactose derivative is preferably linked to the branch point via linkers or spacers.
- a preferred spacer is a flexible hydrophilic spacer (U.S. Patent 5885968; Biessen et al. J. Med. Chern. 1995 Vol. 39 p. 1538-1546).
- a preferred flexible hydrophilic spacer is a PEG spacer.
- a preferred PEG spacer is a PEG3 spacer.
- the branch point can be any small molecule which permits attachment of the three galactose derivatives and further permits attachment of the branch point to the oligomer.
- An exemplary branch point group is a di-lysine.
- a di-lysine molecule contains three amine groups through which three galactose derivatives may be attached and a carboxyl reactive group through which the di-lysine may be attached to the oligomer.
- a preferred spacer is a flexible hydrophilic spacer.
- a preferred flexible hydrophilic spacer is a PEG spacer.
- a preferred PEG spacer is a PEG3 spacer (three ethylene units).
- the galactose cluster may be attached to the 3' or 5' end of the oligomer using methods known in the art.
- a preferred galactose derivative is an N-acetyl-galactosamine (GalNAc).
- Other saccharides having affinity for the asialoglycoprotein receptor may be selected from the list comprising: galactosamine, N-n-butanoylgalactosamine, and N-iso-butanoylgalactosamine.
- the affinities of numerous galactose derivatives for the asialoglycoprotein receptor have been studied (see for example: Jobst, ST. and Drickamer, K. JB.C. 1996,271 ,6686) or are readily determined using methods typical in the art.
- a GalNac conjugate is illustrated in figure 1 . Further examples of the conjugate of the invention are illustrated below:
- the LNA antisense oligomer may, in some emboidments, comprise cET nucleosides, such as (S)-cET nucleosides.
- the carbohydrate conjugate (e.g. GalNAc) may therefore be linked to the oligomer via a linker, such as (poly)ethylene glycol linker (PEG), such as a di, tri, tetra, penta, hexa- ethylene glycol linker.
- a linker such as (poly)ethylene glycol linker (PEG), such as a di, tri, tetra, penta, hexa- ethylene glycol linker.
- a carbohydrate conjugate e.g. GalNAc
- a biocleavable linker such as region B as defined herein, and optionally region Y, which is illustrated as a di-lysine in the above diagrams.
- hydrophobic or lipophilic (or further conjugate) moiety i.e. pharmacokinetic modulator
- BNA or LNA oligomers such as LNA antisense oligonucleotides, optional.
- Each carbohydrate moiety of a GalNAc cluster may therefore be joined to the oligomer via a spacer, such as (poly)ethylene glycol linker (PEG), such as a di, tri, tetra, penta, hexa-ethylene glycol linker.
- PEG polyethylene glycol linker
- the PEG moiety forms a spacer between the galactose sugar moiety and a peptide (trilysine is shown) linker.
- the carbohydrate conjugate(e.g. GalNAc), or carbohydrate-linker moiety may be covalently joined (linked) to the oligomer via a branch point group such as, an amino acid, or peptide, which suitably comprises two or more amino groups (such as 3, 4, or5), such as lysine, di-lysine or tri-lysine or tetra-lysine.
- a tri-lysine molecule contains four amine groups through which three carbohydrate conjugate groups, such as galactose & derivatives (e.g.
- GalNAc GalNAc
- a further conjugate such as a hydrophobic or lipophilic moiety/group may be attached and a carboxyl reactive group through which the tri-lysine may be attached to the oligomer.
- the further conjugate such as lipophilic/hyrodphobic moiety may be attached to the lysine residue that is attached to the oligomer.
- the GalNac cluster comprises a peptide linker, e.g. a Tyr-Asp(Asp) tripeptide or Asp(Asp) dipeptide, which is attached to the oligomer (or to region Y or region B) via a biradical linker, for example the GalNac cluster may comprise the following biradical linkers:
- R 1 is a biradical preferably selected from -C 2 H 4 -, -C 3 H 6 -, -C 4 H 8 -, -C 5 H 10 -, -C 6 H 12 -, 1 ,4- cyclohexyl (-C6H 10-), 1 ,4-phenyl (-C 6 H 4 -), -C 2 H 4 OC 2 H 4 -, -C 2 H 4 (OC 2 H 4 ) 2 - or -C 2 H 4 (OC 2 H 4 ) 3 -, C(0)CH 2 -, -C(0)C 2 H 4 -, - C(0)C 3 H 6 -, - C(0)C 4 H 8 -, - C(O)C 5 H 10 -, - C(0)C 6 H 12 -, 1 ,4-cyclohexyl (-C(0)C6H 10-), 1 ,4-phenyl (-C(0)C 6 H 4 -), - C(0)C 2 H 4 OC
- R 1 is a biradical preferably selected from - C 2 H 4 -, -C 3 H 6 -, -C 4 H 8 -, -C 5 H 10 -, -C 6 H 12 -, 1 ,4-cyclohexyl (-C6H 10-), 1 ,4-phenyl (-C 6 H 4 -), - C 2 H 4 OC 2 H 4 -, -C 2 H 4 (OC 2 H 4 ) 2 - or -C 2 H 4 (OC 2 H 4 ) 3 - .
- the invention further provides for the LNA oligomer intermediates which comprise an antisense LNA oligomer which comprises an (e.g. terminal, 5' or 3') amino alkyl, such as a C2 - C36 amino alkyl group, including, for example C6 and C12 amino alkyl groups.
- the amino alkyl group may be added to the LNA oligomer as part of standard oligonucleotide synthesis, for example using a (e.g. protected) amino alkyl phosphoramidite.
- the linkage group between the amino alkyl and the LNA oligomer may for example be a
- the amino alkyl group may be covalently linked to, for example, the 5' or 3' of the LNA oligomer, such as by the nucleoside linkage group, such as phosphorothioate or phosphodiester linkage.
- the invention also provides a method of synthesis of the LNA oligomer comprising the sequential synthesis of the LNA oligomer, such as solid phase oligonucleotide synthesis, comprising the step of adding an amino alkyl group to the oligomer, such as e.g. during the first or last round of oligonucleotide synthesis.
- the method of synthesis my further comprise the step of reacting the carbohydrate conjugate to the amino alkyl -LNA oligomer (the conjugation step).
- the carbohydrate conjugate may comprise suitable linkers and/or branch point groups, and optionally further conjugate groups, such as hydrophobic or lipophilic groups, as described herein.
- the conjugation step may be performed whilst the oligomer is bound to the solid support (e.g. after oligonucleotide synthesis, but prior to elution of the oligomer from the solid support), or subsequently (i.e. after elution).
- the invention provides for the use of an amino alkyl linker in the synthesis of the oligomer of the invention.
- the invention provides for a method of synthesizing (or manufacture) of an oligomeric compound, such as the oligomeric compound of the invention, said method comprising a step of [sequential] oligonucleotide synthesis of a first region (A) and (optionally, when present), a second region (B), wherein the synthesis step is followed by a step of adding a third region [phosphoramidite comprising] comprising a carbohydrate conjugate group as described herein (X or with a linker X-Y), such as for example a GalNAc conjugate, such as a trivalent GalNAc (e.g.
- Conjl a conjugate selected from the group consisting of Conjl , Conj2, Conj3, Conj 4, Conjl a, Conj2a, Conj3a, and Conj 4a, or other trivalent GalNAc conjugate moieties, such as those disclosed herein.
- Conjl a conjugate selected from the group consisting of Conjl , Conj2, Conj3, Conj 4, Conjl a, Conj2a, Conj3a, and Conj 4a, or other trivalent GalNAc conjugate moieties, such as those disclosed herein.
- the conjuigate region (e.gX or X-Y) may be added after the cleavage from the solid support.
- the method of synthesis may comprise the steps of synthesizing a first (A), and optionally second region (B), followed by the cleavage of the oligomer from the support, with a subsequent step of adding the third region, such as X or X-Y group to the oligomer.
- region B may be a non- nucleotide cleavable linker for example a peptide linker, which may form part of region X (also referred to as region C) or be region Y (or part thereof).
- region X such as C
- X-Y such as the conjugate (e.g. a GalNAc conjugate) comprises an activation group, (an activated functional group) and in the method of synthesis the activated conjugate (or region x, or X-Y) is added to the first and second regions, such as an amino linked oligomer.
- the amino group may be added to the oligomer by standard phosphoramidite chemistry, for example as the final step of oligomer synthesis (which typically will result in amino group at the 5' end of the oligomer).
- a protected amino-alkyl phosphoramidite is used, for example a TFA-aminoC6 phosphoramidite (6- (Trifluoroacetylamino)-hexyl-(2-cyanoethyl)-(N,N-diisopropyl)-phosphoramidite).
- Region X (or region C as referred to herein), such as the conjugate (e.g. a GalNac conjugate) may be activated via NHS ester method and then the aminolinked oligomer is added.
- a N-hydroxysuccinimide (NHS) may be used as activating group for region X (or region C, such as a conjugate, such as a GalNac conjugate moiety.
- the conjugate moiety is hydrophilic.
- the conjugate group does not comprise a lipophilic substituent group, such as a fatty acid substituent group, such as a C8 - C26, such as a palmotyl substituent group, or does not comprise a sterol, e.g. a cholesterol subtituent group.
- part of the invention is based on the suprising discovery that LNA oligomers GalNAC conjugates have remarkable pharmacokinetic properties even without the use of pharmacokinetic modulators, such as fatty acid substituent groups (e.g. >C8 or >C16 fatty acid groups).
- a linkage or linker is a connection between two atoms that links one chemical group or segment of interest to another chemical group or segment of interest via one or more covalent bonds.
- Conjugate moieties can be attached to the oligomeric compound directly or through a linking moiety (linker or tether) - a linker.
- Linkers are bifunctional moieties that serve to covalently connect a third region, e.g. a conjugate moiety, to an oligomeric compound (such as to region B).
- the linker comprises a chain structure or an oligomer of repeating units such as ethylene glyol or amino acid units.
- the linker can have at least two functionalities, one for attaching to the oligomeric compound and the other for attaching to the conjugate moiety.
- Example linker functionalities can be electrophilic for reacting with nucleophilic groups on the oligomer or conjugate moiety, or nucleophilic for reacting with electrophilic groups.
- linker functionalities include amino, hydroxyl, carboxylic acid, thiol, phosphoramidate, phophate, phosphite, unsaturations (e.g., double or triple bonds), and the like.
- Some example linkers include 8-amino-3,6-dioxaoctanoic acid (ADO), succinimidyl 4- (N-maleimidomethyl)cyclohexane-l-carboxylate (SMCC), 6- aminohexanoic acid (AHEX or AHA), 6-aminohexyloxy, 4-aminobutyric acid, 4- aminocyclohexylcarboxylic acid, succinimidyl 4-(N-maleimidomethyl)cyclohexane- l-carboxy-(6-amido-caproate) (LCSMCC), succinimidyl m-maleimido-benzoylate (MBS), succinimidyl N-e-maleimido-caproylate
- EMCS succinimidyl 6-(beta - maleimido-propionamido) hexanoate
- SMPH succinimidyl N-(a-maleimido acetate)
- AMAS succinimidyl 4-(p-maleimidophenyl)butyrate
- SMPB succinimidyl 4-(p-maleimidophenyl)butyrate
- beta -alanine beta -ALA
- PEG phenylglycine
- ACAC 4-aminocyclohexanoic acid
- beta -CYPR amino dodecanoic acid
- ADC amino dodecanoic acid
- linker groups are known in the art that can be useful in the attachment of conjugate moieties to oligomeric compounds.
- a review of many of the useful linker groups can be found in, for example, Antisense Research and Applications, S. T. Crooke and B. Lebleu, Eds., CRC Press, Boca Raton, Fla., 1993, p. 303-350.
- a disulfide linkage has been used to link the 3' terminus of an oligonucleotide to a peptide (Corey, et al., Science 1987, 238, 1401 ; Zuckermann, et al, J Am. Chem. Soc. 1988, 1 10, 1614; and Corey, et al., J Am. Chem. Soc.
- This reagent was also utilized to link a peptide to an oligonucleotide as reported by Judy, et al., Tetrahedron Letters 1991 , 32, 879.
- a similar commercial reagent for linking to the 5 '-terminus of an oligonucleotide is 5'- Amino-Modifier C6. These reagents are available from Glen Research Corporation (Sterling, Va.). These compounds or similar ones were utilized by Krieg, et al, Antisense Research and Development 1991 , 1 , 161 to link fluorescein to the 5'- terminus of an oligonucleotide.
- Linkers and their use in preparation of conjugates of oligomeric compounds are provided throughout the art such as in WO 96/1 1205 and WO 98/52614 and U.S. Pat. Nos. 4,948,882; 5,525,465; 5,541 ,313; 5,545,730; 5,552,538; 5,580,731 ; 5,486,603; 5,608,046; 4,587,044; 4,667,025; 5,254,469; 5,245,022; 5,1 12,963; 5,391 ,723; 5,510475; 5,512,667; 5,574,142; 5,684,142; 5,770,716; 6,096,875; 6,335,432; and 6,335,437,Wo2012/083046 each of which is incorporated by reference in its entirety.
- a physiologically labile bond is a labile bond that is cleavable under conditions normally encountered or analogous to those encountered within a mammalian body (also referred to as a cleavable linker).
- Physiologically labile linkage groups are selected such that they undergo a chemical transformation (e.g., cleavage) when present in certain physiological conditions.
- Mammalian intracellular conditions include chemical conditions such as pH, temperature, oxidative or reductive conditions or agents, and salt concentration found in or analogous to those encountered in mammalian cells. Mammalian intracellular conditions also include the presence of enzymatic activity normally present in a mammalian cell such as from proteolytic or hydrolytic enzymes.
- the cleavable linker is susceptible to nuclease(s) which may for example, be expressed in the target cell - and as such, as detailed herein, the linker may be a short region (e.g. 1 - 10) phosphodiester linked nucleosides, such as DNA nucleosides,
- Chemical transformation may be initiated by the addition of a pharmaceutically acceptable agent to the cell or may occur spontaneously when a molecule containing the labile bond reaches an appropriate intra-and/or extra-cellular environment.
- a pH labile bond may be cleaved when the molecule enters an acidified endosome.
- a pH labile bond may be considered to be an endosomal cleavable bond.
- Enzyme cleavable bonds may be cleaved when exposed to enzymes such as those present in an endosome or lysosome or in the cytoplasm.
- a disulfide bond may be cleaved when the molecule enters the more reducing environment of the cell cytoplasm.
- a disulfide may be considered to be a cytoplasmic cleavable bond.
- a pH-labile bond is a labile bond that is selectively broken under acidic conditions (pH ⁇ 7). Such bonds may also be termed endosomally labile bonds, since cell endosomes and lysosomes have a pH less than 7.
- a linkage or linker is a connection between two atoms that links one chemical group or segment of interest to another chemical group or segment of interest via one or more covalent bonds.
- Conjugate moieties can be attached to the oligomeric compound directly or through a linking moiety (linker or tether) - a linker.
- Linkers are bifunctional moieties that serve to covalently connect a third region, e.g. a conjugate moiety, to an oligomeric compound (such as to region B).
- the linker comprises a chain structure or an oligomer of repeating units such as ethylene glyol or amino acid units.
- the linker can have at least two functionalities, one for attaching to the oligomeric compound and the other for attaching to the conjugate moiety.
- Example linker functionalities can be electrophilic for reacting with nucleophilic groups on the oligomer or conjugate moiety, or nucleophilic for reacting with electrophilic groups.
- linker functionalities include amino, hydroxyl, carboxylic acid, thiol, phosphoramidate, phosphorothioate, phosphate, phosphite, unsaturations (e.g., double or triple bonds), and the like.
- linkers include 8-amino-3,6-dioxaoctanoic acid (ADO), succinimidyl 4-(N-maleimidomethyl)cyclohexane-l-carboxylate (SMCC), 6- aminohexanoic acid (AHEX or AHA), 6-aminohexyloxy, 4-aminobutyric acid, 4- aminocyclohexylcarboxylic acid, succinimidyl 4-(N-maleimidomethyl)cyclohexane- l-carboxy- (6-amido-caproate) (LCSMCC), succinimidyl m-maleimido-benzoylate (MBS), succinimidyl N-e-maleimido-caproylate (EMCS), succinimidyl 6-(beta - maleimido-propionamido) hexanoate (SMPH), succinimidyl N-(a-maleimido acetate) (AMAS), succinimid
- linker groups are known in the art that can be useful in the attachment of conjugate moieties to oligomeric compounds.
- a review of many of the useful linker groups can be found in, for example, Antisense Research and Applications, S. T. Crooke and B. Lebleu, Eds., CRC Press, Boca Raton, Fla., 1993, p. 303-350.
- a disulfide linkage has been used to link the 3' terminus of an oligonucleotide to a peptide (Corey, et al., Science 1987, 238, 1401 ; Zuckermann, et al, J Am. Chem. Soc. 1988, 1 10, 1614; and Corey, et al., J Am. Chem. Soc.
- This reagent was also utilized to link a peptide to an oligonucleotide as reported by Judy, et al., Tetrahedron Letters 1991 , 32, 879.
- a similar commercial reagent for linking to the 5 '-terminus of an oligonucleotide is 5'- Amino-Modifier C6. These reagents are available from Glen Research Corporation (Sterling, Va.). These compounds or similar ones were utilized by Krieg, et al, Antisense Research and Development 1991 , 1 , 161 to link fluorescein to the 5'- terminus of an oligonucleotide.
- a physiologically labile bond is a labile bond that is cleavable under conditions normally encountered or analogous to those encountered within a mammalian body (also referred to as a cleavable linker).
- Physiologically labile linkage groups are selected such that they undergo a chemical transformation (e.g., cleavage) when present in certain physiological conditions.
- Mammalian intracellular conditions include chemical conditions such as pH, temperature, oxidative or reductive conditions or agents, and salt concentration found in or analogous to those encountered in mammalian cells. Mammalian intracellular conditions also include the presence of enzymatic activity normally present in a mammalian cell such as from proteolytic or hydrolytic enzymes.
- the cleavable linker is susceptible to nuclease(s) which may for example, be expressed in the target cell - and as such, as detailed herein, the linker may be a short region (e.g. 1 - 10) phosphodiester linked nucleosides, such as DNA nucleosides,
- Chemical transformation may be initiated by the addition of a pharmaceutically acceptable agent to the cell or may occur spontaneously when a molecule containing the labile bond reaches an appropriate intra-and/or extra-cellular environment.
- a pH labile bond may be cleaved when the molecule enters an acidified endosome.
- a pH labile bond may be considered to be an endosomal cleavable bond.
- Enzyme cleavable bonds may be cleaved when exposed to enzymes such as those present in an endosome or lysosome or in the cytoplasm.
- a disulfide bond may be cleaved when the molecule enters the more reducing environment of the cell cytoplasm.
- a disulfide may be considered to be a cytoplasmic cleavable bond.
- a pH-labile bond is a labile bond that is selectively broken under acidic conditions (pH ⁇ 7). Such bonds may also be termed endosomally labile bonds, since cell endosomes and lysosomes have a pH less than 7.
- the oligomeric compound may optionally, comprise a second region (region B) which is positioned between the oligomer (referred to as region A) and the conjugate (referred to as region C).
- Region B may be a linker such as a cleavable linker (also referred to as a physiologically labile linkage).
- the susceptibility to cleavage in the assays shown in Example 6 may be used to determine whether a linker is biocleavable or physiologically labile.
- Biocleavable linkers according to the present invention refers to linkers which are susceptible to cleavage in a target tissue (i.e. physiologically labile), for example liver and/or kidney. It is preferred that the cleavage rate seen in the target tissue is greater than that found in blood serum. Suitable methods for determining the level (%) of cleavage in tissue (e.g. liver or kidney) and in serum are found in example 6.
- the biocleavable linker (also referred to as the physiologically labile linker, or nuclease susceptible linker), such as region B, in a compound of the invention, are at least about 20% cleaved, such as at least about 30% cleaved, such as at least about 40% cleaved, such as at least about 50% cleaved, such as at least about 60% cleaved, such as at least about 70% cleaved, such as at least about 75% cleaved, in the liver or kidney homogenate assay of Example 9.
- the cleavage (%) in serum, as used in the assay in Example 9 is less than about 30%, is less than about 20%, such as less than about 10%, such as less than 5%, such as less than about 1 %.
- the biocleavable linker also referred to as the physiologically labile linker, or nuclease susceptible linker
- region B in a compound of the invention, are susceptible to S1 nuclease cleavage. Susceptibility to S1 cleavage may be evaluated using the S1 nuclease assay shown in Example 6.
- the biocleavable linker (also referred to as the physiologically labile linker, or nuclease susceptible linker), such as region B, in a compound of the invention, are at least about 30% cleaved, such as at least about 40% cleaved, such as at least about 50% cleaved, such as at least about 60% cleaved, such as at least about 70% cleaved, such as at least about 80% cleaved, such as at least about 90% cleaved, such as at least 95% cleaved after 120min incubation with S1 nuclease according to the assay used in Example 6.
- Nuclease Susceptible Physiological Labile Linkages are at least about 30% cleaved, such as at least about 40% cleaved, such as at least about 50% cleaved, such as at least about 60% cleaved, such as at least about 70% cleaved, such as at least about 80% cleaved,
- the oligomeric compound may optionally, comprise a second region (region B) which is positioned between the LNA oligomer (referred to as region A) and the carbohydrate conjugate (referred to as region C).
- Region B may be a linker such as a cleavable linker (also referred to as a physiologically labile linkage).
- region B comprises between 1 - 10 nucleotides, which is covalently linked to the 5' or 3' nucleotide of the first region, such as via a internucleoside linkage group such as a phosphodiester linkage, wherein either
- the internucleoside linkage between the first and second region is a phosphodiester linkage and the nucleoside of the second region [such as immediately] adjacent to the first region is either DNA or RNA; and/or
- At least 1 nucleoside of the second region is a phosphodiester linked DNA or RNA nucleoside
- region A and region B form a single contiguous nucleotide sequence of 8 - 35 nucleotides in length.
- the internucleoside linkage between the first and second regions may be considered part of the second region.
- a phosphorus containing linkage group between the second and third region.
- the phosphorus linkage group may, for example, be a phosphate (phosphodiester), a phosphorothioate, a phosphorodithioate or a boranophosphate group.
- this phosphorus containing linkage group is positioned between the second region and a linker region which is attached to the third region.
- the phosphate group is a phosphodiester.
- the oligomeric compound comprises at least two phosphodiester groups, wherein at least one is as according to the above statement of invention, and the other is positioned between the second and third regions, optionally between a linker group and the second region.
- the third region is an activation group, such as an activation group for use in conjugation.
- the invention also provides activated oligomers comprising region A and B and a activation group, e.g an intermediate which is suitable for subsequent linking to the third region, such as suitable for conjugation.
- the third region is a reactive group, such as a reactive group for use in conjugation.
- the invention also provides oligomers comprising region A and B and a reactive group, e.g an intermediate which is suitable for subsequent linking to the third region, such as suitable for conjugation.
- the reactive group may, in some embodiments comprise an amine of alcohol group, such as an amine group.
- region A comprises at least one, such as 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, or 24 internucleoside linkages other than phosphodiester, such as internucleoside linkages which are (optionally independently] selected from the group consisting of phosphorothioate, phosphorodithioate,and
- boranophosphate and methylphosphonate, such as phosphorothioate.
- methylphosphonate such as phosphorothioate.
- region A comprises at least one phosphorothioate linkage. In some embodiments at least 50%, such as at least 75%, such as at least 90% of the
- internucleoside linkages such as all the internucleoside linkages within region A are other than phosphodiester, for example are phosphorothioate linkages. In some embodiments, all the internucleoside linkages in region A are other than phosphodiester.
- the oligomeric compound comprises an antisense
- oligonucleotide such as an antisense oligonucleotide conjugate.
- antisense oligonucleotide conjugate such as an antisense oligonucleotide conjugate.
- oligonucleotide may be or may comprise the first region, and optionally the second region.
- region B may form part of a contiguous nucleobase sequence which is complementary to the (nucleic acid) target.
- region B may lack complementarity to the target.
- the invention provides a non- phosphodiester linked, such as a phosphorothioate linked, oligonucleotide (e.g. an antisense oligonucleotide) which has at least one terminal (5' and/or 3') DNA or RNA nucleoside linked to the adjacent nucleoside of the oligonucleotide via a phosphodiester linkage, wherein the terminal DNA or RNA nucleoside is further covalently linked to a conjugate moiety, a targeting moiety or a blocking moiety, optionally via a linker moiety.
- a phosphorothioate linked, oligonucleotide e.g. an antisense oligonucleotide
- the terminal DNA or RNA nucleoside is further covalently linked to a conjugate moiety, a targeting moiety or a blocking moiety, optionally via a linker moiety.
- the first region is covalently linked to the second region, such as via a 5' terminal or 3' terminal internucleoside linkage, such as a phosphodiester linkage.
- the phosphodiester linkage may therefore be positioned between the 5' most nucleoside of region A and the 3' most nucleoside of region B, and/or between the 3' most nucleoside of region A and the 5' most nucleoside of region B.
- the two region Bs may be the same or different, and they may be covalently linked to the same or different third regions, optionally and independently via a linker (Y).
- the oligomers may have a length of 8 - 35 contiguous nucleotides and comprise a first region of e.g. 7 - 23 contiguous nucleotides, and a second region of 1 - 10 contiguous nucleotides, wherein, for example, either the internucleoside linkage between the first and second region is a phosphodiester linked to the first (or only) DNA or RNA nucleoside of the second region, or region B comprises at least one phosphodiester linked DNA or RNA nucleoside.
- the second region may, in some embodiments, comprise further DNA or RNA nucleosides which may be phosphodester linked.
- the second region is further covalently linked to a third region which may, for example, be or comprise the conjugate moiety.
- the second region may comprise or consists of at least one DNA or RNA nucleosides linked to the first region via a phosphodiester linkage.
- the internucleoside linkage between the first and second region is considered as part of region B.
- the second region comprises or consists of at least between 1 and 10 linked nucleosides, such as 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 linked DNA or RNA nucleotides.
- region B also comprises sugar-modified nucleoside analogues, such as those referred to under the first region above.
- the nucleosides of region B are (optionally independently) selected from the group consisting of DNA and RNA. It will be recognised that the nucleosides of region B may comprise naturally occurring or non-naturally occurring nucleobases.
- Region B comprises at least one phosphodiester linked DNA or RNA nucleoside (which may, in some embodiments, be the first nucleoside adjacent to region A). If region B comprises other nucleosides, region B may also comprise of other nucleoside linkages other than phosphodiester, such as (optionally independently) phosphorothioate, phosphodithioate, boranophosphate or methyl phosphonate. However, in other embodiments, all the internucleoside linkages in region B are phosphorothioate. In some embodiments, all the nucleosides of region B comprise (optionally independently) either a 2'-OH ribose sugar (RNA) or a 2'-H sugar - i.e. RNA or DNA.
- RNA 2'-OH ribose sugar
- DNA DNA
- the second region comprises or consists of at least between 1 and 10 (e.g. phosphodiester) linked DNA or RNA nucleosides, such as 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 (e.g. phosphodiester) linked DNA or RNA nucleotides.
- 1 and 10 e.g. phosphodiester linked DNA or RNA nucleosides, such as 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 (e.g. phosphodiester) linked DNA or RNA nucleotides.
- region B comprises no more than 3 or no more than 4 consecutive DNA or RNA nucleosides (such as DNA nucleosides.
- region B may be so short as it does not recruit RNAseH, an aspect which may be important when region B does not form a part of a single contiguous nucleobase sequence which is complementary to the target.
- Shorter region Bs e.g. of 1 - 4nts in length may also be preferable in some embodiments, as they are unlikely to be the target of sequence specific restriction enzymes.
- region B As such it is possible to vary the susceptibility of the region B to endonuclease cleavage, and thereby fine-tune the rate of activation of the active oligomer in vivo, or even intracellular.
- region Bs may be employed and/or region Bs which comprise the recognition sites of (e.g. cell or tissue specific or differentially expressed) restriction enzymes.
- Region B may be conjugated to the conjugate, targeting reactive group, an activation group, or blocking group (X) via a linker group which may, for example, comprise a phosphodiester linkage, and/or optionally a suitable linker group, such as those provided herein.
- a linker group which may, for example, comprise a phosphodiester linkage, and/or optionally a suitable linker group, such as those provided herein.
- a phosphate nucleoside linkage e.g. phosphodiester
- the linkage group is the same as the linkage group between regions A and B, and as such may be a phosphodiester linkage. In some aspects, the linkage group is a phiosphorothioate linkage.
- the DNA or RNA nucleotides of the second region are independently selected from DNA and RNA nucleotides. In some embodiments the DNA or RNA nucleotides of the second region are DNA nucleotides. In some embodiments the DNA or RNA nucleotides of the second region are RNA nucleotides.
- DNA and RNA nucleoside may comprise a naturally occurring or non-naturally occurring base (also referred to as a base analogue or modified base).
- the second region may further comprise other nucleotides or nucleotide analogues.
- the second region comprises only DNA or RNA nucleosides.
- the internucleoside linkages in the second region comprise phosphodiester linkages.
- all the internucleoside linkages in the second region comprise phosphodiester linkages.
- At least two consecutive nucleosides of the second region are DNA nucleosides (such as at least 3 or 4 or 5 consecutive DNA nucleotides). In some embodiments the at least two consecutive nucleosides of the second region are RNA nucleosides (such as at least 3 or 4 or 5 consecutive RNA nucleotides). In some embodiments the at least two consecutive nucleosides of the second region are at least one DNA and at least one RNA nucleoside.
- the internucleoside linkage between region A and region B is a phosphodiester linkage.
- region B comprises more than one nucleoside
- at least one further internucleoside linkage is phosphodiester - such as the linkage group(s) between the 2 (or 3 or 4 or 5) nucleosides adjacent to region A.
- the second region is flanked on one side (either 5' or 3') by the first region, e.g. an antisense oligonucleotide, and on the other side (either 3' or 5' respectfully, via a conjugate moiety or similar group (e.g. a blocking moiety/group, a targeting moiety/group or therapeutic small molecule moiety), optionally via a linker group (i.e. between the second region and the conjugate/blocking group etc. moiety).
- a conjugate moiety or similar group e.g. a blocking moiety/group, a targeting moiety/group or therapeutic small molecule moiety
- linker group i.e. between the second region and the conjugate/blocking group etc. moiety.
- oligonucleotide of the invention may be described according to the following formula:
- A is region A
- PO is a phosphodiester linkage
- B is region B
- Y is an optional linkage group
- X is a conjugate, a targeting, a blocking group or a reactive or activation group.
- region B comprises 3' - 5' or 5'-3': i) a phosphodiester linkage to the 5' nucleoside of region A, ii) a DNA or RNA nucleoside, such as a DNA nucleoside, and iii) a further phosphodiester linkage
- the further phosphodiester linkage link the region B nucleoside with one or more further nucleoside, such as one or more DNA or RNA nucleosides, or may link to X (is a conjugate, a targeting or a blocking group or a reactive or activation group) optionally via a linkage group (Y).
- region B comprises 3' - 5' or 5'-3': i) a phosphodiester linkage to the 5' nucleoside of region A, ii) between 2 - 10 DNA or RNA phosphodiester linked nucleosides, such as a DNA nucleoside, and optionally iii) a further phosphodiester linkage:
- [PO-B]n represents region B, wherein n is 1 - 10, such as 1 , 2, 3,4, 5, 6, 7, 8, 9 or 10, PO is an optional phosphodiester linkage group between region B and X (or Y if present).
- the invention provides compounds according to (or comprising) one of the following formula: 5' [Region A] - PO - [region B] 3' -Y - X
- Region B may for example comprise or consist of:
- region B does not form a complementary sequence when the oligonucleotide region A and B is aligned to the complementary target sequence.
- region B does form a complementary sequence when the oligonucleotide region A and B is aligned to the complementary target sequence.
- region A and B together may form a single contiguous sequence which is complementary to the target sequence.
- the sequence of bases in region B is selected to provide an optimal endonuclease cleavage site, based upon the predominant endonuclease cleavage enzymes present in the target tissue or cell or sub-cellular compartment.
- endonuclease cleavage sequences for use in region B may be selected based upon a preferential cleavage activity in the desired target cell (e.g. liver/hepatocytes) as compared to a non-target cell (e.g. kidney).
- the potency of the compound for target down-regulation may be optimized for the desired tissue/cell.
- region B comprises a dinucleotide of sequence AA, AT, AC, AG, TA, TT, TC, TG, CA, CT, CC, CG, GA, GT, GC, or GG, wherein C may be 5- mthylcytosine, and/or T may be replaced with U.
- region B comprises a trinucleotide of sequence AAA, AAT, AAC, AAG, ATA, ATT, ATC, ATG, ACA, ACT, ACC, ACG, AGA, AGT, AGC, AGG, TAA, TAT, TAC, TAG, TTA, TTT, TTC, TAG, TCA, TCT, TCC, TCG, TGA, TGT, TGC, TGG, CAA, CAT, CAC, CAG, CTA, CTG, CTC, CTT, CCA, CCT, CCC, CCG, CGA, CGT, CGC, CGG, GAA, GAT, GAC, CAG, GTA, GTT, GTC, GTG, GCA, GCT, GCC, GCG, GGA, GGT, GGC, and GGG wherein C may be 5-mthylcytosine and/or T may be replaced with U.
- region B comprises a trinucleotide of sequence AAAX, AATX, AACX, AAGX, ATAX, ATTX, ATCX, ATGX, ACAX, ACTX, ACCX, ACGX, AGAX, AGTX, AGCX, AGGX, TAAX, TATX, TACX, TAGX, TTAX, TTTX, TTCX, TAGX, TCAX, TCTX, TCCX, TCGX, TGAX, TGTX, TGCX, TGGX, CAAX, CATX, CACX, CAGX, CTAX, CTGX, CTCX, CTTX, CCAX, CCTX, CCCX, CCGX, CGAX, CGTX, CGCX, CGGX, GAAX, GATX, GACX, CAGX, GTAX, GTTX, GTCX, GTGX, GCAX, GCTX, GCCX, GCG
- nucleobases A, T, U, G, C these may be substituted with nucleobase analogues which function as the equivalent natural nucleobase (e.g. base pair with the complementary nucleoside).
