WO2008054675A2 - Antidiabetic bicyclic compounds - Google Patents
Antidiabetic bicyclic compounds Download PDFInfo
- Publication number
- WO2008054675A2 WO2008054675A2 PCT/US2007/022650 US2007022650W WO2008054675A2 WO 2008054675 A2 WO2008054675 A2 WO 2008054675A2 US 2007022650 W US2007022650 W US 2007022650W WO 2008054675 A2 WO2008054675 A2 WO 2008054675A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- c3alkyl
- optionally substituted
- group
- independently selected
- compound
- Prior art date
Links
- 0 COc1ccc([C@](CC(*)=O)CCC2)c2c1 Chemical compound COc1ccc([C@](CC(*)=O)CCC2)c2c1 0.000 description 2
- ZGVFIRGMLUVKPD-NSHDSACASA-N CCOC(C[C@H](CCC1)c(cc2)c1cc2O)=O Chemical compound CCOC(C[C@H](CCC1)c(cc2)c1cc2O)=O ZGVFIRGMLUVKPD-NSHDSACASA-N 0.000 description 1
- VROPPALPLJCNIJ-VIFPVBQESA-N CCOC(C[C@H]1c(ccc(O)c2)c2OCC1)=O Chemical compound CCOC(C[C@H]1c(ccc(O)c2)c2OCC1)=O VROPPALPLJCNIJ-VIFPVBQESA-N 0.000 description 1
- PPXYNWAACYDMQJ-UHFFFAOYSA-N CN(C(CC(O)=O)c(c1c2)ccc2Oc(ccc(C(F)(F)F)c2)c2Cl)C1=O Chemical compound CN(C(CC(O)=O)c(c1c2)ccc2Oc(ccc(C(F)(F)F)c2)c2Cl)C1=O PPXYNWAACYDMQJ-UHFFFAOYSA-N 0.000 description 1
- UBTUVNIOAASQFC-UHFFFAOYSA-N OC(CC(CSc1c2)c1ccc2Oc(ccc(C(F)(F)F)c1)c1Cl)=O Chemical compound OC(CC(CSc1c2)c1ccc2Oc(ccc(C(F)(F)F)c1)c1Cl)=O UBTUVNIOAASQFC-UHFFFAOYSA-N 0.000 description 1
- YYUDASQLWSSVBT-UHFFFAOYSA-N OC(CC(c(ccc(Oc(ccc(C(F)(F)F)c1)c1Cl)c1)c1N1)C1=O)=O Chemical compound OC(CC(c(ccc(Oc(ccc(C(F)(F)F)c1)c1Cl)c1)c1N1)C1=O)=O YYUDASQLWSSVBT-UHFFFAOYSA-N 0.000 description 1
- IRWPQRHAEUJQAX-UHFFFAOYSA-N OC(CC1c(ccc(Oc(ccc(C(F)(F)F)c2)c2Cl)c2)c2OC1)=O Chemical compound OC(CC1c(ccc(Oc(ccc(C(F)(F)F)c2)c2Cl)c2)c2OC1)=O IRWPQRHAEUJQAX-UHFFFAOYSA-N 0.000 description 1
- WOYSRXHREMNSMT-LBPRGKRZSA-N OC(C[C@H](CCCc1c2)c1ccc2Oc(ccc(C(F)(F)F)c1)c1Cl)=O Chemical compound OC(C[C@H](CCCc1c2)c1ccc2Oc(ccc(C(F)(F)F)c1)c1Cl)=O WOYSRXHREMNSMT-LBPRGKRZSA-N 0.000 description 1
- DUEXMUSLWHRKCA-LBPRGKRZSA-N OC(C[C@H](CCOc1c2)c1ccc2Oc1ccnc2c1ccc(C(F)(F)F)c2)=O Chemical compound OC(C[C@H](CCOc1c2)c1ccc2Oc1ccnc2c1ccc(C(F)(F)F)c2)=O DUEXMUSLWHRKCA-LBPRGKRZSA-N 0.000 description 1
- SPTHCHDOGHNRHU-QMMMGPOBSA-N OC(C[C@H](CCc1c2)c1ccc2O)=O Chemical compound OC(C[C@H](CCc1c2)c1ccc2O)=O SPTHCHDOGHNRHU-QMMMGPOBSA-N 0.000 description 1
- PRGPQMJBEQHCRN-NSHDSACASA-N OC(C[C@H](CCc1c2)c1ccc2Oc(ccc(C(F)(F)F)c1)c1Cl)=O Chemical compound OC(C[C@H](CCc1c2)c1ccc2Oc(ccc(C(F)(F)F)c1)c1Cl)=O PRGPQMJBEQHCRN-NSHDSACASA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/52—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes
- C07D333/54—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
- C07D333/60—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/48—Drugs for disorders of the endocrine system of the pancreatic hormones
- A61P5/50—Drugs for disorders of the endocrine system of the pancreatic hormones for increasing or potentiating the activity of insulin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C235/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms
- C07C235/42—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings and singly-bound oxygen atoms bound to the same carbon skeleton
- C07C235/44—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings and singly-bound oxygen atoms bound to the same carbon skeleton with carbon atoms of carboxamide groups and singly-bound oxygen atoms bound to carbon atoms of the same non-condensed six-membered aromatic ring
- C07C235/46—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings and singly-bound oxygen atoms bound to the same carbon skeleton with carbon atoms of carboxamide groups and singly-bound oxygen atoms bound to carbon atoms of the same non-condensed six-membered aromatic ring having the nitrogen atoms of the carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/54—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing cyano groups and etherified hydroxy groups bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/72—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings and other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/76—Unsaturated compounds containing keto groups
