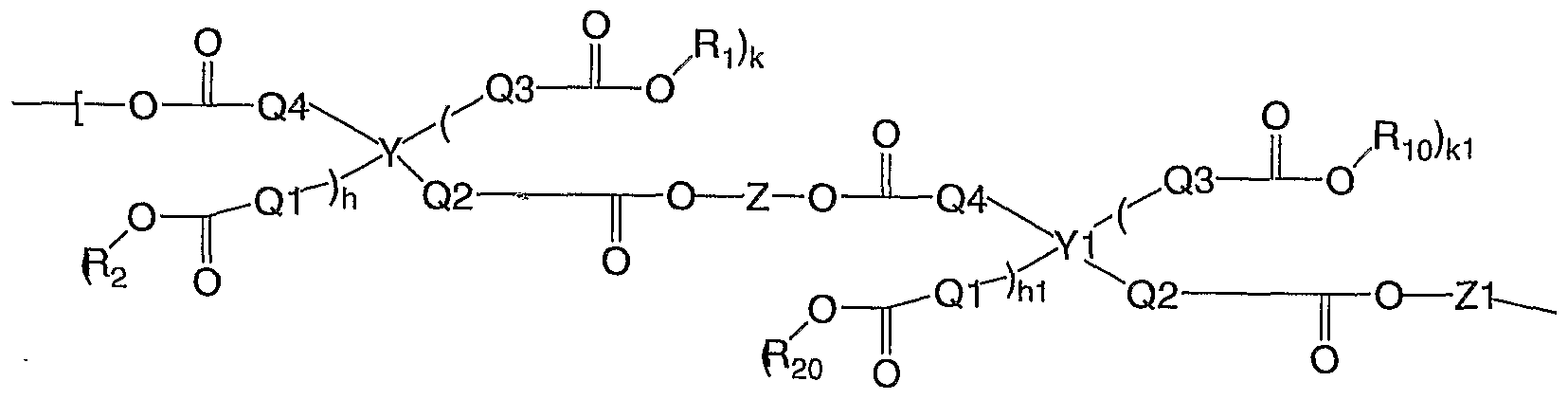WO2010004377A1 - Antireflective coating compositions - Google Patents
Antireflective coating compositions Download PDFInfo
- Publication number
- WO2010004377A1 WO2010004377A1 PCT/IB2009/005458 IB2009005458W WO2010004377A1 WO 2010004377 A1 WO2010004377 A1 WO 2010004377A1 IB 2009005458 W IB2009005458 W IB 2009005458W WO 2010004377 A1 WO2010004377 A1 WO 2010004377A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- containing compound
- independently
- polymer
- structural unit
- coating composition
- Prior art date
Links
- 0 C*OC(CC(C(CC(OCCN(C(N(CCO)C(N1CCOC(CC(C(CC(OC(C(C(OC)=O)IC)C(OC)=O)=O)C(OCC(C)O)=O)C(OCC(CN(C(N(CC2OC2)C(N2CC3OC3)=O)=O)C2=O)O)=O)=O)=O)=O)C1=O)=O)C(OCC(C)O)=O)C(OCC(CN(C(N(CC1OC1)C(N1CC2OC2)=O)=O)C1=O)O)=O)=O Chemical compound C*OC(CC(C(CC(OCCN(C(N(CCO)C(N1CCOC(CC(C(CC(OC(C(C(OC)=O)IC)C(OC)=O)=O)C(OCC(C)O)=O)C(OCC(CN(C(N(CC2OC2)C(N2CC3OC3)=O)=O)C2=O)O)=O)=O)=O)=O)C1=O)=O)C(OCC(C)O)=O)C(OCC(CN(C(N(CC1OC1)C(N1CC2OC2)=O)=O)C1=O)O)=O)=O 0.000 description 2
- JMYMNFUFFIVTIK-ZZXKWVIFSA-N CC/C=C/N(C(N(CCC1OC1)C(N1CCC2OC2)=O)=O)C1=O Chemical compound CC/C=C/N(C(N(CCC1OC1)C(N1CCC2OC2)=O)=O)C1=O JMYMNFUFFIVTIK-ZZXKWVIFSA-N 0.000 description 2
- YKGOGSRVNALHLB-UHFFFAOYSA-N CCCCCN(C(N(CCCC1OC1)C(N1CCCC2OC2)=O)=O)C1=O Chemical compound CCCCCN(C(N(CCCC1OC1)C(N1CCCC2OC2)=O)=O)C1=O YKGOGSRVNALHLB-UHFFFAOYSA-N 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D167/00—Coating compositions based on polyesters obtained by reactions forming a carboxylic ester link in the main chain; Coating compositions based on derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/20—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the epoxy compounds used
- C08G59/32—Epoxy compounds containing three or more epoxy groups
- C08G59/3236—Heterocylic compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D163/00—Coating compositions based on epoxy resins; Coating compositions based on derivatives of epoxy resins
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B1/00—Optical elements characterised by the material of which they are made; Optical coatings for optical elements
- G02B1/10—Optical coatings produced by application to, or surface treatment of, optical elements
- G02B1/11—Anti-reflection coatings
- G02B1/111—Anti-reflection coatings using layers comprising organic materials
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/09—Photosensitive materials characterised by structural details, e.g. supports, auxiliary layers
- G03F7/091—Photosensitive materials characterised by structural details, e.g. supports, auxiliary layers characterised by antireflection means or light filtering or absorbing means, e.g. anti-halation, contrast enhancement
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/68—Polyesters containing atoms other than carbon, hydrogen and oxygen
- C08G63/685—Polyesters containing atoms other than carbon, hydrogen and oxygen containing nitrogen
- C08G63/6854—Polyesters containing atoms other than carbon, hydrogen and oxygen containing nitrogen derived from polycarboxylic acids and polyhydroxy compounds
Definitions
- the present invention relates to a novel antireflective coating composition and its use in image processing by forming a thin layer of the novel antireflective coating composition between a reflective substrate and a photoresist coating.
- compositions are particularly useful in the fabrication of semiconductor devices by photolithographic techniques.
- Photoresist compositions are used in microlithography processes for making miniaturized electronic components such as in the fabrication of computer chips and integrated circuits.
- a thin coating of film of a photoresist composition is first applied to a substrate material, such as silicon wafers used for making integrated circuits.
- the coated substrate is then baked to evaporate any solvent in the photoresist composition and to fix the coating onto the substrate.
- the baked coated surface of the substrate is next subjected to an image-wise exposure to radiation.
- This radiation exposure causes a chemical transformation in the exposed areas of the coated surface.
- Visible light, ultraviolet (UV) light, electron beam and X-ray radiant energy are radiation types commonly used today in microlithographic processes.
- UV light, ultraviolet (UV) light, electron beam and X-ray radiant energy are radiation types commonly used today in microlithographic processes.
- the coated substrate is treated with a developer solution to dissolve and remove either the radiation-exposed or the unexposed areas of the photoresist.
- the trend towards the miniaturization of semiconductor devices has led to the use of new photoresists that are sensitive to lower and lower wavelengths of radiation and has also led to the use of sophisticated multilevel systems to overcome difficulties associated with such miniaturization.
- bottom antireflective coating provides the best solution for the elimination of reflectivity.
- the bottom antireflective coating is applied to the substrate prior to coating with the photoresist and prior to exposure.
- the photoresist is exposed imagewise and developed.
- the antireflective coating in the exposed area is then etched, typically in gaseous plasma, and the photoresist pattern is thus transferred to the substrate.
- the etch rate of the antireflective film should be relatively high in comparison to the photoresist so that the antireflective film is etched without excessive loss of the photoresist film during the etch process.
- Antireflective coatings must also possess the correct absorption and refractive index at the wavelength of exposure to achieve the desired lithographic properties.
- antireflective coating that functions well at exposures less than 300 nm.
- Such antireflective coatings need to have high etch rates and be sufficiently absorbing with the correct refractive index to act as antireflective coatings.
- the invention describes an antireflective coating composition which contains a novel polymer without an aromatic chromophore, which finds applications in anti-reflective coating materials in high NA lithography.
- the materials have ultra high etch rates because of the backbone of the polymer and the absence of aromatic chromophore attached to the polymer.