- the compound of the invention may comprise more than one conjugate group (or more than one functional group X - such as a conjugate, targeting, blocking or activated group or a reactive or activation group), such as 2 or 3 such groups.
- region B is covalently linked, optionally via a [e.g. non-nucleotide] linker group), to at least one functional group, such as two or three functional groups.
- the first region may be covalently linked (e.g. via internucleoside linkages, such as phosphodiester linkages), to two region Bs, for example, one 5' and one 3' to the first region, wherein each region B may be (optionally independently) selected from the region B described herein.
- one region B may have one or more functional groups
- the second region B may have one or more function groups, wherein the functional groups of each region B may be independently selected from a conjugate, a targeting group, a blocking group or a reactive/activation group.
- a region B such as a "PO DNA linker”, as decribed above, between the oligomer and the conjugate moiety (X, or X-Y), is particularly advantageous as it ensures a uniform cleavage of the conjugate moiety from the oligomer sequence, once the oligomer has been delivered to the target cell (e.g. a hepatocyte). Uniform cleavage may be useful in retaining maximal intra-cellular potency of the parent compound, as well as enhancing the safety profile of the oligomer conjugate.
- the invention provides for a poly oligomeric compound which may comprise the first region (region A), (optionally the second region (region B)) and the third region (region C), wherein the first region is covalently linked to at least one further oligomeric compound (region A'), wherein the first region (region A) and region A' are covalently linked via a biocleavable linker (region B'), which may be, by way of example, as according to the second region
- regions B and B' may, in some embodiments have the same structure, e.g. the same number of DNA RNA nucleosides and phosphodiester linkages and/or the same nucleobase sequence. In other embodiments Regions B and B' may be different.
- poly oligomeric compounds may have a structure such as: (5' - 3' or 3' - 5') Conjugate-PO-ON- PO'-ON', wherein conjugate is region C, PO is region B, PO' is region B', and ON 1 is region A, and ON' is region A'
- region A may, in some embodiments, comprise multiple further oligomeric compounds (such as a further 2 or 3 oligomeric compounds) linked in series (or in parallel) via biocleavable linkers, for example: Conjugate-PO-ON-PO-ON'-PO"-ON", or Conjugate-PO-ON-[PO-ON']n, wherein n may, for example be 1 , 2 or 3, and each ON' may be the same or different, and if different may have the same or different targets.
- further oligomeric compounds such as a further 2 or 3 oligomeric compounds linked in series (or in parallel) via biocleavable linkers, for example: Conjugate-PO-ON-PO-ON'-PO"-ON", or Conjugate-PO-ON-[PO-ON']n, wherein n may, for example be 1 , 2 or 3, and each ON' may be the same or different, and if different may have the same or different targets.
- the invention provides for an oligomer of the invention comprising a contiguous nucleotide sequence of formula [LNAs]7-i8-[DNA] 1-5 -[LNAs]7-i8, and a non-nucleobase conjugate, such as a GalNAc conjugate moeity, for example a trivalent GalNAc conjugate conjugate, such as a conjugate moeity selected from the group consisting of any one of Conjl , 2, 3, 4, 1 a, 2a, 3a, 4a, or other trivalent GalNAc conjugates, such as those disclosed herein.
- Subscript s refers to a phosphorothioate linkage.
- At least one internucleoside linkage within or adjacent to the -[DNA] 1-5- region are phosphodiester linkages. In some embodiments, all internucleoside linkages within or adjacent to the -[DNA] 1-5- region are phosphodiester linkages. In some embodiments, the -[DNA] 1-5- region has 2, 3, 4 or 5 contiguous DNA nucleoside which are joined by phosphodiester linkages.
- the internucleoside linkages between the -[DNA] 2- 5- are phosphodiester linkages, and optionally the internucleoside linkages between region -[DNA] 1-5 and the LNA regions [LNA S ] 7-18 are independently phosphorothioate or phosphodiester linkages, such as both phosphodiester or both phosphorothioate, or one phosphodiester and one phosphorothioate.
- the DNA region is a single DNA nucleoside, at least one or both the the the
- internucleoside linkages adjacent to the DNA region is a phosphodiester, and if only a single phosphodiester, the other may be a phosphorothioate.
- the region -[DNA] 1-5 may be as defined as described by region B herein - i.e. may be a physiologically cleavable nucleoside linker region.
- Each [LNA S ] 7-18 is a LNA phosphorothioate oligomer, and may for example be independently selected from the group consisting of an LNA gapmer, an LNA mixmer or an LNA totalmer. For inhibiting the mature miR-122 sequence mixmers or toalmers are preferred.
- the GalNAc conjugate may for example be located 5' or 3' to the contiguous nucleotide sequence.
- at least one of the LNA oligomers, or both the poly oligomer conjugate is a LNA totalmer of 7 - 12, such as 8, 9 or 10 nucleotides in length.
- the LNA totalmer may comprise only LNA nucleotides, such as beta-D-oxy LNA nucleoside, which are linked by phosphorothioate linkages.
- the poly oligomer conjugate may comprise a contiguous nucleositide sequence [LNA s ] 7 -io- [DNA] 1-5 -[LNA s ] 7-10 , such as [LNA S ] 7-10 -[DNA] 2 -[LNA S ] 7-10 or [LNA s ] 7-1 o-[DNA] 3 -[LNA s ] 7-1 o or [LNA s ] 7-1 o-[DNA] 4 -[LNA s ] 7-1 o.
- the contiguous nucleositide sequence comprises [LNA s ] 8 -[DNA] 1-5 -[LNA s ] 8 , such as [LNAJ 8 -[DNA] 2 -[LNA S ] 8 , [LNA S ] 8 -[DNA] 3 -[LNA S ] 8 , or [LNA S ] 8 -[DNA] 4 -[LNA S ] 8 .
- the use of poly-oligomer conjugates allws for enhanced delivery to the target cell. It is also important to avoid complete saturation of cell -suface receptors which are performing an important biological function, the use of the poly-oligomer strategy therefore allows for effective delivery of sufficient compound to ensure relevant pharmacology, whilst reducing the risk of side effects due to receptor saturation/competition by the conjugate moiety.
- the use of the poly-oligomer conjugate therefore provides an effective solution for enhancing the therapeutic index - increased oligomer delivery and activity with a reduction of undesirable side-effects.
- the oligomer of the invention may be used in pharmaceutical formulations and compositions.
- such compositions comprise a pharmaceutically acceptable diluent, carrier, salt or adjuvant.
- WO2007/031091 provides suitable and preferred pharmaceutically acceptable diluent, carrier and adjuvants - which are hereby incorporated by reference.
- Suitable dosages, formulations, administration routes, compositions, dosage forms, combinations with other therapeutic agents, pro-drug formulations are also provided in WO2007/031091 - which are also hereby incorporated by reference.
- Antisense oligonucleotides may be admixed with pharmaceutically acceptable active or inert substances for the preparation of pharmaceutical compositions or formulations.
- Compositions and methods for the formulation of pharmaceutical compositions are dependent upon a number of criteria, including, but not limited to, route of administration, extent of disease, or dose to be administered.
- An antisense compound can be utilized in pharmaceutical compositions by combining the antisense compound with a suitable pharmaceutically acceptable diluent or carrier.
- a pharmaceutically acceptable diluent includes phosphate-buffered saline (PBS).
- PBS is a diluent suitable for use in compositions to be delivered parenterally.
- compositions comprising antisense compounds encompass any pharmaceutically acceptable salts, esters, or salts of such esters, or any other
- oligonucleotide which, upon administration to an animal, including a human, is capable ofproviding (directly or indirectly) the biologically active metabolite or residue thereof.
- the disclosure is also drawn to pharmaceutically acceptable salts of antisense compounds, prodrugs, pharmaceutically acceptable salts of such prodrugs, and other bioequivalents.
- Suitable pharmaceutically acceptable salts include, but are not limited to, sodium and potassium salts.
- the oligomer of the invention is a pro-drug.
- the invention provides for the antisense oligomer conjugate for use in medicine.
- the invention provides for the antisense oligomer conjugate, for use in down-regulating a liver-expressed micro-RNA.-122.
- the invention provides for the LNA antisense oligomer conjugate, for use in treatment of a metabolic disease or disorder, such as a hepatic disease or disorder.
- the invention provides for the LNA antisense oligomer conjugate, for use in treatment of hepatitis C.
- the disease is liver disease or disorder.
- the disease or disorder is, or results in or is associated with liver-fibrosis.
- the disease or disorder is a liver disease or disorder.
- the disease or disorder is a metabolic disorder, which may for example be a liver disease or disorder, and/or in some aspects a cardiovascular disease or disorder).
- Cardiovascular/Metabolic diseases include, for examples, metabolic syndrome, obesity, hyperlipidaemia , atherosclerosis, HDL/LDL cholesterol imbalance, dyslipidemias, e.g., familial combined hyperlipidaemia (FCHL), acquired hyperlipidaemia, statin-resistant, hypercholesterolemia, coronary artery disease (CAD), and coronary heart disease (CHD)., atherosclerosis, heart disease, diabetes (I and/or II), NASH, acute coronary syndrome (ACS), NASH, chronic heart failure, cardiovascular disease, cardio metabolic disease, hyperlipidaemia and related disorders, metabolic syndrome, atherosclerosis, chronic heart failure, vascular disease, peripheral arterial disease, heart disease, ischemia.
- FCHL familial combined hyperlipidaemia
- CAD coronary artery disease
- CHD coronary heart disease
- atherosclerosis heart disease
- diabetes I and/or II
- NASH acute coronary syndrome
- ACS acute coronary syndrome
- chronic heart failure cardiovascular disease
- cardio metabolic disease hyperlipidaemia and related disorders
- metabolic syndrome atheros
- the disease or disorder is selected from the group consisting of metabolic syndrome, obesity, hyperlipidaemia, atherosclerosis, HDL/LDL cholesterol imbalance, dyslipidemias, e.g., familial combined hyperlipidaemia (FCHL), acquired hyperlipidaemia, statin-resistant, hypercholesterolemia, coronary artery disease (CAD), and coronary heart disease (CHD).
- FCHL familial combined hyperlipidaemia
- CAD coronary artery disease
- CHD coronary heart disease
- the disease or disorder is selected from the group consisting of chronic heart failure, cardiovascular disease, cardio metabolic disease, chronic heart failure, vascular disease, peripheral arterial disease, heart disease, ischemia, acute coronary syndrome (ACS).
- chronic heart failure cardiovascular disease
- cardio metabolic disease chronic heart failure
- vascular disease peripheral arterial disease
- heart disease ischemia
- acute coronary syndrome ACS
- the disease or disorder is a viral disease, such viral hepatitis, such as hepatitis C.ln some embodiments, the liver disease may be a disease or disorder selected from the group consisting of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis.
- the invention further provides use of a compound of the invention in the manufacture of a medicament for the treatment of a disease, disorder or condition, such as those as referred to herein.
- An interesting aspect of the invention is directed to the use of the compound as defined herein for the preparation of a medicament for the treatment of a disease, disorder or condition as referred to herein.
- the invention relates to a method of treating a subject, such as a mammal, such as a human suffering from a disease or condition such as those referred to herein.
- a patient who is in need of treatment is a patient suffering from or likely to suffer from the disease or disorder.
- treatment refers to both treatment of an existing disease (e.g. a disease or disorder as herein referred to), or prevention of a disease, i.e. prophylaxis. It will therefore be recognized that treatment as referred to herein may, in some embodiments, be prophylactic.
- the compound of the invention is for use in a combination treatment with another therapeutic agent.
- Inhibitors of HMG CoA reductase such as statins for example are widely used in the treatment of metabolic disease (see WO2009/043354, hereby incorporated by reference for examples of combination treatments).
- Suitable cholesterol lowering compounds may be selected from a compound is selected from the group consisting of bile salt sequestering resins (e.g., cholestyramine, colestipol, and colesevelam hydrochloride), HMGCoA-reductase inhibitors (e.g., lovastatin, cerivastatin, prevastatin, atorvastatin, simvastatin, and fluvastatin), nicotinic acid, fibric acid derivatives (e.g., clofibrate, gemfibrozil, fenofibrate, bezafibrate, and ciprofibrate), probucol, neomycin, dextrothyroxine, plant-stanol esters, cholesterol absorption inhibitors (e.g., ezetimibe), implitapide, inhibitors of bile acid transporters (apical sodium-dependent bile acid
- statins may be particularly preferred.
- the compound of the invention are for use in treating an HCV infection in a subject, wherein said treatment is in combination with a further therapeutic compound, such as an interferon or an anti-viral, such as ribavirin.
- the invention provides a method of treatment of an HCV infection in a subject infected with HCV, said method comprising administering an effective amount of the compound of the invention in combination with an effective amount of a further therapeutic compound, such as an interferon or an anti-viral, such as ribavirin, to the subject infected with HCV.
- the combination treatment may be with a combination with both an interferon and one or more other antivirals.
- the combination treatment may be with ribavirin and an other anti-viral.
- the combination treatment may be with the compound of the invention and an other antiviral (i.e. other than ribavirin).
- the interferons include, but are not limited to pegylated rIFN-alpha 2b, pegylated rIFN-alpha 2a, rIFN-alpha 2b, rIFN-alpha 2a, consensus IFN alpha (infergen), feron, reaferon, intermax alpha, r-IFN-beta, infergen and actimmune, IFN-omega with DUROS, albuferon, locteron, Albuferon, Rebif, oral interferon alpha, IFNalp ha-2b XL, AVI-005, PEG-lnfergen, and pegylated IFN-beta.
- Ribavirin analogs and the ribavirin prodrug viramidine have been administered with interferons to control HCV.
- compound 3 may therefore also comprises antiviral ribavirin analogs and antiviral ribavirin derivatives, as well as ribavirin pro-drugs.
- antivirals for use in combination treatment of the invention examples include NS5A protein inhibitors (see WO2013000856, hereby incorporated by reference, e.g. ledipasvir), NS3/4 protease inhibitor, such as telaprevir or boceprevir (see WO2012/175733, hereby incorporated by reference), or an HS5B RNA dependent RNA polymerase inhibitor (see WO2013/000855, hereby incorporated by reference), including non-nucleoside inhibitors and nucleoside inhibitors, such as Sovaldi (sofosbuvir, previously known as PSI-7977 or Gl- 7977), .
- NS5A protein inhibitors see WO2013000856, hereby incorporated by reference, e.g. ledipasvir
- NS3/4 protease inhibitor such as telaprevir or boceprevir
- HS5B RNA dependent RNA polymerase inhibitor see WO2013/000855, hereby incorporated by reference
- the compound of the invention may be used, either in monotherapy or as part of a combination therapy to treat a subject infected with HCV.
- the subject may be infected with HCV of a genotype selected from the group consisting of 1 a, 1 b, 2, 3, 4, 5 or 6.
- the genotype of the HCV is 1 a.
- the genotype of the HCV is 1 b.
- the subject is treatment naive.
- the subject is subject who is a non-responder, a partial responder, a relapse responder or a null responder to a HCV therapy, such as an interferon, or other HCV therapeutics described herein.
- LNA nucleosides such as beta-D-oxy LNA
- lower case letters represent DNA nucleosides.
- Capital L is a LNA, such as beta-D-oxy
- lower case d is a DNA nucleoside.
- LNA cytosines are optionally 5'methyl cytosine.
- the internucleosides within region A are phosphorothioate, and within region B are
- phosphodiester (as shown).
- the internucleoside linkage between region A and B is phoshodiester, but where region B is >1 DNA nucleotide, may optionally be other than phosphodiester (e.g. may be phosphorothioate).
- Y further linker between region B and region C, such as a C6 linker.
- # refers to SEQ ID No.
- FAM-labeled ASOs with different DNA PO-linkers were subjected to in vitro cleavage either in S1 nuclease extract (Fig. 6A), Liver or kidney homogenates or Serum FAM-labeled ASOs 100 ⁇ with different DNA/PO-linkers were subjected to in vitro cleavage by S1 nuclease in nuclease buffer (60 U pr. 100 ⁇ _) for 20 and 120 minutes (A). The enzymatic activity was stopped by adding EDTA to the buffer solution.
- Sequence of DNA/PO-linkers can modulate the cleavage rate as seen after 20 min in Nuclease S1 extract Sequence selection for region B (e.g.for the DNA/PO-linker) can therefore also be used to modulate the level of cleavage in serum and in cells of target tissues.
- Liver, kidney and Serum were spiked with oligonucleotide SEQ ID NO 16 to concentrations of 200 ⁇ g g tissue.
- Liver and kidney samples collected from NMRI mice were homogenized in a homogenisation buffer (0,5% Igepal CA-630, 25 mM Tris pH 8.0, 100 mM NaCI, pH 8.0 (adjusted with 1 N NaOH). The homogenates were incubated for 24 hours at 37° and thereafter the homogenates were extracted with phenol - chloroform. The content of cleaved and non cleaved oligonucleotide in the extract from liver and kidney and from the serum were determinded against a standard using the above HPLC method.
- the PO linkers results in cleavage of the conjugate (or group C) from the oligonucleotide, in liver or kidney homogenate, but not in serum.
- cleavage in the above assays refers to the cleavage of the cleavable linker, the oligomer or region A should remain functionally intact.
- the susceptibility to cleavage in the above assays can be used to determine whether a linker is biocleavable or physiologically labile.
- LNA such as beta-D-oxy LNA.
- Lower case letters are DNA.
- Subscript s is a phosphorothioate linkage.
- Other internucleoside linkages are phosphodiester
- Conjl a group may be another GalNAc conjugate group, such as those disclosed herein, for example Conj2a.
Abstract
The invention relates conjugated oligonucleotides which target microRNA-122 which comprise an antisense oligomer which is complementary to a microRNA-122 target sequence wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
Description
ANTIMIR-122 OLIGONUCLEOTIDE CARBOHYDRATE CONJUGATES
FIELD OF INVENTION
The invention relates conjugated oligonucleotides which target microRNA-122 which comprise an antisense oligomer which is complementary to a microRNA-122 target sequence wherein the antisense oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
BACKGROUND
Oligonucleotide conjugates have been extensively evaluated for use in siRNAs, where they are considered essential in order to obtain sufficient in vivo potency. For example, see WO2004/044141 refers to modified oligomeric compounds that modulate gene expression via an RNA interference pathway. The oligomeric compounds include one or more conjugate moieties that can modify or enhance the pharmacokinetic and pharmacodynamic properties of the attached oligomeric compound.
WO2012/083046 reports on a galactose cluster-pharmacokinetic modulator targeting moiety for siRNAs.
In contrast, single stranded antisense oligonucleotides are typically administered therapeutically without conjugation or formulation. The main target tissues for antisense oligonucleotides are the liver and the kidney, although a wide range of other tissues are also accessible by the antisense modality, including lymph node, spleen, bone marrow.
According to van Poelgeest et al., (American Journal of Kidney Disease, In Press), the administration of an LNA antisense oligonucleotide in human clinical trials may have resulted in acute kidney injury. According to Swayze et al, NAR, Dec. 2006, advanced online publication antisense oligonucleotides containing locked nucleic acid improve potency but cause significant hepatotoxicity in animals.
Cholesterol conjugates have been used to target siRNAs and antagomirs to the liver - see for example, Soutscheck et al., Nature 2004 vol. 432 173 - 178 and Krijtzfeldt et al., Nature 2005 vol 438, 685 - 689. For the partially phosphorothiolated siRNAs and antagomirs, the use of cholesterol as a liver targeting entity was found to be essential for in vivo activity.
Bhat et al., AASLD November 7 - 1 1 th 2013 (poster) disclosed data from the use of a GalNac conjugated anti-miR, RG-101 , targeting miR-122 for reduction of HCV in preclinical studies. The identity of RG-101 was not disclosed.
Hepatitis C virus (HCV) is the most common infectious cause of chronic liver disease in Europe and the United States. Worldwide, approximately 3% of the population is estimated to be infected; this corresponds to approximately 200 million people at risk of developing serious liver related morbidity.
An unconjugates 15mer LNA mixmer oligonucleotide, miravirsen which targets microRNA- 122, an essential host factor for HCV infection in the liver, is being developed for treatment of HCV and has shown efficacy for the treatment of hepatitis C (HCV) in both chimpanzees and human patients. Miravirsen is a microRNA-122 inhibitor.
microRNA-122 (miR-122) is a liver specific microRNA. miR-122 which is involved in lipid and cholesterol metabolism, and inhibition of miR-122 in vivo in rodents results in a reduction in serum cholesterol levels. The molecular mechanisms whereby miR-122 regulated cholesterol metabolism are apparently, at present unknown. The present invention is based upon the discovery that the potency, bio-distribution and therapeutic index of single stranded antisense oligonucleotides targeting microRNA-122 can be vastly improved by the conjugation of the oligonucleotide to a carbohydrate conjugate, such as a GalNAc conjugate. The invention therefore provides novel therapeutic agents for treatment of HCV or cholesterol associated metabolic disease.
RELATED APPLICATIONS
This application claims priority from EP13153296.2 (filed 2013-01 -30),
EP13157237.2 (filed 2013-02-28), EP13174092.0, (filed 2013-06-27), EP13192931.7 (filed 2013-1 1 -14), EP13192930.9 (filed 2013-1 1 -14), PCT/EP2013/073859 (filed 2013-1 1 -14): These contents of these applications are hereby incorporated by reference.
PCT/EP2014/051781 which refers to LNA oligomer conjugates is also hereby incorporated by reference in its entirity.
SUMMARY OF INVENTION
The invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
The invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to a conjugate group which comprises at least one N-acetylgalactosamine (GalNac) group.
The invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to a conjugate group which comprises a galactose cluster.
The invention provides for an antisense oligonucleotide which targets microRNA-122 for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to a conjugate group which comprises three N-acetylgalactosamine (GalNAc) groups (a trivalent GalNAc cluster).
The antisense oligonucleotide comprises a contiguous nucleotide sequence which is complementary to at least 8 contiguous nucleotides present in a hsa-miR-122 sequence. In some embodiments, the contiguous nucleotide sequence of the antisense oligonucleotide is fully complementary to a corresponding region of hsa-miR-122. Such compounds are are refered to as targeting a microR-122 nucleic acid, such as hsa-miR-122.
In some embodiments, the contiguous nucleotide sequence of the antisense oligonucleotide comprises a region which is fully complementary to the hsa-miR-122 seed region.
In some embodiments, the contiguous nucleotide sequence of the antisense oligonucleotide comprises the sequence 5'-CACACTCC-3' or 5'-ACACTCC-3'.
The invention also provides for a poly oligomeric oligomer targeting miR-122. The antisense oligonucleotide according to the invention may therefore comprise a first region which is complementary to at least 7 consecutive nucleotides of hsa-miR-122, and a second region which is complementary to at least 7 consecutive nucleotides of hsa-miR-122, wherein the first and the second regions are linked via a region physiologically labile nucleotides, such as 1 - 5 phosphodiester linked DNA nucleotides. The first and second regions may each be as according to the oligonucleotide region of the (mono-oligomeric) other compounds of the invention, and may in some embodiments be the same and in some be different oligomer sequences. In some embodiments the first and the second regions are 7 - 10 LNA nucleotides in length which each target the microRNA seed region. Suitably the nucleosides of the first and the second regions may be linked by phosphorothioate linkages.
The invention provides for an antisense oligonucleotide which targets microRNA-122, for the treatment of high cholesterol, wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
In some embodiments the antisense oligonucleotide of the invention comprises LNA nucleotides.
The invention provides for an antisense oligonucleotide according to the invention for the treatment of a metabolic disease, such as a cholesterol associated metabolic disease, such as elevated cholesterol.
The invention provides for an antisense oligonucleotide according to the invention for the treatment of hepatitis C infection.
The invention provides for an antisense oligonucleotide which targets microRNA-122- wherein said antisense oligonucleotide is conjugated to an asialoglycoprotein receptor targeting moiety, for use in medicine.
The invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the treatment of a metabolic disease or viral disease.
The invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the treatment of HCV infection.
The invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the improvement of hepatic function in a subject.
The invention provides for the use of an antisense oligonucleotide according to the invention, for the preparation of a medicament for the reduction in necroinflammation in a subject.
The invention provides for a method of treatment of HCV infection in a subject suffering from hepatitis, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention, to the subject.
The invention provides for a method of treatment of HCV infection in a subject infected with HCV, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention, to the subject.
The invention provides for a method of treatment of a metabolic disease a subject in need of treatment, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the imvention, to the subject.
The uinvention provides for a method of improving the haptic function a subject in need of improved hepatic function, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention to the subject.
The invention provides for a method of reducing necroinflammation a subject in need of reduced necroinflammation, wherein said method comprises the administration of an effective amount of the antisense oligonucleotide according to the invention, to the subject.
The invention provides for an LNA antisense oligomer which targets microRNA-122 (which may be referred to as region A herein) comprising an antisense oligomer and an asialoglycoprotein receptor targeting conjugate moiety, such as a GalNAc moiety, which may form part of a further region (referred to as region C).
The invention provides for an LNA antisense oligomer which targets microRNA-122
(which may be referred to as region A herein) comprising an antisense oligomer and a GalNAc moiety, such as a trivalent GalNAc moiety which may form part of a further region (referred to as region C).
The invention provides for an LNA antisense oligomer which targets microRNA-122 which is covalently joined to (e.g. linked to) a (non-nucleoside) carbohydrate moiety, such as a carbohydrate conjugate moiety. In some embodiments the carbohydrate moiety is not a linear carbohydrate polymer. The carbohydrate moiety may however be multi-valent, such as, for example 2, 3, 4 or 4 identical or non-identical carbohydrate moieties may be covalently joined to the oligomer, optionally via a linker or linkers.
The invention provides for an LNA antisense oligomer (conjugate) comprising an LNA antisense oligomer and a conjugate moiety which comprises a carbohydrate, such as a carbohydrate conjugate moiety.
The invention provides for the oligomeric compound according to the invention for use in the inhibition of miroRNA-122 target in a cell. In some embodiments the use is in vitro. In some embodiments the use is in vivo.
The invention also provides for an oligomer which targets microRNA-122, such as an LNA oligomer which targets microRNA-122, comprising a contiguous region of 7 - 23 phosphorothioate linked nucleosides, and further comprising between 1 and 6 DNA nucleosides which are contiguous with the LNA oligomer, wherein the internucleoside linkages between the DNA, and/or adjacent to the DNA nucleoside(s), is physiologically labile, such as is / are phosphodiester linkages. Such an LNA oligomer may be in the form of a conjugate, as described herein. When conjugated, the conjugate may, for example a carbohydrate, such as a GalNac conjugate, such as a GalNac cluster, e.g. triGalNac, or another conjugate as described herein.
BRIEF DESCRIPTION OF FIGURES
Figure 1 : Examples of tri-GalNac conjugates which may be used. Conjuagtes 1 - 4 illustrate 4 suitable GalNac conjugate moieties, and conjugates 1 a - 4a refer to the same conjugates with an additional linker moiety (Y) which is used to link the conjugate to the oligomer (region
A or to a biocleavable linker, such as region B). The wavy line represents the covalent link to the oligomer.
Figure 2: Silencing of miR-122 in the mouse liver by seed-targeting tiny LNA. (a) RNA blot analysis of liver RNAs from mice after treatment with three intravenous doses of 20 mg/kg tiny antimiR-122, 15-mer antimiR-122 or LNA scramble control or with saline.
Figure 3: Total Cholesterol analysis at pre-dose, day 4 and day7. Cholesterol is upregulated due to decreased miR122.
Figure 4: Expression of Aldo A and Bckdk was measured by standard TaqMan Q-PCR assays. The mRNA levels of these genes are upregulated due to decreased miR122.
Figure 5: ALT was measured from final serum (day 7) to assess tolerability of the compounds.
DESCRIPTION OF THE INVENTION
The invention provides for an antisense oligonucleotide which targets microRNA-122 (miR- 122), such as for use in the treatment of hepatitis C infection (HCV) or a metabolic disorder, wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety. In some embodiments, the invention relates to LNA oligomeric compounds, such as LNA antisense oligonucleotides, which are covalently linked to a non-nucleotide carbohydrate conjugate group.
The Oligomer
In various embodiments, the compound of the invention does not comprise RNA (units). In some embodiments, the oligomer has a single contiguous sequence which is a linear molecule or is synthesized as a linear molecule. The oligomer may therefore be single stranded molecule. In some embodiments, the oligomer does not comprise short regions of, for example, at least 3, 4 or 5 contiguous nucleotides, which are complementary to equivalent regions within the same oligomer (i.e. duplexes). The oligomer, in some embodiments, may be not (essentially) double stranded. The oligomer is essentially not double stranded, such as is not a siRNA. In some embodiments, the oligomeric compound is not in the form of a duplex with a (substantially) complementary oligonucleotide - e.g. is not an siRNA.
The term "oligomer" in the context of the present invention, refers to a molecule formed by covalent linkage of two or more nucleotides (i.e. an oligonucleotide). Herein, a single nucleotide (unit) may also be referred to as a monomer or unit. In some
embodiments, the terms "nucleoside", "nucleotide", "unit" and "monomer" are used
interchangeably. It will be recognized that when referring to a sequence of nucleotides or monomers, what is referred to is the sequence of bases, such as A, T, G, C or U.
The oligomer may consists or comprises of a contiguous nucleotide sequence of from 7 - 30, such as 7 - 26 or 8 - 25, such as 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24, 25 nucleotides in length, such as 10 - 20 nucleotides in length. In some
embodiments, the length of the LNA oligomer is 10 - 16 nucleotides, such as 12, 13 or 14 nucleosides. In some embodiments, the LNA oligomer is 7, 8, 9 nucleosides in length, such as a "Tiny" LNA.
In some embodiments, the oligomers comprise or consist of a contiguous nucleotide sequence of a total of from 10 - 22, such as 12 - 18, such as 13 - 17 or 12 - 16, such as 13, 14, 15, 16 contiguous nucleotides in length.
In some embodiments, the oligomers comprise or consist of a contiguous nucleotide sequence of a total of 10, 1 1 , 12, 13, or 14 contiguous nucleotides in length.
In some embodiments, the oligomer according to the invention consists of no more than 22 nucleotides, such as no more than 20 nucleotides, such as no more than 18 nucleotides, such as 15, 16 or 17 nucleotides. In some embodiments the oligomer of the invention comprises less than 20 nucleotides. It should be understood that when a range is given for an oligomer, or contiguous nucleotide sequence length it includes the lower an upper lengths provided in the range, for example from (or between) 10 - 30, includes both 10 and 30.
In some embodiments, the use of the carbohydrate conjugates according to the invention have been found to be particularly suitable for short LNA oligomers, including short gapmers, mixmers or totalmers (tinys) such as LNA oligomers of less than 20, such as less than 18, such as 16nts or less or 15 or 14nts or less.
LNA Oligomers
In some embodiments, the invention relates to LNA oligomeric compounds, such as LNA antisense oligonucleotides, which are covalently linked to a non-nucleotide carbohydrate conjugate group. An LNA oligomer is an oligomer which comprises at least one LNA nucleoside, and may typically comprise at least 20% LNA nucleosides, such as at least 30% LNA nucleosides, such as at least 40% such as at least 50% LNA (not including region B). In some embodiments, short LNA oligomers (or region A) may be 7 - 12 nucleotides wherein at least 70% of the nucleotides are LNA (excluding region B), such as at least 80% LNA, such as at least 90% LNA, or all nucleotides are LNA nucleotides. Such short LNAs
may be joined together via region B to form multiple oligomer conjugates (poly oligomer conjugates).
In some embodiments, the invention provides LNA antisense oligomer conjugate, comprising a LNA antisense oligomer and a conjugate moiety which comprises a
carbohydrate, such as a carbohydrate conjugate moiety, covalently bound to the LNA antisense oligomer.
The present invention employs LNA oligomeric compounds (also referred herein as LNA oligomers or LNA oligonucleotides) for use in modulating, such as inhibiting a target nucleic acid in a cell. An LNA oligomer comprises at least one "Locked Nucleic Acid" (LNA) nucleoside, such as a nucleoside which comprises a covalent bridge (also referred to a radical) between the 2' and 4' position (a 2' - 4' bridge). LNA nucleosides are also referred to as "bicyclic nucleosides". The LNA oligomer is typically a single stranded antisense oligonucleotide.
In some embodiments the LNA oligomer comprises or is a gapmer. In some embodiments the LNA oligomer comprises or is a mixmer. In some embodiments the LNA oligomer comprises or is a totalmer.
In some embodiments, the nucleoside analogues present in the oligomer are all LNA, and the oligomer may, optionally further comprise RNA or DNA, such as DNA nucleosides (e.g. in a gapmer or mixmer).
Internucleoside Linkages
In some embodiments, the internucleoside linkages of the oligomer comprise at least one internucleoside linkage other than phosphodiester, such as at least one, such as at least 50%, such as at least 75%, such as at least 90%, such as 100% of the internucleoside linkages in the oligomer (or oligomer region or region A) are other than phosphodiester. In some embodiments, the internucleoside linkages other than phosphodiester are sulphur containing internucleoside linkages, such as phosphorothioate, phosphorodithioate such as phosphorothioate.
The oligomer may comprise at least one phosphorothioate internucleoside linkage, such as at least two, three or four phosphorothioate linkages, and in some embodiments at least 50% of the internucleoside linkages may be phosphorothioate, such as at least 75%, at least 90% or all internucleoside linkages (other than those, optionally present in a cleavable linker) may be phosphorothioate. In some embodiments the internucleoside linkages between the two terminal nucleosides at the 5' end, the 3' end or both the 5' and 3' end (other than in region B, when present), are sulphur containing internucleoside linkages, such
as phosphorothioate. In some embodiments the oligomer comprises at least one region of consecutive DNA nucleosides, such as a region of 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12
consecutive DNA nucleosides, wherein the internucleoside linkage between the DNA nucleosides is a sulphur containing internucleoside linkage such as phosphorothiote. In some embodiments, the central region of a gapmer (Υ'), which typically comprises a region of consecutive DNA nucleosides has are sulphur containing internucleoside linkages, such as phosphorothioate, between the nucleosides, such as between consecutive DNA nucleosides and/or between DNA nucleosides and nucleic acid analogue nucleosides, such as the sugar modified nucleosides referred to herein, such as LNA.
Other examples of internucleoside linkages which may be used in the oligomer include methylphosphonates (CH3P=0) and methylthionophosphate (CH3P=S) and
boranophosphate.