- C07C59/90—Unsaturated compounds containing keto groups containing singly bound oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/34—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/30—Indoles; Hydrogenated indoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to carbon atoms of the hetero ring
- C07D209/32—Oxygen atoms
- C07D209/34—Oxygen atoms in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/44—Iso-indoles; Hydrogenated iso-indoles
- C07D209/46—Iso-indoles; Hydrogenated iso-indoles with an oxygen atom in position 1
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/61—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/20—Oxygen atoms
- C07D215/22—Oxygen atoms attached in position 2 or 4
- C07D215/233—Oxygen atoms attached in position 2 or 4 only one oxygen atom which is attached in position 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/22—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the nitrogen-containing ring
- C07D217/24—Oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/79—Benzo [b] furans; Hydrogenated benzo [b] furans with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
- C07D307/80—Radicals substituted by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D311/04—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring
- C07D311/58—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring other than with oxygen or sulphur atoms in position 2 or 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/46—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings substituted on the ring sulfur atom
- C07D333/48—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings substituted on the ring sulfur atom by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/08—One of the condensed rings being a six-membered aromatic ring the other ring being five-membered, e.g. indane
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/10—One of the condensed rings being a six-membered aromatic ring the other ring being six-membered, e.g. tetraline
Definitions
- the instant invention is concerned with diaryl ether compounds in which one of the aryl groups is fused to a cycloalkyl or hetercyclic ring that is connected to an acetic acid moiety, including pharmaceutically acceptable salts and prodrugs thereof.
- the compounds are agonists of G-protein coupled receptor 40 (GPR40) and are useful as therapeutic compounds, particularly in the treatment of Type 2 diabetes melhtus, and of conditions that often accompany this disease, including insulin resistance, obesity and lipid disorders.
- GPR40 G-protein coupled receptor 40
- Diabetes is a disease derived from multiple causative factors and characterized by elevated levels of plasma glucose (hyperglycemia) in the fasting state or after administration of glucose during an oral glucose tolerance test.
- diabetes There are two generally recognized forms of diabetes.
- type 1 diabetes or insulin-dependent diabetes melhtus (IDDM)
- IDDM insulin-dependent diabetes melhtus
- NIDDM noninsuhn-dependent diabetes melhtus
- insulin is still produced in the body.
- Patients having type 2 diabetes have a resistance to the effects of insulin in stimulating glucose and lipid metabolism in the mam insulin-sensitive tissues, which are muscle, liver and adipose tissues.
- Insulin resistance is not primarily caused by a diminished number of insulin receptors but rather by a post-msulin receptor binding defect that is not yet completely understood. This lack of responsiveness to insulin results in insufficient insulin-mediated activation of uptake, oxidation and storage of glucose in muscle, and inadequate msuhn-mediated repression of hpolysis in adipose tissue and of glucose production and secretion in the liver.