- the anti reflective coating composition comprising a polymer which does not contain an aromatic chromophore, an acid generator, and optionally a crosslinking agent, where the polymer comprises a structural unit derived from a diacid, a triacid, a dianhydride or its corresponding tetraacid or a mixture thereof and a structural unit derived from a diol, a dithiol, a triol, a trithiol, a diepoxy containing compound, a triepoxy containing compound, or mixture thereof, where the diol, dithiol, triol, trithiol, the diepoxy containing compound, or triepoxy containing compound contains one or more nitrogen and/or sulfur atoms or contains one or more alkylene groups.
- the polymer can comprise a repetitive unit of structure (1) or (2)
- each of Y and Y1 independently are a non-aromatic linking moiety; each of Z and Z1 independently are the structural unit derived from a diol, a dithiol, a triol, a trithiol, a diepoxy containing compound, or a triepoxy containing compound; each of R 1 and Ri 0 independently are hydrogen or the structural unit derived from a monoepoxy containing compound; each of R 2 and R 2 o independently are Ri or the structural unit derived from a dithiol, a trithiol, a diepoxy containing compound, or a triepoxy containing compound; each of Q1 , Q2, Q3, and Q4 are independently a direct bond or an unsubstituted or substituted Ci -4 alkylene; each of h and hi independently are 0 or 1; and each of k or k1 independently are 0 or 1.
- the present invention is also related to a polymer which does not contain an aromatic chromophore comprising a structural unit derived from a diacid, a triacid, a dianhydride or its corresponding tetraacid or a mixture thereof and a structural unit derived from a diol, a dithiol, a triol, a trithiol, a diepoxy containing compound, a triepoxy containing compound, or mixture thereof, where the diol, dithiol, triol, trithiol, the diepoxy containing compound, or triepoxy containing compound contains one or more nitrogen and/or sulfur atoms or contains one or more alkylene groups.
- the present invention also relates to a coated substrate comprising a substrate having thereon an antireflective coating layer formed from the antireflective coating composition disclosed herein, where the antireflective coating layer has an absorption parameter (k) in the range of 0.01 ⁇ k ⁇ 0.35 when measured at 193 nm.
- the present invention also relates to a process for forming an image comprising, a) coating and baking a substrate with the antireflective coating composition disclosed herein; b) coating and baking a photoresist film on top of the antireflective coating; c) imagewise exposing the photoresist; d) developing an image in the photoresist; e) optionally, baking the substrate after the exposing step.
- Fig. 1 illustrates the principle of anomalous dispersion with the following meanings: Ca: Cauchy correlation Ex ⁇ : Exposure ⁇ (high n low k region) ab: absorption band ha: half measure
- the invention describes an antireflective coating composition which contains a novel polymer without an aromatic chromophore, which finds applications in anti-reflective coating materials in high NA lithography.
- the materials have ultra high etch rates because of the backbone of the polymer and the absence of aromatic chromophore attached to the polymer.
- the antirefiective coating composition comprising a polymer which does not contain an aromatic chromophore, an acid generator, and optionally a crosslinking agent, where the polymer comprises a structural unit derived from a diacid, a triacid, a dianhydride or its corresponding tetraacid or a mixture thereof and a structural unit derived from a diol, a dithiol, a triol, a trithiol, a diepoxy containing compound, a triepoxy containing compound, or mixture thereof, where the diol, dithiol, triol, trithiol, the diepoxy containing compound, or triepoxy containing compound contains one or more nitrogen and/or sulfur atoms or contains one or more alkylene groups.
- the polymer can comprise a repetitive unit of structure (1) or (2)
- each of Y and Y1 independently are a non-aromatic linking moiety; each of Z and Z1 independently are the structural unit derived from a diol, a dithiol, a triol, a trithiol, a diepoxy containing compound, or a triepoxy containing compound; each of Ri and Ri 0 independently are hydrogen or the structural unit derived from a monoepoxy containing compound; each of R 2 and R20 independently are Ri or the structural unit derived from a dithiol, a trithiol, a diepoxy containing compound, or a triepoxy containing compound; each of Q1 , Q2, Q3, and Q4 are independently a direct bond or an unsubstituted or substituted Ci -4 alkylene; each of h and hi independently are 0 or 1; and each of k or k1 independently are 0 or 1.
- the present invention is also related to a polymer which does not contain an aromatic chromophore comprising a structural unit derived from a diacid, a triacid, a dianhydride or its corresponding tetraacid or a mixture thereof and a structural unit derived from a diol, a dithiol, a triol, a trithiol, a diepoxy containing compound, a triepoxy containing compound, or mixture thereof, where the diol, dithiol, triol, trithiol, the diepoxy containing compound, or triepoxy containing compound contains one or more nitrogen and/or sulfur atoms or contains one or more alkylene groups.
- the present invention also relates to a coated substrate comprising a substrate having thereon an anti reflective coating layer formed from the antireflective coating composition disclosed herein, where the anti reflective coating layer has an absorption parameter (k) in the range of 0.01 ⁇ k ⁇ 0.35 when measured at 193 nm.
- the present invention also relates to a process for forming an image comprising, a) coating and baking a substrate with the antireflective coating composition disclosed herein; b) coating and baking a photoresist film on top of the antireflective coating; c) imagewise exposing the photoresist; d) developing an image in the photoresist; e) optionally, baking the substrate after the exposing step.
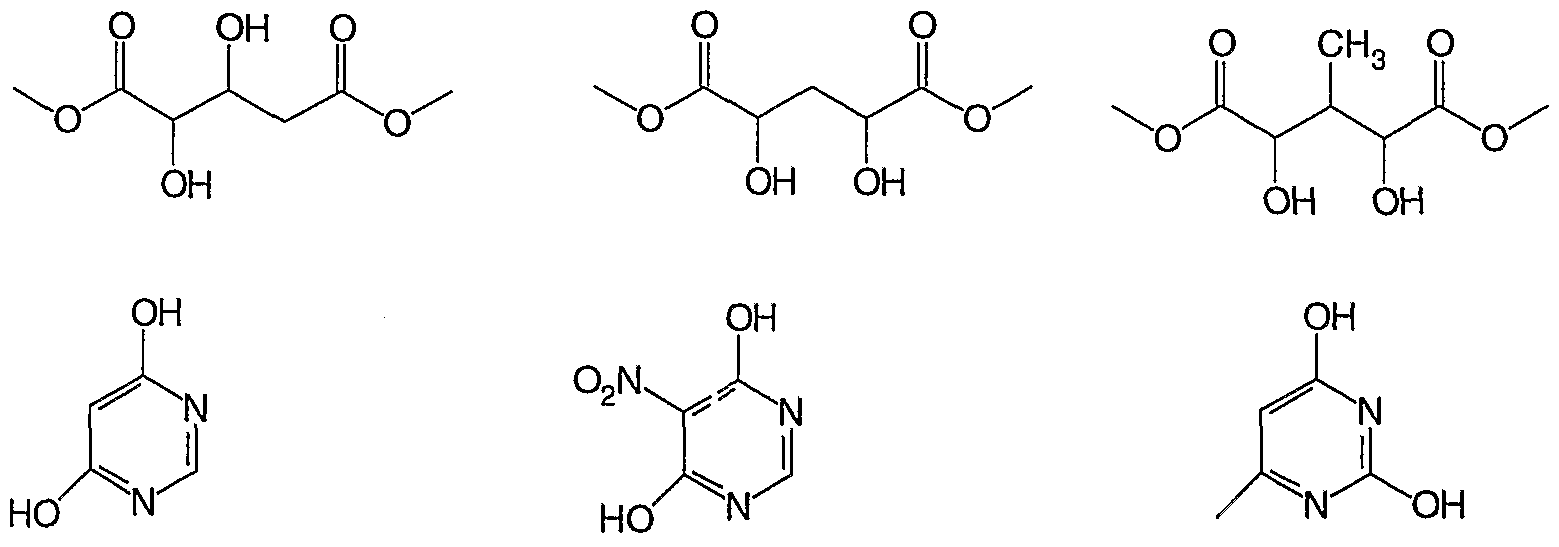
- the polymers useful in the present invention are formed by reacting a dianhydride (e.g., 1 ,2,3,4-butanetetracarboxylic acid dianhydride, 1 ,2,3,4- pentanetetracarboxylic acid dianhydride, 1 ,2,4,5-cyclohexanetetracarboxylic acid dianhydride, 1 ,2,3,4-cyclopentanetetracarboxylic acid dianhydride, 1 ,2,3,4- bicyclohexenetetracarboxylic acid dianhydride, 2,3,4,5- tetrahydrofurantetracarboxylic acid dianhydride, 1 ,2,3,4- cyclobutanetetracarboxylic acid dianhydride, 1 ,2,5,6-cycloocta-1 ,5- dienetetracarboxylic acid dianhydride, 2,3,5-tricarboxycyclopentylacetic acid dianhydride, 3,5,6-tricarboxynorborn
- neopentyl glycol, dialkyl tartrate, tris(2-hydroxyethyl) cyanuric acid 1 ,4- dithiane-2,5-diol, 2-butene-1 ,4-diol, 2,3-dibromo-2-butene-1 ,4-diol, 1 ,3- propanediol, 1 ,6-hexanediol, 2,2-dimethyl-1 ,3-propanediol, diethylene glycol, triethylene glycol, and the like, etc in the presence of tetraalkylammonium chloride catalyst.