Locked Nucleic Acid Nucleosides (LNA)
Bicyclic nucleoside analogues (LNA nucleosides) include nucleoside analogues typically which comprise a bridge (or biradical) linking the second and forth carbon of the ribose ring, (C4*-C2* bridge or biradical). The presence of the biradical between the 2nd and 4th carbon locks the ribose into a 3' endo- (north) confirmation, and as such bicyclic nucleoside analogues with a C2*-C4* biradical are often referred to as Locked nucleic acid (LNA), or bicyclic nucleic acids (BNA). The terms LNA and BNA are used interchangeably herein.
In some embodiments, some or all of the nucleosides of the LNA oligomer may be modified nucleosides, also referred to as nucleoside analogues herein, such as sugar modified nucleoside analogues, for example bicyclic nucleoside analogues (e.g. LNA) and/or 2'substituted nucleoside analogues. In some embodiments, the nucleoside analogues present in the oligomer all comprise the same sugar modification, for example are all bicyclic nucleoside analogues, such as they may be (optionally independently) selected from the group consisting of beta-D-X-LNA or alpha-L-X-LNA (wherein X is oxy, amino or thio), or other LNAs disclosed herein including, but not limited to,(R/S) cET, cMOE or 5'-Me-LNA.
In some embodiments, the oligomer may comprise at least one bicyclic nucleoside (LNA) and at least one further nucleoside analogue, such as one or more 2'substituted nucleoside. In some embodiments, some or all of the nucleosides of the oligomer may be modified nucleosides, also referred to as nucleoside analogues herein.
In some embodiments, the oligomer or oligomer region (region A) comprises at least one, such as at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at
least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21 , at least 22, at least 23, at least 24 or 25 nucleoside analogues. In some embodiments the nucleoside analogues are (optionally independently) selected from the group consisting of bicyclic nucleoside analogues (such as LNA), and/or 2' substituted nucleoside analogues, such as (optionally independently) selected from the group consisting of 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino- DNA units, 2'-AP, 2'-FANA, 2'-(3-hydroxy)propyl, and 2'-fluoro-DNA units, and/or other (optionally) sugar modified nucleoside analogues such as morpholino, peptide nucleic acid (PNA), CeNA, unlinked nucleic acid (UNA), hexitol nucleoic acid (HNA). bicyclo-HNA (see e.g. WO2009/100320), In some embodiments, the further nucleoside analogues increase the affinity of the first region for its target nucleic acid (or a complementary DNA or RNA sequence). Various nucleoside analogues are disclosed in Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, hereby incorporated by reference.
In some embodiments, the LNA oligomer comprises at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, for example all nucleoside analogues (or in a totalmer all nucleosides) bicyclic nucleoside analogues, such as LNA, e.g. beta-D-X-LNA or alpha-L- X-LNA (wherein X is oxy, amino or thio, such as oxy), or other LNAs disclosed herein including, but not limited to,(R/S) cET, cMOE or 5'-Me-LNA. In some embodiments, the LNA oligomer, comprises of DNA and sugar modified nucleoside analogues, such as bicyclic nucleoside analogues and/or 2'substituted nucleoside analogues. In some embodiments, the oligomer comprises of DNA and LNA nucleoside units.
WO05/013901 , WO07/027775, WO07027894 refers to fully 2'substituted oligomers, such as fully 2'-0-MOE. In some embodiments, the first region of the oligomer may comprise of 2' substituted nucleosides. WO07/027775 also refers to MOE, LNA, DNA mixmers for use in targeting microRNAs.
The Target
The oligomer of the invention targets the liver expressed microRNA-122, such as hsa- miR-122. Oligomer's targeting miR-122 are disclosed in WO2007/1 12754,
WO2007/1 12753, WO2009/043353, and may be mixmers, such as SPC3649, also referred to as miravirsen (which has the sequence 5'-CcAttGTcaCaCtCC-3' (SEQ ID NO 1 ) , where capital letters are beta-D-oxy LNA, small letters are DNA, fully phosphorothioate and LNA C are 5-methyl cyctosine), or a tiny LNA, such as those disclosed in WO2009/043353 (e.g. 5'-
ACACTCC-3', 5'-CACACTCC-3\ 5'-TCACACTCC-3', SEQ ID NOs 2-5) where capital letters are (optionally beta-D_oxy) LNA, fully phosphorothioate and LNA Cs are, optionally5-methyl cyctosine). In some embodiments, the miR-122 targeting oligomers have a length of 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17 or 18 nucleotides in length. In some embodiments, the miR- 122 targeting oligomers a sequence which is fully complementary to miR-122 as measured accross the length of the oligomer, and preferably include the sequence 5'-CACACTCC-3'. According to miRBase, the mature microRNA-122 sequence is 5'
uggagugugacaaugguguuugu 3' (SEQ ID NO 6). In some embodiments, the oligomer targeting a microRNA such as miR-122, is complementary to a corresponding region of the microRNA accorss the length of the oligomer and in some embodiments the 3' nucleoside of the oligomer is compelmentary to (i.e. aligns to) the first, second, third or fourth 5' nucleotides of the microRNA, such as miR-122, such as the second 5' nucleotide of the microRNA, such as miR-122.
In some embodiments, such as when the target is microRNA-122, the disease may be a viral disease, such as hepatitis, including hepatitis B and hepatitis C, or a metabolic disease related to elevated cholesterol, such as atherosclosis and hyperlipidaemia and related disorders. Oligomers targeting miR-122 may also be used for the improvement of hepatic function (see e.g. PCT/EP2012/071934), for the treatment of treatment of necroinflammation, for improving blood serum biomarkers of liver function, for preventing loss (or reducing the rate of loss) of liver function in a human subject who may or may not be infected with HCV and is at risk of deteriorating liver function, improving liver function in a human subject who is or who is not infected with HCV, and is in need of improved liver function. The liver disease may be a disease or disorder selected from the group consisting of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis; or from the group consisting of a disease or disorder selected from the group consisting of cytomegalovirus infection, schistosomiasis infection and Leptospirosis infection.
The invention provides a method of down-regulating or inhibiting the expression of the target in a cell which is expressing the target, said method comprising administering the oligomer or conjugate according to the invention to said cell to down-regulating or inhibiting the expression of the target in said cell. Suitably the cell is a mammalian cell such as a human cell. The administration may occur, in some embodiments, in vitro. The
administration may occur, in some embodiments, in vivo.
In some embodiments, the first region (or first and second region) form a single contiguous nucleobase sequence which is complementary, to a corresponding region of a microRNA- 122 such as miR-122a (i.e. targets), such as the hsa-miR-122 sequences (miRBase release 20: MI0000442), such as:
>hsa-mir-122 MI0000442
CCUUAGCAGAGCUGUGGAGUGUGACAAUGGUGUUUGUGUCUAAACUAUCAAACGCCAUUAUCACACUAAAUAGCU ACUGCUAGGC SEQ ID NO 7
>hsa-miR-122-5p MIMAT0000421
UGGAGUGUGACAAUGGUGUUUG SEQ ID NO 8
miR-122 has been indicated in HCV infection, where it is an essential host factor required for maintenance of the infection. Inhibitors of miR-122 may therefore be used in the treatment of hepatitis C infection.
Compounds of the invention which target miR-122 may be used in the treatment of HCV infection. The invention therefore provides for the oligomer according to the invention which targets miR-122 for use in the treatment of HCV infection. The invention further provides for a method of treatment of HCV infection, wherein said method comprises the administration of the oligomer of the invention to a subject in need to said treatment.
The invention provides for the use of an oligomer of the invention which targets miR- 122, for the manufacture of a medicament for the treatment of HCV infection.
The invention provides for a method of treating HCV infection, said method
comprising administering an effective amount of an oligomer according to the invention which targets miR-122, to a patient suffering from HCV infection.
The invention provides for an in vivo or in vitro method for the inhibition of miR-122 in a cell which is expressing miR-122, such as an HCV infected cell or a HCV replicon expressing cell, said method comprising administering an oligomer or conjugate or pharmaceutical composition according to the invention to said cell so as to inhibit miR-122 in said cell.
miR-122 has also been indicated in cholesterol metabolism, and it has been suggested that inhibition of miR-122 may be used for a treatment to reduce plasma cholesterol levels (Esau, Cell Metab. 2006 Feb;3(2):87-98.)
Inhibitors of miR-122 may therefore be used in a treatment to reduce plasma cholesterol levels, or in the treatment of a metabolic disease associated with elevated levels of cholesterol (related disorders), such as indications selected from the group consisting of liver steatosis,_atherosclerosis, hyperlipidaemia, hypercholesterolemia, familiar
hypercholesterolemia, dyslipidemias, coronary artery disease (CAD), and coronary heart disease (CHD)
Compounds of the invention which target miR-122 may be used in the treatment of elevated cholesterol levels or related disorders. The invention therefore provides for the oligomer according to the invention which targets miR-122 for use in the treatment of elevated cholesterol levels or related disorders. The invention further provides for a method of treatment of elevated cholesterol levels or related disorders, wherein said method comprises the administration of the oligomer of the invention to a subject in need to said treatment.
The invention provides for the use of an oligomer of the invention which targets miR- 122, for the manufacture of a medicament for the treatment of elevated cholesterol levels or related disorders.
The invention provides for a method of treating elevated cholesterol levels or related disorders, said method comprising administering an effective amount of an oligomer according to the invention which targets miR-122, to a patient suffering from said disorder.
The invention provides for an in vivo or in vitro method for the inhibition of miR-122 in a cell which is expressing miR-122, such as an HCV infected cell or a HCV replicon expressing cell, said method comprising administering an oligomer or conjugate or pharmaceutical composition according to the invention to said cell so as to inhibit miR-122 in said cell.
Oligomer's targeting miR-122 are disclosed in WO2007/1 12754, WO2007/1 12753,
WO2009/043353, and may be mixmers, such as SPC3649, also referred to as miravirsen see below, or a tiny LNA, such as those disclosed in WO2009/043353 (e.g. 5'-ACACTCC- 3', 5'-CACACTCC-3', 5'-TCACACTCC-3', (SEQ ID NOs 3 - 5) where capital letters are beta- D_oxy LNA, fully phosphorothioate and LNA C are 5-methyl cyctosine). In some
embodiments, the miR-122 targeting oligomers have a length of 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17 or 18 (or 19, 20, 21 , 22 or 23 nucleotides) in length. In some embodiments, the miR- 122 targeting oligomers a sequence which is fully complementary to miR-122 as measured accross the length of the oligomer, and preferably include the sequence 5'-CACACTCC-3' (SEQ ID NO 3). In some embodiments, the oligomer targeting a microRNA such as miR- 122, is complementary to a corresponding region of the microRNA accorss the length of the oligomer and in some embodiments the 3' nucleoside of the oligomer is compelmentary to (i.e. aligns to) the first, second, third or fourth 5' nucleotides of the microRNA, such as miR- 122, such as the second 5' nucleotide of the microRNA, such as miR-122.
The following is an oligomers which targets the hsa-miR-122 (human miR-122), and may be used as region A in the compounds of the invention.
Miravirsen: 5'- mCsCsAststsGsTscsasmCsasmCstsmCsmC -3' (SEQ ID NO 1 )
Other miR-122 targeting compounds which may be used in the context of the present invention (region A) are disclosed in WO2007/027894, WO2007/027775.
In some embodiments, the contiguous nucleotide sequence comprises no more than a single mismatch when hybridizing to the target sequence. Region B may however be non- complementary and may therefore be disregarded when determining the degree of complementarity.
In determining the degree of "complementarity" between oligomers of the invention (or regions thereof) and the target region of the nucleic acid, such as those disclosed herein, the degree of "complementarity" (also, "homology" or "identity") is expressed as the percentage identity (or percentage homology) between the sequence of the oligomer (or region thereof) and the sequence of the target region (or the reverse complement of the target region) that best aligns therewith. The percentage is calculated by counting the number of aligned bases that are identical between the 2 sequences, dividing by the total number of contiguous monomers in the oligomer, and multiplying by 100. In such a comparison, if gaps exist, it is preferable that such gaps are merely mismatches rather than areas where the number of monomers within the gap differs between the oligomer of the invention and the target region.
As used herein, the terms "homologous" and "homology" are interchangeable with the terms "identity" and "identical".
The terms "corresponding to" and "corresponds to" refer to the comparison between the nucleotide sequence of the oligomer (i.e. the nucleobase or base sequence) or contiguous nucleotide sequence (a first region) and the equivalent contiguous nucleotide sequence of a further sequence selected from either i) a sub-sequence of the reverse complement of the nucleic acid target. Nucleotide analogues are compared directly to their equivalent or corresponding nucleotides. A first sequence which corresponds to a further sequence under i) or ii) typically is identical to that sequence over the length of the first sequence (such as the contiguous nucleotide sequence) or, as described herein may, in some embodiments, is at least 80% homologous to a corresponding sequence, such as at least 85%, at least 90%, at least 91 %, at least 92%at least 93%, at least 94%, at least 95%, at least 96% homologous, such as 100% homologous (identical).
The terms "corresponding nucleotide analogue" and "corresponding nucleotide" are intended to indicate that the nucleotide in the nucleotide analogue and the naturally
occurring nucleotide are identical. For example, when the 2-deoxyribose unit of the nucleotide is linked to an adenine, the "corresponding nucleotide analogue" contains a pentose unit (different from 2-deoxyribose) linked to an adenine.
The terms "reverse complement", "reverse complementary" and "reverse
complementarity" as used herein are interchangeable with the terms "complement", "complementary" and "complementarity".
The contiguous nucleobase sequence of the oligomer may therefore be
complementary to a target, such as those referred to herein.
Nucleosides and Nucleoside analogues
The term "nucleotide" as used herein, refers to a glycoside comprising a sugar moiety
(or analogue thereof), a base moiety and a covalently linked group (linkage group), such as a phosphate or phosphorothioate internucleotide linkage group, and covers both naturally occurring nucleotides, such as DNA or RNA, and non-naturally occurring nucleotides comprising modified sugar and/or base moieties, which are also referred to as "nucleotide analogues" herein. Herein, a single nucleotide (unit) may also be referred to as a monomer or nucleic acid unit.
It will be recognized that in the context of the present invention the term nucleoside and nucleotide are used to refer to both naturally occurring nucleotides/sides, such as DNA and RNA, as well as nucleotide/side analogues.
In field of biochemistry, the term "nucleoside" is commonly used to refer to a glycoside comprising a sugar moiety and a base moiety, and may therefore be used when referring to the nucleotide units, which are covalently linked by the internucleoside linkages between the nucleotides of the oligomer. In the field of biotechnology, the term "nucleotide" is often used to refer to a nucleic acid monomer or unit, and as such in the context of an oligonucleotide may refer to the base - such as the "nucleotide sequence", typically refer to the nucleobase sequence (i.e. the presence of the sugar backbone and internucleoside linkages are implicit). Likewise, particularly in the case of oligonucleotides where one or more of the internucleoside linkage groups are modified, the term "nucleotide" may refer to a
"nucleoside" for example the term "nucleotide" may be used, even when specifying the presence or nature of the linkages between the nucleosides.
As one of ordinary skill in the art would recognise, the 5' terminal nucleotide of an oligonucleotide does not comprise a 5' internucleoside linkage group, although may or may not comprise a 5' terminal group. Non-naturally occurring nucleotides include nucleotides
which have modified sugar moieties, such as bicyclic nucleotides or 2' modified nucleotides, such as 2' substituted nucleotides.
"Nucleotide analogues" are variants of natural nucleotides, such as DNA or RNA nucleotides, by virtue of modifications in the sugar and/or base moieties. Analogues could in principle be merely "silent" or "equivalent" to the natural nucleotides in the context of the oligonucleotide, i.e. have no functional effect on the way the oligonucleotide works to inhibit target gene expression. Such "equivalent" analogues may nevertheless be useful if, for example, they are easier or cheaper to manufacture, or are more stable to storage or manufacturing conditions, or represent a tag or label. Preferably, however, the analogues will have a functional effect on the way in which the oligomer works to inhibit expression; for example by producing increased binding affinity to the target and/or increased resistance to intracellular nucleases and/or increased ease of transport into the cell. Specific examples of nucleoside analogues are described by e.g. Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, and in Scheme 1 :
Boranop osp ates
Scheme 1
The oligomer may thus comprise or consist of a simple sequence of natural occurring nucleotides - preferably 2'-deoxynucleotides (referred here generally as "DNA"), but also possibly ribonucleotides (referred here generally as "RNA"), or a combination of such naturally occurring nucleotides and one or more non-naturally occurring nucleotides, i.e. nucleotide analogues. Such nucleotide analogues may suitably enhance the affinity of the oligomer for the target sequence.
Examples of suitable and preferred nucleotide analogues are provided by WO2007/031091 or are referenced therein. Other nucleotide analogues which may be used in the oligomer of the invention include tricyclic nucleic acids, for example please see WO2013154798 and WO2013154798 which are hereby incorporated by reference.
Oligomeric compounds, such as antisense oligonucleotides, such as the compounds referred to herein, including region A, and in some optional embodiments, region B, may contain one or more nucleosides wherein the sugar group has been modified. Such sugar modified nucleosides (nucleoside analogues) may impart enhanced nuclease stability, increased binding affinity, or some other beneficial biological property to the antisense compounds. In some embodiments, nucleosides comprise a chemically modified
ribofiiranose ring moiety.
In some embodiments, the oligomer, or first region thereof, comprises at least one , such as at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21 , at least 22, at least 23, at least 24 or 25 nucleoside analogues, such as sugar modified nucleoside analogues.
In some embodiments the nucleoside analogues are (optionally independently selected from the group consisting of bicyclic nucleoside analogues (such as LNA), and/or 2' substituted nucleoside analogues, such as (optionally independently) selected from the group consisting of 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino-DNA units, 2'-AP, 2'- FANA, 2'-(3-hydroxy)propyl, and 2'-fluoro-DNA units, and/or other (optionally) sugar modified nucleoside analogues such as morpholino, peptide nucleic acid (PNA), CeNA, unlinked nucleic acid (UNA), hexitol nucleoic acid (HNA). bicyclo-HNA (see e.g.
WO2009/100320), In some embodiments, the nucleoside analogues increase the affinity of the first region for its target nucleic acid (or a complementary DNA or RNA sequence).
In some embodiments, the oligomer comprises at least one bicyclic nucleotide analogue, such as LNA. In some embodiments, the first region comprises of at least one bicyclic nucleoside analogues (e.g. LNA) and/or 2'substituted nucleoside analogues. In
some embodiments, the nucleoside analogues present in the oligomer all comprise the same sugar modification. In some embodiments, at least one nucleoside analogue present in the first region is a bicyclic nucleoside analogue, such as at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 1 1 , at least 12, at least 13, at least 14, at least 15, at least 16, for example all nucleoside analogues (except the DNA and or RNA nucleosides of region B) are sugar modified nucleoside analogues, such as such as bicyclic nucleoside analogues, such as LNA, e.g. beta-D-X-LNA or alpha-L- X-LNA (wherein X is oxy, amino or thio), or other LNAs disclosed herein including, but not limited to,(R/S) cET, cMOE or 5'-Me-LNA.
Examples of chemically modified ribofiiranose rings include, without limitation, addition of substituent groups (including 5' and 2' substituent groups); bridging of non-geminal ring atoms to form bicyclic nucleic acids (BNA); replacement of the ribosyl ring oxygen atom with S, N(R), or C(Ri)(R2) (R = H, Ci -C2 alkyl or a protecting group); and combinations thereof. Examples of chemically modified sugars include, 2'-F-5'-methyl substituted nucleoside (see, PCT International Application WO 2008/101 157, published on 8/21/08 for other disclosed 5', 2'-bis substituted nucleosides), replacement of the ribosyl ring oxygen atom with S with further substitution at the 2'-position (see, published U.S. Patent Application
US2005/0130923, published on June 16, 2005), or, alternatively, 5'-substitution of a BNA (see, PCT International Application WO 2007/134181 , published on 1 1/22/07, wherein LNA is substituted with, for example, a 5'-methyl or a 5 -vinyl group).
Examples of nucleosides having modified sugar moieties include, without limitation, nucleosides comprising 5'-vinyl, 5'-methyl (R or S), 4'-S, 2'-F, 2'-OCH3, and 2'-0(CH2)2 O CH3 substituent groups. The substituent at the 2' position can also be selected from allyl, amino, azido, thio, O-allyl, O-C Ci0 alkyl, OCF3, O (CH2)2SCH3, O (CH2)2- O -N(Rm)(Rn), and O -CH2-C(=0)-N(Rm)(Rn), where each Rm and Rn is, independently, H or substituted or unsubstituted C1-C10 alkyl.
As used herein, "bicyclic nucleosides" refer to modified nucleosides comprising a bicyclic sugar moiety. Examples of bicyclic nucleosides include, without limitation, nucleosides comprising a bridge between the 4' and the 2' ribosyl ring atoms. In some embodiments, compounds provided herein include one or more bicyclic nucleosides wherein the bridge comprises a 4' to 2' bicyclic nucleoside. Examples of such 4' to 2' bicyclic nucleosides, include, but are not limited to, one of the formulae: 4'-(CH2)- O -2' (LNA); 4'- (CH2)-S-2'; 4 -(CH2)2- O -2' (ENA); 4'-CH(CH3)- O -2' and 4'-CH(CH2OCH3)-0-2*, and analogs thereof (see, U.S. Patent 7,399,845, issued on July 15, 2008); 4'-C(CH3)(CH3)-0-2',
and analogs thereof (see, published PCT International Application WO2009/006478, published January 8, 2009); 4'-CH2-N(OCH3)-2', and analogs thereof (see, published PCT International Application WO2008/150729, published December 1 1 , 2008); 4'-CH2-0- N(CH3)-2' (see, published U.S. Patent Application US2004/0171570, published September 2, 2004); 4'-CH2-N(R)- O -2', wherein R is H, C Ci0 alkyl, or a protecting group (see, U.S. Patent 7,427,672, issued on September 23, 2008); 4'-CH2-C(H)(CH3)-2' (see,
Chattopadhyaya, et al, J. Org. Chem.,2009, 74, 1 18-134); and 4'-CH2-C(=CH2)-2', and analogs thereof (see, published PCT International Application WO 2008/154401 , published on December 8, 2008). Also see, for example: Singh et al., Chem. Commun., 1998, 4, 455- 456; Koshkin et al., Tetrahedron, 1998, 54, 3607-3630; Wahlestedt et al., Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 5633-5638; Kumar et al., Bioorg. Med. Chem. Lett., 1998, 8, 2219- 2222; Singh et al., J. Org. Chem., 1998, 63, 10035-10039; Srivastava et al., J. Am. Chem. Soc, 129(26) 8362-8379 (Jul. 4, 2007); Elayadi et al., Curr. Opinion Invens. Drugs, 2001 , 2, 558-561 ; Braasch et al., Chem. Biol, 2001 , 8, 1 -7; Oram et al, Curr. Opinion Mol. Ther., 2001 , 3, 239-243; U.S. Patent Nos U.S. 6,670,461 , 7,053,207, 6,268,490, 6,770,748,
6,794,499, 7,034,133, 6,525,191 , 7,399,845; published PCT International applications WO 2004/106356, WO 94/14226, WO 2005/021570, and WO 2007/134181 ; U.S. Patent Publication Nos. US2004/0171570, US2007/0287831 , and US2008/0039618; and U.S. Patent Serial Nos. 12/129,154, 60/989,574, 61/026,995, 61/026,998, 61/056,564,
61/086,231 , 61/097,787, and 61/099,844; and PCT International Application Nos.
PCT/US2008/064591 , PCT/US2008/066154, and PCT/US2008/068922. Each of the foregoing bicyclic nucleosides can be prepared having one or more stereochemical sugar configurations including for example a-L-ribofuranose and beta -D-ribofuranose (see PCT international application PCT DK98/00393, published on March 25, 1999 as WO 99/14226).
In some embodiments, bicyclic sugar moieties of BNA nucleosides include, but are not limited to, compounds having at least one bridge between the 4' and the 2' position of the pentofuranosyl sugar moiety wherein such bridges independently comprises 1 or from 2 to 4 linked groups independently selected from - [CiRaXRb)],,-, -C(Ra)=C(Rb)-, -C(Ra)=N-, - C(=NRa)-, -C(=0)-, -C(=S)-, - O -, -Si(Ra)2-, -S(=0)x-, and -N(Ra)-; wherein: x is 0, 1 , or 2; n is 1 , 2, 3, or 4; each Ra and Rb is, independently, H, a protecting group, hydroxyl, C Ci2 alkyl, substituted C Ci2 alkyl, C2-Ci2 alkenyl, substituted C2-Ci2 alkenyl, C2-Ci2 alkynyl, substituted C2-Ci2 alkynyl, C5-C20 aryl, substituted C5-C20 aryl, heterocycle radical, substituted heterocycle radical, heteroaryl, substituted heteroaryl, C5-C7 alicyclic radical, substituted C5-C7 alicyclic radical, halogen, OJi, NJiJ2, SJi, N3, COOJ1, acyl (C(=0)- H),
substituted acyl, CN, sulfonyl (S(=0)2-Ji), or sulfoxyl
and J2 is, independently, H, CrC6 alkyl, substituted C1-C12 alkyl, C2-C12 alkenyl, substituted C2-Ci2 alkenyl, C2-Ci2 alkynyl, substituted C2-Ci2 alkynyl, C5-C20 aryl, substituted C5-C2o aryl, acyl (C(=0)- H), substituted acyl, a heterocycle radical, a substituted heterocycle radical, C1-Ci2 aminoalkyl, substituted C Ci2 aminoalkyl, or a protecting group.
In some embodiments, the bridge of a bicyclic sugar moiety is, -[C(Ra)(Rb)]n-, - [C(Ra)( b)]n- O -, -C(RaRb)-N(R)- O - or, -C(RaRb)- O -N(R)-. In some embodiments, the bridge is 4'-CH2-2', 4'-(CH2)2-2', 4'- (CH2)3-2', 4'-CH2- O -2', 4*-(CH2)2- O -2', 4'-CH2- O - N(R)-2', and 4'-CH2-N(R)- O -2'-, wherein each R is, independently, H, a protecting group, or Ci-Ci2 alkyl.
In some embodiments, bicyclic nucleosides are further defined by isomeric
configuration. For example, a nucleoside comprising a 4'-2' methylene-oxy bridge, may be in the a-L configuration or in the beta - D configuration. Previously, a-L-methyleneoxy (4'-CH2- 0-2') BNA's have been incorporated into antisense oligonucleotides that showed antisense activity (Frieden et al, Nucleic Acids Research, 2003, 21 , 6365- 6372).
In some embodiments, bicyclic nucleosides include, but are not limited to, (A) a-L- Methyleneoxy (4'-CH2-0-2') BNA, (B) beta -D-Methyleneoxy (4'-CH2-0-2') BNA, (C)
Ethyleneoxy (4'-(CH2)2-0-2') BNA, (D) Aminooxy (4'-CH2-0-N(R)-2') BNA, (E) Oxyamino (4'- CH2-N(R)-0-2') BNA, (F), Methyl(methyleneoxy) (4'-CH(CH3)-0-2') BNA, (G) methylene-thio (4'-CH2-S-2') BNA, (H) methylene- amino (4'-CH2-N(R)-2') BNA, (I) methyl carbocyclic (4'- CH2-CH(CH3)-2') BNA, and (J) propylene carbocyclic (4'-(CH2)3-2') BNA as depicted below.
wherein Bx is the base moiety and R is, independently, H, a protecting group or Ci-C2 alkyl. odiments, bicyclic nucleoside having Formula I:
wherein:
Bx is a heterocyclic base moiety;
-Qa-Qb-Qc- is -CH2-N(Rc)-CH2-, -C(=0)-N(Rc)-CH2-, -CH2-0-N(Rc)-, -CH2-N(Rc)-0-, or - N(Rc)-0-CH2;
Rc is Ci-Ci2 alkyl or an amino protecting group; and
Ta and Tb are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium.
In some embodiments, bicyclic nucleoside having Formula II:
wherein:
Bx is a heterocyclic base moiety;
Ta and Tb are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium; Za is CrC6 alkyi, C2-C6 alkenyl, C2-C6 alkynyl, substituted CrC6 alkyi, substituted C2-C6 alkenyl, substituted C2-C6 alkynyl, acyl, substituted acyl, substituted amide, thiol, or substituted thio.
In some embodiments, each of the substituted groups is, independently, mono or poly substituted with substituent groups independently selected from halogen, oxo, hydroxyl, OJc NJ d, SJc, N3, OC(=X)Jc, and NJeC(=X)NJcJd, wherein each Jc, Jd, and Je is, independently, H, Ci-C6 alkyi, or substituted C C6 alkyi and X is O or NJC.
I nts, bicyclic nucleoside having Formula III:
wherein:
Bx is a heterocyclic base moiety;
Ta and Tb are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium;
Rd is Ci-C6 alkyi, C2-C6 alkenyl, C2-C6 alkynyl, substituted C C6 alkyi, substituted C2-C6 alkenyl, substituted C2-C6 alkynyl, or substituted acyl (C(=0)-).
In some embodiments, bicyclic nucleoside having Formula IV:
wherein:
Bx is a heterocyclic base moiety;
Ta and Tb are each, independently H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium;
Rd is Ci-C6 alkyl, substituted Ci-C6 alkyl, C2-C6 alkenyl, substituted C2-C6 alkenyl, C2-C6 alkynyl, substituted C2-C6 alkynyl; each qb, qc and qd is, independently, H, halogen, Ci-C6 alkyl, substituted Ci-C6 alkyl, C2-Ce alkenyl, substituted C2-C6 alkenyl, C2-C6 alkynyl, or substituted C2-C6 alkynyl, CrC6 alkoxyl, substituted Q- C6 alkoxyl, acyl, substituted acyl, d- C6 aminoalkyl, or substituted Ci-C6 aminoalkyl;
In some embodiments bicyclic nucleoside having Formula V:
wherein:
Bx is a heterocyclic base moiety;
Ta and Tb are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium; qa, qb, qc and qf are each, independently, hydrogen, halogen, C Ci2 alkyl, substituted C Ci2 alkyl, C2- Ci2 alkenyl, substituted C2-Ci2 alkenyl, C2-Ci2 alkynyl, substituted C2-Ci2 alkynyl, C Ci2 alkoxy, substituted C Ci2 alkoxy, OJj, SJj, SOJj, S02Jj, NJjJk, N3, CN, C(=0)OJj, C(=0)NJjJk, C(=0)Jj, 0-C(=0)NJjJk, N(H)C(=NH)NJjJk,
N(H)C(=0)NJjJk or N(H)C(=S)NJjJk; or qe and qf together are =C(qg)(qh); qg and qh are each, independently, H, halogen, d- Ci2 alkyl, or substituted C Ci2 alkyl.
The synthesis and preparation of the methyleneoxy (4'-CH2-0-2') BNA monomers adenine, cytosine, guanine, 5-methyl-cytosine, thymine, and uracil, along with their oligomerization, and nucleic acid recognition properties have been described (see, e.g., Koshkin et al., Tetrahedron, 1998, 54, 3607-3630). BNAs and preparation thereof are also described in WO 98/39352 and WO 99/14226.
Analogs of methyleneoxy (4'-CH2-0-2') BNA, methyleneoxy (4'-CH2-0-2') BNA, and 2'-thio- BNAs, have also been prepared {see, e.g., Kumar et al., Bioorg. Med. Chem. Lett., 1998, 8, 2219-2222). Preparation of locked nucleoside analogs comprising oligodeoxynbonucleotide duplexes as substrates for nucleic acid polymerases has also been described (see, e.g., Wengel et al., WO 99/14226). Furthermore, synthesis of 2'-amino-BNA, a novel
comformationally restricted high-affinity oligonucleotide analog, has been described in the art (see, e.g., Singh et al., J. Org. Chem., 1998, 63, 10035-10039). In addition, 2'- amino- and 2'-methylamino-BNA's have been prepared and the thermal stability of their duplexes with complementary RNA and DNA strands has been previously reported.
In some embodiments, the bic clic nucleoside has Formula VI:
wherein:
Bx is a heterocyclic base moiety;
Ta and Tb are each, independently, H, a hydroxyl protecting group, a conjugate group, a reactive phosphorus group, a phosphorus moiety, or a covalent attachment to a support medium; each qj, qj, qk and ql is, independently, H, halogen, C1-C12 alkyl, substituted C1-C12 alkyl, C2-C12 alkenyl, substituted C2-C12 alkenyl, C2-Ci2 alkynyl, substituted C2-Ci2 alkynyl, Ci-Ci2 alkoxyl, substituted C2- Ci2 alkoxyl, OJj, SJ,, SOJj, S02Jj, NJjJk, N3, CN, C(=0)OJj, C(=0)NJjJk, C(=0)Jj, 0-C(=0)NJjJk, N(H)C(=NH)NJjJk, N(H)C(=0)NJjJk, or (H)C(=S)NJjJk; and qi and q, or ql and qk together are =C(qg)(qh), wherein qg and qh are each,
independently, H, halogen, C Ci2 alkyl, or substituted Ci-C6 alkyl.
One carbocyclic bicyclic nucleoside having a 4'-(CH2)3-2' bridge and the alkenyl analog, bridge 4'- CH=CH-CH2-2', have been described (see, e.g., Freier et al, Nucleic Acids Research, 1997, 25(22), 4429- 4443 and Albaek et al, J. Org. Chem., 2006, 71 , 7731 -77 '40). The synthesis and preparation of carbocyclic bicyclic nucleosides along with their oligomerization and biochemical studies have also been described (see, e.g., Srivastava et al, J. Am. Chem. Soc. 2007, 129(26), 8362-8379).
As used herein, "4'-2' bicyclic nucleoside" or "4' to 2' bicyclic nucleoside" refers to a bicyclic nucleoside comprising a furanose ring comprising a bridge connecting the 2' carbon atom and the 4' carbon atom.