- Persistent or uncontrolled hyperglycemia that occurs with diabetes is associated with increased and premature morbidity and mortality. Often abnormal glucose homeostasis is associated both directly and indirectly with obesity, hypertension, and alterations of the lipid, lipoprotein and apolipoprotem metabolism, as well as other metabolic and hemodynamic disease. Patients with type 2 diabetes melhtus have a significantly increased ⁇ sk of macrovascular and microvascular complications, including atherosclerosis, coronary heart disease, stroke, peripheral vascular disease, hypertension, nephropathy, neuropathy, and retinopathy. Therefore, therapeutic control of glucose homeostasis, lipid metabolism, obesity, and hypertension are critically important in the clinical management and treatment of diabetes melhtus.
- the biguanides are a class of drugs that are widely used to treat type 2 diabetes.
- the two best known biguamdes, phenformin and metformin cause some correction of hyperglycemia.
- the biguamdes act primarily by inhibiting hepatic glucose production, and they also are believed to modestly improve insulin sensitivity.
- the biguamdes can be used as monotherapy or in combination with other anti-diabetic drugs, such as insulin or an msulm secretagogues, without increasing the risk of hypoglycemia.
- phenformin and metformin can induce lactic acidosis and nausea/diarrhea.
- Metformin has a lower risk of side effects than phenformin and is widely prescribed for the treatment of Type 2 diabetes.
- the ghtazones are a newer class of compounds that can ameliorate hyperglycemia and other symptoms of type 2 diabetes.
- the ghtazones that are currently marketed are agonists of the peroxisome prohferator activated receptor (PPAR) gamma subtype.
- PPAR peroxisome prohferator activated receptor
- the PPAR-gamma agonists substantially increase insulin sensitivity in muscle, liver and adipose tissue in several animal models of type 2 diabetes, resulting in partial or complete correction of elevated plasma glucose levels without the occurrence of hypoglycemia.
- PPAR-gamma agonism is believed to be responsible for the improved insulin sensitization that is observed in human patients who are treated with the ghtazones.
- New PPAR agonists are currently being developed. Many of the newer PPAR compounds are agonists of one or more of the PPAR alpha, gamma and delta subtypes. Compounds that are agonists of both the PPAR alpha and PPAR gamma subtypes (PPAR alpha/gamma dual agonists) are promising because they reduce hyperglycemia and also improve lipid metabolism.
- the currently marketed PPAR gamma agonists are modestly effective in reducing plasma glucose and HemoglobmAlC.
- the currently marketed compounds do not greatly improve lipid metabolism and may actually have a negative effect on the lipid profile.
- the PPAR compounds represent an important advance in diabetic therapy, but further improvements are still needed.
- Another widely used drug treatment involves the administration of insulin secretagogues, such as the sulfonylureas (e.g. tolbutamide and glipizide). These drugs increase the plasma level of insulin by stimulating the pancreatic ⁇ -cells to secrete more insulin. Insulin secretion in the pancreatic ⁇ -cell is under strict regulation by glucose and an array of metabolic, neural and hormonal signals.
- Glucose stimulates insulin production and secretion through its metabolism to generate ATP and other signaling molecules, whereas other extracellular signals act as potentiators or inhibitors of insulin secretion through GPCR's present on the plasma membrane.
- Sulfonylureas and related insulin secretagogues act by blocking the ATP-dependent K+ channel in /3-cells, which causes depolarization of the cell and the opening of the voltage-dependent Ca2+ channels with stimulation of insulin release. This mechanism is non-glucose dependent, and hence insulin secretion can occur regardless of the ambient glucose levels. This can cause insulin secretion even if the glucose level is low, resulting in hypoglycemia, which can be fatal in severe cases. The administration of insulin secretagogues must therefore be carefully controlled.
- the insulin secretagogues are often used as a first-lme drug treatment for Type 2 diabetes.
- GPCR G-protein coupled receptors
- GPR40 agonists that are active in the islets may have the potential to restore or preserve islet function. This would be highly advantageous, because long term diabetes therapy often leads to the gradual diminution of islet activity, so that after extended periods of treatment, it is often necessary to treat type 2 diabetic patients with daily insulin injections. By restoring or preserving islet function, GPR40 agonists may delay or prevent the diminution and loss of islet function in a type 2 diabetic patient.