- these polymers can also be made by reacting diacids or triacids with diols, dithiols, triols, trithiols, diepoxy and triepoxy compounds.
- diacids and triacids include 1 ,4-butane dicarboxylic acid, 1 ,8-octane dicarboxylic acid, 1 ,4-cyclohexane dicarboxylic acid, fumaric acid, muconic acid, and the like.
- Examples of the dianhydrides, diacids, and triacids include where R 6 is linear or branched alkylene chain of 2 to 20 carbon atoms, which may also contain an ether oxygen or ester groups, and R 7 is hydrogen or methyl.
- R 6 examples include
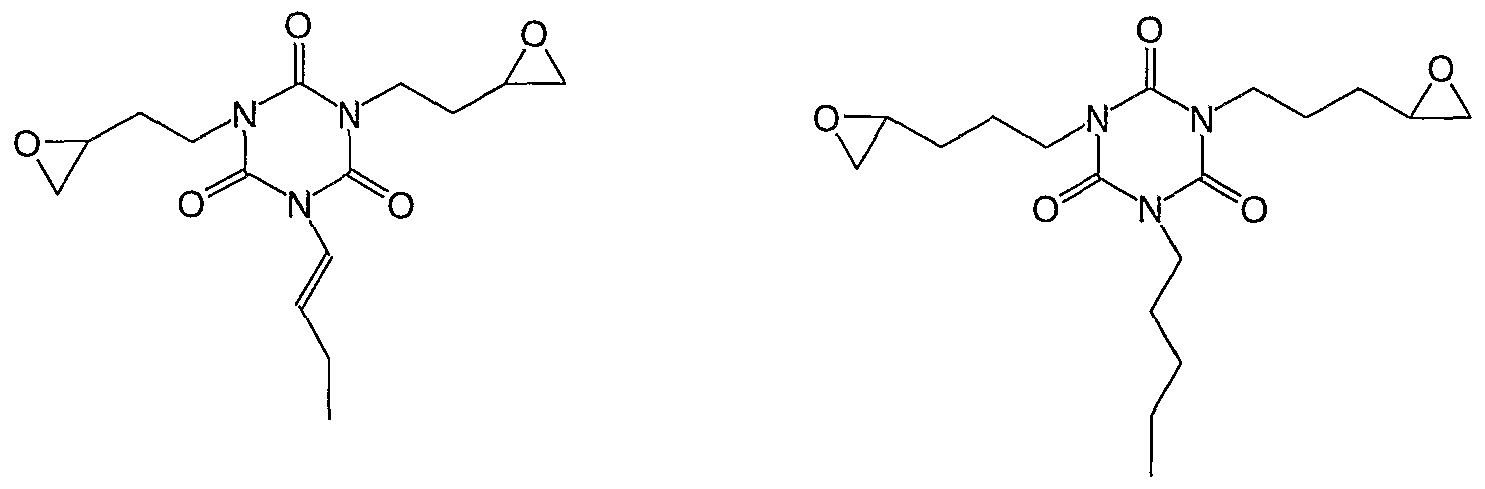
- diepoxy and triepoxy compounds examples include
- diols, dithiols, triols, and trithiols examples include
- R 1 00 is hydrogen, alkyl, or alkenyl.
- the carboxylic acid groups in the prepolymer then undergo ring opening reactions with propylene oxide to give final product.
- a molecule with more than one epoxy functional groups e.g. ethylene glycol diglycidyl ether. 1 ,4- butanediol diglycidyl ether or tris(2,3-epoxypropyl) isocyanurate
- Polyester can also be formed by reaction of a compound containing multiple carboxylic acid groups with a di-/tri-epoxy monomer or a diol/triol monomer.
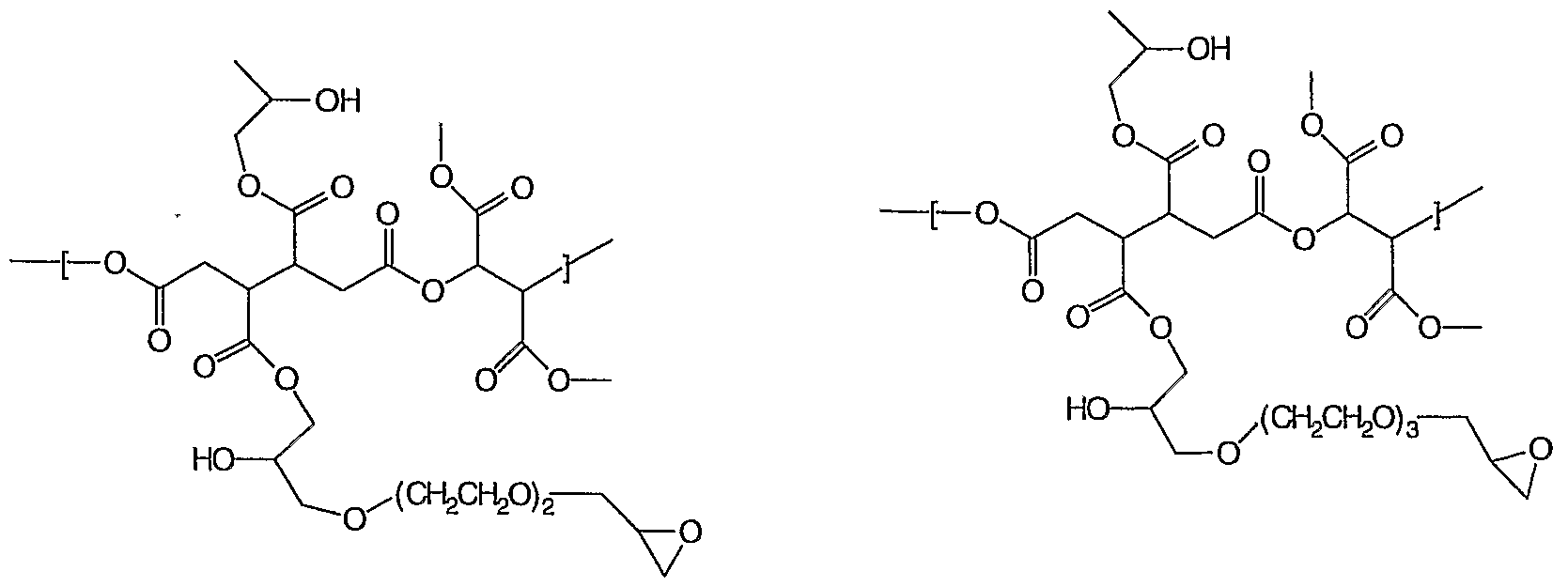
- Examples of Y and Y1 for structures (1 ) and (2) include
- repeat units of structure (1) and (2) include
- the optional crosslinking agent can be an aminoplast.