As used herein, "monocylic nucleosides" refer to nucleosides comprising modified sugar moieties that are not bicyclic sugar moieties. In some embodiments, the sugar moiety, or sugar moiety analogue, of a nucleoside may be modified or substituted at any position. As used herein, "2'-modified sugar" means a furanosyl sugar modified at the 2' position. In some embodiments, such modifications include substituents selected from: a halide, including, but not limited to substituted and unsubstituted alkoxy, substituted and
unsubstituted thioalkyl, substituted and unsubstituted amino alkyl, substituted and unsubstituted alkyl, substituted and unsubstituted allyl, and substituted and unsubstituted alkynyl. In some embodiments, 2' modifications are selected from substituents including, but not limited to: 0[(CH2)nO]mCH3, 0(CH2)„NH2, 0(CH2)„CH3, 0(CH2)„ONH2,
OCH2C(=0)N(H)CH3, and 0(CH2)nON[(CH2)nCH3]2, where n and m are from 1 to about 10. Other 2'- substituent groups can also be selected from: C Ci2 alkyl; substituted alkyl;
alkenyl; alkynyl; alkaryl; aralkyl; O-alkaryl or O-aralkyl; SH; SCH3; OCN ; CI ; Br; CN; CF3; OCF3; SOCH3; S02CH3; ON02; N02; N3; NH2; heterocycloalkyl; heterocycloalkaryl;
aminoalkylamino; polyalkylamino; substituted silyl; an R; a cleaving group; a reporter group; an intercalator; a group for improving pharmacokinetic properties; and a group for improving the pharmacodynamic properties of an antisense compound, and other substituents having similar properties. In some embodiments, modified nucleosides comprise a 2'-MOE side chain {see, e.g., Baker et al., J. Biol. Chem., 1997, 272, 1 1944-12000). Such 2'-MOE substitution have been described as having improved binding affinity compared to unmodified nucleosides and to other modified nucleosides, such as 2'- O- methyl, O-propyl, and O-aminopropyl. Oligonucleotides having the 2 -MOE substituent also have been shown to be antisense inhibitors of gene expression with promising features for in vivo use {see, e.g., Martin, P., He/v. Chim. Acta, 1995, 78, 486-504; Altmann et al., Chimia, 1996, 50, 168-
176; Altmann et al., Biochem. Soc. Trans., 1996, 24, 630-637; and Altmann et al.,
Nucleosides Nucleotides, 1997, 16, 917-926).
As used herein, a "modified tetrahydropyran nucleoside" or "modified THP nucleoside" means a nucleoside having a six-membered tetrahydropyran "sugar" substituted in for the pentofuranosyl residue in normal nucleosides (a sugar surrogate). Modified ?THP
nucleosides include, but are not limited to, what is referred to in the art as hexitol nucleic acid (HNA), anitol nucleic acid (ANA), manitol nucleic acid (MNA) {see Leumann, CJ. Bioorg. and Med. Chem. (2002) 10:841 -854), fluoro HNA (F-HNA), or those compounds having Formula X:
Formula
X wherein independently for each of said at least one tetrahydropyran nucleoside analog of Formula X:
Bx is a heterocyclic base moiety;
T3 and T4 are each, independently, an internucleoside linking group linking the
tetrahydropyran nucleoside analog to the antisense compound or one of T3 and T4 is an internucleoside linking group linking the tetrahydropyran nucleoside analog to the antisense compound and the other of T3 and T4 is H, a hydroxyl protecting group, a linked conjugate group, or a 5' or 3'-terminal group; qi q2 q3 q4 qs, q6 and q7 are each, independently, H, CrC6 alkyl, substituted Ci-C6 alkyl, C2-C6 alkenyl, substituted C2-C6 alkenyl, C2-C6 alkynyl, or substituted C2-C6 alkynyl; and one of Ri and R2 is hydrogen and the other is selected from halogen, substituted or unsubstituted alkoxy, NJ,J2, SJ,, N3,
OC(=X)NJ1J2, NJ3C(=X) NJ^, and CN, wherein X is O, S, or N^ and each J2, and J3 is, independently, H or Ci-C6 alkyl.
In some embodiments, the modified THP nucleosides of Formula X are provided wherein qm, qn, qP, qr, qs, qt, and qu are each H. In some embodiments, at least one of qm, qn, q , qr, qs, qt and qu is other than H. In some embodiments, at least one of qm, qn, q , qr, qs, qtand qu is methyl. In some embodiments, THP nucleosides of Formula X are provided wherein one of
Ri and R2 is F. In some embodiments, Riis fluoro and R2 is H, R-i is methoxy and R2 is H, and Ri is methoxyethoxy and R2 is H.
As used herein, "2'-modified" or "2'-substituted" refers to a nucleoside comprising a sugar comprising a substituent at the 2' position other than H or OH. 2'-modified
nucleosides, include, but are not limited to nucleosides with non-bridging 2'substituents, such as allyl, amino, azido, thio, O-allyl, O-C Ci0 alkyl, -OCF3, 0-(CH2)2-0-CH3, 2'- 0(CH2)2SCH3, 0-(CH2)2-0- N(Rm)(Rn), or 0-CH2-C(=0)-N(Rm)(Rn), where each Rm and R„ is, independently, H or substituted or unsubstituted C-I-C-IO alkyl. 2'-modifed nucleosides may further comprise other modifications, for example, at other positions of the sugar and/or at the nucleobase.
As used herein, "2'-F" refers to a sugar comprising a fluoro group at the 2' position.
As used herein, "2'-OMe" or "2'-OCH3" or "2'-0-methyl" each refers to a nucleoside comprising a sugar comprising an -OCH3 group at the 2' position of the sugar ring.
As used herein, "oligonucleotide" refers to a compound comprising a plurality of linked nucleosides.
In some embodiments, one or more of the plurality of nucleosides is modified. In some embodiments, an oligonucleotide comprises one or more ribonucleosides (RNA) and/or deoxyribonucleosides (DNA).
Many other bicyclo and tricyclo sugar surrogate ring systems are also known in the art that can be used to modify nucleosides for incorporation into antisense compounds {see, e.g., review article: Leumann, J. C, Bioorganic and Medicinal Chemistry, 2002, 10, 841 -854). Such ring systems can undergo various additional substitutions to enhance activity. Methods for the preparations of modified sugars are well known to those skilled in the art. In nucleotides having modified sugar moieties, the nucleobase moieties (natural, modified, or a combination thereof) are maintained for hybridization with an appropriate nucleic acid target.
In some embodiments, antisense compounds comprise one or more nucleotides having modified sugar moieties. In some embodiments, the modified sugar moiety is 2'- MOE. In some embodiments, the 2'-MOE modified nucleotides are arranged in a gapmer motif. In some embodiments, the modified sugar moiety is a cEt. In some embodiments, the cEt modified nucleotides are arranged throughout the wings of a gapmer motif.
In some embodiments, in the BNA (LNA), R4* and R2* together designate the biradical -0-CH(CH2OCH3)- (2'0-methoxyethyl bicyclic nucleic acid - Seth at al., 2010, J. Org. Chem) - in either the R- or S- configuration.
In some embodiments, in the BNA (LNA), R4* and R2* together designate the biradical -0-CH(CH2CH3)- (2'0-ethyl bicyclic nucleic acid - Seth at al., 2010, J. Org. Chem). - in either the R- or S- configuration.
In some embodiments, in the BNA (LNA), R4* and R2* together designate the biradical -0-CH(CH3)-. - in either the R- or S- configuration. In some embodiments, R4* and R2* together designate the biradical -0-CH2-0-CH2- - (Seth at al., 2010, J. Org. Chem).
In some embodiments, in the BNA (LNA), R4* and R2* together designate the biradical -O-NR-CH3- (Seth at al., 2010, J. Org. Chem) .
In some embodiments, the LNA units have a structure selected from the following group:
(R,S)-cEt (R,S)-cMOE (7?,S 5'-Me-LNA
We have evaluated the nephrotoxicity of a cET compound (using (S)-cET, with the sequence (Compound I D 6/41 1847 of WO2009/12495 and a comparative beta-D-oxy LNA compound (6/392063 of WO2009/12495) and found that the cET compounds elicit surprisingly high nephrotoxicity as compared to the beta-D-oxy LNA control. The study was a single dose study, with sacrifice after 3 days (see EP1984381 example 41 for the methodology, although we used NMRI mice). Nephrotoxicity was confirmed by histological analysis. Notably signs of nephrotoxicity we seen at dosages of the cET compound below those where serum ALT was noted, indicating that for cET compounds, nephrotoxicity may be a particular problem. The use of the conjugates of the present invention, such as trivalent GalNAc conjugates are therefore highly useful in reducing the nephrotoxicity of LNA compounds, such as cET compounds.
The oligomer may thus comprise or consist of a simple sequence of natural occurring nucleotides - preferably 2'-deoxynucleotides (referred to here generally as "DNA"), but also possibly ribonucleotides (referred to here generally as "RNA"), or a combination of such naturally occurring nucleotides and one or more non-naturally occurring nucleotides, i.e. nucleotide analogues. Such nucleotide analogues may suitably enhance the affinity of the oligomer for the target sequence.
Incorporation of affinity-enhancing nucleotide analogues in the oligomer, such as BNA, (e.g.) LNA or 2'-substituted sugars, can allow the size of the specifically binding oligomer to be reduced, and may also reduce the upper limit to the size of the oligomer before nonspecific or aberrant binding takes place.
In some embodiments, the oligomer comprises at least 1 nucleoside analogue. In some embodiments the oligomer comprises at least 2 nucleotide analogues. In some embodiments, the oligomer comprises from 3-8 nucleotide analogues, e.g. 6 or 7 nucleotide analogues. In the by far most preferred embodiments, at least one of said nucleotide analogues is a BNA, such as locked nucleic acid (LNA); for example at least 3 or at least 4, or at least 5, or at least 6, or at least 7, or 8, of the nucleotide analogues may be BNA, such as LNA. In some embodiments all the nucleotides analogues may be BNA, such as LNA.
It will be recognised that when referring to a preferred nucleotide sequence motif or nucleotide sequence, which consists of only nucleotides, the oligomers of the invention which are defined by that sequence may comprise a corresponding nucleotide analogue in place of one or more of the nucleotides present in said sequence, such as BNA units or other nucleotide analogues, which raise the duplex stability/Tm of the oligomer/target duplex (i.e. affinity enhancing nucleotide analogues).
A preferred nucleotide analogue is LNA, such as oxy-LNA (such as beta-D-oxy-LNA, and alpha-L-oxy-LNA), and/or amino-LNA (such as beta-D-amino-LNA and alpha-L-amino- LNA) and/or thio-LNA (such as beta-D-thio-LNA and alpha-L-thio-LNA) and/or ENA (such as beta-D-ENA and alpha-L-ENA).
In some embodiments the further nucleotide analogues present within the oligomer of the invention are independently selected from, for example: 2'-0-alkyl-RNA units, 2'-amino- DNA units, 2'-fluoro-DNA units, BNA units, e.g. LNA units, arabino nucleic acid (ANA) units, 2'-fluoro-ANA units, HNA units, INA (intercalating nucleic acid -Christensen, 2002. Nucl.
Acids. Res. 2002 30: 4918-4925, hereby incorporated by reference) units and 2'MOE units. In some embodiments there is only one of the above types of nucleotide analogues present in the oligomer of the invention, such as the first region, or contiguous nucleotide sequence thereof.
In some embodiments the further nucleotide analogues are 2'-0-methoxyethyl-RNA
(2'MOE), 2'-fluoro-DNA monomers or LNA nucleotide analogues, and as such the oligonucleotide of the invention may comprise nucleotide analogues which are
independently selected from these three types of analogue, or may comprise only one type of analogue selected from the three types. In some embodiments at least one of said
nucleotide analogues is 2'-MOE-RNA, such as 2, 3, 4, 5, 6, 7, 8, 9 or 10 2'-MOE-RNA nucleotide units. In some embodiments at least one of said nucleotide analogues is 2'-fluoro DNA, such as 2, 3, 4, 5, 6, 7, 8, 9 or 10 2'-fluoro-DNA nucleotide units.
The oligomer according to the invention comprises at least one BNA, e.g. Locked Nucleic Acid (LNA) unit, such as 1 , 2, 3, 4, 5, 6, 7, or 8 BNA LNA units, such as from 3 - 7 or 4 to 8 BNA LNA units, or 3, 4, 5, 6 or 7 BNA/LNA units. In some embodiments, all the nucleotide analogues are BNA, such as LNA. In some embodiments, the oligomer may comprise both beta-D-oxy-LNA, and one or more of the following LNA units: thio-LNA, amino-LNA, oxy-LNA, and/or ENA in either the beta-D or alpha-L configurations or combinations thereof. In some embodiments all BNA, such as LNA, cytosine units are 5'methyl-Cytosine. In some embodiments of the invention, the oligomer (such as the first and optionally second regions) may comprise both BNA and LNA and DNA units. In some embodiments, the combined total of LNA and DNA units is 10-25, such as 10 - 24, preferably 10-20, such as 10 - 18, such as 12-16. In some embodiments of the invention, the nucleotide sequence of the oligomer, of first region thereof, such as the contiguous nucleotide sequence consists of at least one BNA, e.g. LNA and the remaining nucleotide units are DNA units. In some embodiments the oligomer, or first region thereof, comprises only BNA, e.g. LNA, nucleotide analogues and naturally occurring nucleotides (such as RNA or DNA, most preferably DNA nucleotides), optionally with modified internucleotide linkages such as phosphorothioate.
The term "nucleobase" refers to the base moiety of a nucleotide and covers both naturally occurring a well as non-naturally occurring variants. Thus, "nucleobase" covers not only the known purine and pyrimidine heterocycles but also heterocyclic analogues and tautomeres thereof. It will be recognised that the DNA or RNA nucleosides of region B may have a naturally occurring and/or non-naturally occurring nucleobase(s), such as DNA nucleobases independently selected from the group A, C, T and G, or the group C, T and G.
Examples of nucleobases include, but are not limited to adenine, guanine, cytosine, thymidine, uracil, xanthine, hypoxanthine, 5-methylcytosine, isocytosine, pseudoisocytosine, 5-bromouracil, 5-propynyluracil, 6-aminopurine, 2-aminopurine, inosine, diaminopurine, and 2-chloro-6-aminopurine. In some embodiments the nucleobases may be independently selected from the group consisting of adenine, guanine, cytosine, thymidine, uracil, 5- methylcytosine. In some embodiments the nucleobases may be independently selected from the group consisting of adenine, guanine, cytosine, thymidine, and 5-methylcytosine.
In some embodiments, at least one of the nucleobases present in the oligomer is a modified nucleobase selected from the group consisting of 5-methylcytosine, isocytosine, pseudoisocytosine, 5-bromouracil, 5-propynyluracil, 6-aminopurine, 2-aminopurine, inosine, diaminopurine, and 2-chloro-6-aminopurine.
LNA
The term "LNA" refers to a bicyclic nucleoside analogue which comprises a C2* - C4* biradical (a bridge), and is known as "Locked Nucleic Acid". It may refer to an LNA monomer, or, when used in the context of an "LNA oligonucleotide", LNA refers to an oligonucleotide containing one or more such bicyclic nucleotide analogues. In some aspects bicyclic nucleoside analogues are LNA nucleotides, and these terms may therefore be used interchangeably, and is such embodiments, both are be characterised by the presence of a linker group (such as a bridge) between C2' and C4' of the ribose sugar ring.
In some embodiments the LNA used in the oligonucleotide compounds of the invention preferably has the structure of the general formula II:
wherein Y is selected from the group consisting of -0-, -CH20-, -S-, -NH-, N(Re) and/or - CH2-; Z and Z* are independently selected among an internucleotide linkage, RH, a terminal group or a protecting group; B constitutes a natural or non-natural nucleotide base moiety (nucleobase), and RH is selected from hydrogen and Ci-4-alkyl; Ra, Rb Rc, Rd and Re are, optionally independently, selected from the group consisting of hydrogen, optionally substituted Ci-i2-alkyl, optionally substituted C2-i2-alkenyl, optionally substituted C2-i2-alkynyl, hydroxy, Ci-i2-alkoxy, C2-i2-alkoxyalkyl, C2-i2-alkenyloxy, carboxy, Ci-i2-alkoxycarbonyl, Ci_i2- alkylcarbonyl, formyl, aryl, aryloxy-carbonyl, aryloxy, arylcarbonyl, heteroaryl, heteroaryloxy- carbonyl, heteroaryloxy, heteroarylcarbonyl, amino, mono- and di(Ci-6-alkyl)amino, carbamoyl, mono- and di(Ci-6-alkyl)-amino-carbonyl, amino-Ci-6-alkyl-aminocarbonyl, mono- and di(Ci-6-alkyl)amino-Ci-6-alkyl-aminocarbonyl, Ci-6-alkyl-carbonylamino, carbamido, Ci-6- alkanoyloxy, sulphono, Ci-6-alkylsulphonyloxy, nitro, azido, sulphanyl, Ci-6-alkylthio, halogen, DNA intercalators, photochemically active groups, thermochemically active groups, chelating
groups, reporter groups, and ligands, where aryl and heteroaryl may be optionally substituted and where two geminal substituents Ra and Rb together may designate optionally substituted methylene (=CH2); and RH is selected from hydrogen and Ci-4-alkyl. In some embodiments Ra, Rb Rc, Rd and Re are, optionally independently, selected from the group consisting of hydrogen and Ci-6 alkyl, such as methyl. For all chiral centers, asymmetric groups may be found in either R or S orientation, for example, two exemplary
stereochemical isomers include the beta-D and alpha-L isoforms, which may be illustrated as follows:
Specific exemplary LNA units are shown below:
β-D-oxy-LNA
β-D-thio-LNA
β-D-ENA
The term "thio-LNA" comprises a locked nucleotide in which Y in the general formula above is selected from S or -CH2-S-. Thio-LNA can be in both beta-D and alpha-L- configuration.
The term "amino-LNA" comprises a locked nucleotide in which Y in the general formula above is selected from -N(H)-, N(R)-, CH2-N(H)-, and -CH2-N(R)- where R is selected from hydrogen and Ci-4-alkyl. Amino-LNA can be in both beta-D and alpha-L- configuration.
The term "oxy-LNA" comprises a locked nucleotide in which Y in the general formula above represents -0-. Oxy-LNA can be in both beta-D and alpha-L-configuration.
The term "ENA" comprises a locked nucleotide in which Y in the general formula above is -CH2-0- (where the oxygen atom of -CH2-0- is attached to the 2'-position relative to the base B). Re is hydrogen or methyl.
In some exemplary embodiments LNA is selected from beta-D-oxy-LNA, alpha-L-oxy-LNA, beta-D-amino-LNA and beta-D-thio-LNA, in particular beta-D-oxy-LNA.
RNAse recruitment
It is recognised that an oligomeric compound may function via non RNase mediated degradation of target mRNA, such as by steric hindrance of translation, or other methods, In some embodiments, the oligomers of the invention are capable of recruiting an
endoribonuclease (RNase), such as RNase H.
In some embodiments, an oligomer is deemed essentially incapable of recruiting
RNaseH if, when provided with the complementary RNA target, and RNaseH, the RNaseH initial rate, as measured in pmol/l/min, is less than 1 %, such as less than 5%, such as less than 10% or less than 20% of the initial rate determined using the equivalent DNA only oligonucleotide, with no 2' substitutions, with phosphorothioate linkage groups between all nucleotides in the oligonucleotide, using the methodology provided by Example 91 - 95 of EP 1 222 309.
In other embodiments, an oligomer is deemed capable of recruiting RNaseH if, when provided with the complementary RNA target, and RNaseH, the RNaseH initial rate, as measured in pmol/l/min, is at least 20%, such as at least 40 %, such as at least 60 %, such as at least 80 % of the initial rate determined using the equivalent DNA only oligonucleotide, with no 2' substitutions, with phosphorothioate linkage groups between all nucleotides in the oligonucleotide, using the methodology provided by Example 91 - 95 of EP 1 222 309. Mixmers
Most antisense oligonucleotides are compounds which are designed to recruit RNase enzymes (such as RNaseH) to degrade their intended target. Such compounds include DNA phosphorothioate oligonucleotides and gapmer, headmers and tailmers. These compounds typically comprise a region of at least 5 or 6 DNA nucleotides, and in the case of gapmers are flanked on either side by affinity enhancing nucleotide analogues.
The oligomers of the present invention may operate via an RNase (such as RNaseH) independent mechanism. Examples of oligomers which operate via a non-RNaseH (or non- RNase) mechanism are mixmers and totalmers.
The term 'mixmer' refers to oligomers which comprise both naturally and non-naturally occurring nucleotides, where, as opposed to gapmers, tailmers, and headmers there is no contiguous sequence of more than 5, and in some embodiments no more than 4
consecutive, such as no more than three consecutive, naturally occurring nucleotides, such as DNA units. In some embodiments, the mixmer does not comprise more than 5 consecutive nucleoside analogues, such as BNA (LNA), and in some embodiments no more than 4 consecutive, such as no more than three consecutive, consecutive nucleoside analogues, such as BNA (LNA). In such mixmers the remaining nucleosides may, for example be DNA nucleosides, and/or in non-bicyclic nucleoside analogues, such as those referred to herein, for example, 2' substituted nucleoside analogues, such as 2'-0-MOE and or 2'fluoro.
The oligomer according to the invention maybe mixmers - indeed various mixmer designs are highly effective as oligomer or first region thereof, particularly when targeting microRNA (antimiRs), microRNA binding sites on mRNAs (Blockmirs) or as splice switching oligomers (SSOs). See for example WO2007/1 12754 (LNA-AntimiRs™), WO2008/131807 (LNA splice switching oligos),
In some embodiments, the oligomer or mixmer may comprise of BNA and 2' substituted nucleoside analogues, optionally with DNA nucleosides - see for example see WO07027894 and WO2007/1 12754 which are hereby incorporated by reference. Specific examples include oligomers or first regions which comprise LNA, 2'-0-MOE and DNA, LNA, 2'fluoro and 2 -O-MOE, 2'-0-MOE and 2'fluoro, 2'-0-MOE and 2'fluoro and LNA, or LNA and 2'-0-MOE and LNA and DNA.
In some embodiments, the oligomer or mixmer comprises or consists of a contiguous nucleotide sequence of repeating pattern of nucleotide analogue and naturally occurring nucleotides, or one type of nucleotide analogue and a second type of nucleotide analogues. The repeating pattern, may, for instance be every second or every third nucleotide is a
nucleotide analogue, such as BNA (LNA), and the remaining nucleotides are naturally occurring nucleotides, such as DNA, or are a 2'substituted nucleotide analogue such as 2'MOE of 2'fluoro analogues as referred to herein, or, in some embodiments selected form the groups of nucleotide analogues referred to herein. It is recognised that the repeating pattern of nucleotide analogues, such as LNA units, may be combined with nucleotide analogues at fixed positions - e.g. at the 5' or 3' termini.
In some embodiments the first nucleotide of oligomer or mixmer, counting from the 3' end, is a nucleotide analogue, such as an LNA nucleotide.
In some embodiments, which maybe the same or different, the second nucleotide of the oligomer or mixmer, counting from the 3' end, is a nucleotide analogue, such as an LNA nucleotide.
In some embodiments, which maybe the same or different, the seventh and/or eighth nucleotide of the oligomer or mixmer In some embodiments, which maybe the same or different, the ninth and/or the tenth nucleotides of the oligomer or mixmer, counting from the 3' end, are nucleotide analogues, such as LNA nucleotides.
In some embodiments, which maybe the same or different, the 5' terminal of olifgmer or mixmer is a nucleotide analogue, such as an LNA nucleotide.
The above design features may, in some embodiments be incorporated into the mixmer design, such as antimiR mixmers.
In some embodiments, the oligomer or mixmer does not comprise a region of more than 4 consecutive DNA nucleotide units or 3 consecutive DNA nucleotide units. In some embodiments, the mixmer does not comprise a region of more than 2 consecutive DNA nucleotide units.
In some embodiments, the oligomer or mixmer comprises at least a region consisting of at least two consecutive nucleotide analogue units, such as at least two consecutive LNA units.
In some embodiments, the oligomer or mixmer comprises at least a region consisting of at least three consecutive nucleotide analogue units, such as at least three consecutive LNA units.
In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 7 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 6 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 5 consecutive
nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 4 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 3 consecutive nucleotide analogue units, such as LNA units. In some embodiments, the oligomer or mixmer of the invention does not comprise a region of more than 2 consecutive nucleotide analogue units, such as LNA units. The following embodiments may apply to mixmers or totalmer oligomers (e.g. as region A): The oligomer (e.g. region A) of the invention may, in some embodiments, comprise of at least two alternating regions of LNA and non-LNA nucleotides (such as DNA or 2' substituted nucleotide analogues).
The oligomer of the invention may, in some embodiments, comprise a contiguous sequence of formula: 5' ([LNA nucleotides]i-5 and [non-LNA nucleotides]i-4)2 - i2- 3'.
In some embodiments, the 5' nucleotide of the contiguous nucleotide sequence (or the oligomer) is an LNA nucleotide.
In some embodiments, the 3' nucleotide of the contiguous nucleotide sequence is a nucleotide analogue, such as LNA, or the 2, 3, 4, 5 3' nucleotides are nucleotide analogues, such as LNA nucleotides, or other nucleotide analogues which confer enhanced serum stability to the oligomer.
In some embodiments, the contiguous nucleotide sequence of the oligomer has a formula 5' ([LNA nucleotides]i-5 - [non-LNA nucleotides]i-4)2 - n - [LNA nucleotides]i-5 3'.
In some embodiments, the contiguous nucleotide sequence of the oligomer has 2, 3 or 4 contiguous regions of LNA and non-LNA nucleotides - e.g. comprises formula 5' ([LNA nucleotides]i-5 and [non-LNA nucleotides]i-4)2 - 3, optionally with a further 3' LNA region [LNA nucleotides]i-5.
In some embodiments, the contiguous nucleotide sequence of the oligomer comprises 5' ([LNA nucleotides]i-3 and [non-LNA nucleotides]i-3)2 - 5, optionally with a further 3' LNA region [LNA nucleotides]i-3.
In some embodiments, the contiguous nucleotide sequence of the oligomer comprises 5' ([LNA nucleotides]i-3 and [non-LNA nucleotides]i-3)3, optionally with a further 3' LNA region [LNA nucleotides]i-3.
In some embodiments the non-LNA nucleotides are all DNA nucleotides.
In some embodiments, the non-LNA nucleotides are independently or dependency selected from the group consisting of DNA units, RNA units, 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino-DNA units, and 2'-fluoro-DNA units.
In some embodiments the non-LNA nucleotides are (optionally independently selected from the group consisting of 2' substituted nucleoside analogues, such as (optionally
independently) selected from the group consisting of 2'-0-alkyl-RNA units, 2'-OMe-RNA units, 2'-amino-DNA units, 2'-AP, 2'-FANA, 2'-(3-hydroxy)propyl, and 2'-fluoro-DNA units, and/or other (optionally) sugar modified nucleoside analogues such as morpholino, peptide nucleic acid (PNA), CeNA, unlinked nucleic acid (UNA), hexitol nucleoic acid (HNA). bicyclo- HNA (see e.g. WO2009/100320), In some embodiments, the nucleoside analogues increase the affinity of the first region for its target nucleic acid (or a complementary DNA or RNA sequence). Various nucleoside analogues are disclosed in Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, hereby incorporated by reference.
In some embodiments, the non-LNA nucleotides are DNA nucleotides. In some
embodiments, the oligomer or contiguous nucleotide sequence comprises of LNA nucleotides and optionally other nucleotide analogues (such as the nucleotide analogues listed under non-LNA nucleotides) which may be affinity enhancing nucleotide analogues and/or nucleotide analogues which enhance serum stability.
In some embodiments, the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of said nucleotide analogues.
In some embodiments, the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of LNA nucleotides.
In some embodiments, the oligomer or contiguous nucleotide sequence is 8 - 12, such as 8 - 10, or 10 - 20, such as 12 - 18 or 14 - 16 nts in length.
In some embodiments, the oligomer or contiguous nucleotide sequence is capable of forming a duplex with a complementary single stranded RNA nucleic acid molecule with phosphodiester internucleoside linkages, wherein the duplex has a Tm of at least about 60°C, such as at least 65°C.
Example of a Tm Assay: The oligonucleotide: Oligonucleotide and RNA target (PO) duplexes are diluted to 3 mM in 500 ml RNase-free water and mixed with 500 ml 2x Tm- buffer (200mM NaCI, 0.2mM EDTA, 20mM Naphosphate, pH 7.0). The solution is heated to 95°C for 3 min and then allowed to anneal in room temperature for 30 min. The duplex melting temperatures (Tm) is measured on a Lambda 40 UVA IS Spectrophotometer equipped with a Peltier temperature programmer PTP6 using PE Templab software (Perkin Elmer). The temperature is ramped up from 20°C to 95°C and then down to 25°C, recording
absorption at 260 nm. First derivative and the local maximums of both the melting and annealing are used to assess the duplex Tm.
Total mers
A totalmer is a single stranded oligomer which only comprises non-naturally occurring nucleosides, such as sugar-modified nucleoside analogues.
The first region according to the invention maybe totalmers - indeed various totalmer designs are highly effective as oligomers or first region thereofs, e.g. particularly when targeting microRNA (antimiRs). In some embodiments, the totalmer comprises or consists of at least one XYX or YXY sequence motif, such as a repeated sequence XYX or YXY, wherein X is LNA and Y is an alternative (i.e. non LNA) nucleotide analogue, such as a 2'-0- MOE RNA unit and 2'-fluoro DNA unit. The above sequence motif may, in some
embodiments, be XXY, XYX, YXY or YYX for example.
In some embodiments, the totalmer may comprise or consist of a contiguous nucleotide sequence of between 7 and 16 nucleotides, such as 9, 10, 1 1 , 12, 13, 14, or 15 nucleotides, such as between 7 and 12 nucleotides.
In some embodiments, the contiguous nucleotide sequence of the totolmer comprises of at least 30%, such as at least 40%, such as at least 50%, such as at least 60%, such as at least 70%, such as at least 80%, such as at least 90%, such as 95%, such as 100% BNA (LNA) units. The remaining units may be selected from the non-LNA nucleotide analogues referred to herein in, such those selected from the group consisting of 2'-0_alkyl-RNA unit, 2'-OMe-RNA unit, 2'-amino-DNA unit, 2'-fluoro-DNA unit, LNA unit, PNA unit, HNA unit, INA unit, and a 2'MOE RNA unit, or the group 2'-OMe RNA unit and 2'-fluoro DNA unit.
In some embodiments the totalmer consist or comprises of a contiguous nucleotide sequence which consists only of LNA units. In some embodiments, the totalmer, such as the LNA totalmer, is between 7 - 12 nucleoside units in length. In some embodiments, the totalmer (as the oligomer or first region thereof) may be targeted against a microRNA (i.e. be antimiRs) - as referred to WO2009/043353, which are hereby incorporated by reference. In some embodiments, the oligomer or contiguous nucleotide sequence comprises of LNA nucleotides and optionally other nucleotide analogues which may be affinity enhancing nucleotide analogues and/or nucleotide analogues which enhance serum stability.
In some embodiments, the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of said nucleotide analogues.
In some embodiments, the oligomer or contiguous nucleotide sequence thereof consists of a contiguous nucleotide sequence of LNA nucleotides.
MicroRNA modulation via the oligomer of the invention
In some embodiments, the oligomer or first region thereof is an oligomer, such as an LNA-antimiR®, which comprises or consists of a contiguous nucleotide sequence which is corresponds to or is fully complementary to a mature microRNA or part thereof. The oligomer may in some embodiments, target (i.e. comprises or consists of a contiguous nucleotide sequence which is fully complementary to (a corresponding region of) a microRNA. The microRNA may be a liver expressed microRNA, such as miR-122.
In some embodiments the oligomer according to the invention, consists or comprises of a contiguous nucleotide sequence which corresponds to or is fully complementary to hsa- miR122 (NR_029667.1 Gl:262205241 ), such as the mature hsa-miR-122. In some embodiments the oligomer according to the invention, consists or comprises of a contiguous nucleotide sequence which corresponds to or is fully complementary to hsa-miR122
(NR_029667.1 Gl:262205241 ), such as the mature hsa-miR-122 across the length of the oligomer.
In some embodiments when the oligomer or first region thereof targets miR-122, the oligomer is for the use in the treatment of hepatitis C infection.
AntimiR oligomers
Preferred oligomer or first region thereof 'antimiR' designs and oligomers are disclosed in WO2007/1 12754, WO2007/1 12753, PCT/DK2008/000344 and US provisional applications 60/979217 and 61/028062, all of which are hereby incorporated by reference. In some embodiments, the oligomer or first region thereof is an antimiR which is a mixmer or a totalmer. The term AntimiR may therefore be replaces with the term oligomer.
AntimiR oligomers are oligomers which consist or comprise of a contiguous nucleotide sequence which is fully complementary to, or essentially complementary to (i.e. may comprise one or two mismatches), to a microRNA sequence, or a corresponding subsequence thereof. In this regards it is considered that the antimiR may be comprise a contiguous nucleotide sequence which is complementary or essentially complementary to the entire mature microRNA, or the antimiR may be comprise a contiguous nucleotide sequence which is complementary or essentially complementary to a sub-sequence of the mature microRNA or pre-microRNA - such a sub-sequence (and therefore the
corresponding contiguous nucleotide sequence) is typically at least 8 nucleotides in length, such as between 8 and 25 nucleotides, such as 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24 nucleotides in length, such as between 10-17 or 10-16 nucleotides, such as between 12 - 15 nucleotides.
Numerous designs of AnitmiRs have been suggested, and typically antimiRs for therapeutic use, such as the contiguous nucleotide sequence thereof comprise one or more nucleotide analogues units.
In some embodiments the antimiR may have a gapmer structure as herein described. However, as explained in WO2007/1 12754 and WO2007/1 12753, other designs may be preferable, such as mixmers, or totalmers.
WO2007/1 12754 and WO2007/1 12753, both hereby incorporated by reference, provide antimiR oligomers and antimiR oligomer designs where the oligomers which are complementary to mature microRNA
In some embodiments, a subsequence of the antimiR corresponds to the miRNA seed region. In some embodiments, the first or second 3' nucleobase of the oligomer corresponds to the second 5' nucleotide of the microRNA sequence.
In some antimiR embodiments, nucleobase units 1 to 6 (inclusive) of the oligomer as measured from the 3' end the region of the oligomer are complementary to the microRNA seed region sequence.