- the class of compounds described herein is a new class of GPR40 agonists.
- the compounds are useful in the treatment of diseases that are modulated by GPR40 agonists, including type 2 diabetes and hyperglycemia that may be associated with type 2 diabetes or pre-diabetic insulin resistance.
- the present invention is directed to a compound of formula I, or a pharmaceutically acceptable salt thereof, including individual diastereomers and enantiomers, and mixtures of diastereomers and/or enantiomers:
- Heterocycle is a 5-6 membered saturated or partly saturated monocyclic heterocyclic ring having 1-3 heteroatoms independently selected from O, N and S;
- Heteroaryl is a 5-6 membered monocyclic heteroaromatic ring having 1-3 heteroatoms independently selected from O, N and S;
- -C(O)Phenyl, -C(O)Naphthyl, -C(0)Heterocyle, Heterocycle, Heteroaryl, C3-C7Cycloalkyl, Phenyl and Naphthyl either as Rl, R2, R3, R4, and R 5 or as substituents on Rl, R2, R3 ; R4 ; an d R 5 are optionally substituted with 1-4 substituents independently selected from halogen, -CN, -NO2, -OH, -Ci- C3alkyl, -C( 0)Ci-C3alkyl, -S(O)2Ci-C3alkyl, and -0Ci-C3alkyl, wherein the -Ci-C3alkyl, -OCi- C3alkyl, -S(O)2Ci-C3alkyl, and -C(O)C i-C3alkyl substituents are
- Each R6 is independently selected from the group consisting of H and -Ci-Cgalkyl;
- R7 is selected from the group consisting of H and -Ci-C3alkyl;
- R°" is selected from the group consisting of H, -OH, -Ci-C3alkyl optionally substituted with 1-3 halogens, and -OC 1-C3 alkyl optionally substituted with 1-3 halogens and optionally with one group -C(O)OR 1 O;
- R9 and RlO are independently selected from the group consisting of H and -Ci -C ⁇ alkyl, wherein -Ci -C ⁇ alkyl is optionally substituted with 1-5 halogens.
- the invention has numerous embodiments, which are summarized below.
- the invention includes the compounds as shown. It also includes individual diastereomers, enantiomers, and epimers of the compounds; mixtures of diastereomers and/or enantiomers; and racemates.
- the invention also includes pharmaceutically acceptable salts of the compounds, and pharmaceutical compositions comprising the compounds and a pharmaceutically acceptable carrier.
- the compounds are useful in treating type 2 diabetes and insulin resistance.
- the compounds may also be useful in treating or controlling obesity.
- alkyl groups may be either linear or branched, unless otherwise defined.
- each R6 IS independently selected from the group consisting of H and -Ci-C3alkyl.
- each R6 group is independently selected from H and CH3.
- B is selected from the group consisting of -CH2-, -CH2CH2-, and -CH(CH3)-.
- Z is -CR?R8CO2H
- R7 is H
- A is selected from -CH2-, -0-, -S-, -S(0)2-, -C(O)-, and -CH(OH)-; and B is selected from -CH2-, -CH2CH2-, and -CH(CH3)-;
- R2 IS selected from the group consisting of H, halogen, Ci-C 3 alkyl optionally substituted with 1-3 F, -OCi-C3alkyl optionally substituted with 1-3 F, and Phenyl optionally substituted with 1-3 groups independently selected from halogen, CH3, CF3, -OCH3, and -OCF3;
- R3 IS selected from H, halogen, CH3, CF3, -OCH3, and -OCF3;
- R7 IS H; R8 IS selected from the group consisting of H, -OH, CH 3 , and -OCH2C( 0)ORlO; and
- RlO is selected from the group consisting of H and -Ci-C4alkyl.
- At least one of the groups R*, R ⁇ , R3 ; R4 ; or R5 that is present is not H.
- at least two of the groups R ⁇ , R ⁇ , R ⁇ , R4 ; ar ⁇ j R ⁇ that are present are not H.
- 2-3 of the groups R ⁇ , R ⁇ , R ⁇ , R4 ; an( j R5 that are present are not H.