- the aminoplast can be substituted by two or more alkoxy groups can be based on aminoplasts such as, for example, glycoluril-aldehyde resins, melamine-aldehyde resins, benzoguanamine-aldehyde resins, and urea-aldehyde resins.
- Examples of the aldehyde include formaldehyde, acetaldehyde, etc. In some instances, three or four alkoxy groups are useful.
- Monomeric, methylated glycoluril-formaldehyde resins are an example.
- One example is tetra(alkoxymethyl)glycoluril.
- tetra(alkoxymethyl)glycoluril may include, e.g., tetra(methoxymethyl)glycoluril, tetra(ethoxymethyl)glycoluril, tetra(n-propoxymethyl)glycoluril, tetra(i- propoxymethyl)glycoluril, tetra(n-butoxymethy1)glycoluril and tetra(t- butoxymethyl)glycoluril.
- Tetra(methoxymethyl)glycoluril is available under the trademark POWDERLiNK from Cytec Industries (e.g., POWDERLINK 1174).
- Other examples include methylpropyltetramethoxymethyl glycoluril, and methylphenyltetramethoxymethyl glycoluril.
- aminoplasts are commercially available from Cytec Industries under the trademark CYMEL and from Monsanto Chemical Co. under the trademark RESIMENE.
- Condensation products of other amines and amides can also be employed, for example, aldehyde condensates of triazines, diazines, diazoles, guanidines, guanimines and alkyl- and aryl-substituted derivatives of such compounds, including alkyl- and aryl-substituted melamines.
- Some examples of such compounds are N,N'-dimethyl urea, benzourea, dicyandiamide, formaguanamine, acetoguanamine, ammeline, 2-chloro-4,6-diamino-1 ,3,5- triazine, 6-methyl-2,4-diamino,1 ,3,5-traizine, 3,5-diaminotriazole, triaminopyrimidine,2-mercapto-4,6-diamino-pyrimidine, 3,4,6-tris(ethylamino)- 1 ,3,5-triazine, tris(alkoxycarbonylamino)triazine, N, N, N', N'- tetramethoxym ethyl urea and the like.
- aminoplasts include compounds having the following structures:
- the acid generator used with the present invention preferably a thermal acid generator is a compound which, when heated to temperatures greater than 9O 0 C. and less than 250 0 C, generates an acid.
- the acid in combination with the cros ⁇ ilinker, crosslinks the polymer.
- the anti reflective coating layer after heat treatment becomes insoluble in the solvents used for coating photoresists, and furthermore, is also insoluble in the alkaline developer used to image the photoresist.
- the thermal acid generator is activated at 90 0 C. and more preferably at above 120 0 C, and even more preferably at above 150 0 C.
- the antireflective coating layer is heated for a sufficient length of time to crosslink the coating.
- acids and thermal acid generators are butane sulfonic acid, triflic acid, nanofluorobutane sulfonic acid, nitrobenzyl tosylates, such as 2- nitrobenzyl tosylate, 2,4-dinitrobenzyl tosylate, 2,6-dinitrobenzyl tosylate, 4- nitrobenzyl tosylate; benzenesulfonates such as 2-trifluoromethyl-6-nitrobenzyl 4- chlorobenzenesulfonate, 2-trifluoromethyl-6-nitrobenzyl 4-nitro benzenesulfonate; phenolic sulfonate esters such as phenyl, 4-methoxybenzenesulfonate; alkyl ammonium salts of organic acids, such as triethylammonium salt of 10- camphorsulfonic acid, and the like.
- nitrobenzyl tosylates such as 2- nitrobenzyl tosylate, 2,4-
- Thermal acid generators are preferred over free acids, although free acids may also be used, in the novel antireflective composition, since it is possible that over time the shelf stability of the antireflective solution will be effected by the presence of the acid, if the polymer were to crosslink in solution. Thermal acid generators are only activated when the antireflective film is heated on the substrate. Additionally, mixtures of thermal acids and free acids may be used. Although thermal acid generators are preferred for crosslinking the polymer efficiently, an anti-reflective coating composition comprising the polymer and crosslinking agent may also be used, where heating crosslinks the polymer. Examples of a free acid are, without limitation, strong acids, such as sulfonic acids. Sulfonic acids such as toluene sulfonic acid, triflic acid or mixtures of these are preferred.
- the novel composition may further contain a photoacid generator, examples of which without limitation, are onium salts, sulfonate compounds, nitrobenzyl esters, triazines, etc.
- the preferred photoacid generators are onium salts and sulfonate esters of hydoxyimides, specifically diphenyl iodnium salts, triphenyl sulfonium salts, dialkyl iodonium salts, triakylsulfonium salts, and mixtures thereof.
- One or more cross-linking catalysts can be used in the composition.
- solvents for the coating composition include alcohols, esters, glymes, ethers, glycol ethers, glycol ether esters, ketones, lactones, cyclic ketones, and mixtures thereof.
- solvents include, but are not limited to, propylene glycol methyl ether, propylene glycol methyl ether acetate, cyclohexanone, 2-heptanone, ethyl 3-ethoxy-propionate, propylene glycol methyl ether acetate, ethyl lactate, gamma valerolactone, methyl 3-methoxypropionate, and mixtures thereof.
- the solvent is typically present in an amount of from about 40 to about 99 weight percent.
- the addition of lactone solvents is useful in helping flow characteristics of the antireflective coating composition when used in layered systems.
- the lactone solvent comprises about 1 to about 10% of the solvent system.
- ⁇ -valerolactone is a useful lactone solvent.
- the amount of the polymer in the present composition can vary from about 100 weight % to about 1 weight %relative to the solid portion of the composition.
- the amount of the crosslinker in the present composition when used, can vary from 0 weight % to about 50 weight % relative to the solid portion of the composition.
- the amount of the acid generator in the present composition can vary from 0.1 weight % to about 10 weight % relative to the solid portion of the composition.
- the present composition can optionally comprise additional materials typically found in antireflective coating compositions such as, for example, monomeric dyes, lower alcohols, surface leveling agents, adhesion promoters, antifoaming agents, etc, provided that the performance is not negatively impacted.
- additional materials typically found in antireflective coating compositions such as, for example, monomeric dyes, lower alcohols, surface leveling agents, adhesion promoters, antifoaming agents, etc, provided that the performance is not negatively impacted.
- composition is coated on top of the substrate and is further subjected to dry etching, it is envisioned that the composition is of sufficiently low metal ion level and purity that the properties of the semiconductor device are not adversely affected.
- Treatments such as passing a solution of the polymer, or compositions containing such polymers, through an ion exchange column, filtration, and extraction processes can be used to reduce the concentration of metal ions and to reduce particles.
- the optical characteristics of the antireflective coating are optimized for the exposure wavelength and other desired lithographic characteristics.
- the absorption parameter (k) of the novel composition for 193 nm exposure ranges from about 0.01 to about 1.0, preferably from about 0.1 to about 0.35 as measured using ellipsometry.
- the value of the refractive index (n) ranges from about 1.25 to about 2.0, preferably from about 1.7 to about 2.0. Due to the good absorption characteristics of this composition at 193 nm, very thin antireflective films of the order of about 40 nm may be used. This is particularly advantageous when using a nonaromatic photoresist, such as those sensitive at 193 nm, 157 nm and lower wavelengths, where the photoresist films are thin and must act as an etch mask for the antireflective film.
- the substrates over which the antireflective coatings are formed can be any of those typically used in the semiconductor industry. Suitable substrates include, without limitation, silicon, silicon substrate coated with a metal surface, copper coated silicon wafer, copper, substrate coated with antireflective coating, aluminum, polymeric resins, silicon dioxide, metals, doped silicon dioxide, silicon nitride, tantalum, polysilicon, ceramics, aluminum/copper mixtures; gallium arsenide and other such Group Ill/V compounds.
- the substrate may comprise any number of layers made from the materials described above.
- the coating composition can be coated on the substrate using techniques well known to those skilled in the art, such as dipping, spincoating or spraying.
- the film thickness of the anti-reflective coating ranges from about 0.01 ⁇ m to about 1 ⁇ m.
- the coating can be heated on a hot plate or convection oven or other well known heating methods to remove any residual solvent and induce crosslinking if desired, and insolubilizing the anti-reflective coatings to prevent intermixing between the anti-reflective coating and the photoresist.