In some antimiR embodiments, nucleobase units 1 to 7 (inclusive) of the oligomer as measured from the 3' end the region of the oligomer are complementary to the microRNA seed region sequence.
In some e antimiR embodiments, nucleobase units 2 to 7 (inclusive) of the oligomer as measured from the 3' end the region of the oligomer are complementary to the microRNA seed region sequence.
In some embodiments, the antimiR oligomer comprises at least one nucleotide analogue unit, such as at least one LNA unit, in a position which is within the region complementary to the miRNA seed region. The antimiR oligomer may, in some
embodiments comprise at between one and 6 or between 1 and 7 nucleotide analogue units, such as between 1 and 6 and 1 and 7 LNA units, in a position which is within the region complementary to the miRNA seed region.
In some embodiments, the antimiR of the invention is 7, 8 or 9 nucleotides long, and comprises a contiguous nucleotide sequence which is complementary to a seed region of a human or viral microRNA, and wherein at least 80 %, such as 85%, such as 90%, such as 95%, such as 100% of the nucleotides are LNA.
In some embodiments, the antimiR of the invention is 7, 8 or 9 nucleotides long, and comprises a contiguous nucleotide sequence which is complementary to a seed region of a human or viral microRNA, and wherein at least 80 % of the nucleotides are LNA, and
wherein at least 80%, such as 85%, such as 90%, such as 95%, such as 100% of the internucleotide bonds are phosphorothioate bonds.
In some embodiments, the antimiR comprises one or two LNA units in positions three to eight, counting from the 3' end. This is considered advantageous for the stability of the A- helix formed by the oligo:microRNA duplex, a duplex resembling an RNA:RNA duplex in structure.
The table on pages 48 line 15 to page 51 , line 9 of WO2007/1 12754 provides examples of anti microRNA oligomers (i.e. antimiRs which may be the oligomer or first region thereof) and is hereby specifically incorporated by reference.
Internucleotide Linkages
The nucleoside monomers of the oligomers (e.g. first and second regions) described herein are coupled together via [internucleoside] linkage groups. Suitably, each monomer is linked to the 3' adjacent monomer via a linkage group.
The person having ordinary skill in the art would understand that, in the context of the present invention, the 5' monomer at the end of an oligomer does not comprise a 5' linkage group, although it may or may not comprise a 5' terminal group.
The terms "linkage group" or "internucleotide linkage" are intended to mean a group capable of covalently coupling together two nucleotides. Specific and preferred examples include phosphate groups and phosphorothioate groups.
The nucleotides of the oligomer of the invention or contiguous nucleotides sequence thereof are coupled together via linkage groups. Suitably each nucleotide is linked to the 3' adjacent nucleotide via a linkage group.
Suitable internucleotide linkages include those listed within WO2007/031091 , for example the internucleotide linkages listed on the first paragraph of page 34 of
WO2007/031091 (hereby incorporated by reference).
It is, in some embodiments, other than the phosphodiester linkage(s) or region B, the preferred to modify the internucleotide linkage from its normal phosphodiester to one that is more resistant to nuclease attack, such as phosphorothioate or boranophosphate - these two, being cleavable by RNase H, also allow that route of antisense inhibition in reducing the expression of the target gene.
Suitable sulphur (S) containing internucleotide linkages as provided herein may be preferred, such as phosphorothioate or phosphodithioate. Phosphorothioate internucleotide linkages are also preferred, particularly for the first region, such as in gapmers, mixmers, antimirs splice switching oligomers, and totalmers.
For gapmers, the internucleotide linkages in the oligomer may, for example be phosphorothioate or boranophosphate so as to allow RNase H cleavage of targeted RNA. Phosphorothioate is preferred, for improved nuclease resistance and other reasons, such as ease of manufacture.
In one aspect, with the exception of the phosphodiester linkage between the first and second region, and optionally within region B, the remaining internucleoside linkages of the oligomer of the invention, the nucleotides and/or nucleotide analogues are linked to each other by means of phosphorothioate groups. In some embodiments, at least 50%, such as at least 70%, such as at least 80%, such as at least 90% such as all the internucleoside linkages between nucleosides in the first region are other than phosphodiester (phosphate), such as are selected from the group consisting of phosphorothioate phosphorodithioate, or boranophosphate. In some embodiments, at least 50%, such as at least 70%, such as at least 80%, such as at least 90% such as all the internucleoside linkages between
nucleosides in the first region are phosphorothioate.
WO09124238 refers to oligomeric compounds having at least one bicyclic nucleoside attached to the 3' or 5' termini by a neutral internucleoside linkage. The oligomers of the invention may therefore have at least one bicyclic nucleoside attached to the 3' or 5' termini by a neutral internucleoside linkage, such as one or more phosphotriester,
methylphosphonate, MMI, amide-3, formacetal or thioformacetal. The remaining linkages may be phosphorothioate.
Conjugates, targeting moieties and blocking groups
The term "conjugate" is intended to indicate a heterogenous molecule formed by the covalent attachment ("conjugation") of the oligomer as described herein has one or more non-nucleotide, or non-polynucleotide moieties conjugated thereto.
Carbohydrate conjugates
In some embodiments, the conjugate group is a carbohydrate moiety.
In some embodiments, the conjugate is or may comprise a carbohydrate or comprises a carbohydrate group. In some embodiments, the carbohydrate is selected from the group consisting of galactose, lactose, n-acetylgalactosamine, mannose, and mannose-6- phosphate. In some embodiments, the conjugate group is or may comprise mannose or mannose-6-phosphate. Carbohydrate conjugates may be used to enhance delivery or activity in a range of tissues, such as liver and/or muscle. See, for example, EP1495769,
W099/65925, Yang et al., Bioconjug Chem (2009) 20(2): 213-21. Zatsepin & Oretskaya Chem Biodivers. (2004) 1 (10): 1401 -17.
In addition, the oligomer may further comprise one or more additional conjugate moieties, of which lipophilic or hydrophobic moieties are particularly interesting. These may for example, act as pharmacokinetic modulators, and may be covalently linked to either the carbohydrate conjugate, a linker linking the carbohydrate conjugate to the oligomer or a linker linking multiple carbohydrate conjugates (multi-valent) conjugates, or to the oligomer, optionally via a linker, such as a bio cleavable linker.
Pharmacokinetic Modulators
The compound of the invention may further comprise one or more additional conjugate moieties, of which lipophilic or hydrophobic moieties are particularly interesting, such as when the conjugate group is a carbohydrate moiety. Such lipophilic or hydrophobic moieties may act as pharmacokinetic modulators, and may be covalently linked to either the carbohydrate conjugate, a linker linking the carbohydrate conjugate to the oligomer or a linker linking multiple carbohydrate conjugates (multi-valent) conjugates, or to the oligomer, optionally via a linker, such as a bio cleavable linker.
The oligomer or conjugate moiety may therefore comprise a pharmacokinetic modulator, such as a lipophilic or hydrophobic moieties. Such moieties are disclosed within the context of siRNA conjugates in WO2012/082046. The hydrophobic moiety may comprise a C8 - C36 fatty acid, which may be saturated or un-saturated. In some embodiments, C10, C12, C14, C16, C18, C20, C22, C24, C26, C28, C30, C32 and C34 fatty acids may be used. The hydrophobic group may have 16 or more carbon atoms. Exemplary suitable hydrophobic groups may be selected from the group comprising:sterol, cholesterol, palmitoyl, hexadec-8-enoyl, oleyl, (9E, 12E)-octadeca-9,12-dienoyl, dioctanoyl, and C16- C20 acyl. According to WO'346, hydrophobic groups having fewer than 16 carbon atoms are less effective in enhancing polynucleotide targeting, but they may be used in multiple copies (e.g. 2x, such as 2x C8 or C10, C12 or C14) to enhance efficacy. Pharmacokinetic modulators useful as polynucleotide targeting moieties may be selected from the group consisting of: cholesterol, alkyl group, alkenyl group, alkynyl group, aryl group, aralkyi group, aralkenyl group, and aralkynyl group, each of which may be linear, branched, or cyclic. Pharmacokinetic modulators are preferably hydrocarbons, containing only carbon and hydrogen atoms. However, substitutions or heteroatoms which maintain hydrophobicity, for example fluorine, may be permitted.
Surprisingly, the present inventors have found that GalNac conjugates for use with LNA oligomers do not require a pharmacokinetic modulator, and as such, in some embodiments, the GalNac conjugate is not covalently linked to a lipophilic or hydrophobic moiety, such as those described here in, e.g. do not comprise a C8 - C36 fatty acid or a sterol. The invention therefore also provides for LNA oligomer GalNac conjugates which do not comprise a lipophilic or hydrophobic pharmacokinetic modulator or conjugate moiety/group.
GalNAc Conjugates
The invention provides LNA antisense oligonucleotides which are conjugated to an asialoglycoprotein receptor targeting moiety. In some embodiments, the conjugate moiety (such as the third region or region C) comprises an asialoglycoprotein receptor targeting moiety, such as galactose, galactosamine, N-formyl-galactosamine, Nacetylgalactosamine, N-propionyl-galactosamine, N-n-butanoyl-galactosamine, and N-isobutanoylgalactos-amine. In some embodiments the conjugate comprises a galactose cluster, such as N- acetylgalactosamine trimer. In some embodiments, the conjugate moiety comprises an GalNAc (N-acetylgalactosamine), such as a mono-valent, di-valent , tri-valent of tetra-valent GalNAc. Trivalent GalNAc conjugates may be used to target the compound to the liver. GalNAc conjugates have been used with methylphosphonate and PNA antisense oligonucleotides (e.g. US 5,994517 and Hangeland et al., Bioconjug Chem. 1995 Nov- Dec;6(6):695-701 ) and siRNAs (e.g. WO2009/126933, WO2012/089352 &
WO2012/083046). The GalNAc references and the specific conjugates used therein are hereby incorporated by reference. Other GalNAc conjugate moieties can include, for example, oligosaccharides and carbohydrate clusters such as Tyr-Glu-Glu-(aminohexyl GalNAc)3 (YEE(ahGalNAc)3; a glycotripeptide that binds to Gal/Gal N Ac receptors on hepatocytes, see, e.g., Duff, et al., Methods Enzymol, 2000, 313, 297); lysine-based galactose clusters (e.g., L3G4; Biessen, et al., Cardovasc. Med., 1999, 214); and cholane- based galactose clusters (e.g., carbohydrate recognition motif for asialoglycoprotein receptor). Further suitable conjugates can include oligosaccharides that can bind to carbohydrate recognition domains (CRD) found on the asiologlycoprotein-receptor (ASGP- R). Example conjugate moieties containing oligosaccharides and/or carbohydrate complexes are provided in U.S. Pat. No. 6,525,031 , which is incorporated herein by reference in its entirity.
WO2012/083046 discloses siRNAs withGalNAc conjugate moieties which comprise cleavable pharmacokinetic modulators, which are suitable for use in the present invention, the preferred pharmacokinetic modulators are C16 hydrophobic groups such as palmitoyl,
hexadec-8-enoyl, oleyl, (9E, 12E)-octadeca-9,12-dienoyl, dioctanoyi, and C16-C20 acyl. The Ό46 cleavable pharmacokinetic modulators may also be cholesterol.
The 'targeting moieties (conjugate moieties) may be selected from the group consisting of: galactose, galactosamine, N-formyl-galactosamine, N-acetylgalactosamine, Npropionyl- galactosamine, N-n-butanoyl-galactosamine, N-iso-butanoylgalactos-amine, galactose cluster, and N-acetylgalactosamine trimer and may have a pharmacokinetic modulator selected from the group consisting of: hydrophobic group having 16 or more carbon atoms, hydrophobic group having 16-20 carbon atoms, palmitoyl, hexadec-8-enoyl, oleyl, (9E,12E)-octadeca-9,12dienoyl, dioctanoyi, and C16-C20 acyl, and cholesterol.
Certain GalNac clusters disclosed in Ό46 include: (E)-hexadec-8-enoyl (C16), oleyl (C18), (9,E,12E)-octadeca-9,12-dienoyl (C18), octanoyl (C8), dodececanoyl (C12), C-20 acyl, C24 acyl, dioctanoyi (2xC8). The targeting moiety-pharmacokinetic modulator targeting moiety may be linked to the polynucleotide via a physiologically labile bond or, e.g. a disulfide bond, or a PEG linker. The invention also relates to the use of phospodiester linkers, such as DNA phosphodiester linkers, between the oligomer and the conjugate group (these are referred to as region B herein, and suitably are positioned between the LNA oligomer and the carbohydrate conjugate group).
For targeting hepatocytes in liver, a preferred targeting ligand is a galactose cluster. A galactose cluster comprises a molecule having e.g. comprising two to four terminal galactose derivatives. As used herein, the term galactose derivative includes both galactose and derivatives of galactose having affinity for the asialoglycoprotein receptor equal to or greater than that of galactose. A terminal galactose derivative is attached to a molecule through its C-l carbon. The asialoglycoprotein receptor (ASGPr) is unique to hepatocytes and binds branched galactose-terminal glycoproteins. A preferred galactose cluster has three terminal galactosamines or galactosamine derivatives each having affinity for the asialoglycoprotein receptor. A more preferred galactose cluster has three terminal N-acetyl- galactosamines. Other terms common in the art include tri-antennary galactose, tri-valent galactose and galactose trimer. It is known that tri-antennary galactose derivative clusters are bound to the ASGPr with greater affinity than bi-antennary or mono-antennary galactose derivative structures (Baenziger and Fiete, 1980, Cell, 22, 61 1 -620; Connolly et al., 1982,1. Biol. Chern., 257,939-945). Multivalency is required to achieve nM affinity. According to WO 2012/083046 the attachment of a single galactose derivative having affinity for the asialoglycoprotein receptor does not enable functional delivery of the RNAi polynucleotide to hepatocytes in vivo when co-administered with the delivery polymer.
A galactose cluster may comprise two or preferably three galactose derivatives each linked to a central branch point. The galactose derivatives are attached to the central branch point through the C-l carbons of the saccharides. The galactose derivative is preferably linked to the branch point via linkers or spacers. A preferred spacer is a flexible hydrophilic spacer (U.S. Patent 5885968; Biessen et al. J. Med. Chern. 1995 Vol. 39 p. 1538-1546). A preferred flexible hydrophilic spacer is a PEG spacer. A preferred PEG spacer is a PEG3 spacer. The branch point can be any small molecule which permits attachment of the three galactose derivatives and further permits attachment of the branch point to the oligomer. An exemplary branch point group is a di-lysine. A di-lysine molecule contains three amine groups through which three galactose derivatives may be attached and a carboxyl reactive group through which the di-lysine may be attached to the oligomer. Attachment of the branch point to oligomer may occur through a linker or spacer. A preferred spacer is a flexible hydrophilic spacer. A preferred flexible hydrophilic spacer is a PEG spacer. A preferred PEG spacer is a PEG3 spacer (three ethylene units). The galactose cluster may be attached to the 3' or 5' end of the oligomer using methods known in the art.
A preferred galactose derivative is an N-acetyl-galactosamine (GalNAc). Other saccharides having affinity for the asialoglycoprotein receptor may be selected from the list comprising: galactosamine, N-n-butanoylgalactosamine, and N-iso-butanoylgalactosamine. The affinities of numerous galactose derivatives for the asialoglycoprotein receptor have been studied (see for example: Jobst, ST. and Drickamer, K. JB.C. 1996,271 ,6686) or are readily determined using methods typical in the art.
Galactose cluster with PEG spacer between branch point and nucleic acid
A GalNac conjugate is illustrated in figure 1 . Further examples of the conjugate of the invention are illustrated below:
The LNA antisense oligomer may, in some emboidments, comprise cET nucleosides, such as (S)-cET nucleosides.
The carbohydrate conjugate (e.g. GalNAc) may therefore be linked to the oligomer via a linker, such as (poly)ethylene glycol linker (PEG), such as a di, tri, tetra, penta, hexa- ethylene glycol linker.
As described herein, a carbohydrate conjugate (e.g. GalNAc) may therefore be linked to the oligomer via a biocleavable linker, such as region B as defined herein, and optionally region Y, which is illustrated as a di-lysine in the above diagrams.
Where at the hydrophobic or lipophilic (or further conjugate) moiety (i.e. pharmacokinetic modulator) in the above GalNac cluster conjugates is, when using BNA or LNA oligomers, such as LNA antisense oligonucleotides, optional.
See the figures for specific Galnac clusters used in the present study, Conj 1 , 2, 3, 4 and Conjl a, 2a, 3a and 4a (which are shown with an optional C6 linker which joins the GalNac cluster to the oligomer).
Each carbohydrate moiety of a GalNAc cluster (e.g. GalNAc) may therefore be joined to the oligomer via a spacer, such as (poly)ethylene glycol linker (PEG), such as a di, tri, tetra, penta, hexa-ethylene glycol linker. As is shown above the PEG moiety forms a spacer between the galactose sugar moiety and a peptide (trilysine is shown) linker.
The carbohydrate conjugate(e.g. GalNAc), or carbohydrate-linker moiety (e.g. carbohydrate- PEG moiety) may be covalently joined (linked) to the oligomer via a branch point group such as, an amino acid, or peptide, which suitably comprises two or more amino groups (such as 3, 4, or5), such as lysine, di-lysine or tri-lysine or tetra-lysine. A tri-lysine molecule contains four amine groups through which three carbohydrate conjugate groups, such as galactose & derivatives (e.g. GalNAc) and a further conjugate such as a hydrophobic or lipophilic moiety/group may be attached and a carboxyl reactive group through which the tri-lysine may be attached to the oligomer. The further conjugate, such as lipophilic/hyrodphobic moiety may be attached to the lysine residue that is attached to the oligomer.
In some embodiments, the GalNac cluster comprises a peptide linker, e.g. a Tyr-Asp(Asp) tripeptide or Asp(Asp) dipeptide, which is attached to the oligomer (or to region Y or region B) via a biradical linker, for example the GalNac cluster may comprise the following biradical linkers:
R1 is a biradical preferably selected from -C2H4-, -C3H6-, -C4H8-, -C5H10-, -C6H12-, 1 ,4- cyclohexyl (-C6H 10-), 1 ,4-phenyl (-C6H4-), -C2H4OC2H4-, -C2H4(OC2H4)2- or -C2H4(OC2H4)3-, C(0)CH2-, -C(0)C2H4-, - C(0)C3H6-, - C(0)C4H8-, - C(O)C5H10-, - C(0)C6H12-, 1 ,4-cyclohexyl (-C(0)C6H 10-), 1 ,4-phenyl (-C(0)C6H4-), - C(0)C2H4OC2H4-, - C(0)C2H4(OC2H4)2- or - C(0)C2H4(OC2H4)3- . In some embodiments, R1 is a biradical preferably selected from - C2H4-, -C3H6-, -C4H8-, -C5H10-, -C6H12-, 1 ,4-cyclohexyl (-C6H 10-), 1 ,4-phenyl (-C6H4-), - C2H4OC2H4-, -C2H4(OC2H4)2- or -C2H4(OC2H4)3- .
Amino alkyl Intermediates
The invention further provides for the LNA oligomer intermediates which comprise an antisense LNA oligomer which comprises an (e.g. terminal, 5' or 3') amino alkyl, such as a C2 - C36 amino alkyl group, including, for example C6 and C12 amino alkyl groups. The amino alkyl group may be added to the LNA oligomer as part of standard oligonucleotide synthesis, for example using a (e.g. protected) amino alkyl phosphoramidite. The linkage group between the amino alkyl and the LNA oligomer may for example be a
phosphorothioate or a phosphodiester, or one of the other nucleoside linkage groups referred to herein, for example. The amino alkyl group may be covalently linked to, for example, the 5' or 3' of the LNA oligomer, such as by the nucleoside linkage group, such as phosphorothioate or phosphodiester linkage.
The invention also provides a method of synthesis of the LNA oligomer comprising the sequential synthesis of the LNA oligomer, such as solid phase oligonucleotide synthesis, comprising the step of adding an amino alkyl group to the oligomer, such as e.g. during the first or last round of oligonucleotide synthesis. The method of synthesis my further comprise the step of reacting the carbohydrate conjugate to the amino alkyl -LNA oligomer (the conjugation step). The carbohydrate conjugate may comprise suitable linkers and/or branch point groups, and optionally further conjugate groups, such as hydrophobic or lipophilic
groups, as described herein. The conjugation step may be performed whilst the oligomer is bound to the solid support (e.g. after oligonucleotide synthesis, but prior to elution of the oligomer from the solid support), or subsequently (i.e. after elution). The invention provides for the use of an amino alkyl linker in the synthesis of the oligomer of the invention.
The invention provides for a method of synthesizing (or manufacture) of an oligomeric compound, such as the oligomeric compound of the invention, said method comprising a step of [sequential] oligonucleotide synthesis of a first region (A) and (optionally, when present), a second region (B), wherein the synthesis step is followed by a step of adding a third region [phosphoramidite comprising] comprising a carbohydrate conjugate group as described herein (X or with a linker X-Y), such as for example a GalNAc conjugate, such as a trivalent GalNAc (e.g. a conjugate selected from the group consisting of Conjl , Conj2, Conj3, Conj 4, Conjl a, Conj2a, Conj3a, and Conj 4a, or other trivalent GalNAc conjugate moieties, such as those disclosed herein. Followed by the cleavage of the oligomeric compound from the [solid phase] support.
It is however recognized that the conjuigate region (e.gX or X-Y) may be added after the cleavage from the solid support. Alternatively, the method of synthesis may comprise the steps of synthesizing a first (A), and optionally second region (B), followed by the cleavage of the oligomer from the support, with a subsequent step of adding the third region, such as X or X-Y group to the oligomer. The addition of the third region may be achieved, by example, by adding an amino phosphoramidite unit in the final step of oligomer synthesis (on the support), which can, after cleavage from the support, be used to join to the X or X-Y group, optionally via an activation group on the X or Y (when present) group. In the embodiments where the cleavable linker is not a nucleotide region, region B may be a non- nucleotide cleavable linker for example a peptide linker, which may form part of region X (also referred to as region C) or be region Y (or part thereof).
In some embodiments of the method, region X (such as C) or (X-Y), such as the conjugate (e.g. a GalNAc conjugate) comprises an activation group, (an activated functional group) and in the method of synthesis the activated conjugate (or region x, or X-Y) is added to the first and second regions, such as an amino linked oligomer. The amino group may be added to the oligomer by standard phosphoramidite chemistry, for example as the final step of oligomer synthesis (which typically will result in amino group at the 5' end of the oligomer). For example during the last step of the oligonucleotide synthesis a protected amino-alkyl phosphoramidite is used, for example a TFA-aminoC6 phosphoramidite (6- (Trifluoroacetylamino)-hexyl-(2-cyanoethyl)-(N,N-diisopropyl)-phosphoramidite).
Region X (or region C as referred to herein), such as the conjugate (e.g. a GalNac conjugate) may be activated via NHS ester method and then the aminolinked oligomer is added. For example a N-hydroxysuccinimide (NHS) may be used as activating group for region X (or region C, such as a conjugate, such as a GalNac conjugate moiety.
In some embodiments, the conjugate moiety is hydrophilic. In some embodiments, the conjugate group does not comprise a lipophilic substituent group, such as a fatty acid substituent group, such as a C8 - C26, such as a palmotyl substituent group, or does not comprise a sterol, e.g. a cholesterol subtituent group. In this regards, part of the invention is based on the suprising discovery that LNA oligomers GalNAC conjugates have remarkable pharmacokinetic properties even without the use of pharmacokinetic modulators, such as fatty acid substituent groups (e.g. >C8 or >C16 fatty acid groups).
Linkers
A linkage or linker is a connection between two atoms that links one chemical group or segment of interest to another chemical group or segment of interest via one or more covalent bonds. Conjugate moieties (or targeting or blocking moieties) can be attached to the oligomeric compound directly or through a linking moiety (linker or tether) - a linker. Linkers are bifunctional moieties that serve to covalently connect a third region, e.g. a conjugate moiety, to an oligomeric compound (such as to region B). In some embodiments, the linker comprises a chain structure or an oligomer of repeating units such as ethylene glyol or amino acid units. The linker can have at least two functionalities, one for attaching to the oligomeric compound and the other for attaching to the conjugate moiety. Example linker functionalities can be electrophilic for reacting with nucleophilic groups on the oligomer or conjugate moiety, or nucleophilic for reacting with electrophilic groups. In some
embodiments, linker functionalities include amino, hydroxyl, carboxylic acid, thiol, phosphoramidate, phophate, phosphite, unsaturations (e.g., double or triple bonds), and the like. Some example linkers include 8-amino-3,6-dioxaoctanoic acid (ADO), succinimidyl 4- (N-maleimidomethyl)cyclohexane-l-carboxylate (SMCC), 6- aminohexanoic acid (AHEX or AHA), 6-aminohexyloxy, 4-aminobutyric acid, 4- aminocyclohexylcarboxylic acid, succinimidyl 4-(N-maleimidomethyl)cyclohexane- l-carboxy-(6-amido-caproate) (LCSMCC), succinimidyl m-maleimido-benzoylate (MBS), succinimidyl N-e-maleimido-caproylate
(EMCS), succinimidyl 6-(beta - maleimido-propionamido) hexanoate (SMPH), succinimidyl N-(a-maleimido acetate) (AMAS), succinimidyl 4-(p-maleimidophenyl)butyrate (SMPB), beta -alanine (beta -ALA), phenylglycine (PHG), 4-aminocyclohexanoic acid (ACHC), beta -
(cyclopropyl) alanine (beta -CYPR), amino dodecanoic acid (ADC), alylene diols,
polyethylene glycols, amino acids, and the like.
A wide variety of further linker groups are known in the art that can be useful in the attachment of conjugate moieties to oligomeric compounds. A review of many of the useful linker groups can be found in, for example, Antisense Research and Applications, S. T. Crooke and B. Lebleu, Eds., CRC Press, Boca Raton, Fla., 1993, p. 303-350. A disulfide linkage has been used to link the 3' terminus of an oligonucleotide to a peptide (Corey, et al., Science 1987, 238, 1401 ; Zuckermann, et al, J Am. Chem. Soc. 1988, 1 10, 1614; and Corey, et al., J Am. Chem. Soc. 1989, 1 1 1 , 8524). Nelson, et al., Nuc. Acids Res. 1989, 17, 7187 describe a linking reagent for attaching biotin to the 3'-terminus of an oligonucleotide. This reagent, N-Fmoc-O- DMT-3 -amino- 1 ,2-propanediol is commercially available from Clontech Laboratories (Palo Alto, Calif.) under the name 3'-Amine. It is also commercially available under the name 3'-Amino-Modifier reagent from Glen Research Corporation (Sterling, Va.). This reagent was also utilized to link a peptide to an oligonucleotide as reported by Judy, et al., Tetrahedron Letters 1991 , 32, 879. A similar commercial reagent for linking to the 5 '-terminus of an oligonucleotide is 5'- Amino-Modifier C6. These reagents are available from Glen Research Corporation (Sterling, Va.). These compounds or similar ones were utilized by Krieg, et al, Antisense Research and Development 1991 , 1 , 161 to link fluorescein to the 5'- terminus of an oligonucleotide. Other compounds such as acridine have been attached to the 3 '-terminal phosphate group of an oligonucleotide via a polymethylene linkage (Asseline, et al., Proc. Natl. Acad. Sci. USA 1984, 81 , 3297). [0074] Any of the above groups can be used as a single linker or in combination with one or more further linkers.
Linkers and their use in preparation of conjugates of oligomeric compounds are provided throughout the art such as in WO 96/1 1205 and WO 98/52614 and U.S. Pat. Nos. 4,948,882; 5,525,465; 5,541 ,313; 5,545,730; 5,552,538; 5,580,731 ; 5,486,603; 5,608,046; 4,587,044; 4,667,025; 5,254,469; 5,245,022; 5,1 12,963; 5,391 ,723; 5,510475; 5,512,667; 5,574,142; 5,684,142; 5,770,716; 6,096,875; 6,335,432; and 6,335,437,Wo2012/083046 each of which is incorporated by reference in its entirety.
As used herein, a physiologically labile bond is a labile bond that is cleavable under conditions normally encountered or analogous to those encountered within a mammalian body (also referred to as a cleavable linker). Physiologically labile linkage groups are selected such that they undergo a chemical transformation (e.g., cleavage) when present in
certain physiological conditions. Mammalian intracellular conditions include chemical conditions such as pH, temperature, oxidative or reductive conditions or agents, and salt concentration found in or analogous to those encountered in mammalian cells. Mammalian intracellular conditions also include the presence of enzymatic activity normally present in a mammalian cell such as from proteolytic or hydrolytic enzymes. In some embodiments, the cleavable linker is susceptible to nuclease(s) which may for example, be expressed in the target cell - and as such, as detailed herein, the linker may be a short region (e.g. 1 - 10) phosphodiester linked nucleosides, such as DNA nucleosides,
Chemical transformation (cleavage of the labile bond) may be initiated by the addition of a pharmaceutically acceptable agent to the cell or may occur spontaneously when a molecule containing the labile bond reaches an appropriate intra-and/or extra-cellular environment. For example, a pH labile bond may be cleaved when the molecule enters an acidified endosome. Thus, a pH labile bond may be considered to be an endosomal cleavable bond. Enzyme cleavable bonds may be cleaved when exposed to enzymes such as those present in an endosome or lysosome or in the cytoplasm. A disulfide bond may be cleaved when the molecule enters the more reducing environment of the cell cytoplasm. Thus, a disulfide may be considered to be a cytoplasmic cleavable bond. As used herein, a pH-labile bond is a labile bond that is selectively broken under acidic conditions (pH<7). Such bonds may also be termed endosomally labile bonds, since cell endosomes and lysosomes have a pH less than 7.
Linkers (e.g. Region Y)
A linkage or linker is a connection between two atoms that links one chemical group or segment of interest to another chemical group or segment of interest via one or more covalent bonds. Conjugate moieties (or targeting or blocking moieties) can be attached to the oligomeric compound directly or through a linking moiety (linker or tether) - a linker. Linkers are bifunctional moieties that serve to covalently connect a third region, e.g. a conjugate moiety, to an oligomeric compound (such as to region B). In some embodiments, the linker comprises a chain structure or an oligomer of repeating units such as ethylene glyol or amino acid units. The linker can have at least two functionalities, one for attaching to the oligomeric compound and the other for attaching to the conjugate moiety. Example linker functionalities can be electrophilic for reacting with nucleophilic groups on the oligomer or conjugate moiety, or nucleophilic for reacting with electrophilic groups. In some
embodiments, linker functionalities include amino, hydroxyl, carboxylic acid, thiol,
phosphoramidate, phosphorothioate, phosphate, phosphite, unsaturations (e.g., double or triple bonds), and the like. Some example linkers include 8-amino-3,6-dioxaoctanoic acid (ADO), succinimidyl 4-(N-maleimidomethyl)cyclohexane-l-carboxylate (SMCC), 6- aminohexanoic acid (AHEX or AHA), 6-aminohexyloxy, 4-aminobutyric acid, 4- aminocyclohexylcarboxylic acid, succinimidyl 4-(N-maleimidomethyl)cyclohexane- l-carboxy- (6-amido-caproate) (LCSMCC), succinimidyl m-maleimido-benzoylate (MBS), succinimidyl N-e-maleimido-caproylate (EMCS), succinimidyl 6-(beta - maleimido-propionamido) hexanoate (SMPH), succinimidyl N-(a-maleimido acetate) (AMAS), succinimidyl 4-(p- maleimidophenyl)butyrate (SMPB), beta -alanine (beta -ALA), phenylglycine (PHG), 4- aminocyclohexanoic acid (ACHC), beta -(cyclopropyl) alanine (beta -CYPR), amino dodecanoic acid (ADC), alylene diols, polyethylene glycols, amino acids, and the like.
A wide variety of further linker groups are known in the art that can be useful in the attachment of conjugate moieties to oligomeric compounds. A review of many of the useful linker groups can be found in, for example, Antisense Research and Applications, S. T. Crooke and B. Lebleu, Eds., CRC Press, Boca Raton, Fla., 1993, p. 303-350. A disulfide linkage has been used to link the 3' terminus of an oligonucleotide to a peptide (Corey, et al., Science 1987, 238, 1401 ; Zuckermann, et al, J Am. Chem. Soc. 1988, 1 10, 1614; and Corey, et al., J Am. Chem. Soc. 1989, 1 1 1 , 8524). Nelson, et al., Nuc. Acids Res. 1989, 17, 7187 describe a linking reagent for attaching biotin to the 3'-terminus of an oligonucleotide. This reagent, N-Fmoc-O- DMT-3 -amino- 1 ,2-propanediol is commercially available from Clontech Laboratories (Palo Alto, Calif.) under the name 3'-Amine. It is also commercially available under the name 3'-Amino-Modifier reagent from Glen Research Corporation (Sterling, Va.). This reagent was also utilized to link a peptide to an oligonucleotide as reported by Judy, et al., Tetrahedron Letters 1991 , 32, 879. A similar commercial reagent for linking to the 5 '-terminus of an oligonucleotide is 5'- Amino-Modifier C6. These reagents are available from Glen Research Corporation (Sterling, Va.). These compounds or similar ones were utilized by Krieg, et al, Antisense Research and Development 1991 , 1 , 161 to link fluorescein to the 5'- terminus of an oligonucleotide. Other compounds such as acridine have been attached to the 3 '-terminal phosphate group of an oligonucleotide via a polymethylene linkage (Asseline, et al., Proc. Natl. Acad. Sci. USA 1984, 81 , 3297). [0074] Any of the above groups can be used as a single linker or in combination with one or more further linkers.
Linkers and their use in preparation of conjugates of oligomeric compounds are provided throughout the art such as in WO 96/1 1205 and WO 98/52614 and U.S. Pat. Nos. 4,948,882; 5,525,465; 5,541 ,313; 5,545,730; 5,552,538; 5,580,731 ; 5,486,603; 5,608,046; 4,587,044; 4,667,025; 5,254,469; 5,245,022; 5,1 12,963; 5,391 ,723; 5,510475; 5,512,667; 5,574,142; 5,684,142; 5,770,716; 6,096,875; 6,335,432; and 6,335,437,Wo2012/083046 each of which is incorporated by reference in its entirety.