- stereoisomers including diastereomers, enantiomers, epimers, and mixtures of these, including racemates, may also have utility m treating GPR40 mediated diseases.
- Inactive or less active diastereoisomers and enantiomers are also useful for scientific studies relating to the receptor and the mechanism of activation.
- the compounds of this invention may be used in pharmaceutical compositions comprising (a) the compound(s) or pharmaceutically acceptable salts thereof, and (b) a pharmaceutically acceptable carrier.
- the compounds of this invention may be used in pharmaceutical compositions that include one or more other active pharmaceutical ingredients.
- the compounds of this invention may also be used m pharmaceutical compositions in which the compound of Formula I or a pharmaceutically acceptable salt thereof is the only active ingredient.
- a compound of Formula I, or a pharmaceutically acceptable salt thereof may be used in the manufacture of a medicament for the treatment of type 2 diabetes melhtus in a human or other mammalian patient.
- a method of treating type 2 diabetes comprises the administration of a therapeutically effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound, to a patient in need of treatment.
- Other medical uses of the compounds of Formula I are described hereinafter.
- Alkyl means saturated carbon chains which may be linear or branched or combinations thereof, unless the carbon chain is defined otherwise.
- alkyl groups include methyl, ethyl, propyl, isopropyl, butyl, sec- and tert-butyl, pentyl, hexyl, heptyl, octyl, nonyl, and the like.
- alkenyl means carbon chains which contain at least one carbon-carbon double bond, and which may be linear or branched or combinations thereof.
- alkenyl include vinyl, allyl, isopropenyl, pentenyl, hexenyl, heptenyl, 1-propenyl, 2-butenyl, 2-methyl-2-butenyl, and the like
- alkynyl means carbon chains which contain at least one carbon-carbon triple bond, and which may be linear or branched or combinations thereof. Examples of alkynyl include ethynyl, propargyl, 3 -methyl- 1-pentynyl, 2-heptynyl and the like.
- Cycloalkyl means a saturated carbocyclic ring, having a specified number of carbon atoms. The term may also be used to describe a carbocyclic ring fused to an aryl group. Examples of cycloalkyl include cyclopropyl, cyclopentyl, cyclohexyl, cycloheptyl, and the like. Cycloalkenyl rings comp ⁇ se a double bond in the ring.
- Aryl is commonly used to refer to carbocyclic aromatic structures. The most common aryl groups are phenyl and naphthyl. Phenyl is generally the most preferred aryl group.
- Heterocycle means a fully or partially saturated 5-6-membered nng containing 1-3 heteroatoms independently selected from N, S and O. Examples of heterocycles include tetrahydrofuran, piperazine, pipendme, and morphohne.
- Heteroaryl means a 5-6-membered aromatic ⁇ ng containing 1-3 heteroatoms independently selected from N, O and S.
- heteroaryl examples include pyrrolyl, isoxazolyl, isothiazolyl, pyrazolyl, py ⁇ dyl, oxazolyl, oxadiazolyl, thiadiazolyl, thiazolyl, lmidazolyl, triazolyl, tetrazolyl, furanyl, t ⁇ azmyl, thienyl, pyrimidyl, py ⁇ dazinyl, and pyrazinyl.
- "Halogen” includes fluo ⁇ ne, chlorine, bromine and iodine.
- composition as m pharmaceutical composition, is intended to encompass a product comprising the active mgredient(s), and the inert ⁇ ngredient(s) that make up the carrier, as well as any product which results, directly or indirectly, from combination, complexation or aggregation of any two or more of the ingredients, or from dissociation of one or more of the ingredients, or from other types of reactions or interactions of one or more of the ingredients.
- the pharmaceutical compositions of the present invention encompass any composition made by admixing a compound of the present invention and a pharmaceutically acceptable carrier.
- tetrazole means a 2H-tetrazol-5-yl substituent group and tautomers thereof.
- Compounds of Formula I may contain one or more asymmetric centers and can thus occur as racemates, racemic mixtures, single enantiomers, individual diastereomers, and mixtures of diastereomers and/or enantiomers.
- the present invention is meant to comprehend all such isomeric forms of the compounds of Formula I.
- the compounds of the instant invention have at least one asymmetric center, which is on the ⁇ ng that is fused to the phenyl ⁇ ng at the point where the acetic acid moiety is attached. There may also be a second asymmetric center on substituent groups.