- the preferred range of temperature is from about 90 0 C to about 250 0 C. If the temperature is below 90 0 C then insufficient loss of solvent or insufficient amount of crosslinking takes place, and at temperatures above 300 0 C the composition may become chemically unstable.
- a film of photoresist is then coated on top of the uppermost antireflective coating and baked to substantially remove the photoresist solvent.
- An edge bead remover may be applied after the coating steps to clean the edges of the substrate using
- photoresist compositions there are two types, negative-working and positive-working.
- negative-working photoresist compositions When negative-working photoresist compositions are exposed image-wise to radiation, the areas of the resist composition exposed to the radiation become less soluble to a developer solution (e.g. a cross-linking reaction occurs) while the unexposed areas of the photoresist coating remain relatively soluble to such a solution.
- a developer solution e.g. a cross-linking reaction occurs
- treatment of an exposed negative- working resist with a developer causes removal of the non-exposed areas of the photoresist coating and the creation of a negative image in the coating, thereby uncovering a desired portion of the underlying substrate surface on which the photoresist composition was deposited.
- Negative working photoresist and positive working photoresist compositions and their use are well known to those skilled in the art.
- 193 nm immersion lithography is a viable solution for nodes down to 45 nm node and beyond.
- low "k" BARCs are more suitable for optimum substrate reflectivity control based on simulations.
- the BARC film needs to be thin enough to have desired ⁇ tch selectivity for very small features in thin film lithography.
- the low film thickness requires high n value for BARCs.
- This invention describes an antireflective coating composition which contains a novel polymer with non- aromatic dyes.
- the dye is less absorbing at 193 nm than that of conventional 193 nm BARC, e.g. phenyl or its derivatives.
- the invention takes advantage of anomalous dispersion effects near the absorption maxima ⁇ max (excluding ⁇ max ) by a judicious choice of the dye.
- the dye with absorbance maxima lower than the exposure absorbance (193 nm) is used to achieve hyper n value. Theoretically, a n value that is above the value predicted from the Cauchy correlation is considered high, which covers entire half of the absorbance band in high wavelength area.
- the high n low k material should ideally have absorption maxima, ⁇ max , such that the actinic wavelength is the same as the wavelength ⁇ + that is at half height of the absorption band on the higher wavelength side of the absorption band (Rg. 1).
- the absorption maxima ⁇ max moving from that position in the shorter wavelength direction up to a half length ( ⁇ max - ⁇ .) forces exposure wavelength ⁇ to fall into a typical high n low k region indicated in Fig. 1.
- the amplitude of the refractive index fluctuation is not only determined by the position of ⁇ max but also influenced by the strength of absorbance based on a Kramers-Kronic relation.
- n value can be achieved as long as the dye has extremely strong absorbance corresponding to the anomalous dispersion area.
- the low k requirement of the organic BARC limits the amplitude of n enhancement and adds challenges in dye selection and material development.
- dyes with absorption maxima between 160- 190 nm, preferably between 170-190 nm such as cyanuric acid into the polymer structure.
- optical indices used in the antireflective coating is not the same as the absorption property of organic compound in its pure form or in its liquid solution.
- the absorption spectrum of a dye in coating may shift due to changes of chemical and physical environment such as solvent, additives and possible chemical reactions.
- a dye behaves ideally in solution may not be right for antireflective coating.
- the invention has studied many low k BARC materials with various carefully selected dyes and the structures are presented in this work.
- a process of the instant invention comprises coating a substrate with a coating composition comprising a polymer of the present invention and heating the substrate on a hotplate or convection oven or other well known heating methods at a sufficient temperature for sufficient length of time to remove the coating solvent, and crosslink the polymer if necessary, to a sufficient extent so that the coating is not soluble in the coating solution of a photoresist or in a aqueous alkaline developer.
- An edge bead remover may be applied to clean the edges of the substrate using processes well known in the art.
- the heating ranges in temperature from about 70°C to about 250°C.
- a film of a photoresist composition is then coated on top of the anti- reflective coating and baked to substantially remove the photoresist solvent.
- the photoresist is image-wise exposed and developed in an aqueous developer to remove the treated resist.
- An optional heating step can be incorporated into the process prior to development and after exposure.
- the process of coating and imaging photoresists is well known to those skilled in the art and is optimized for the specific type of resist used.
- the patterned substrate can then be dry etched in a suitable etch chamber to remove the exposed portions of the anti-reflective film, with the remaining photoresist acting as an etch mask.
- Various gases are known in the art for etching organic anti reflective coatings, such as O2, Cb, F2 and CF 4 . This process is generally known as a bilayer process.
- An intermediate layer may be placed between the antireflective coating and the photoresist to prevent intermixing, and is envisioned as lying within the scope of this invention.
- the intermediate layer is an inert polymer cast from a solvent, where examples of the polymer are polysulfones and polyimides.
- a multilayer system for example, a trilayer system, or process is also envisioned within the scope of the invention.
- a trilayer process for example, an organic film is formed on a substrate, an antireflection film is formed on the organic film, and a photoresist film is formed on the antireflection film.
- the organic film can also act as an antireflection film.
- the organic film is formed on a substrate as a lower resist film by spin coating method etc.
- the organic film may or may not then crosslinked with heat or acid after application by spin coating method etc.
- the antireflection film for example that which is disclosed herein, as an intermediate resist film.
- an organic solvent is evaporated, and baking is carried out in order to promote crosslinking reaction to prevent the antireflection film from intermixing with an overlying photoresist film.
- the photoresist film is formed thereon as an upper resist film.
- Spin coating method can be used for forming the photoresist film as with forming the antireflection film.
- pre-baking is carried out. After that, a pattern circuit area is exposed, and post exposure baking (PEB) and development with a developer are carried out to obtain a resist pattern.
- Another trilayer resist process is such when a bottom layer is formed with a carbon etch mask.
- an intermediate layer is formed by using an intermediate resist layer composition containing silicon atoms.
- an antireflection layer based on the antireflection coating composition of the present invention, is formed.
- a top layer is formed by using a top resist layer composition of a photoresist composition.
- the composition for forming the intermediate layer may include polysilsesquioxane-based silicone polymer, tetraorthosilicate glass (TEOS), and the like.
- the top resist layer composition of a photoresist composition preferably comprises a polymer without a silicon atom.
- a top resist layer comprising a polymer without a silicon atom has an advantage of providing superior resolution to a top resist layer comprising a polymer containing silicon atoms.
- PEB post exposure baking
- the wafer was submitted for evaluation of optical parameters using a spectroscopic Ellipsometer.
- the optimized refractive index "n” at 193 nm and the absorption parameter "k” are listed in Table 1.
- PGMEA/PGME 70/30 solvent to make a 3.3 wt% solution.
- 0.1 g of 10 % nanofluorobutanesulfonic acid /triethylamine in of PGMEA/PGME 70/30 solvent was added in the polymer solution.
- the mixture was filtered through a micro filter with a pore size of 0.2 ⁇ m and coated on a silicon wafer and baked at 200°C for 90 seconds.
- the wafer was submitted for evaluation of optical parameters using a spectroscopic Ellipsometer.
- the optimized refractive index "n" at 193 nm and the absorption parameter "k" are listed in Table 1.
- Synthesis Example B were dissolved in 20 g of PGMEA/PGME (70/30) solvent to make a ⁇ 5 wt% solution. 1% of nanofluorobatane sulfonic acid /triethylamine was added in the polymer solution. The mixture then was filtered through a micro filter with a pore size of 0.2 ⁇ m.
- Formulation Example 1 was evaluated using T83472 photoresist (product of AZ Electronic Materials USA Corp., Somerville, NJ). About 140 nm thick film of Litho Formulation Example 1 was coated and baked at 200 0 C for 90 seconds on a silicon wafer. Then a 190 nm thick T83472 photoresist solution was coated and baked at 115°C for 60 seconds. The wafer was then imagewise exposed using a Nikon NSR-306D 193 nm scanner with 0.85 NA, under dipole Y illumination of 0.9 sigma with PSM mask. The exposed wafer was baked at 110 0 C for 60 seconds and developed in AZ® 300 MIF developer (available from AZ Electronic Materials USA Corp., Somerville, NJ) for 30 seconds. The cleaned wafer was then examined under scanning electron microscope. Line and space patterns showed no standing waves, no footing and no scumming, indicating efficacy of the bottom anti-reflective coating.