As used herein, a physiologically labile bond is a labile bond that is cleavable under conditions normally encountered or analogous to those encountered within a mammalian body (also referred to as a cleavable linker). Physiologically labile linkage groups are selected such that they undergo a chemical transformation (e.g., cleavage) when present in certain physiological conditions. Mammalian intracellular conditions include chemical conditions such as pH, temperature, oxidative or reductive conditions or agents, and salt concentration found in or analogous to those encountered in mammalian cells. Mammalian intracellular conditions also include the presence of enzymatic activity normally present in a mammalian cell such as from proteolytic or hydrolytic enzymes. In some embodiments, the cleavable linker is susceptible to nuclease(s) which may for example, be expressed in the target cell - and as such, as detailed herein, the linker may be a short region (e.g. 1 - 10) phosphodiester linked nucleosides, such as DNA nucleosides,
Chemical transformation (cleavage of the labile bond) may be initiated by the addition of a pharmaceutically acceptable agent to the cell or may occur spontaneously when a molecule containing the labile bond reaches an appropriate intra-and/or extra-cellular environment. For example, a pH labile bond may be cleaved when the molecule enters an acidified endosome. Thus, a pH labile bond may be considered to be an endosomal cleavable bond. Enzyme cleavable bonds may be cleaved when exposed to enzymes such as those present in an endosome or lysosome or in the cytoplasm. A disulfide bond may be cleaved when the molecule enters the more reducing environment of the cell cytoplasm. Thus, a disulfide may be considered to be a cytoplasmic cleavable bond. As used herein, a pH-labile bond is a labile bond that is selectively broken under acidic conditions (pH<7). Such bonds may also be termed endosomally labile bonds, since cell endosomes and lysosomes have a pH less than 7.
Oligomer linked biocleavable conjugates
The oligomeric compound may optionally, comprise a second region (region B) which is positioned between the oligomer (referred to as region A) and the conjugate (referred to as region C). Region B may be a linker such as a cleavable linker (also referred to as a physiologically labile linkage).
The susceptibility to cleavage in the assays shown in Example 6 may be used to determine whether a linker is biocleavable or physiologically labile.
Biocleavable linkers according to the present invention refers to linkers which are susceptible to cleavage in a target tissue (i.e. physiologically labile), for example liver and/or kidney. It is preferred that the cleavage rate seen in the target tissue is greater than that found in blood serum. Suitable methods for determining the level (%) of cleavage in tissue (e.g. liver or kidney) and in serum are found in example 6. In some embodiments, the biocleavable linker (also referred to as the physiologically labile linker, or nuclease susceptible linker), such as region B, in a compound of the invention, are at least about 20% cleaved, such as at least about 30% cleaved, such as at least about 40% cleaved, such as at least about 50% cleaved, such as at least about 60% cleaved, such as at least about 70% cleaved, such as at least about 75% cleaved, in the liver or kidney homogenate assay of Example 9. In some embodiments, the cleavage (%) in serum, as used in the assay in Example 9, is less than about 30%, is less than about 20%, such as less than about 10%, such as less than 5%, such as less than about 1 %.
In some embodiments, which may be the same of different, the biocleavable linker (also referred to as the physiologically labile linker, or nuclease susceptible linker), such as region B, in a compound of the invention, are susceptible to S1 nuclease cleavage. Susceptibility to S1 cleavage may be evaluated using the S1 nuclease assay shown in Example 6. In some embodiments, the biocleavable linker (also referred to as the physiologically labile linker, or nuclease susceptible linker), such as region B, in a compound of the invention, are at least about 30% cleaved, such as at least about 40% cleaved, such as at least about 50% cleaved, such as at least about 60% cleaved, such as at least about 70% cleaved, such as at least about 80% cleaved, such as at least about 90% cleaved, such as at least 95% cleaved after 120min incubation with S1 nuclease according to the assay used in Example 6.
Nuclease Susceptible Physiological Labile Linkages
The oligomeric compound may optionally, comprise a second region (region B) which is positioned between the LNA oligomer (referred to as region A) and the carbohydrate conjugate (referred to as region C). Region B may be a linker such as a cleavable linker (also referred to as a physiologically labile linkage).
In some embodiments, region B comprises between 1 - 10 nucleotides, which is covalently linked to the 5' or 3' nucleotide of the first region, such as via a internucleoside linkage group such as a phosphodiester linkage, wherein either
a. the internucleoside linkage between the first and second region is a phosphodiester linkage and the nucleoside of the second region [such as immediately] adjacent to the first region is either DNA or RNA; and/or
b. at least 1 nucleoside of the second region is a phosphodiester linked DNA or RNA nucleoside;
In some embodiments, region A and region B form a single contiguous nucleotide sequence of 8 - 35 nucleotides in length. In some aspects the internucleoside linkage between the first and second regions may be considered part of the second region.
In some embodiments, there is a phosphorus containing linkage group between the second and third region. The phosphorus linkage group, may, for example, be a phosphate (phosphodiester), a phosphorothioate, a phosphorodithioate or a boranophosphate group. In some embodiments, this phosphorus containing linkage group is positioned between the second region and a linker region which is attached to the third region. In some
embodiments, the phosphate group is a phosphodiester.
Therefore, in some aspects the oligomeric compound comprises at least two phosphodiester groups, wherein at least one is as according to the above statement of invention, and the other is positioned between the second and third regions, optionally between a linker group and the second region.
In some embodiments, the third region is an activation group, such as an activation group for use in conjugation. In this respect, the invention also provides activated oligomers comprising region A and B and a activation group, e.g an intermediate which is suitable for subsequent linking to the third region, such as suitable for conjugation.
In some embodiments, the third region is a reactive group, such as a reactive group for use in conjugation. In this respect, the invention also provides oligomers comprising region A and B and a reactive group, e.g an intermediate which is suitable for subsequent
linking to the third region, such as suitable for conjugation. The reactive group may, in some embodiments comprise an amine of alcohol group, such as an amine group.
In some embodiments region A comprises at least one, such as 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, or 24 internucleoside linkages other than phosphodiester, such as internucleoside linkages which are (optionally independently] selected from the group consisting of phosphorothioate, phosphorodithioate,and
boranophosphate, and methylphosphonate, such as phosphorothioate. In some
embodiments region A comprises at least one phosphorothioate linkage. In some embodiments at least 50%, such as at least 75%, such as at least 90% of the
internucleoside linkages, such as all the internucleoside linkages within region A are other than phosphodiester, for example are phosphorothioate linkages. In some embodiments, all the internucleoside linkages in region A are other than phosphodiester.
In some embodiments, the oligomeric compound comprises an antisense
oligonucleotide, such as an antisense oligonucleotide conjugate. The antisense
oligonucleotide may be or may comprise the first region, and optionally the second region. In this respect, in some embodiments, region B may form part of a contiguous nucleobase sequence which is complementary to the (nucleic acid) target. In other embodiments, region B may lack complementarity to the target.
Alternatively stated, in some embodiments, the invention provides a non- phosphodieser linked, such as a phosphorothioate linked, oligonucleotide (e.g. an antisense oligonucleotide) which has at least one terminal (5' and/or 3') DNA or RNA nucleoside linked to the adjacent nucleoside of the oligonucleotide via a phosphodiester linkage, wherein the terminal DNA or RNA nucleoside is further covalently linked to a conjugate moiety, a targeting moiety or a blocking moiety, optionally via a linker moiety.
The first region is covalently linked to the second region, such as via a 5' terminal or 3' terminal internucleoside linkage, such as a phosphodiester linkage. The phosphodiester linkage may therefore be positioned between the 5' most nucleoside of region A and the 3' most nucleoside of region B, and/or between the 3' most nucleoside of region A and the 5' most nucleoside of region B. In this respect, in some embodiments, there may be two region B covalently joined to region A, one at the 5' terminus of region A and one at the 3' terminus of region A. The two region Bs may be the same or different, and they may be covalently linked to the same or different third regions, optionally and independently via a linker (Y).
The oligomers may have a length of 8 - 35 contiguous nucleotides and comprise a first region of e.g. 7 - 23 contiguous nucleotides, and a second region of 1 - 10 contiguous nucleotides, wherein, for example, either the internucleoside linkage between the first and second region is a phosphodiester linked to the first (or only) DNA or RNA nucleoside of the second region, or region B comprises at least one phosphodiester linked DNA or RNA nucleoside..
The second region may, in some embodiments, comprise further DNA or RNA nucleosides which may be phosphodester linked. The second region is further covalently linked to a third region which may, for example, be or comprise the conjugate moiety.
The second region may comprise or consists of at least one DNA or RNA nucleosides linked to the first region via a phosphodiester linkage. In some aspects, the internucleoside linkage between the first and second region is considered as part of region B.
In some embodiments, the second region comprises or consists of at least between 1 and 10 linked nucleosides, such as 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 linked DNA or RNA nucleotides. Whilst a region of DNA RNA phosphodiester is considered important in the provision of a cleavable linker, it is possible that region B also comprises sugar-modified nucleoside analogues, such as those referred to under the first region above. However in some embodiments, the nucleosides of region B are (optionally independently) selected from the group consisting of DNA and RNA. It will be recognised that the nucleosides of region B may comprise naturally occurring or non-naturally occurring nucleobases. Region B comprises at least one phosphodiester linked DNA or RNA nucleoside (which may, in some embodiments, be the first nucleoside adjacent to region A). If region B comprises other nucleosides, region B may also comprise of other nucleoside linkages other than phosphodiester, such as (optionally independently) phosphorothioate, phosphodithioate, boranophosphate or methyl phosphonate. However, in other embodiments, all the internucleoside linkages in region B are phosphorothioate. In some embodiments, all the nucleosides of region B comprise (optionally independently) either a 2'-OH ribose sugar (RNA) or a 2'-H sugar - i.e. RNA or DNA.
In some embodiments, the second region comprises or consists of at least between 1 and 10 (e.g. phosphodiester) linked DNA or RNA nucleosides, such as 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 (e.g. phosphodiester) linked DNA or RNA nucleotides.
In some embodiments, region B comprises no more than 3 or no more than 4 consecutive DNA or RNA nucleosides (such as DNA nucleosides. As such region B may be so short as it does not recruit RNAseH, an aspect which may be important when region B
does not form a part of a single contiguous nucleobase sequence which is complementary to the target. Shorter region Bs, e.g. of 1 - 4nts in length may also be preferable in some embodiments, as they are unlikely to be the target of sequence specific restriction enzymes. As such it is possible to vary the susceptibility of the region B to endonuclease cleavage, and thereby fine-tune the rate of activation of the active oligomer in vivo, or even intracellular. Suitably, if very rapid activation is required, longer region Bs may be employed and/or region Bs which comprise the recognition sites of (e.g. cell or tissue specific or differentially expressed) restriction enzymes.
Region B may be conjugated to the conjugate, targeting reactive group, an activation group, or blocking group (X) via a linker group which may, for example, comprise a phosphodiester linkage, and/or optionally a suitable linker group, such as those provided herein. For example a phosphate nucleoside linkage (e.g. phosphodiester,
phosphorothioate, phosphodithioate, boranophosphate or methylphosphonate) or a triazol group. In some aspects, the linkage group is the same as the linkage group between regions A and B, and as such may be a phosphodiester linkage. In some aspects, the linkage group is a phiosphorothioate linkage.
In some embodiments the DNA or RNA nucleotides of the second region are independently selected from DNA and RNA nucleotides. In some embodiments the DNA or RNA nucleotides of the second region are DNA nucleotides. In some embodiments the DNA or RNA nucleotides of the second region are RNA nucleotides.
In the context of the second region, the term DNA and RNA nucleoside may comprise a naturally occurring or non-naturally occurring base (also referred to as a base analogue or modified base).
It will be recognized that, in some embodiments, the second region may further comprise other nucleotides or nucleotide analogues. In some embodiments, the second region comprises only DNA or RNA nucleosides. In some embodiments, when the second region comprises more than one nucleoside, the internucleoside linkages in the second region comprise phosphodiester linkages. In some embodiments, when the second region comprises more than one nucleoside, all the internucleoside linkages in the second region comprise phosphodiester linkages.
In some embodiments, at least two consecutive nucleosides of the second region are DNA nucleosides (such as at least 3 or 4 or 5 consecutive DNA nucleotides). In some embodiments the at least two consecutive nucleosides of the second region are RNA nucleosides (such as at least 3 or 4 or 5 consecutive RNA nucleotides). In some
embodiments the at least two consecutive nucleosides of the second region are at least one DNA and at least one RNA nucleoside. The internucleoside linkage between region A and region B is a phosphodiester linkage. In some embodiments, when region B comprises more than one nucleoside, at least one further internucleoside linkage is phosphodiester - such as the linkage group(s) between the 2 (or 3 or 4 or 5) nucleosides adjacent to region A.
The second region is flanked on one side (either 5' or 3') by the first region, e.g. an antisense oligonucleotide, and on the other side (either 3' or 5' respectfully, via a conjugate moiety or similar group (e.g. a blocking moiety/group, a targeting moiety/group or therapeutic small molecule moiety), optionally via a linker group (i.e. between the second region and the conjugate/blocking group etc. moiety).
In such an embodiment, the oligonucleotide of the invention may be described according to the following formula:
5 -A-PO-B [Y)X- 3' or 3 -A-PO-B [Y)X- 5'
wherein A is region A, PO is a phosphodiester linkage, B is region B, Y is an optional linkage group, and X is a conjugate, a targeting, a blocking group or a reactive or activation group.
In some embodiments, region B comprises 3' - 5' or 5'-3': i) a phosphodiester linkage to the 5' nucleoside of region A, ii) a DNA or RNA nucleoside, such as a DNA nucleoside, and iii) a further phosphodiester linkage
5 -A-PO-B - PO- 3' or 3 -A-PO-B - PO- 5'
The further phosphodiester linkage link the region B nucleoside with one or more further nucleoside, such as one or more DNA or RNA nucleosides, or may link to X (is a conjugate, a targeting or a blocking group or a reactive or activation group) optionally via a linkage group (Y).
In some embodiments, region B comprises 3' - 5' or 5'-3': i) a phosphodiester linkage to the 5' nucleoside of region A, ii) between 2 - 10 DNA or RNA phosphodiester linked nucleosides, such as a DNA nucleoside, and optionally iii) a further phosphodiester linkage:
5'-A-[PO-B]n - [Y]-X 3' or 3'-A-[PO-B]n -[Y]-X 5'
5'-A-[PO-B]n - PO-[Y]-X 3' or 3'-A-[PO-B]n - PO-[Y]-X 5'
Wherein A represent region A, [PO-B]n represents region B, wherein n is 1 - 10, such as 1 , 2, 3,4, 5, 6, 7, 8, 9 or 10, PO is an optional phosphodiester linkage group between region B and X (or Y if present).
In some embodiments the invention provides compounds according to (or comprising) one of the following formula:
5' [Region A] - PO - [region B] 3' -Y - X
5' [Region A] - PO - [region B] -P0 3' -Y - X
5' [Region A] - PO - [region B] 3' - X
5' [Region A] - PO - [region B] -P0 3' - X
3' [Region A] - PO - [region B] 5' -Y - X
3' [Region A] - PO - [region B] -PO 5' -Y - X
3' [Region A] - PO - [region B] 5' - X
3' [Region A] - PO - [region B] -PO 5' - X
Region B, may for example comprise or consist of:
5' DNA3'
3' DNA 5'
5' DNA-PO-DNA-3'
3' DNA-PO-DNA-5'
5' DNA-PO-DNA-PO-DNA 3'
3' DNA-PO-DNA-PO-DNA 5'
5' DNA-PO-DNA-PO-DNA-PO-DNA 3'
3' DNA-PO-DNA-PO-DNA-PO-DNA 5'
5' DNA-PO-DNA-PO-DNA-PO-DNA-PO-DNA 3'
3' DNA-PO-DNA-PO-DNA-PO-DNA-PO-DNA 5'
Sequence selection in the second region:
In some embodiments, region B does not form a complementary sequence when the oligonucleotide region A and B is aligned to the complementary target sequence.
In some embodiments, region B does form a complementary sequence when the oligonucleotide region A and B is aligned to the complementary target sequence. In this respect region A and B together may form a single contiguous sequence which is complementary to the target sequence.
In some embodiments, the sequence of bases in region B is selected to provide an optimal endonuclease cleavage site, based upon the predominant endonuclease cleavage enzymes present in the target tissue or cell or sub-cellular compartment. In this respect, by isolating cell extracts from target tissues and non-target tissues, endonuclease cleavage sequences for use in region B may be selected based upon a preferential cleavage activity in the desired target cell (e.g. liver/hepatocytes) as compared to a non-target cell (e.g. kidney). In this respect, the potency of the compound for target down-regulation may be optimized for the desired tissue/cell.
In some embodiments region B comprises a dinucleotide of sequence AA, AT, AC, AG, TA, TT, TC, TG, CA, CT, CC, CG, GA, GT, GC, or GG, wherein C may be 5- mthylcytosine, and/or T may be replaced with U. In some embodiments region B comprises a trinucleotide of sequence AAA, AAT, AAC, AAG, ATA, ATT, ATC, ATG, ACA, ACT, ACC, ACG, AGA, AGT, AGC, AGG, TAA, TAT, TAC, TAG, TTA, TTT, TTC, TAG, TCA, TCT, TCC, TCG, TGA, TGT, TGC, TGG, CAA, CAT, CAC, CAG, CTA, CTG, CTC, CTT, CCA, CCT, CCC, CCG, CGA, CGT, CGC, CGG, GAA, GAT, GAC, CAG, GTA, GTT, GTC, GTG, GCA, GCT, GCC, GCG, GGA, GGT, GGC, and GGG wherein C may be 5-mthylcytosine and/or T may be replaced with U. In some embodiments region B comprises a trinucleotide of sequence AAAX, AATX, AACX, AAGX, ATAX, ATTX, ATCX, ATGX, ACAX, ACTX, ACCX, ACGX, AGAX, AGTX, AGCX, AGGX, TAAX, TATX, TACX, TAGX, TTAX, TTTX, TTCX, TAGX, TCAX, TCTX, TCCX, TCGX, TGAX, TGTX, TGCX, TGGX, CAAX, CATX, CACX, CAGX, CTAX, CTGX, CTCX, CTTX, CCAX, CCTX, CCCX, CCGX, CGAX, CGTX, CGCX, CGGX, GAAX, GATX, GACX, CAGX, GTAX, GTTX, GTCX, GTGX, GCAX, GCTX, GCCX, GCGX, GGAX, GGTX, GGCX, and GGGX, wherein X may be selected from the group consisting of A, T, U, G, C and analogues thereof, wherein C may be 5-mthylcytosine and/or T may be replaced with U. It will be recognised that when referring to (naturally occurring) nucleobases A, T, U, G, C, these may be substituted with nucleobase analogues which function as the equivalent natural nucleobase (e.g. base pair with the complementary nucleoside).
In some embodiments, the compound of the invention may comprise more than one conjugate group (or more than one functional group X - such as a conjugate, targeting, blocking or activated group or a reactive or activation group), such as 2 or 3 such groups. In some embodiments, region B is covalently linked, optionally via a [e.g. non-nucleotide] linker group), to at least one functional group, such as two or three functional groups. In some embodiments, the first region may be covalently linked (e.g. via internucleoside linkages, such as phosphodiester linkages), to two region Bs, for example, one 5' and one 3' to the first region, wherein each region B may be (optionally independently) selected from the region B described herein. In this respect one region B may have one or more functional groups, and the second region B may have one or more function groups, wherein the functional groups of each region B may be independently selected from a conjugate, a targeting group, a blocking group or a reactive/activation group.
The use of a region B, such as a "PO DNA linker", as decribed above, between the oligomer and the conjugate moiety (X, or X-Y), is particularly advantageous as it ensures a
uniform cleavage of the conjugate moiety from the oligomer sequence, once the oligomer has been delivered to the target cell (e.g. a hepatocyte). Uniform cleavage may be useful in retaining maximal intra-cellular potency of the parent compound, as well as enhancing the safety profile of the oligomer conjugate.
Poly oligomeric compounds
The invention provides for a poly oligomeric compound which may comprise the first region (region A), (optionally the second region (region B)) and the third region (region C), wherein the first region is covalently linked to at least one further oligomeric compound (region A'), wherein the first region (region A) and region A' are covalently linked via a biocleavable linker (region B'), which may be, by way of example, as according to the second region
(reion B) as disclosed here, for example a region of at least one phosphodiester linked DNA or RNA (such as DNA), such as two, three, four or five phosphodiester linked DNA or RNA nucleosides (such as DNA nucleosides). In embodiments comprising a region B, regions B and B' may, in some embodiments have the same structure, e.g. the same number of DNA RNA nucleosides and phosphodiester linkages and/or the same nucleobase sequence. In other embodiments Regions B and B' may be different. By way of example such poly oligomeric compounds may have a structure such as: (5' - 3' or 3' - 5') Conjugate-PO-ON- PO'-ON', wherein conjugate is region C, PO is region B, PO' is region B', and ON 1 is region A, and ON' is region A'
It should be understood that region A may, in some embodiments, comprise multiple further oligomeric compounds (such as a further 2 or 3 oligomeric compounds) linked in series (or in parallel) via biocleavable linkers, for example: Conjugate-PO-ON-PO-ON'-PO"-ON", or Conjugate-PO-ON-[PO-ON']n, wherein n may, for example be 1 , 2 or 3, and each ON' may be the same or different, and if different may have the same or different targets.
The invention provides for an oligomer of the invention comprising a contiguous nucleotide sequence of formula [LNAs]7-i8-[DNA]1-5-[LNAs]7-i8, and a non-nucleobase conjugate, such as a GalNAc conjugate moeity, for example a trivalent GalNAc conjugate conjugate, such as a conjugate moeity selected from the group consisting of any one of Conjl , 2, 3, 4, 1 a, 2a, 3a, 4a, or other trivalent GalNAc conjugates, such as those disclosed herein. Subscript s refers to a phosphorothioate linkage. At least one internucleoside linkage within or adjacent to the -[DNA]1-5- region are phosphodiester linkages. In some embodiments, all internucleoside linkages within or adjacent to the -[DNA]1-5- region are phosphodiester linkages. In some embodiments, the -[DNA]1-5- region has 2, 3, 4 or 5 contiguous DNA nucleoside which are
joined by phosphodiester linkages. In such an embodiment, the internucleoside linkages between the -[DNA]2-5- are phosphodiester linkages, and optionally the internucleoside linkages between region -[DNA]1-5 and the LNA regions [LNAS]7-18 are independently phosphorothioate or phosphodiester linkages, such as both phosphodiester or both phosphorothioate, or one phosphodiester and one phosphorothioate. In the embodiment when the DNA region is a single DNA nucleoside, at least one or both the the
internucleoside linkages adjacent to the DNA region is a phosphodiester, and if only a single phosphodiester, the other may be a phosphorothioate. The region -[DNA]1-5 may be as defined as described by region B herein - i.e. may be a physiologically cleavable nucleoside linker region. Each [LNAS]7-18 is a LNA phosphorothioate oligomer, and may for example be independently selected from the group consisting of an LNA gapmer, an LNA mixmer or an LNA totalmer. For inhibiting the mature miR-122 sequence mixmers or toalmers are preferred. The GalNAc conjugate may for example be located 5' or 3' to the contiguous nucleotide sequence. In a preferred embodiment, at least one of the LNA oligomers, or both the poly oligomer conjugate is a LNA totalmer of 7 - 12, such as 8, 9 or 10 nucleotides in length. In some embodiments, the LNA totalmer may comprise only LNA nucleotides, such as beta-D-oxy LNA nucleoside, which are linked by phosphorothioate linkages. For example the poly oligomer conjugate may comprise a contiguous nucleositide sequence [LNAs]7-io- [DNA]1-5-[LNAs]7-10, such as [LNAS]7-10-[DNA]2-[LNAS]7-10 or [LNAs]7-1o-[DNA]3-[LNAs]7-1o or [LNAs]7-1o-[DNA]4-[LNAs]7-1o. In one embodiment the contiguous nucleositide sequence comprises [LNAs]8-[DNA]1-5-[LNAs]8, such as [LNAJ8-[DNA]2-[LNAS]8, [LNAS]8-[DNA]3-[LNAS]8, or [LNAS]8-[DNA]4-[LNAS]8.
This of particular importance for receptor mediate conjugate targeting, such as with asialoglycoprotein receptor conjugates, where the receptor mediated uptake of e.g. GalNAc conjugated oligomers is limited by the availability of free receptors on the surface of the target cell, the use of poly-oligomer conjugates allws for enhanced delivery to the target cell. It is also important to avoid complete saturation of cell -suface receptors which are performing an important biological function, the use of the poly-oligomer strategy therefore allows for effective delivery of sufficient compound to ensure relevant pharmacology, whilst reducing the risk of side effects due to receptor saturation/competition by the conjugate moiety. The use of the poly-oligomer conjugate therefore provides an effective solution for enhancing the therapeutic index - increased oligomer delivery and activity with a reduction of undesirable side-effects.
Compositions
The oligomer of the invention may be used in pharmaceutical formulations and compositions. Suitably, such compositions comprise a pharmaceutically acceptable diluent, carrier, salt or adjuvant. WO2007/031091 provides suitable and preferred pharmaceutically acceptable diluent, carrier and adjuvants - which are hereby incorporated by reference. Suitable dosages, formulations, administration routes, compositions, dosage forms, combinations with other therapeutic agents, pro-drug formulations are also provided in WO2007/031091 - which are also hereby incorporated by reference.
Antisense oligonucleotides may be admixed with pharmaceutically acceptable active or inert substances for the preparation of pharmaceutical compositions or formulations. Compositions and methods for the formulation of pharmaceutical compositions are dependent upon a number of criteria, including, but not limited to, route of administration, extent of disease, or dose to be administered.
An antisense compound can be utilized in pharmaceutical compositions by combining the antisense compound with a suitable pharmaceutically acceptable diluent or carrier. A pharmaceutically acceptable diluent includes phosphate-buffered saline (PBS). PBS is a diluent suitable for use in compositions to be delivered parenterally.
Pharmaceutical compositions comprising antisense compounds encompass any pharmaceutically acceptable salts, esters, or salts of such esters, or any other
oligonucleotide which, upon administration to an animal, including a human, is capable ofproviding (directly or indirectly) the biologically active metabolite or residue thereof.
Accordingly, for example, the disclosure is also drawn to pharmaceutically acceptable salts of antisense compounds, prodrugs, pharmaceutically acceptable salts of such prodrugs, and other bioequivalents. Suitable pharmaceutically acceptable salts include, but are not limited to, sodium and potassium salts. In some embodiments, the oligomer of the invention is a pro-drug.
Medical Indications
The invention provides for the antisense oligomer conjugate for use in medicine.
The invention provides for the antisense oligomer conjugate, for use in down-regulating a liver-expressed micro-RNA.-122. The invention provides for the LNA antisense oligomer conjugate, for use in treatment of a metabolic disease or disorder, such as a hepatic disease or disorder. The invention provides for the LNA antisense oligomer conjugate, for use in treatment of hepatitis C.
In some embodiments, the disease is liver disease or disorder. In some embodiments, the disease or disorder is, or results in or is associated with liver-fibrosis.
In some embodiments, the disease or disorder is a liver disease or disorder.
In some embodiments the disease or disorder is a metabolic disorder, which may for example be a liver disease or disorder, and/or in some aspects a cardiovascular disease or disorder).
Cardiovascular/Metabolic diseases include, for examples, metabolic syndrome, obesity, hyperlipidaemia , atherosclerosis, HDL/LDL cholesterol imbalance, dyslipidemias, e.g., familial combined hyperlipidaemia (FCHL), acquired hyperlipidaemia, statin-resistant, hypercholesterolemia, coronary artery disease (CAD), and coronary heart disease (CHD)., atherosclerosis, heart disease, diabetes (I and/or II), NASH, acute coronary syndrome (ACS), NASH, chronic heart failure, cardiovascular disease, cardio metabolic disease, hyperlipidaemia and related disorders, metabolic syndrome, atherosclerosis, chronic heart failure, vascular disease, peripheral arterial disease, heart disease, ischemia. In some embodiments, the disease or disorder is selected from the group consisting of metabolic syndrome, obesity, hyperlipidaemia, atherosclerosis, HDL/LDL cholesterol imbalance, dyslipidemias, e.g., familial combined hyperlipidaemia (FCHL), acquired hyperlipidaemia, statin-resistant, hypercholesterolemia, coronary artery disease (CAD), and coronary heart disease (CHD).
In some embodiments, the disease or disorder is selected from the group consisting of chronic heart failure, cardiovascular disease, cardio metabolic disease, chronic heart failure, vascular disease, peripheral arterial disease, heart disease, ischemia, acute coronary syndrome (ACS).
In some embodiments, the disease or disorder is a viral disease, such viral hepatitis, such as hepatitis C.ln some embodiments, the liver disease may be a disease or disorder selected from the group consisting of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis.
The invention further provides use of a compound of the invention in the manufacture of a medicament for the treatment of a disease, disorder or condition, such as those as referred to herein.
An interesting aspect of the invention is directed to the use of the compound as defined herein for the preparation of a medicament for the treatment of a disease, disorder or condition as referred to herein.
Moreover, the invention relates to a method of treating a subject, such as a mammal, such as a human suffering from a disease or condition such as those referred to herein.
A patient who is in need of treatment is a patient suffering from or likely to suffer from the disease or disorder.
In some embodiments, the term 'treatment' as used herein refers to both treatment of an existing disease (e.g. a disease or disorder as herein referred to), or prevention of a disease, i.e. prophylaxis. It will therefore be recognized that treatment as referred to herein may, in some embodiments, be prophylactic.
Combination Treatments
In some embodiments the compound of the invention is for use in a combination treatment with another therapeutic agent. Inhibitors of HMG CoA reductase, such as statins for example are widely used in the treatment of metabolic disease (see WO2009/043354, hereby incorporated by reference for examples of combination treatments). Suitable cholesterol lowering compounds may be selected from a compound is selected from the group consisting of bile salt sequestering resins (e.g., cholestyramine, colestipol, and colesevelam hydrochloride), HMGCoA-reductase inhibitors (e.g., lovastatin, cerivastatin, prevastatin, atorvastatin, simvastatin, and fluvastatin), nicotinic acid, fibric acid derivatives (e.g., clofibrate, gemfibrozil, fenofibrate, bezafibrate, and ciprofibrate), probucol, neomycin, dextrothyroxine, plant-stanol esters, cholesterol absorption inhibitors (e.g., ezetimibe), implitapide, inhibitors of bile acid transporters (apical sodium-dependent bile acid
transporters), regulators of hepatic CYP7a, estrogen replacement therapeutics (e.g., tamoxifen), and anti-inflammatories (e.g., glucocorticoids). Combinations with statins may be particularly preferred.
In some embodiments, the compound of the invention are for use in treating an HCV infection in a subject, wherein said treatment is in combination with a further therapeutic compound, such as an interferon or an anti-viral, such as ribavirin. In some embodiments, the invention provides a method of treatment of an HCV infection in a subject infected with HCV, said method comprising administering an effective amount of the compound of the invention in combination with an effective amount of a further therapeutic compound, such as an interferon or an anti-viral, such as ribavirin, to the subject infected with HCV. The combination treatment may be with a combination with both an interferon and one or more other antivirals. The combination treatment may be with ribavirin and an other anti-viral. The combination treatment may be with the compound of the invention and an other antiviral
(i.e. other than ribavirin). Examples of the interferons include, but are not limited to pegylated rIFN-alpha 2b, pegylated rIFN-alpha 2a, rIFN-alpha 2b, rIFN-alpha 2a, consensus IFN alpha (infergen), feron, reaferon, intermax alpha, r-IFN-beta, infergen and actimmune, IFN-omega with DUROS, albuferon, locteron, Albuferon, Rebif, oral interferon alpha, IFNalp ha-2b XL, AVI-005, PEG-lnfergen, and pegylated IFN-beta.
Ribavirin analogs and the ribavirin prodrug viramidine (taribavirin) have been administered with interferons to control HCV. In some embodiments, compound 3 may therefore also comprises antiviral ribavirin analogs and antiviral ribavirin derivatives, as well as ribavirin pro-drugs.
Examples of other antivirals for use in combination treatment of the invention include NS5A protein inhibitors (see WO2013000856, hereby incorporated by reference, e.g. ledipasvir), NS3/4 protease inhibitor, such as telaprevir or boceprevir (see WO2012/175733, hereby incorporated by reference), or an HS5B RNA dependent RNA polymerase inhibitor (see WO2013/000855, hereby incorporated by reference), including non-nucleoside inhibitors and nucleoside inhibitors, such as Sovaldi (sofosbuvir, previously known as PSI-7977 or Gl- 7977), .
For the use in treatment of HCV infection, the compound of the invention may be used, either in monotherapy or as part of a combination therapy to treat a subject infected with HCV. The subject may be infected with HCV of a genotype selected from the group consisting of 1 a, 1 b, 2, 3, 4, 5 or 6. In some embodiments the genotype of the HCV is 1 a. In some embodiments the genotype of the HCV is 1 b. In some embodiments the subject is treatment naive. In some embodiments, the subject is subject who is a non-responder, a partial responder, a relapse responder or a null responder to a HCV therapy, such as an interferon, or other HCV therapeutics described herein.
EXAMPLES
Examples 1 - 5 and 7 - 1 1 - see PCT/EP2013/073858 Examples 1 - 1 1 , hereby
incorporated by reference. See also Examples 1 - 12 of PCT/EP2014/051781 , which are hereby specifically incorporated by reference.
Oligonucleotide List
In the following list, Capital letters represent LNA nucleosides, such as beta-D-oxy LNA, lower case letters represent DNA nucleosides. Capital L is a LNA, such as beta-D-oxy, and
lower case d is a DNA nucleoside. LNA cytosines are optionally 5'methyl cytosine. The internucleosides within region A are phosphorothioate, and within region B are
phosphodiester (as shown). The internucleoside linkage between region A and B is phoshodiester, but where region B is >1 DNA nucleotide, may optionally be other than phosphodiester (e.g. may be phosphorothioate). There is, optionally a further linker (Y), between region B and region C, such as a C6 linker. # refers to SEQ ID No.