- Each such asymmetric center will independently produce two optical isomers, and it is intended that all of the possible optical isomers, stereoisomers, and diastereomers in mixtures and as pure or partially purified compounds are included within the scope of this invention (i.e. all possible combinations of the asymmetric centers as pure compounds or in mixtures).
- Some of the compounds descnbed herein may contain olefinic double bonds, and unless specified otherwise, are meant to include both E and Z geometric isomers.
- tautomers Some of the compounds descnbed herein may exist with different points of attachment of hydrogen, referred to as tautomers.
- An example is a ketone and its enol form, known as keto-enol tautomers.
- keto-enol tautomers The individual tautomers as well as mixtures thereof are encompassed with compounds of Formula I.
- Compounds of Formula I having one or more asymmetric centers may be separated into diastereoisomers, enantiomers, and the like by methods well known in the art.
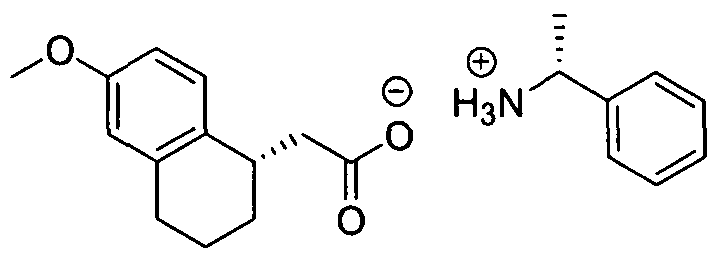
- enantiomers and other compounds with chiral centers may be synthesized by stereospecific synthesis using optically pure starting materials and/or reagents of known configuration.
- salts refers to salts prepared from pharmaceutically acceptable non-toxic bases or acids including inorganic or organic bases and inorganic or organic acids.
- Salts derived from inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic salts, manganous, potassium, sodium, zinc, and the like. Particularly preferred are the ammonium, calcium, magnesium, potassium, and sodium salts. Salts m the solid form may exist in more than one crystal structure, and may also be in the form of hydrates.
- Salts derived from pharmaceutically acceptable organic non-toxic bases include salts of primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, and basic ion exchange resins, such as arginine, betaine, caffeine, choline, N,N'- dibenzylethylenediamme, diethylamine, 2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamme, N-ethyl-morpholine, N-ethylpipe ⁇ dine, glucamine, glucosamine, histidine, hydrabamme, isopropylamine, lysine, methylglucamine, morpholine, piperazine, pipe ⁇ dine, polyamine resms, procaine, purines, theobromine, t ⁇ ethylamine, t ⁇ methylamine, t ⁇ propylamme, tromethamine, and the
- Therapeutically active metabolites where the metabolites themselves fall within the scope of the claimed invention, are also compounds of the current invention.
- Prodrugs which are compounds that are converted to the claimed compounds as they are being administered to a patient or after they have been administered to a patient, are compounds of this invention.
- the claimed chemical structures of this application m some cases may themselves be prodrugs.
- Compounds of the present invention are potent agonists of the GPR40 receptor.
- the compounds of the invention, and pharmaceutically acceptable salts thereof, may be efficacious in the treatment of diseases that are modulated by GPR40 ligands and agonists. Many of these diseases are summarized below.
- One or more of the following diseases may be treated by the administration of a therapeutically effective amount of a compound of this invention, or a pharmaceutically acceptable salt thereof, to a patient in need of treatment.
- the compounds of the invention may be used for the manufacture of a medicament for treating one or more of these diseases:
- non-insulin dependent diabetes mellitus type 2 diabetes
- hypertriglyceridemia (elevated levels of triglyceride-rich-lipoproteins);
- Preferred uses of the compounds are for the treatment of one or more of the following diseases by administering a therapeutically effective amount to a patient in need of treatment.
- the compounds may be used for manufacturing a medicament for the treatment of one or more of these diseases:
- Type 2 diabetes and specifically hyperglycemia
- the compounds are expected to be effective in lowering glucose and lipids in diabetic patients and in non-diabetic patients who have impaired glucose tolerance and/or are in a pre-diabetic condition.