- T83472 photoresist product of AZ Electronic Materials USA Corp.,
- Litho performance evaluation of Litho Formulation Example 2 The performance of the anti-reflective coating formulation from Litho Formulation Example 2 was evaluated using T83472 photoresist (product of AZ Electronic Materials USA Corp., Somerville, NJ). About 140 nm thick film of Litho Formulation Example 1 was coated and baked at 200 0 C for 90 seconds on a silicon wafer. Then a 190 nm thick T83472 photoresist solution was coated and baked at 115°C for 60 seconds. The wafer was then imagewise exposed using a Nikon NSR-306D 193 nm scanner with 0.85 NA, under dipole Y illumination of 0.9 sigma with PSM mask.
- T83472 photoresist product of AZ Electronic Materials USA Corp., Somerville, NJ.
- the exposed wafer was baked at 110 0 C for 60 seconds and developed in AZ® 300 MIF developer (available from AZ Electronic Materials USA Corp., Somerville, NJ) for 30 seconds.
- the cleaned wafer was then examined under scanning electron microscope. Line and space patterns showed no standing waves, no footing and no scumming, indicating efficacy of the bottom anti-reflective coating.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020107027320A KR101429326B1 (en) | 2008-07-08 | 2009-04-29 | Antireflective coating compositions |
| CN200980120772.6A CN102056969B (en) | 2008-07-08 | 2009-04-29 | Antireflective coating compositions |
| JP2011517253A JP5568791B2 (en) | 2008-07-08 | 2009-04-29 | Anti-reflective coating composition |
| EP09785890A EP2300518B1 (en) | 2008-07-08 | 2009-04-29 | Antireflective coating compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/133,562 US8221965B2 (en) | 2008-07-08 | 2008-07-08 | Antireflective coating compositions |
| US12/133,562 | 2008-07-08 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010004377A1 true WO2010004377A1 (en) | 2010-01-14 |
| WO2010004377A8 WO2010004377A8 (en) | 2011-03-31 |
Family
ID=40886504
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2009/005458 WO2010004377A1 (en) | 2008-07-08 | 2009-04-29 | Antireflective coating compositions |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US8221965B2 (en) |
| EP (1) | EP2300518B1 (en) |
| JP (1) | JP5568791B2 (en) |
| KR (1) | KR101429326B1 (en) |
| CN (1) | CN102056969B (en) |
| MY (1) | MY155242A (en) |
| TW (1) | TWI512063B (en) |
| WO (1) | WO2010004377A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2258691A1 (en) * | 2009-05-20 | 2010-12-08 | Rohm and Haas Electronic Materials, L.L.C. | Coating compositions for use with an overcoated photoresist |
| WO2012017948A1 (en) * | 2010-08-05 | 2012-02-09 | 日産化学工業株式会社 | Epoxy compound with nitrogen-containing ring |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8329387B2 (en) * | 2008-07-08 | 2012-12-11 | Az Electronic Materials Usa Corp. | Antireflective coating compositions |
| US20100092894A1 (en) * | 2008-10-14 | 2010-04-15 | Weihong Liu | Bottom Antireflective Coating Compositions |
| EP2397507B1 (en) * | 2009-02-10 | 2017-09-20 | Nissan Chemical Industries, Ltd. | Long chain alkylene group-containing epoxy compound |
| US9244352B2 (en) * | 2009-05-20 | 2016-01-26 | Rohm And Haas Electronic Materials, Llc | Coating compositions for use with an overcoated photoresist |
| US8551686B2 (en) * | 2009-10-30 | 2013-10-08 | Az Electronic Materials Usa Corp. | Antireflective composition for photoresists |
| US8507192B2 (en) * | 2010-02-18 | 2013-08-13 | Az Electronic Materials Usa Corp. | Antireflective compositions and methods of using same |
| EP2788398B1 (en) * | 2011-12-15 | 2018-07-25 | Sun Chemical Corporation | Sulfide extended epoxy resins and barrier coatings applications thereof |
| US9170494B2 (en) | 2012-06-19 | 2015-10-27 | Az Electronic Materials (Luxembourg) S.A.R.L. | Antireflective compositions and methods of using same |
| KR101993472B1 (en) | 2012-09-12 | 2019-09-30 | 주식회사 동진쎄미켐 | Compound and composition for forming under layer of resist pattern and method for forming under layer using the same |
| JP5923472B2 (en) * | 2013-09-18 | 2016-05-24 | 四国化成工業株式会社 | Mercaptoalkylglycolurils and their uses |
| JP6207424B2 (en) * | 2014-02-27 | 2017-10-04 | 四国化成工業株式会社 | Mercaptoalkylglycolurils and their uses |
| JP6492509B2 (en) * | 2014-07-28 | 2019-04-03 | Jsr株式会社 | Liquid crystal aligning agent, liquid crystal alignment film, liquid crystal display element, and method for manufacturing liquid crystal display element |
| US10203602B2 (en) * | 2016-09-30 | 2019-02-12 | Rohm And Haas Electronic Materials Korea Ltd. | Coating compositions for use with an overcoated photoresist |
| KR102487404B1 (en) * | 2017-07-26 | 2023-01-12 | 에스케이이노베이션 주식회사 | Polymer for organic bottom anti-reflective coating and bottom anti-reflective coating composition containing the same |
| KR102414150B1 (en) * | 2017-10-17 | 2022-06-29 | 에스케이이노베이션 주식회사 | Polymer for organic bottom anti-reflective coating, bottom anti-reflective coating composition containing the same and process for forming organic bottom anti-reflective coating using the composition |
| KR102264693B1 (en) | 2018-06-11 | 2021-06-11 | 삼성에스디아이 주식회사 | Resist underlayer composition, and method of forming patterns using the composition |
| US20210271168A1 (en) * | 2018-06-26 | 2021-09-02 | Nissan Chemical Corporation | Resist underlying film-forming composition comprising a reaction product with a glycidyl ester compound |
| WO2020026834A1 (en) * | 2018-07-31 | 2020-02-06 | 日産化学株式会社 | Resist underlayer film-forming composition |
| KR102288386B1 (en) * | 2018-09-06 | 2021-08-10 | 삼성에스디아이 주식회사 | Resist underlayer composition, and method of forming patterns using the composition |
| KR20230082015A (en) * | 2020-10-07 | 2023-06-08 | 닛산 가가쿠 가부시키가이샤 | Resist underlayer film-forming composition containing a reaction product of a trifunctional compound |
| CN117603529B (en) * | 2024-01-18 | 2024-03-22 | 汕头市嘉祥塑料制品厂有限公司 | Antibacterial milk powder cover and preparation method thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008017954A2 (en) | 2006-08-10 | 2008-02-14 | Az Electronic Materials Usa Corp. | Antireflective composition for photoresists |
Family Cites Families (82)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3215758A (en) * | 1961-05-10 | 1965-11-02 | Gulf Oil Corp | Condensation polymers |
| US3279940A (en) * | 1963-05-13 | 1966-10-18 | Gulf Oil Corp | Polyethylene and polypropylene containers coated with a polyester resin |
| US3448084A (en) * | 1964-06-10 | 1969-06-03 | Gulf Oil Corp | Polyesters from tris(2-hydroxyethyl) isocyanurate |
| US3477996A (en) * | 1965-03-29 | 1969-11-11 | Allied Chem | Polyesters prepared from tris - (2-hydroxyalkyl) isocyanurates and ethylenically unsaturated dicarboxylic acid anhydrides |
| US3474054A (en) | 1966-09-13 | 1969-10-21 | Permalac Corp The | Surface coating compositions containing pyridine salts or aromatic sulfonic acids |
| US3476718A (en) | 1967-07-17 | 1969-11-04 | Hercules Inc | Polymers of epoxy cyclic sulfones |
| US3711391A (en) | 1971-05-18 | 1973-01-16 | American Can Co | Photopolymerizable epoxy systems containing sulfoxide gelation inhibitors |