ApoB Targeting Compounds with FAM label conjugates
Example 6. In vitro cleavage of different DNA/PO-linkers
FAM-labeled ASOs with different DNA PO-linkers (PO linkers) were subjected to in vitro cleavage either in S1 nuclease extract (Fig. 6A), Liver or kidney homogenates or Serum FAM-labeled ASOs 100 μΜ with different DNA/PO-linkers were subjected to in vitro cleavage by S1 nuclease in nuclease buffer (60 U pr. 100 μΙ_) for 20 and 120 minutes (A). The enzymatic activity was stopped by adding EDTA to the buffer solution. The solutions were then subjected to AIE HPLC analyses on a Dionex Ultimate 3000 using an Dionex DNApac p-100 column and a gradient ranging from 10mM - 1 M sodium perchlorate at pH 7.5. The content of cleaved and non cleaved oligonucleotide were determinded against a standard using both a fluoresense detector at 615 nm and a uv detector at 260 nm.
Conclusion: The PO linkers (or region B as referred to herein) results in the conjugate (or group C) being cleaved off, and both the length and/or the sequence composition of the linker can be used tomodulate susceptibility to nucleolytic cleavage of region B. The
Sequence of DNA/PO-linkers can modulate the cleavage rate as seen after 20 min in
Nuclease S1 extract Sequence selection for region B (e.g.for the DNA/PO-linker) can therefore also be used to modulate the level of cleavage in serum and in cells of target tissues.
Liver, kidney and Serum (B) were spiked with oligonucleotide SEQ ID NO 16 to concentrations of 200 μg g tissue. Liver and kidney samples collected from NMRI mice were homogenized in a homogenisation buffer (0,5% Igepal CA-630, 25 mM Tris pH 8.0, 100 mM NaCI, pH 8.0 (adjusted with 1 N NaOH). The homogenates were incubated for 24 hours at 37° and thereafter the homogenates were extracted with phenol - chloroform. The content of cleaved and non cleaved oligonucleotide in the extract from liver and kidney and from the serum were determinded against a standard using the above HPLC method.
Conclusion: The PO linkers (or region B as referred to herein) results in cleavage of the conjugate (or group C) from the oligonucleotide, in liver or kidney homogenate, but not in serum.
Note: cleavage in the above assays refers to the cleavage of the cleavable linker, the oligomer or region A should remain functionally intact. The susceptibility to cleavage in the above assays can be used to determine whether a linker is biocleavable or physiologically labile.
Example 12: LNA antimiRs GalNac Poly-Oligo Conjugates
Compounds
Capital letters are LNA, such as beta-D-oxy LNA. Lower case letters are DNA. Subscript s is a phosphorothioate linkage. Other internucleoside linkages are phosphodiester
(phosphate) linkages. Superscript m before a C represents LNA 5-methyl cytosine. In some embodiments, the compounds may also be made with LNA cytosine. In some embodiments, the Conjl a group may be another GalNAc conjugate group, such as those disclosed herein, for example Conj2a. miR-122 (Tiny) 5'- mCsAs mCsAs mCsTs mCsmC -3' (Compound no 51 ) SEQ ID NO 14
GalNAc-tiny 5'- Conjl a mCsAs mCsAs mCsTs mCsmC -3' (Compound no 52) SEQ ID NO 15 tiny-linker-tiny5'- mCsAs mCsAs mCsTs mCsmCcamCsAs mCsAs mCsTs mCsmC -3' (Compound no 53) SEQ ID NO 16
GalNac-tiny-linker-tiny 5'- Conjl a mCsAs mCsAs mCsTs mCsmCcamCsAs mCsAs mCsTs mCsmC -3' (Compound no 54) SEQ ID NO 17
An in vivo mouse study was performed using a total of 9 groups of mice (n=5). Each mouse was dosed i.v. on days 0, 2 and 4 with either 0,5 mg/kg or 2 mg/kg or equimolar doses of the GalNAc conjugated LNAs compared to parent LNA compound. A saline control group was included (see study set up in Tab. 1 ). Serum samples were taken 4 days pre administration, interim at day 4 and at endpoint day 7. Liver and kidney samples were stored in RNA later. Validation of miR122 knock down of miR122 was done as described in Obad Nat Genet. 201 1 Mar 20;43(4):371 -8 (Fig.2). The cholesterol level in serum were analyzed as described in Elmen J, et al. LNA-mediated microRNA silencing in non-human primates. Nature. 2008;452:896-899. (Fig.3) and mRNA levels of two miR122 down regulated genes (Aldo A and Bckdk) were analyzed using standard QPCR assay techniques (Fig.4). ALT was measured to assess tolerability of the compounds (Fig.5).
Study Set-up
Conclusions: Conjugation of GalNAc to anti-miR122 (Compound 52 and 54) showed a remarkable improvement of miR122 knock down in the liver indicated by decreased total cholesterol levels (Fig.2) and up regulation of Aldo A and Bckdk mRNA already at in the low dose group (Fig.3, 3x 0,5 mg/kg). No effect of the anti- miR122 oligonucleotide was seen in
the kidney. An increase in ALT was measured for Compound 52 which showed a tendency to improve by conjugation of 2 oligonucleotides to one GalNAc (Compound 54).
Claims
1 . An antisense oligonucleotide which targets microRNAR-122 (miR-122) for use in the treatment of hepatitis C infection (HCV), wherein the oligomer is conjugated to an asialoglycoprotein receptor targeting moiety.
2. The antisense oligonucleotide according to claim 1 , wherein the asialoglycoprotein receptor targeting moiety comprises N-acetylgalactosamine (GalNac).
3. The antisense oligonucleotide according to claim 1 wherein the asialoglycoprotein receptor targeting moiety comprises a galactose cluster.
4. The antisense oligonucleotide according to claim 1 , wherein the asialoglycoprotein receptor targeting moiety comprises a trivalent GalNac cluster.
5. An antisense oligonucleotide according to any one of claims 1 - 4 for the treatment of a cholesterol associated metabolic disease or a hepatitis C infection.
6. The antisense oligonucleotide according to claim 5, wherein the asialoglycoprotein receptor targeting moiety comprises N-acetylgalactosamine (GalNac).
7. The antisense oligonucleotide according to claim 5 wherein the asialoglycoprotein receptor targeting moiety comprises a galactose cluster.
8. The antisense oligonucleotide according to claim 5, wherein the asialoglycoprotein receptor targeting moiety comprises a trivalent GalNac cluster.
9. The antisense oligonucleotide according to any one of claims 1 - 8 wherein the
contiguous nucleotide sequence of the antisense oligonucleotide is fully
complementary to a corresponding region of hsa-miR-122.
10. The antisense oligonucleotide according to any one of claims 1 - 9 wherein the
contiguous nucleotide sequence of the antisense oligonucleotide comprises a region which is fully complementary to the seed region of miR-122.
1 1 . The antisense oligonucleotide according to any one of claims 1 - 10 wherein the contiguous nucleotide sequence of the antisense oligonucleotide is fully
complementary to a corresponding region of hsa-miR-122.
12. The antisense oligonucleotide according to any one of claims 1 - 1 1 , wherein the contiguous nucleotide sequence of the antisense oligonucleotide comprises the sequence 5'-CACACTCC-3' or 5'-ACACTCC-3'.
13. The antisense oligonucleotide according to any one of claims 10 - 12, wherein the antisense oligomer comprises 1 - 7 LNA units in the region which is fully
complementary to the seed region of has-miR-122.
14. The use according to any one of claims 1 - 13, wherein nucleotides 1 - 7 or 2 - 8 of the oligomer, as measured from the 3' end, are complementary to the seed region of has-miR-122.
15. The antisense oligonucleotide according to any one of claims 1 - 14, wherein the antisense oligonucleotide has a length of 10 - 30 nucleotides.
16. The antisense oligonucleotide according to any one of claims 1 - 14, wherein the antisense oligonucleotide has a length of 8 - 18 nucleotides.
17. The antisense oligonucleotide according to any one of claims 10 - 14, wherein the antisense oligonucleotide has a length of 7, 8, or 9 nucleotides and wherein at least 80% of the nucleotides are LNA nucleotides, or all nucleotides are LNA nucleotides, linkages with the optional exception of between 1 - 5 DNA consecutive
phosphodiester linked DNA nucleotides (region B).
18. The antisense oligonucleotide according to any one of claims 1 - 17, wherein at least 80% of the internucleoside linkages of the antisense oligonucleotide are
phosphorothioate linkages.
19. The antisense oligonucleotide according to claim 18, wherein all the internucleoside linkages of the antisense oligonucleotide are phosphorothioate linkages with the optional exception of between 1 - 5 DNA consecutive phosphodiester linked DNA nucleotides (region B).
20. The antisense oligonucleotide according to any one of claims 1 - 19, wherein the antisense oligonucleotide comprises a sequence selected from the group consisting of 5'-CcAttGTcaCaCtCC-3', 5'-ACACTCC-3', 5'-CACACTCC-3', and 5 - TCACACTCC-3'; wherin capital letters are beta-D-oxy LNA, small letters are DNA, all internucleoside linkages are phosphorothioate and LNA C are 5-methyl cyctosine.
21 . An antisense oligonucleotide which targets miR-122 wherein said antisense
oligonucleotide is conjugated to an asialoglycoprotein receptor targeting moiety, for use in medicine.
22. The use of the antisense oligonucleotide as defined in any one of claims 1 - 20, for the preparation of a medicament for the treatment of a metabolic disease or viral disease.
23. The use of the antisense oligonucleotide according to claim 22, wherein the use is for the preparation of a medicament for the treatment of HCV infection.
24. The use according to claim 23, wherein said use is in combination with a further therapeutic agent such as an interferon and/or an antiviral agent.
Applications Claiming Priority (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP13153296.2 | 2013-01-30 | ||
| EP13153296 | 2013-01-30 | ||
| EP13157237.2 | 2013-02-28 | ||
| EP13157237 | 2013-02-28 | ||
| EP13174092.0 | 2013-06-27 | ||
| EP13174092 | 2013-06-27 | ||
| EP13192930 | 2013-11-14 | ||
| EP13192930.9 | 2013-11-14 | ||
| EPPCT/EP2013/073858 | 2013-11-14 | ||
| PCT/EP2013/073858 WO2014076195A1 (en) | 2012-11-15 | 2013-11-14 | Oligonucleotide conjugates |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014118272A1 true WO2014118272A1 (en) | 2014-08-07 |
Family
ID=51261486
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2014/051793 WO2014118272A1 (en) | 2013-01-30 | 2014-01-30 | Antimir-122 oligonucleotide carbohydrate conjugates |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2014118272A1 (en) |
Cited By (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9127276B2 (en) | 2013-05-01 | 2015-09-08 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US9157083B2 (en) | 2013-05-01 | 2015-10-13 | Regulus Therapeutics Inc. | MicroRNA compounds and methods for modulating miR-122 |
| WO2016075584A1 (en) * | 2014-11-10 | 2016-05-19 | Glaxosmithkline Intellectual Property (No.2) Limited | Combination long acting compositions and methods for hepatitis c |
| WO2016075583A1 (en) * | 2014-11-10 | 2016-05-19 | Glaxosmithkline Intelle Ctual Property (No.2) Limited | Long acting pharmaceutical compositions for hepatitis c |
| US9382540B2 (en) | 2014-05-01 | 2016-07-05 | Isis Pharmaceuticals, Inc | Compositions and methods for modulating angiopoietin-like 3 expression |
| US9487783B2 (en) | 2014-08-07 | 2016-11-08 | Regulus Therapeutics Inc. | Targeting microRNAs for metabolic disorders |
| US9506030B2 (en) | 2013-05-01 | 2016-11-29 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| WO2016205749A1 (en) | 2015-06-18 | 2016-12-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017055423A1 (en) * | 2015-10-02 | 2017-04-06 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugation process |
| WO2017083368A1 (en) * | 2015-11-12 | 2017-05-18 | Pfizer Inc. | Tissue-specific genome engineering using crispr-cas9 |
| EP3009511B1 (en) | 2015-06-18 | 2017-05-31 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| WO2017106657A1 (en) | 2015-12-18 | 2017-06-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017157899A1 (en) * | 2016-03-14 | 2017-09-21 | F. Hoffmann-La Roche Ag | Oligonucleotides for reduction of pd-l1 expression |
| WO2017184768A1 (en) | 2016-04-19 | 2017-10-26 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017184786A1 (en) | 2016-04-19 | 2017-10-26 | The Broad Institute Inc. | Cpf1 complexes with reduced indel activity |
| WO2017189308A1 (en) | 2016-04-19 | 2017-11-02 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2018035388A1 (en) | 2016-08-17 | 2018-02-22 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| WO2018035387A1 (en) | 2016-08-17 | 2018-02-22 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| WO2018064371A1 (en) | 2016-09-30 | 2018-04-05 | The Regents Of The University Of California | Rna-guided nucleic acid modifying enzymes and methods of use thereof |
| US9982257B2 (en) | 2012-07-13 | 2018-05-29 | Wave Life Sciences Ltd. | Chiral control |
| US9994855B2 (en) | 2014-05-01 | 2018-06-12 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| US10144933B2 (en) | 2014-01-15 | 2018-12-04 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having immunity induction activity, and immunity induction activator |
| US10149905B2 (en) | 2014-01-15 | 2018-12-11 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having antitumor effect and antitumor agent |
| US10160969B2 (en) | 2014-01-16 | 2018-12-25 | Wave Life Sciences Ltd. | Chiral design |
| US10167309B2 (en) | 2012-07-13 | 2019-01-01 | Wave Life Sciences Ltd. | Asymmetric auxiliary group |
| WO2019060746A1 (en) | 2017-09-21 | 2019-03-28 | The Broad Institute, Inc. | Systems, methods, and compositions for targeted nucleic acid editing |
| US10246709B2 (en) | 2016-03-07 | 2019-04-02 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands for therapeutic compounds |
| US10280423B2 (en) | 2014-05-01 | 2019-05-07 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor B expression |
| US10280192B2 (en) | 2011-07-19 | 2019-05-07 | Wave Life Sciences Ltd. | Methods for the synthesis of functionalized nucleic acids |
| WO2019094983A1 (en) | 2017-11-13 | 2019-05-16 | The Broad Institute, Inc. | Methods and compositions for treating cancer by targeting the clec2d-klrb1 pathway |
| US10294474B2 (en) | 2016-09-02 | 2019-05-21 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands |
| WO2019099943A1 (en) | 2017-11-16 | 2019-05-23 | Astrazeneca Ab | Compositions and methods for improving the efficacy of cas9-based knock-in strategies |
| US10307434B2 (en) | 2009-07-06 | 2019-06-04 | Wave Life Sciences Ltd. | Nucleic acid prodrugs and methods of use thereof |
| US10329318B2 (en) | 2008-12-02 | 2019-06-25 | Wave Life Sciences Ltd. | Method for the synthesis of phosphorus atom modified nucleic acids |
| US10428019B2 (en) | 2010-09-24 | 2019-10-01 | Wave Life Sciences Ltd. | Chiral auxiliaries |
| US10450568B2 (en) | 2015-10-09 | 2019-10-22 | Wave Life Sciences Ltd. | Oligonucleotide compositions and methods thereof |
| WO2019217369A1 (en) * | 2018-05-08 | 2019-11-14 | Regulus Therapeutics Inc. | Galnac conjugated modified oligonucleotide as mir-122 inhibitor having hcv antiviral activity with reduced hyperbilirubinemia side-effect |
| US10557137B2 (en) | 2015-11-06 | 2020-02-11 | Ionis Pharmaceuticals, Inc. | Modulating apolipoprotein (a) expression |
| WO2020033601A1 (en) | 2018-08-07 | 2020-02-13 | The Broad Institute, Inc. | Novel cas12b enzymes and systems |
| WO2020049158A1 (en) | 2018-09-07 | 2020-03-12 | Astrazeneca Ab | Compositions and methods for improved nucleases |
| US10648020B2 (en) | 2015-06-18 | 2020-05-12 | The Broad Institute, Inc. | CRISPR enzymes and systems |
| WO2020191102A1 (en) | 2019-03-18 | 2020-09-24 | The Broad Institute, Inc. | Type vii crispr proteins and systems |
| WO2020236972A2 (en) | 2019-05-20 | 2020-11-26 | The Broad Institute, Inc. | Non-class i multi-component nucleic acid targeting systems |
| WO2020236967A1 (en) | 2019-05-20 | 2020-11-26 | The Broad Institute, Inc. | Random crispr-cas deletion mutant |
| WO2021055855A1 (en) | 2019-09-20 | 2021-03-25 | The Broad Institute, Inc. | Compositions and methods for delivering cargo to a target cell |
| US11053482B2 (en) | 2015-04-16 | 2021-07-06 | Wageningen Universiteit | Vector systems for nuclease-mediated genome editing |
| US11174470B2 (en) | 2019-01-04 | 2021-11-16 | Mammoth Biosciences, Inc. | Programmable nuclease improvements and compositions and methods for nucleic acid amplification and detection |
| US11207416B2 (en) | 2020-03-04 | 2021-12-28 | Verve Therapeutics, Inc. | Compositions and methods for targeted RNA delivery |
| EP3929287A2 (en) | 2015-06-18 | 2021-12-29 | The Broad Institute, Inc. | Crispr enzyme mutations reducing off-target effects |
| DE212020000516U1 (en) | 2019-03-07 | 2022-01-17 | The Regents of the University of California | CRISPR-CAS effector polypeptides |
| US11273442B1 (en) | 2018-08-01 | 2022-03-15 | Mammoth Biosciences, Inc. | Programmable nuclease compositions and methods of use thereof |
| US11400161B2 (en) | 2016-10-06 | 2022-08-02 | Ionis Pharmaceuticals, Inc. | Method of conjugating oligomeric compounds |
| US11447527B2 (en) | 2018-09-18 | 2022-09-20 | Vnv Newco Inc. | Endogenous Gag-based capsids and uses thereof |
| WO2023052508A2 (en) | 2021-09-30 | 2023-04-06 | Astrazeneca Ab | Use of inhibitors to increase efficiency of crispr/cas insertions |
| WO2023196818A1 (en) | 2022-04-04 | 2023-10-12 | The Regents Of The University Of California | Genetic complementation compositions and methods |
Citations (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2699808A (en) | 1944-10-06 | 1955-01-18 | Mark W Lowe | Apparatus for peeling tomatoes |
| US4587044A (en) | 1983-09-01 | 1986-05-06 | The Johns Hopkins University | Linkage of proteins to nucleic acids |
| US4667025A (en) | 1982-08-09 | 1987-05-19 | Wakunaga Seiyaku Kabushiki Kaisha | Oligonucleotide derivatives |
| US4948882A (en) | 1983-02-22 | 1990-08-14 | Syngene, Inc. | Single-stranded labelled oligonucleotides, reactive monomers and methods of synthesis |
| US5112963A (en) | 1987-11-12 | 1992-05-12 | Max-Planck-Gesellschaft Zur Foerderung Der Wissenschaften E.V. | Modified oligonucleotides |
| US5245022A (en) | 1990-08-03 | 1993-09-14 | Sterling Drug, Inc. | Exonuclease resistant terminally substituted oligonucleotides |
| US5254469A (en) | 1989-09-12 | 1993-10-19 | Eastman Kodak Company | Oligonucleotide-enzyme conjugate that can be used as a probe in hybridization assays and polymerase chain reaction procedures |
| WO1994014226A1 (en) | 1992-12-14 | 1994-06-23 | Honeywell Inc. | Motor system with individually controlled redundant windings |
| US5391723A (en) | 1989-05-31 | 1995-02-21 | Neorx Corporation | Oligonucleotide conjugates |
| US5486603A (en) | 1990-01-08 | 1996-01-23 | Gilead Sciences, Inc. | Oligonucleotide having enhanced binding affinity |
| WO1996011205A1 (en) | 1994-10-06 | 1996-04-18 | Isis Pharmaceuticals, Inc. | Peptide nucleic acid conjugates |
| US5510475A (en) | 1990-11-08 | 1996-04-23 | Hybridon, Inc. | Oligonucleotide multiple reporter precursors |
| US5512667A (en) | 1990-08-28 | 1996-04-30 | Reed; Michael W. | Trifunctional intermediates for preparing 3'-tailed oligonucleotides |
| US5525465A (en) | 1987-10-28 | 1996-06-11 | Howard Florey Institute Of Experimental Physiology And Medicine | Oligonucleotide-polyamide conjugates and methods of production and applications of the same |
| US5545730A (en) | 1984-10-16 | 1996-08-13 | Chiron Corporation | Multifunctional nucleic acid monomer |
| US5574142A (en) | 1992-12-15 | 1996-11-12 | Microprobe Corporation | Peptide linkers for improved oligonucleotide delivery |
| US5580731A (en) | 1994-08-25 | 1996-12-03 | Chiron Corporation | N-4 modified pyrimidine deoxynucleotides and oligonucleotide probes synthesized therewith |
| US5608046A (en) | 1990-07-27 | 1997-03-04 | Isis Pharmaceuticals, Inc. | Conjugated 4'-desmethyl nucleoside analog compounds |
| US5656408A (en) | 1996-04-29 | 1997-08-12 | Xerox Corporation | Coated carrier particles |
| US5684142A (en) | 1995-06-07 | 1997-11-04 | Oncor, Inc. | Modified nucleotides for nucleic acid labeling |
| US5770716A (en) | 1997-04-10 | 1998-06-23 | The Perkin-Elmer Corporation | Substituted propargylethoxyamido nucleosides, oligonucleotides and methods for using same |
| WO1998039352A1 (en) | 1997-03-07 | 1998-09-11 | Takeshi Imanishi | Novel bicyclonucleoside and oligonucleotide analogues |
| WO1998052614A2 (en) | 1997-05-21 | 1998-11-26 | The Board Of Trustees Of The Leland Stanford Junior University | Composition and method for enhancing transport across biological membranes |
| US5885968A (en) | 1992-08-11 | 1999-03-23 | Rijksuniversiteit Te Leiden | Triantennary cluster glycosides, their preparation and use |
| WO1999014226A2 (en) | 1997-09-12 | 1999-03-25 | Exiqon A/S | Bi- and tri-cyclic nucleoside, nucleotide and oligonucleotide analogues |
| US5994517A (en) | 1995-11-22 | 1999-11-30 | Paul O. P. Ts'o | Ligands to enhance cellular uptake of biomolecules |
| WO1999065925A1 (en) | 1998-06-16 | 1999-12-23 | Isis Pharmaceuticals, Inc. | Targeted oligonucleotide conjugates |
| US6096875A (en) | 1998-05-29 | 2000-08-01 | The Perlein-Elmer Corporation | Nucleotide compounds including a rigid linker |
| US6335437B1 (en) | 1998-09-07 | 2002-01-01 | Isis Pharmaceuticals, Inc. | Methods for the preparation of conjugated oligomers |
| US6335432B1 (en) | 1998-08-07 | 2002-01-01 | Bio-Red Laboratories, Inc. | Structural analogs of amine bases and nucleosides |
| EP1222309A1 (en) | 1999-09-30 | 2002-07-17 | Isis Pharmaceuticals, Inc. | Human rnase h and oligonucleotide compositions thereof |
| US6525191B1 (en) | 1999-05-11 | 2003-02-25 | Kanda S. Ramasamy | Conformationally constrained L-nucleosides |
| US6670461B1 (en) | 1997-09-12 | 2003-12-30 | Exiqon A/S | Oligonucleotide analogues |
| WO2004044141A2 (en) | 2002-11-05 | 2004-05-27 | Isis Pharmaceuticals, Inc. | Conjugated oligomeric compounds and their use in gene modulation |
| US6770748B2 (en) | 1997-03-07 | 2004-08-03 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogue |
| US20040171570A1 (en) | 2002-11-05 | 2004-09-02 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| WO2004106356A1 (en) | 2003-05-27 | 2004-12-09 | Syddansk Universitet | Functionalized nucleotide derivatives |
| EP1495769A1 (en) | 2003-07-11 | 2005-01-12 | LBR Medbiotech B.V. | Mannose-6-phosphate receptor mediated gene transfer into muscle cells |
| WO2005013901A2 (en) | 2003-07-31 | 2005-02-17 | Isis Pharmaceuticals, Inc. | Oligomeric compounds and compositions for use in modulation of small non-coding rnas |
| WO2005021570A1 (en) | 2003-08-28 | 2005-03-10 | Gene Design, Inc. | Novel artificial nucleic acids of n-o bond crosslinkage type |
| US20050130923A1 (en) | 2003-09-18 | 2005-06-16 | Balkrishen Bhat | 4'-thionucleosides and oligomeric compounds |
| US7053207B2 (en) | 1999-05-04 | 2006-05-30 | Exiqon A/S | L-ribo-LNA analogues |
| WO2007021896A2 (en) * | 2005-08-10 | 2007-02-22 | Alnylam Pharmaceuticals, Inc. | Chemically modified oligonucleotides for use in modulating micro rna and uses thereof |
| WO2007027894A2 (en) | 2005-08-29 | 2007-03-08 | Isis Pharmaceuticals, Inc. | Antisense compounds having enhanced anti-microrna activity |
| WO2007027775A2 (en) | 2005-08-29 | 2007-03-08 | Isis Pharmaceuticals, Inc. | Methods for use in modulating mir-122a |
| WO2007031091A2 (en) | 2005-09-15 | 2007-03-22 | Santaris Pharma A/S | Rna antagonist compounds for the modulation of p21 ras expression |
| WO2007112754A2 (en) | 2006-04-03 | 2007-10-11 | Santaris Pharma A/S | Pharmaceutical compositions comprising anti-mirna antisense oligonucleotides |
| WO2007134181A2 (en) | 2006-05-11 | 2007-11-22 | Isis Pharmaceuticals, Inc. | 5'-modified bicyclic nucleic acid analogs |
| US20080039618A1 (en) | 2002-11-05 | 2008-02-14 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| US7399845B2 (en) | 2006-01-27 | 2008-07-15 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| WO2008101157A1 (en) | 2007-02-15 | 2008-08-21 | Isis Pharmaceuticals, Inc. | 5'-substituted-2'-f modified nucleosides and oligomeric compounds prepared therefrom |
| WO2008131807A2 (en) | 2007-05-01 | 2008-11-06 | Santaris Pharma A/S | Splice switching oligomers for tnf superfamily receptors and their use in treatment of disease |
| WO2008150729A2 (en) | 2007-05-30 | 2008-12-11 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| WO2008154401A2 (en) | 2007-06-08 | 2008-12-18 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| WO2009006478A2 (en) | 2007-07-05 | 2009-01-08 | Isis Pharmaceuticals, Inc. | 6-disubstituted bicyclic nucleic acid analogs |
| WO2009012495A1 (en) | 2007-07-19 | 2009-01-22 | Datakey Electronics, Inc. | Rf token and receptacle system and method |
| WO2009043353A2 (en) | 2007-10-04 | 2009-04-09 | Santaris Pharma A/S | Micromirs |
| WO2009100320A2 (en) | 2008-02-07 | 2009-08-13 | Isis Pharmaceuticals, Inc. | Bicyclic cyclohexitol nucleic acid analogs |
| WO2009124238A1 (en) | 2008-04-04 | 2009-10-08 | Isis Pharmaceuticals, Inc. | Oligomeric compounds comprising neutrally linked terminal bicyclic nucleosides |
| WO2009126933A2 (en) | 2008-04-11 | 2009-10-15 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| WO2012083046A2 (en) | 2010-12-17 | 2012-06-21 | Arrowhead Research Corporation | Galactose cluster-pharmacokinetic modulator targeting moiety for sirna |
| WO2012082046A1 (en) | 2010-12-13 | 2012-06-21 | Telefonaktiebolaget L M Ericsson (Publ) | Exchange of parameters relating to measurement periods |
| WO2012089352A1 (en) | 2010-12-29 | 2012-07-05 | F. Hoffmann-La Roche Ag | Small molecule conjugates for intracellular delivery of nucleic acids |
| WO2012175733A1 (en) | 2011-06-23 | 2012-12-27 | Santaris Pharma A/S | Hcv combination therapy |
| WO2013000855A1 (en) | 2011-06-30 | 2013-01-03 | Santaris Pharma A/S | Hcv combination therapy |
| WO2013000856A1 (en) | 2011-06-30 | 2013-01-03 | Santaris Pharma A/S | Hcv combination therapy |
| WO2013154798A1 (en) | 2012-04-09 | 2013-10-17 | Isis Pharmaceuticals, Inc. | Tricyclic nucleic acid analogs |
| US9984408B1 (en) | 2012-05-30 | 2018-05-29 | Amazon Technologies, Inc. | Method, medium, and system for live video cooperative shopping |
-
2014
- 2014-01-30 WO PCT/EP2014/051793 patent/WO2014118272A1/en active Application Filing
Patent Citations (79)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2699808A (en) | 1944-10-06 | 1955-01-18 | Mark W Lowe | Apparatus for peeling tomatoes |
| US4667025A (en) | 1982-08-09 | 1987-05-19 | Wakunaga Seiyaku Kabushiki Kaisha | Oligonucleotide derivatives |
| US4948882A (en) | 1983-02-22 | 1990-08-14 | Syngene, Inc. | Single-stranded labelled oligonucleotides, reactive monomers and methods of synthesis |
| US5541313A (en) | 1983-02-22 | 1996-07-30 | Molecular Biosystems, Inc. | Single-stranded labelled oligonucleotides of preselected sequence |
| US4587044A (en) | 1983-09-01 | 1986-05-06 | The Johns Hopkins University | Linkage of proteins to nucleic acids |
| US5552538A (en) | 1984-10-16 | 1996-09-03 | Chiron Corporation | Oligonucleotides with cleavable sites |
| US5545730A (en) | 1984-10-16 | 1996-08-13 | Chiron Corporation | Multifunctional nucleic acid monomer |
| US5525465A (en) | 1987-10-28 | 1996-06-11 | Howard Florey Institute Of Experimental Physiology And Medicine | Oligonucleotide-polyamide conjugates and methods of production and applications of the same |
| US5112963A (en) | 1987-11-12 | 1992-05-12 | Max-Planck-Gesellschaft Zur Foerderung Der Wissenschaften E.V. | Modified oligonucleotides |
| US5391723A (en) | 1989-05-31 | 1995-02-21 | Neorx Corporation | Oligonucleotide conjugates |
| US5254469A (en) | 1989-09-12 | 1993-10-19 | Eastman Kodak Company | Oligonucleotide-enzyme conjugate that can be used as a probe in hybridization assays and polymerase chain reaction procedures |
| US5486603A (en) | 1990-01-08 | 1996-01-23 | Gilead Sciences, Inc. | Oligonucleotide having enhanced binding affinity |
| US5608046A (en) | 1990-07-27 | 1997-03-04 | Isis Pharmaceuticals, Inc. | Conjugated 4'-desmethyl nucleoside analog compounds |
| US5245022A (en) | 1990-08-03 | 1993-09-14 | Sterling Drug, Inc. | Exonuclease resistant terminally substituted oligonucleotides |
| US5512667A (en) | 1990-08-28 | 1996-04-30 | Reed; Michael W. | Trifunctional intermediates for preparing 3'-tailed oligonucleotides |
| US5510475A (en) | 1990-11-08 | 1996-04-23 | Hybridon, Inc. | Oligonucleotide multiple reporter precursors |
| US5885968A (en) | 1992-08-11 | 1999-03-23 | Rijksuniversiteit Te Leiden | Triantennary cluster glycosides, their preparation and use |
| WO1994014226A1 (en) | 1992-12-14 | 1994-06-23 | Honeywell Inc. | Motor system with individually controlled redundant windings |
| US5574142A (en) | 1992-12-15 | 1996-11-12 | Microprobe Corporation | Peptide linkers for improved oligonucleotide delivery |
| US5580731A (en) | 1994-08-25 | 1996-12-03 | Chiron Corporation | N-4 modified pyrimidine deoxynucleotides and oligonucleotide probes synthesized therewith |
| WO1996011205A1 (en) | 1994-10-06 | 1996-04-18 | Isis Pharmaceuticals, Inc. | Peptide nucleic acid conjugates |
| US5684142A (en) | 1995-06-07 | 1997-11-04 | Oncor, Inc. | Modified nucleotides for nucleic acid labeling |
| US5994517A (en) | 1995-11-22 | 1999-11-30 | Paul O. P. Ts'o | Ligands to enhance cellular uptake of biomolecules |
| US5656408A (en) | 1996-04-29 | 1997-08-12 | Xerox Corporation | Coated carrier particles |
| WO1998039352A1 (en) | 1997-03-07 | 1998-09-11 | Takeshi Imanishi | Novel bicyclonucleoside and oligonucleotide analogues |
| US6268490B1 (en) | 1997-03-07 | 2001-07-31 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogues |
| US6770748B2 (en) | 1997-03-07 | 2004-08-03 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogue |
| US5770716A (en) | 1997-04-10 | 1998-06-23 | The Perkin-Elmer Corporation | Substituted propargylethoxyamido nucleosides, oligonucleotides and methods for using same |
| WO1998052614A2 (en) | 1997-05-21 | 1998-11-26 | The Board Of Trustees Of The Leland Stanford Junior University | Composition and method for enhancing transport across biological membranes |
| US6670461B1 (en) | 1997-09-12 | 2003-12-30 | Exiqon A/S | Oligonucleotide analogues |
| WO1999014226A2 (en) | 1997-09-12 | 1999-03-25 | Exiqon A/S | Bi- and tri-cyclic nucleoside, nucleotide and oligonucleotide analogues |
| US6794499B2 (en) | 1997-09-12 | 2004-09-21 | Exiqon A/S | Oligonucleotide analogues |
| US7034133B2 (en) | 1997-09-12 | 2006-04-25 | Exiqon A/S | Oligonucleotide analogues |
| US6096875A (en) | 1998-05-29 | 2000-08-01 | The Perlein-Elmer Corporation | Nucleotide compounds including a rigid linker |
| WO1999065925A1 (en) | 1998-06-16 | 1999-12-23 | Isis Pharmaceuticals, Inc. | Targeted oligonucleotide conjugates |
| US6525031B2 (en) | 1998-06-16 | 2003-02-25 | Isis Pharmaceuticals, Inc. | Targeted Oligonucleotide conjugates |
| US6335432B1 (en) | 1998-08-07 | 2002-01-01 | Bio-Red Laboratories, Inc. | Structural analogs of amine bases and nucleosides |
| US6335437B1 (en) | 1998-09-07 | 2002-01-01 | Isis Pharmaceuticals, Inc. | Methods for the preparation of conjugated oligomers |
| US7053207B2 (en) | 1999-05-04 | 2006-05-30 | Exiqon A/S | L-ribo-LNA analogues |
| US6525191B1 (en) | 1999-05-11 | 2003-02-25 | Kanda S. Ramasamy | Conformationally constrained L-nucleosides |
| EP1222309A1 (en) | 1999-09-30 | 2002-07-17 | Isis Pharmaceuticals, Inc. | Human rnase h and oligonucleotide compositions thereof |
| WO2004044141A2 (en) | 2002-11-05 | 2004-05-27 | Isis Pharmaceuticals, Inc. | Conjugated oligomeric compounds and their use in gene modulation |
| US20080039618A1 (en) | 2002-11-05 | 2008-02-14 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| US20040171570A1 (en) | 2002-11-05 | 2004-09-02 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| WO2004106356A1 (en) | 2003-05-27 | 2004-12-09 | Syddansk Universitet | Functionalized nucleotide derivatives |
| EP1495769A1 (en) | 2003-07-11 | 2005-01-12 | LBR Medbiotech B.V. | Mannose-6-phosphate receptor mediated gene transfer into muscle cells |
| WO2005013901A2 (en) | 2003-07-31 | 2005-02-17 | Isis Pharmaceuticals, Inc. | Oligomeric compounds and compositions for use in modulation of small non-coding rnas |
| WO2005021570A1 (en) | 2003-08-28 | 2005-03-10 | Gene Design, Inc. | Novel artificial nucleic acids of n-o bond crosslinkage type |
| US7427672B2 (en) | 2003-08-28 | 2008-09-23 | Takeshi Imanishi | Artificial nucleic acids of n-o bond crosslinkage type |
| US20050130923A1 (en) | 2003-09-18 | 2005-06-16 | Balkrishen Bhat | 4'-thionucleosides and oligomeric compounds |
| WO2007021896A2 (en) * | 2005-08-10 | 2007-02-22 | Alnylam Pharmaceuticals, Inc. | Chemically modified oligonucleotides for use in modulating micro rna and uses thereof |
| WO2007027894A2 (en) | 2005-08-29 | 2007-03-08 | Isis Pharmaceuticals, Inc. | Antisense compounds having enhanced anti-microrna activity |
| WO2007027775A2 (en) | 2005-08-29 | 2007-03-08 | Isis Pharmaceuticals, Inc. | Methods for use in modulating mir-122a |
| WO2007031091A2 (en) | 2005-09-15 | 2007-03-22 | Santaris Pharma A/S | Rna antagonist compounds for the modulation of p21 ras expression |
| EP1984381A2 (en) | 2006-01-27 | 2008-10-29 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| US7399845B2 (en) | 2006-01-27 | 2008-07-15 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| WO2007112753A2 (en) | 2006-04-03 | 2007-10-11 | Santaris Pharma A/S | Pharmaceutical composition comprising anti-mirna antisense oligonucleotides |
| WO2007112754A2 (en) | 2006-04-03 | 2007-10-11 | Santaris Pharma A/S | Pharmaceutical compositions comprising anti-mirna antisense oligonucleotides |
| US20070287831A1 (en) | 2006-05-11 | 2007-12-13 | Isis Pharmaceuticals, Inc | 5'-modified bicyclic nucleic acid analogs |
| WO2007134181A2 (en) | 2006-05-11 | 2007-11-22 | Isis Pharmaceuticals, Inc. | 5'-modified bicyclic nucleic acid analogs |
| WO2008101157A1 (en) | 2007-02-15 | 2008-08-21 | Isis Pharmaceuticals, Inc. | 5'-substituted-2'-f modified nucleosides and oligomeric compounds prepared therefrom |
| WO2008131807A2 (en) | 2007-05-01 | 2008-11-06 | Santaris Pharma A/S | Splice switching oligomers for tnf superfamily receptors and their use in treatment of disease |
| WO2008150729A2 (en) | 2007-05-30 | 2008-12-11 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| WO2008154401A2 (en) | 2007-06-08 | 2008-12-18 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| WO2009006478A2 (en) | 2007-07-05 | 2009-01-08 | Isis Pharmaceuticals, Inc. | 6-disubstituted bicyclic nucleic acid analogs |
| WO2009012495A1 (en) | 2007-07-19 | 2009-01-22 | Datakey Electronics, Inc. | Rf token and receptacle system and method |
| WO2009043354A2 (en) | 2007-10-04 | 2009-04-09 | Santaris Pharma A/S | Combination treatment for the treatment of hepatitis c virus infection |
| WO2009043353A2 (en) | 2007-10-04 | 2009-04-09 | Santaris Pharma A/S | Micromirs |
| WO2009100320A2 (en) | 2008-02-07 | 2009-08-13 | Isis Pharmaceuticals, Inc. | Bicyclic cyclohexitol nucleic acid analogs |
| WO2009124238A1 (en) | 2008-04-04 | 2009-10-08 | Isis Pharmaceuticals, Inc. | Oligomeric compounds comprising neutrally linked terminal bicyclic nucleosides |
| WO2009126933A2 (en) | 2008-04-11 | 2009-10-15 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| WO2012082046A1 (en) | 2010-12-13 | 2012-06-21 | Telefonaktiebolaget L M Ericsson (Publ) | Exchange of parameters relating to measurement periods |
| WO2012083046A2 (en) | 2010-12-17 | 2012-06-21 | Arrowhead Research Corporation | Galactose cluster-pharmacokinetic modulator targeting moiety for sirna |
| WO2012089352A1 (en) | 2010-12-29 | 2012-07-05 | F. Hoffmann-La Roche Ag | Small molecule conjugates for intracellular delivery of nucleic acids |
| WO2012175733A1 (en) | 2011-06-23 | 2012-12-27 | Santaris Pharma A/S | Hcv combination therapy |
| WO2013000855A1 (en) | 2011-06-30 | 2013-01-03 | Santaris Pharma A/S | Hcv combination therapy |
| WO2013000856A1 (en) | 2011-06-30 | 2013-01-03 | Santaris Pharma A/S | Hcv combination therapy |
| WO2013154798A1 (en) | 2012-04-09 | 2013-10-17 | Isis Pharmaceuticals, Inc. | Tricyclic nucleic acid analogs |
| US9984408B1 (en) | 2012-05-30 | 2018-05-29 | Amazon Technologies, Inc. | Method, medium, and system for live video cooperative shopping |
Non-Patent Citations (58)
| Title |
|---|
| "Antisense Research and Applications", 1993, CRC PRESS, pages: 303 - 350 |
| "Positive Preclinical Profile of RG-101, a GalNAc-conjugated anti-miR Targeting microRNA- 122, Supports Clinical Development for the Treatment of HCV", 4 November 2013 (2013-11-04), La Jolla, USA, XP055113426, Retrieved from the Internet <URL:http://ir.regulusrx.com/common/download/download.cfm?companyid=AMDA-14LKIJ&fileid=702853&filekey=4983a1e5-ad0e-403d-84e0-18467463f58f&filename=803979.pdf> [retrieved on 20140410] * |
| "Regulus Provides Update on 'Road to the Clinic' Strategy and Reports First Quarter 2013 Financial Results and Recent Highlights", 14 May 2013 (2013-05-14), La Jolla, USA, XP055113339, Retrieved from the Internet <URL:http://ir.regulusrx.com/common/download/download.cfm?companyid=AMDA-14LKIJ&fileid=663964&filekey=95d522e5-6d8d-4998-b01f-1afcfe6b9c30&filename=764723.pdf> [retrieved on 20140410] * |
| ALBAEK ET AL., J. ORG. CHEM., vol. 71, 2006, pages 7731 - 7740 |
| ALTMAN ET AL., NUCLEOSIDES NUCLEOTIDES, vol. 16, 1997, pages 917 - 926 |
| ALTMANN ET AL., BIOCHEM. SOC. TRANS., vol. 24, 1996, pages 630 - 637 |
| ALTMANN ET AL., CHIMIA, vol. 50, 1996, pages 168 - 176 |
| ASSELINE ET AL., PROC. NATL. ACAD. SCI. USA, vol. 81, 1984, pages 3297 |
| BAENZIGER; FIETE, CELL, vol. 22, 1980, pages 611 - 620 |
| BAKER ET AL., J. BIOL. CHEM., vol. 272, no. 1, 1997, pages 1944 - 12000 |
| BALKRISHEN BHAT ET AL: "RG-101, a GalNAc-conjugated anti-miR Employinga Unique Mechanism of Action by Targeting Host Factor MicroRNA-122 ( miR-122),Demonstrates Potent Activity and Reduction of HCV in Preclinical Studies", 4 November 2013 (2013-11-04), XP055113633, Retrieved from the Internet <URL:http://www.regulusrx.com/wp-content/uploads/2013/11/RT13-002-Neben_AASLD-LFP_final.pdf> [retrieved on 20140411] * |
| BHAT BALKRISHEN ET AL: "RG-101, a GalNAC-conjugated anti-miR employing a unique mechanism of action by targeting host factor microRNA-122 (miR-122), demonstrates potent activity and reduction of HCV in preclinical studies", HEPATOLOGY, vol. 58, no. 6, 1 December 2013 (2013-12-01), & 64TH ANNUAL MEETING OF THE AMERICAN-ASSOCIATION-FOR-THE-STUDY-OF-LIVER-DISEASES; WASHINGTON, DC, USA; NOVEMBER 01 -05, 2013, pages 1393A, XP002723153 * |
| BHAT ET AL., AASLD, 7 November 2013 (2013-11-07) |
| BIESSEN ET AL., J. MED. CHERN., vol. 39, 1995, pages 1538 - 1546 |
| BRAASCH ET AL., CHEM. BIOL, vol. 8, 2001, pages 1 - 7 |
| CHATTOPADHYAYA ET AL., J. ORG. CHEM., vol. 74, 2009, pages 118 - 134 |
| CHRISTENSEN, NUCL. ACIDS. RES., vol. 30, 2002, pages 4918 - 4925 |
| CONNOLLY ET AL., 1. BIOL. CHERN., vol. 257, 1982, pages 939 - 945 |
| COREY ET AL., J AM. CHEM. SOC., vol. 111, 1989, pages 8524 |
| COREY ET AL., SCIENCE, vol. 238, 1987, pages 1401 |
| DUFF ET AL., METHODS ENZYMOL, vol. 313, 2000, pages 297 |
| ELAYADI ET AL., CURR. OPINION INVENS. DRUGS, vol. 2, 2001, pages 558 - 561 |
| ELMEN J ET AL.: "LNA-mediated microRNA silencing in non-human primates", NATURE, vol. 452, 2008, pages 896 - 899 |
| ERIK A.L. BIESSEN ET AL: "Targeted delivery of oligodeoxynucleotides to parenchymal liver cells in vivo", BIOCHEMICAL JOURNAL, vol. 340, no. 3, 15 June 1999 (1999-06-15), pages 783 - 792, XP055101395, ISSN: 0264-6021, DOI: 10.1042/0264-6021:3400783 * |
| ESAU, CELL METAB., vol. 3, no. 2, February 2006 (2006-02-01), pages 87 - 98 |
| FREIER ET AL., NUCLEIC ACIDS RESEARCH, vol. 25, no. 22, 1997, pages 4429 - 4443 |
| FREIER; ALTMANN, NUCL. ACID RES., vol. 25, 1997, pages 4429 - 4443 |
| FRIEDEN ET AL., NUCLEIC ACIDS RESEARCH, vol. 21, 2003, pages 6365 - 6372 |
| HANGELAND ET AL., BIOCONJUG CHEM., vol. 6, no. 6, November 1995 (1995-11-01), pages 695 - 701 |
| JOBST, S.T.; DRICKAMER, K., JB.C., vol. 271, 1996, pages 6686 |
| JUDY ET AL., TETRAHEDRON LETTERS, vol. 32, 1991, pages 879 |
| KOSHKIN ET AL., TETRAHEDRON, vol. 54, 1998, pages 3607 - 3630 |
| KRIEG ET AL., ANTISENSE RESEARCH AND DEVELOPMENT, vol. 1, 1991, pages 161 |
| KRÜTZFELDT ET AL., NATURE, vol. 438, 2005, pages 685 - 689 |
| KUMAR ET AL., BIOORG. MED. CHEM. LETT., vol. 8, 1998, pages 2219 - 2222 |
| LEUMANN, CJ., BIOORG. AND MED. CHEM., vol. 10, 2002, pages 841 - 854 |
| LEUMANN, J. C, BIOORGANIC AND MEDICINAL CHEMISTRY, vol. 10, 2002, pages 841 - 854 |
| LIN ZHU ET AL: "Site-Specific Delivery of Oligonucleotides to Hepatocytes after Systemic Administration", BIOCONJUGATE CHEMISTRY, vol. 19, no. 1, 1 January 2008 (2008-01-01), pages 290 - 298, XP055101407, ISSN: 1043-1802, DOI: 10.1021/bc070126m * |
| MAIER M A ET AL: "Synthesis of antisense oligonucleotides conjugated to a multivalent carbohydrate cluster for cellular targeting", BIOCONJUGATE CHEMISTRY, ACS, WASHINGTON, DC, US, vol. 14, 1 January 2003 (2003-01-01), pages 18 - 29, XP002510288, ISSN: 1043-1802, [retrieved on 20021203], DOI: 10.1021/BC020028V * |
| MAKINO NAOKI ET AL: "Intravenous injection with antisense oligodeoxynucleotides against angiotensin decreases blood pressure in spontaneously hypertensive rats", 19980501; 19980500, vol. 31, no. 5, 1 May 1998 (1998-05-01), pages 1166 - 1170, XP002336133 * |
| MARTIN, P., HE/V. CHIM. ACTA, vol. 78, 1995, pages 486 - 504 |
| NELSON ET AL., NUC. ACIDS RES., vol. 17, 1989, pages 7187 |
| OBAD, NAT GENET., vol. 43, no. 4, 20 March 2011 (2011-03-20), pages 371 - 8 |
| ORAM ET AL., CURR. OPINION MOL. THER., vol. 3, 2001, pages 239 - 243 |
| S. B. RAJUR ET AL: "Covalent Protein-Oligonucleotide Conjugates for Efficient Delivery of Antisense Molecules", BIOCONJUGATE CHEMISTRY, vol. 8, no. 6, 1 November 1997 (1997-11-01), pages 935 - 940, XP055095373, ISSN: 1043-1802, DOI: 10.1021/bc970172u * |
| SETH, J. ORG. CHEM, 2010 |
| SINGH ET AL., CHEM. COMMUN., vol. 4, 1998, pages 455 - 456 |
| SINGH ET AL., J. ORG. CHEM., vol. 63, 1998, pages 10035 - 10039 |
| SOUTSCHECK ET AL., NATURE, vol. 432, 2004, pages 173 - 178 |
| SRIVASTAVA ET AL., J. AM. CHEM. SOC, vol. 129, no. 26, 4 July 2007 (2007-07-04), pages 8362 - 8379 |
| SRIVASTAVA ET AL., J. AM. CHEM. SOC., vol. 129, no. 26, 2007, pages 8362 - 8379 |
| UHLMANN, CURR. OPINION IN DRUG DEVELOPMENT, vol. 3, no. 2, 2000, pages 293 - 213 |
| VAN POELGEEST ET AL., AMERICAN JOURNAL OF KIDNEY DISEASE |
| WAHLESTEDT ET AL., PROC. NATL. ACAD. SCI. U. S. A., vol. 97, 2000, pages 5633 - 5638 |
| YANG ET AL., BIOCONJUG CHEM, vol. 20, no. 2, 2009, pages 213 - 21 |
| ZATSEPIN; ORETSKAYA, CHEM BIODIVERS, vol. 1, no. 10, 2004, pages 1401 - 17 |
| ZHENG SU-JUN ET AL: "Distribution and anti-HBV effects of antisense oligodeoxynucleotides conjugated to galactosylated poly-L-lysine", WORLD JOURNAL OF GASTROENTEROLOGY, WJG PRESS, CN, vol. 9, no. 6, 1 January 2003 (2003-01-01), pages 1251 - 1255, XP002510287, ISSN: 1007-9327 * |
| ZUCKERMANN ET AL., J AM. CHEM. SOC., vol. 110, 1988, pages 1614 |
Cited By (128)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10329318B2 (en) | 2008-12-02 | 2019-06-25 | Wave Life Sciences Ltd. | Method for the synthesis of phosphorus atom modified nucleic acids |
| US10307434B2 (en) | 2009-07-06 | 2019-06-04 | Wave Life Sciences Ltd. | Nucleic acid prodrugs and methods of use thereof |
| US10428019B2 (en) | 2010-09-24 | 2019-10-01 | Wave Life Sciences Ltd. | Chiral auxiliaries |
| US10280192B2 (en) | 2011-07-19 | 2019-05-07 | Wave Life Sciences Ltd. | Methods for the synthesis of functionalized nucleic acids |
| US9982257B2 (en) | 2012-07-13 | 2018-05-29 | Wave Life Sciences Ltd. | Chiral control |
| US10590413B2 (en) | 2012-07-13 | 2020-03-17 | Wave Life Sciences Ltd. | Chiral control |
| US10167309B2 (en) | 2012-07-13 | 2019-01-01 | Wave Life Sciences Ltd. | Asymmetric auxiliary group |
| US10941400B2 (en) | 2013-05-01 | 2021-03-09 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| US9714421B2 (en) | 2013-05-01 | 2017-07-25 | Ionis Pharmaceuticals, Inc. | Compositions and methods |
| US10150967B2 (en) | 2013-05-01 | 2018-12-11 | Regulus Therapeutics Inc. | MicroRNA compounds and methods for modulating miR-122 |
| US9932581B2 (en) | 2013-05-01 | 2018-04-03 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein C-III expression |
| US9506030B2 (en) | 2013-05-01 | 2016-11-29 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| US9145558B2 (en) | 2013-05-01 | 2015-09-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating HBV expression |
| US9574194B2 (en) | 2013-05-01 | 2017-02-21 | Regulus Therapeutics Inc. | MicroRNA compounds and methods for modulating miR-122 |
| US11851655B2 (en) | 2013-05-01 | 2023-12-26 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| US9157083B2 (en) | 2013-05-01 | 2015-10-13 | Regulus Therapeutics Inc. | MicroRNA compounds and methods for modulating miR-122 |
| US9163239B2 (en) | 2013-05-01 | 2015-10-20 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein C-III expression |
| US9181549B2 (en) | 2013-05-01 | 2015-11-10 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US10883104B2 (en) | 2013-05-01 | 2021-01-05 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| US10683499B2 (en) | 2013-05-01 | 2020-06-16 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating TTR expression |
| US9932580B2 (en) | 2013-05-01 | 2018-04-03 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating HBV expression |
| US9309513B2 (en) | 2013-05-01 | 2016-04-12 | Regulus Therapeutics Inc. | MicroRNA compounds and methods for modulating miR-122 |
| US10240151B2 (en) | 2013-05-01 | 2019-03-26 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| US9181550B2 (en) | 2013-05-01 | 2015-11-10 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| US11299736B1 (en) | 2013-05-01 | 2022-04-12 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US9127276B2 (en) | 2013-05-01 | 2015-09-08 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US9957504B2 (en) | 2013-05-01 | 2018-05-01 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| US10144933B2 (en) | 2014-01-15 | 2018-12-04 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having immunity induction activity, and immunity induction activator |
| US10149905B2 (en) | 2014-01-15 | 2018-12-11 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having antitumor effect and antitumor agent |
| US10160969B2 (en) | 2014-01-16 | 2018-12-25 | Wave Life Sciences Ltd. | Chiral design |
| US10280423B2 (en) | 2014-05-01 | 2019-05-07 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor B expression |
| US11312964B2 (en) | 2014-05-01 | 2022-04-26 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| US9957292B2 (en) | 2014-05-01 | 2018-05-01 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating angiopoietin-like 3 expression |
| US10875884B2 (en) | 2014-05-01 | 2020-12-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating angiopoietin-like 3 expression |
| US9994855B2 (en) | 2014-05-01 | 2018-06-12 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| US11732265B2 (en) | 2014-05-01 | 2023-08-22 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor B expression |
| US10793862B2 (en) | 2014-05-01 | 2020-10-06 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| US9382540B2 (en) | 2014-05-01 | 2016-07-05 | Isis Pharmaceuticals, Inc | Compositions and methods for modulating angiopoietin-like 3 expression |
| US9862950B2 (en) | 2014-08-07 | 2018-01-09 | Regulus Therapeutics Inc. | Targeting microRNAs for metabolic disorders |
| US10138484B2 (en) | 2014-08-07 | 2018-11-27 | Regulus Therapeutics Inc. | Targeting microRNAs for metabolic disorders |
| US9487783B2 (en) | 2014-08-07 | 2016-11-08 | Regulus Therapeutics Inc. | Targeting microRNAs for metabolic disorders |
| CN107206089A (en) * | 2014-11-10 | 2017-09-26 | 葛兰素史克知识产权第二有限公司 | Joint durable action composition and method for hepatitis C |
| US10308937B2 (en) | 2014-11-10 | 2019-06-04 | Glaxosmithkline Intellectual Property (No. 2) Limited | Combination long acting compositions and methods for Hepatitis C |
| WO2016075583A1 (en) * | 2014-11-10 | 2016-05-19 | Glaxosmithkline Intelle Ctual Property (No.2) Limited | Long acting pharmaceutical compositions for hepatitis c |
| JP2017534651A (en) * | 2014-11-10 | 2017-11-24 | グラクソスミスクライン、インテレクチュアル、プロパティー、(ナンバー2)、リミテッドGlaxosmithkline Intellectual Property (No.2) Limited | Long-acting pharmaceutical composition for hepatitis C |
| WO2016075584A1 (en) * | 2014-11-10 | 2016-05-19 | Glaxosmithkline Intellectual Property (No.2) Limited | Combination long acting compositions and methods for hepatitis c |
| US11760985B2 (en) | 2015-04-16 | 2023-09-19 | Wageningen Universiteit | CRISPR-Cas complex |
| US11053482B2 (en) | 2015-04-16 | 2021-07-06 | Wageningen Universiteit | Vector systems for nuclease-mediated genome editing |
| US11479761B2 (en) | 2015-04-16 | 2022-10-25 | Wageningen Universiteit | Nuclease-mediated genome editing |
| EP3009511B1 (en) | 2015-06-18 | 2017-05-31 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| US11781172B2 (en) | 2015-06-18 | 2023-10-10 | The Broad Institute, Inc. | Crispr enzymes and systems |
| US11634755B2 (en) | 2015-06-18 | 2023-04-25 | The Broad Institute, Inc. | Crispr enzymes and systems |
| US11180751B2 (en) | 2015-06-18 | 2021-11-23 | The Broad Institute, Inc. | CRISPR enzymes and systems |
| US11773432B2 (en) | 2015-06-18 | 2023-10-03 | The Broad Institute Inc. | CRISPR enzymes and systems |
| EP3929287A2 (en) | 2015-06-18 | 2021-12-29 | The Broad Institute, Inc. | Crispr enzyme mutations reducing off-target effects |
| EP3502253A1 (en) | 2015-06-18 | 2019-06-26 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| US11091798B2 (en) | 2015-06-18 | 2021-08-17 | The Broad Institute, Inc. | CRISPR enzymes and systems |
| US10669540B2 (en) | 2015-06-18 | 2020-06-02 | The Board Institute, Inc. | CRISPR enzymes and systems |
| EP3502253B1 (en) | 2015-06-18 | 2020-05-27 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| US10648020B2 (en) | 2015-06-18 | 2020-05-12 | The Broad Institute, Inc. | CRISPR enzymes and systems |
| EP3470519B1 (en) | 2015-06-18 | 2020-02-12 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| EP3310917B1 (en) | 2015-06-18 | 2020-02-12 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| WO2016205749A1 (en) | 2015-06-18 | 2016-12-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| US10577388B2 (en) | 2015-10-02 | 2020-03-03 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugation process |
| US11555050B2 (en) | 2015-10-02 | 2023-01-17 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugation process |
| WO2017055423A1 (en) * | 2015-10-02 | 2017-04-06 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugation process |
| CN108136041A (en) * | 2015-10-02 | 2018-06-08 | 罗氏创新中心哥本哈根有限公司 | Oligonucleotides conjugation methods |
| CN108136041B (en) * | 2015-10-02 | 2022-12-30 | 罗氏创新中心哥本哈根有限公司 | Oligonucleotide conjugation methods |
| US10450568B2 (en) | 2015-10-09 | 2019-10-22 | Wave Life Sciences Ltd. | Oligonucleotide compositions and methods thereof |
| US10557137B2 (en) | 2015-11-06 | 2020-02-11 | Ionis Pharmaceuticals, Inc. | Modulating apolipoprotein (a) expression |
| US11319536B2 (en) | 2015-11-06 | 2022-05-03 | Ionis Pharmacueticals, Inc. | Modulating apolipoprotein (a) expression |
| WO2017083368A1 (en) * | 2015-11-12 | 2017-05-18 | Pfizer Inc. | Tissue-specific genome engineering using crispr-cas9 |
| JP7268103B2 (en) | 2015-11-12 | 2023-05-02 | ファイザー・インコーポレイテッド | Tissue-specific genome manipulation using CRISPR-Cas9 |
| JP2022000440A (en) * | 2015-11-12 | 2022-01-04 | ファイザー・インコーポレイテッド | TISSUE-SPECIFIC GENOME ENGINEERING USING CRISPR-Cas9 |
| JP2018537448A (en) * | 2015-11-12 | 2018-12-20 | ファイザー・インコーポレイテッド | Tissue-specific genome manipulation using CRISPR-Cas9 |
| US10851367B2 (en) | 2015-11-12 | 2020-12-01 | Pfizer Inc. | Tissue-specific genome engineering using CRISPR-Cas9 |
| WO2017106657A1 (en) | 2015-12-18 | 2017-06-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| US10246709B2 (en) | 2016-03-07 | 2019-04-02 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands for therapeutic compounds |
| EP3786297A1 (en) * | 2016-03-14 | 2021-03-03 | F. Hoffmann-La Roche AG | Oligonucleotides for reduction of pd-l1 expression |
| CN114085836A (en) * | 2016-03-14 | 2022-02-25 | 豪夫迈·罗氏有限公司 | Oligonucleotides for reducing PD-L1 expression |
| CN114085836B (en) * | 2016-03-14 | 2024-01-26 | 豪夫迈·罗氏有限公司 | Oligonucleotides for reducing PD-L1 expression |
| KR102580776B1 (en) | 2016-03-14 | 2023-09-20 | 에프. 호프만-라 로슈 아게 | Oligonucleotides for reduction of pd-l1 expression |
| WO2017157899A1 (en) * | 2016-03-14 | 2017-09-21 | F. Hoffmann-La Roche Ag | Oligonucleotides for reduction of pd-l1 expression |
| US10745480B2 (en) | 2016-03-14 | 2020-08-18 | Hoffmann-La Roche, Inc. | Oligonucleotides for reduction of PD-L1 expression |
| RU2747822C2 (en) * | 2016-03-14 | 2021-05-14 | Ф. Хоффманн-Ля Рош Аг | Oligonucleotides for lowering pd-l1 expression |
| US11466081B2 (en) | 2016-03-14 | 2022-10-11 | Hoffmann-La Roche Inc. | Oligonucleotides for reduction of PD-L1 expression |
| CN108779465A (en) * | 2016-03-14 | 2018-11-09 | 豪夫迈·罗氏有限公司 | Oligonucleotides for reducing PD-L1 expression |
| KR20210120131A (en) * | 2016-03-14 | 2021-10-06 | 에프. 호프만-라 로슈 아게 | Oligonucleotides for reduction of pd-l1 expression |
| KR20220100095A (en) * | 2016-03-14 | 2022-07-14 | 에프. 호프만-라 로슈 아게 | Oligonucleotides for reduction of pd-l1 expression |
| CN114736901A (en) * | 2016-03-14 | 2022-07-12 | 豪夫迈·罗氏有限公司 | Oligonucleotides for reducing PD-L1 expression |
| CN114736900A (en) * | 2016-03-14 | 2022-07-12 | 豪夫迈·罗氏有限公司 | Oligonucleotides for reducing PD-L1 expression |
| KR102417646B1 (en) | 2016-03-14 | 2022-07-07 | 에프. 호프만-라 로슈 아게 | Oligonucleotides for reduction of pd-l1 expression |
| CN108779465B (en) * | 2016-03-14 | 2022-05-13 | 豪夫迈·罗氏有限公司 | Oligonucleotides for reducing PD-L1 expression |
| US10829555B2 (en) | 2016-03-14 | 2020-11-10 | Hoffman-La Roche Inc. | Oligonucleotides for reduction of PD-L1 expression |
| JP2020172488A (en) * | 2016-03-14 | 2020-10-22 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | Oligonucleotides for reduction of pd-l1 expression |
| JP7002603B2 (en) | 2016-03-14 | 2022-02-10 | エフ.ホフマン-ラ ロシュ アーゲー | Oligonucleotide for reducing PD-L1 expression |
| WO2017184786A1 (en) | 2016-04-19 | 2017-10-26 | The Broad Institute Inc. | Cpf1 complexes with reduced indel activity |
| WO2017189308A1 (en) | 2016-04-19 | 2017-11-02 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| US11286478B2 (en) | 2016-04-19 | 2022-03-29 | The Broad Institute, Inc. | Cpf1 complexes with reduced indel activity |
| WO2017184768A1 (en) | 2016-04-19 | 2017-10-26 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2018035387A1 (en) | 2016-08-17 | 2018-02-22 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| WO2018035388A1 (en) | 2016-08-17 | 2018-02-22 | The Broad Institute, Inc. | Novel crispr enzymes and systems |
| US11174481B2 (en) | 2016-09-02 | 2021-11-16 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands |
| US10294474B2 (en) | 2016-09-02 | 2019-05-21 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands |
| WO2018064371A1 (en) | 2016-09-30 | 2018-04-05 | The Regents Of The University Of California | Rna-guided nucleic acid modifying enzymes and methods of use thereof |
| US11400161B2 (en) | 2016-10-06 | 2022-08-02 | Ionis Pharmaceuticals, Inc. | Method of conjugating oligomeric compounds |
| WO2019060746A1 (en) | 2017-09-21 | 2019-03-28 | The Broad Institute, Inc. | Systems, methods, and compositions for targeted nucleic acid editing |
| WO2019094983A1 (en) | 2017-11-13 | 2019-05-16 | The Broad Institute, Inc. | Methods and compositions for treating cancer by targeting the clec2d-klrb1 pathway |
| WO2019099943A1 (en) | 2017-11-16 | 2019-05-23 | Astrazeneca Ab | Compositions and methods for improving the efficacy of cas9-based knock-in strategies |
| CN112400020A (en) * | 2018-05-08 | 2021-02-23 | 莱古路斯治疗法股份有限公司 | GALNAC conjugated modified oligonucleotides as MIR-122 inhibitors with HCV antiviral activity with reduced hyperbilirubinemia side effects |
| WO2019217369A1 (en) * | 2018-05-08 | 2019-11-14 | Regulus Therapeutics Inc. | Galnac conjugated modified oligonucleotide as mir-122 inhibitor having hcv antiviral activity with reduced hyperbilirubinemia side-effect |
| US11761029B2 (en) | 2018-08-01 | 2023-09-19 | Mammoth Biosciences, Inc. | Programmable nuclease compositions and methods of use thereof |
| US11273442B1 (en) | 2018-08-01 | 2022-03-15 | Mammoth Biosciences, Inc. | Programmable nuclease compositions and methods of use thereof |
| WO2020033601A1 (en) | 2018-08-07 | 2020-02-13 | The Broad Institute, Inc. | Novel cas12b enzymes and systems |
| WO2020049158A1 (en) | 2018-09-07 | 2020-03-12 | Astrazeneca Ab | Compositions and methods for improved nucleases |
| US11505578B2 (en) | 2018-09-18 | 2022-11-22 | Vnv Newco Inc. | Endogenous Gag-based capsids and uses thereof |
| US11447527B2 (en) | 2018-09-18 | 2022-09-20 | Vnv Newco Inc. | Endogenous Gag-based capsids and uses thereof |
| US11174470B2 (en) | 2019-01-04 | 2021-11-16 | Mammoth Biosciences, Inc. | Programmable nuclease improvements and compositions and methods for nucleic acid amplification and detection |
| EP4219700A1 (en) | 2019-03-07 | 2023-08-02 | The Regents of the University of California | Crispr-cas effector polypeptides and methods of use thereof |
| DE212020000516U1 (en) | 2019-03-07 | 2022-01-17 | The Regents of the University of California | CRISPR-CAS effector polypeptides |
| WO2020191102A1 (en) | 2019-03-18 | 2020-09-24 | The Broad Institute, Inc. | Type vii crispr proteins and systems |
| WO2020236967A1 (en) | 2019-05-20 | 2020-11-26 | The Broad Institute, Inc. | Random crispr-cas deletion mutant |
| WO2020236972A2 (en) | 2019-05-20 | 2020-11-26 | The Broad Institute, Inc. | Non-class i multi-component nucleic acid targeting systems |
| WO2021055855A1 (en) | 2019-09-20 | 2021-03-25 | The Broad Institute, Inc. | Compositions and methods for delivering cargo to a target cell |
| US11801306B2 (en) | 2020-03-04 | 2023-10-31 | Verve Therapeutics, Inc. | Compositions and methods for targeted RNA delivery |
| US11207416B2 (en) | 2020-03-04 | 2021-12-28 | Verve Therapeutics, Inc. | Compositions and methods for targeted RNA delivery |
| WO2023052508A2 (en) | 2021-09-30 | 2023-04-06 | Astrazeneca Ab | Use of inhibitors to increase efficiency of crispr/cas insertions |
| WO2023196818A1 (en) | 2022-04-04 | 2023-10-12 | The Regents Of The University Of California | Genetic complementation compositions and methods |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11739332B2 (en) | Antisense oligomers targeting PCSK9 | |
| WO2014118272A1 (en) | Antimir-122 oligonucleotide carbohydrate conjugates | |
| EP2920307B1 (en) | Anti apob antisense conjugate compounds | |
| EP2951305B1 (en) | Lna oligonucleotide carbohydrate conjugates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14702541 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12016500291 Country of ref document: PH |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14702541 Country of ref document: EP Kind code of ref document: A1 |