- the compounds may ameliorate hyperinsulinemia, which often occurs in diabetic or pre- diabetic patients, by modulating the swings in the level of serum glucose that often occurs in these patients.
- the compounds may also be effective in treating or reducing insulin resistance.
- the compounds may be effective in treating or preventing gestational diabetes.
- the compounds, compositions, and medicaments as described herein may also be effective in reducing the risks of adverse sequelae associated with metabolic syndrome, and in reducing the risk of developing atherosclerosis, delaying the onset of atherosclerosis, and/or reducing the ⁇ sk of sequelae of atherosclerosis.
- Sequelae of atherosclerosis include angina, claudication, heart attack, stroke, and others.
- the compounds may also be effective in delaying or preventing vascular restenosis and diabetic retinopathy.
- the compounds of this invention may also have utility m improving or restoring jS-cell function, so that they may be useful in treating type 1 diabetes or in delaying or preventing a patient with type 2 diabetes from needing insulin therapy.
- the compounds generally may be efficacious in treating one or more of the following diseases: (1) type 2 diabetes (also known as non-insulin dependent diabetes mellitus, or NIDDM), (2) hyperglycemia, (3) impaired glucose tolerance, (4) insulin resistance, (5) obesity, (6) lipid disorders, (7) dyshpidemia, (8) hyperhpidemia, (9) hypertriglyceridemia, (10) hypercholesterolemia, (11) low HDL levels, (12) high LDL levels, (13) atherosclerosis and its sequelae, (14) vascular restenosis, (15) abdominal obesity, (16) retinopathy, (17) metabolic syndrome, (18) high blood pressure, and (19) insulin resistance.
- type 2 diabetes also known as non-insulin dependent diabetes mellitus, or NIDDM
- hyperglycemia also known as non-insulin dependent diabetes mellitus, or NIDDM
- impaired glucose tolerance (4) insulin resistance
- One aspect of the invention provides a method for the treatment and control of mixed or diabetic dyshpidemia, hypercholesterolemia, atherosclerosis, low HDL levels, high LDL levels, hyperhpidemia, and/or hypertriglyceridemia, which comprises administering to a patient in need of such treatment a therapeutically effective amount of a compound having formula I.
- the compound may be used alone or advantageously may be administered with a cholesterol biosynthesis inhibitor, particularly an HMG-CoA reductase inhibitor such as lovastatm, simvastatin, rosuvastatin, pravastatin, fluvastatm, atorvastatin, ⁇ vastatm, itavastatin, or ZD-4522
- a cholesterol biosynthesis inhibitor particularly an HMG-CoA reductase inhibitor such as lovastatm, simvastatin, rosuvastatin, pravastatin, fluvastatm, atorvastatin, ⁇ vastatm, itavastatin, or ZD-4522
- the compound may also be used advantageously in combination with other lipid lowering drugs such as cholesterol absorption inhibitors (for example stanol esters, sterol glycosides such as tiqueside, and azetidinones such as ezetimibe), ACAT inhibitors (such as avasimibe), CETP inhibitors (for example torcetrapi
- Any suitable route of administration may be employed for providing a mammal, especially a human, with an effective dose of a compound of the present invention.
- oral, rectal, topical, parenteral, ocular, pulmonary, nasal, and the like may be employed.
- Dosage forms include tablets, troches, dispersions, suspensions, solutions, capsules, creams, ointments, aerosols, and the like.
- compounds of Formula I are administered orally.
- the effective dosage of active ingredient employed may vary depending on the particular compound employed, the mode of administration, the condition being treated and the severity of the condition being treated. Such dosage may be ascertained readily by a person skilled in the art.
- a daily dosage of from about 0.1 milligram to about 100 milligram per kilogram of animal body weight, preferably given as a single daily dose or in divided doses two to six times a day, or in sustained release form.
- the total daily dosage is from about 1.0 milligrams to about 1000 milligrams.
- the total daily dose will generally be from about 1 milligram to about 500 milligrams.
- the dosage for an adult human may be as low as 0.1 mg
- the dosage regimen may be adjusted within this range or even outside of this range to provide the optimal therapeutic response
- Oral administration will usually be carried out using tablets or capsules. Examples of doses in tablets and capsules are 0.1 mg, 0.25 mg, 0.5 mg, 1 mg, 2 mg, 5 mg, 10 mg, 25 mg, 50 mg, 100 mg, 200 mg, 300 mg, 400 mg, and 500 mg.