| US4058537A (en) | 1976-01-05 | 1977-11-15 | Ciba-Geigy Corporation | Esters of anhydride aromatic polycarboxylic acids with perfluoroalkyl alcohols |
| US4064191A (en) | 1976-03-10 | 1977-12-20 | American Cyanamid Company | Coating composition containing an alkylated glycoluril, a polymeric non-self-crosslinking compound and an acid catalyst |
| US4118437A (en) | 1976-04-08 | 1978-10-03 | American Cyanamid Company | Cross linkable powder coating compositions |
| US4200729A (en) | 1978-05-22 | 1980-04-29 | King Industries, Inc | Curing amino resins with aromatic sulfonic acid oxa-azacyclopentane adducts |
| US4251665A (en) | 1978-05-22 | 1981-02-17 | King Industries, Inc. | Aromatic sulfonic acid oxa-azacyclopentane adducts |
| US4255558A (en) | 1979-06-18 | 1981-03-10 | Scm Corporation | Self-curing thermosetting powder paints |
| US4309529A (en) * | 1980-05-12 | 1982-01-05 | Minnesota Mining And Manufacturing Company | Water-dispersible energy curable heterocyclic group-containing polyesters |
| EP0058638B1 (en) | 1981-02-13 | 1985-08-28 | Ciba-Geigy Ag | Curable compositions containing an acid-curable resin, and process for curing them |
| US4491628A (en) | 1982-08-23 | 1985-01-01 | International Business Machines Corporation | Positive- and negative-working resist compositions with acid generating photoinitiator and polymer with acid labile groups pendant from polymer backbone |
| JPH01293339A (en) | 1988-05-23 | 1989-11-27 | Tosoh Corp | Photoresist composition |
| DE69125634T2 (en) | 1990-01-30 | 1998-01-02 | Wako Pure Chem Ind Ltd | Chemically reinforced photoresist material |
| US5187019A (en) | 1991-09-06 | 1993-02-16 | King Industries, Inc. | Latent catalysts |
| US5380804A (en) | 1993-01-27 | 1995-01-10 | Cytec Technology Corp. | 1,3,5-tris-(2-carboxyethyl) isocyanurate crosslinking agent for polyepoxide coatings |
| US5693691A (en) | 1995-08-21 | 1997-12-02 | Brewer Science, Inc. | Thermosetting anti-reflective coatings compositions |
| KR100500499B1 (en) | 1996-03-07 | 2005-12-30 | 에이제토 엘렉토로닉 마티리알즈 가부시키가이샤 | Bottom antireflective coatings through refractive index modification by anomalous dispersion |
| US5998099A (en) | 1996-03-08 | 1999-12-07 | Lucent Technologies Inc. | Energy-sensitive resist material and a process for device fabrication using an energy-sensitive resist material |
| US5843624A (en) | 1996-03-08 | 1998-12-01 | Lucent Technologies Inc. | Energy-sensitive resist material and a process for device fabrication using an energy-sensitive resist material |
| EP0851300B1 (en) | 1996-12-24 | 2001-10-24 | Fuji Photo Film Co., Ltd. | Bottom anti-reflective coating material composition and method of forming resist pattern using the same |
| KR100265597B1 (en) | 1996-12-30 | 2000-09-15 | 김영환 | Arf photosensitive resin and manufacturing method thereof |
| US6808859B1 (en) | 1996-12-31 | 2004-10-26 | Hyundai Electronics Industries Co., Ltd. | ArF photoresist copolymers |
| US6274295B1 (en) | 1997-03-06 | 2001-08-14 | Clariant Finance (Bvi) Limited | Light-absorbing antireflective layers with improved performance due to refractive index optimization |
| US5935760A (en) | 1997-10-20 | 1999-08-10 | Brewer Science Inc. | Thermosetting polyester anti-reflective coatings for multilayer photoresist processes |
| US6849377B2 (en) | 1998-09-23 | 2005-02-01 | E. I. Du Pont De Nemours And Company | Photoresists, polymers and processes for microlithography |
| US6790587B1 (en) | 1999-05-04 | 2004-09-14 | E. I. Du Pont De Nemours And Company | Fluorinated polymers, photoresists and processes for microlithography |
| CN1247674C (en) | 1999-10-27 | 2006-03-29 | 钟渊化学工业株式会社 | Extruded styrene resin foam and process for producing the same |
| US6686124B1 (en) | 2000-03-14 | 2004-02-03 | International Business Machines Corporation | Multifunctional polymeric materials and use thereof |
| US6323310B1 (en) | 2000-04-19 | 2001-11-27 | Brewer Science, Inc. | Anti-reflective coating compositions comprising polymerized aminoplasts |
| AU2001274579A1 (en) | 2000-06-21 | 2002-01-02 | Asahi Glass Company, Limited | Resist composition |
| US6447980B1 (en) | 2000-07-19 | 2002-09-10 | Clariant Finance (Bvi) Limited | Photoresist composition for deep UV and process thereof |
| KR100734249B1 (en) | 2000-09-07 | 2007-07-02 | 삼성전자주식회사 | Photosensitive polymers having protective group including fused aromatic ring and resist compositions comprising the same |
| US6509417B1 (en) | 2000-10-31 | 2003-01-21 | Lilly Industries, Inc. | Coating of fatty acid-modified glycidyl copolymer, OH polymer and optional anhydride polymer |
| CN1221861C (en) | 2001-02-09 | 2005-10-05 | 旭硝子株式会社 | Protective film composition |
| JP3804792B2 (en) | 2001-04-10 | 2006-08-02 | 日産化学工業株式会社 | Antireflection film forming composition for lithography |
| TW591341B (en) | 2001-09-26 | 2004-06-11 | Shipley Co Llc | Coating compositions for use with an overcoated photoresist |
| US6723488B2 (en) | 2001-11-07 | 2004-04-20 | Clariant Finance (Bvi) Ltd | Photoresist composition for deep UV radiation containing an additive |
| EP1315043A1 (en) | 2001-11-27 | 2003-05-28 | Fujitsu Limited | Resist pattern thickening material, resist pattern and forming process thereof, and semiconductor device and manufacturing process thereof |
| US6806026B2 (en) | 2002-05-31 | 2004-10-19 | International Business Machines Corporation | Photoresist composition |
| JP2004014474A (en) | 2002-06-11 | 2004-01-15 | Sumitomo Wiring Syst Ltd | Molding method of wire harness and molded wire harness |
| AU2003271123A1 (en) | 2002-10-09 | 2004-05-04 | Nissan Chemical Industries, Ltd. | Composition for forming antireflection film for lithography |
| US7038328B2 (en) | 2002-10-15 | 2006-05-02 | Brewer Science Inc. | Anti-reflective compositions comprising triazine compounds |
| US7264913B2 (en) | 2002-11-21 | 2007-09-04 | Az Electronic Materials Usa Corp. | Antireflective compositions for photoresists |
| US7794919B2 (en) | 2003-04-02 | 2010-09-14 | Nissan Chemical Industries, Ltd. | Composition for forming underlayer coating for lithography containing epoxy compound and carboxylic acid compound |
| US7186789B2 (en) * | 2003-06-11 | 2007-03-06 | Advanced Cardiovascular Systems, Inc. | Bioabsorbable, biobeneficial polyester polymers for use in drug eluting stent coatings |
| TWI363251B (en) | 2003-07-30 | 2012-05-01 | Nissan Chemical Ind Ltd | Sublayer coating-forming composition for lithography containing compound having protected carboxy group |
| TWI358612B (en) | 2003-08-28 | 2012-02-21 | Nissan Chemical Ind Ltd | Polyamic acid-containing composition for forming a |
| TW200533692A (en) | 2003-11-06 | 2005-10-16 | Showa Denko Kk | Curable polyester having an oxetanyl group at end and process for preparing the same, resist composition, jet printing ink composition, curing methods and uses thereof |
| WO2005097883A2 (en) | 2004-03-26 | 2005-10-20 | King Industries, Inc. | Method of producing a crosslinked coating in the manufacture of integrated circuits |
| US7081511B2 (en) * | 2004-04-05 | 2006-07-25 | Az Electronic Materials Usa Corp. | Process for making polyesters |
| JP4702559B2 (en) | 2004-04-09 | 2011-06-15 | 日産化学工業株式会社 | Antireflection film for semiconductor having condensed polymer |
| US20060057491A1 (en) | 2004-05-18 | 2006-03-16 | Rohm And Haas Electronic Materials, L.L.C. | Coating compositions for use with an overcoated photoresist |
| US7691556B2 (en) | 2004-09-15 | 2010-04-06 | Az Electronic Materials Usa Corp. | Antireflective compositions for photoresists |
| US7326523B2 (en) | 2004-12-16 | 2008-02-05 | International Business Machines Corporation | Low refractive index polymers as underlayers for silicon-containing photoresists |
| EP1691238A3 (en) | 2005-02-05 | 2009-01-21 | Rohm and Haas Electronic Materials, L.L.C. | Coating compositions for use with an overcoated photoresist |
| EP1705519B1 (en) | 2005-03-20 | 2016-07-06 | Rohm and Haas Electronic Materials, L.L.C. | Method of treating a microelectronic substrate |
| EP1742108B1 (en) | 2005-07-05 | 2015-10-28 | Rohm and Haas Electronic Materials, L.L.C. | Coating compositions for use with an overcoated photoresist |
| US7470500B2 (en) * | 2005-07-19 | 2008-12-30 | Az Electronic Materials Usa Corp. | Organic bottom antireflective polymer compositions |
| US11372330B2 (en) | 2005-09-27 | 2022-06-28 | Nissan Chemical Industries, Ltd. | Anti-reflective coating forming composition containing reaction product of isocyanuric acid compound with benzoic acid compound |
| US7553905B2 (en) | 2005-10-31 | 2009-06-30 | Az Electronic Materials Usa Corp. | Anti-reflective coatings |
| JP4666166B2 (en) | 2005-11-28 | 2011-04-06 | 信越化学工業株式会社 | Resist underlayer film material and pattern forming method |
| KR101423056B1 (en) * | 2006-06-19 | 2014-07-25 | 닛산 가가쿠 고교 가부시키 가이샤 | Composition containing hydroxylated condensation resin for forminng film under resist |
| CN101506736B (en) | 2006-08-28 | 2013-07-10 | 日产化学工业株式会社 | Composition containing liquid additive for forming resist underlayer film, underlayer film forming method and semiconductor device manufacture method |
| US7416834B2 (en) | 2006-09-27 | 2008-08-26 | Az Electronic Materials Usa Corp. | Antireflective coating compositions |
| US20080175882A1 (en) * | 2007-01-23 | 2008-07-24 | Trollsas Mikael O | Polymers of aliphatic thioester |
| US20090035704A1 (en) | 2007-08-03 | 2009-02-05 | Hong Zhuang | Underlayer Coating Composition Based on a Crosslinkable Polymer |
| US20090042133A1 (en) | 2007-08-10 | 2009-02-12 | Zhong Xiang | Antireflective Coating Composition |
| US8435721B2 (en) * | 2008-02-21 | 2013-05-07 | Nissan Chemical Industries, Ltd. | Resist underlayer film forming composition and forming method of resist pattern using the same |
| US7989144B2 (en) | 2008-04-01 | 2011-08-02 | Az Electronic Materials Usa Corp | Antireflective coating composition |
| US20090274974A1 (en) | 2008-04-30 | 2009-11-05 | David Abdallah | Spin-on graded k silicon antireflective coating |
| US7932018B2 (en) | 2008-05-06 | 2011-04-26 | Az Electronic Materials Usa Corp. | Antireflective coating composition |
| US8329387B2 (en) | 2008-07-08 | 2012-12-11 | Az Electronic Materials Usa Corp. | Antireflective coating compositions |
| US20100092894A1 (en) | 2008-10-14 | 2010-04-15 | Weihong Liu | Bottom Antireflective Coating Compositions |
| ES2610627T3 (en) * | 2008-10-27 | 2017-04-28 | Unilever N.V. | Antiperspirant or deodorant compositions |
| US8501877B2 (en) * | 2009-04-03 | 2013-08-06 | Ccp Composites Us Llc | Thermosetting compositions containing isocyanurate rings |
| US8551686B2 (en) | 2009-10-30 | 2013-10-08 | Az Electronic Materials Usa Corp. | Antireflective composition for photoresists |
| US8507192B2 (en) | 2010-02-18 | 2013-08-13 | Az Electronic Materials Usa Corp. | Antireflective compositions and methods of using same |
-
2008
- 2008-07-08 US US12/133,562 patent/US8221965B2/en active Active
-
2009
- 2009-04-29 CN CN200980120772.6A patent/CN102056969B/en active Active
- 2009-04-29 WO PCT/IB2009/005458 patent/WO2010004377A1/en active Application Filing
- 2009-04-29 KR KR1020107027320A patent/KR101429326B1/en active IP Right Grant
- 2009-04-29 EP EP09785890A patent/EP2300518B1/en not_active Not-in-force
- 2009-04-29 JP JP2011517253A patent/JP5568791B2/en not_active Expired - Fee Related
- 2009-04-29 MY MYPI2011000055A patent/MY155242A/en unknown
- 2009-04-30 TW TW098114508A patent/TWI512063B/en active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008017954A2 (en) | 2006-08-10 | 2008-02-14 | Az Electronic Materials Usa Corp. | Antireflective composition for photoresists |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2258691A1 (en) * | 2009-05-20 | 2010-12-08 | Rohm and Haas Electronic Materials, L.L.C. | Coating compositions for use with an overcoated photoresist |
| WO2012017948A1 (en) * | 2010-08-05 | 2012-02-09 | 日産化学工業株式会社 | Epoxy compound with nitrogen-containing ring |
| CN103068822A (en) * | 2010-08-05 | 2013-04-24 | 日产化学工业株式会社 | Epoxy compound with nitrogen-containing ring |
| EP2602257A1 (en) * | 2010-08-05 | 2013-06-12 | Nissan Chemical Industries, Ltd. | Epoxy compound with nitrogen-containing ring |
| EP2602257A4 (en) * | 2010-08-05 | 2014-01-08 | Nissan Chemical Ind Ltd | Epoxy compound with nitrogen-containing ring |
| JP5804282B2 (en) * | 2010-08-05 | 2015-11-04 | 日産化学工業株式会社 | Epoxy compounds having nitrogen-containing rings |
| KR101821574B1 (en) * | 2010-08-05 | 2018-01-25 | 닛산 가가쿠 고교 가부시키 가이샤 | Epoxy compound with nitrogen-containing ring |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2300518A1 (en) | 2011-03-30 |
| JP5568791B2 (en) | 2014-08-13 |
| TWI512063B (en) | 2015-12-11 |
| JP2011527460A (en) | 2011-10-27 |
| TW201002793A (en) | 2010-01-16 |
| KR20110042262A (en) | 2011-04-26 |
| MY155242A (en) | 2015-09-30 |
| US8221965B2 (en) | 2012-07-17 |
| WO2010004377A8 (en) | 2011-03-31 |
| KR101429326B1 (en) | 2014-08-12 |
| EP2300518B1 (en) | 2012-08-15 |
| CN102056969B (en) | 2014-09-24 |
| US20100009293A1 (en) | 2010-01-14 |
| CN102056969A (en) | 2011-05-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2300518B1 (en) | Antireflective coating compositions | |
| US8329387B2 (en) | Antireflective coating compositions | |
| US20100092894A1 (en) | Bottom Antireflective Coating Compositions | |
| KR101441705B1 (en) | Antireflective composition for photoresists | |
| KR101486841B1 (en) | Underlayer coating composition based on a crosslinkable polymer | |
| EP1563343B1 (en) | Antireflective compositions for photoresists | |
| EP2493991B1 (en) | Antireflective composition for photoresists | |
| US20150227043A1 (en) | Bottom antireflective materials and compositions | |
| EP2577361B1 (en) | Antireflective coating composition and process for manufacturing microelectronic device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980120772.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09785890 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20107027320 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2011517253 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009785890 Country of ref document: EP |