- Other oral forms may also have the same or similar dosages.
- compositions which comprise a compound of Formula I and a pharmaceutically acceptable carrier.
- the pharmaceutical compositions of the present invention comprise a compound of Formula I or a pharmaceutically acceptable salt as an active ingredient, as well as a pharmaceutically acceptable carrier and optionally other therapeutic ingredients.
- pharmaceutically acceptable salts refers to salts prepared from pharmaceutically acceptable non-toxic bases or acids including inorganic bases or acids and organic bases or acids.
- a pharmaceutical composition may also comp ⁇ se a prodrug, or a pharmaceutically acceptable salt thereof, if a prodrug is administered.
- the compounds of Formula I can be combined as the active ingredient in intimate admixture with a pharmaceutical carrier according to conventional pharmaceutical compounding techniques.
- the carrier may take a wide variety of forms depending on the form of preparation desired for administration, e.g., oral or parenteral (including intravenous).
- any of the usual pharmaceutical media may be employed, such as, for example, water, glycols, oils, alcohols, flavoring agents, preservatives, coloring agents and the like in the case of oral liquid preparations, such as, for example, suspensions, elixirs and solutions; or carriers such as starches, sugars, microcrystalline cellulose, diluents, granulating agents, lubricants, binders, disintegrating agents and the like in the case of oral solid preparations such as, for example, powders, hard and soft capsules and tablets, with the solid oral preparations being preferred over the liquid preparations.
- oral liquid preparations such as, for example, suspensions, elixirs and solutions
- carriers such as starches, sugars, microcrystalline cellulose, diluents, granulating agents, lubricants, binders, disintegrating agents and the like in the case of oral solid preparations such as, for example, powders, hard and soft capsules and tablets, with the solid oral preparations being preferred over the liquid preparation
- tablets and capsules represent the most advantageous oral dosage unit form in which case solid pharmaceutical carriers are obviously employed. If desired, tablets may be coated by standard aqueous or nonaqueous techniques. Such compositions and preparations should contain at least 0.1 percent of active compound. The percentage of active compound in these compositions may, of course, be varied and may conveniently be between about 2 percent to about 60 percent of the weight of the unit. The amount of active compound in such therapeutically useful compositions is such that an effective dosage will be obtained.
- the active compounds can also be administered intranasally as, for example, liquid drops or spray.
- the tablets, pills, capsules, and the like may also contain a binder such as gum tragacanth, acacia, corn starch or gelatin; excipients such as dicalcium phosphate; a disintegrating agent such as corn starch, potato starch, alginic acid, a lubricant such as magnesium stearate; and a sweetening agent such as sucrose, lactose or saccharin.
- a dosage unit form is a capsule, it may contain, in addition to materials of the above type, a liquid carrier such as a fatty oil.
- the compound or salt may be advantageous to formulate the compound or salt as a solution in an oil such as a triglyceride of one or more medium chain fatty acids, a lipophilic solvent such as triacetin, a hydrophilic solvent (e.g propylene glycol), or a mixture of two or more of these, also optionally including one or more ionic or nomonic surfactants, such as sodium lauryl sulfate, polysorbate 80, polyethoxylated triglycerides, and mono and/or diglyce ⁇ des of one or more medium chain fatty acids.
- an oil such as a triglyceride of one or more medium chain fatty acids, a lipophilic solvent such as triacetin, a hydrophilic solvent (e.g propylene glycol), or a mixture of two or more of these, also optionally including one or more ionic or nomonic surfactants, such as sodium lauryl sulfate, polysorbate 80, polyethoxylated trig
- Solutions containing surfactants will form emulsions or microemulsions on contact with water.
- the compound may also be formulated in a water soluble polymer in which it has been dispersed as an amorphous phase by such methods as hot melt extrusion and spray drying, such polymers including HPMCAS, HPMCS, and polyvinylpyrrohdinones.
- Va ⁇ ous other mate ⁇ als may be present as coatings or to modify the physical form of the dosage unit.
- tablets may be coated with shellac, sugar or both.
- a syrup or elixir may contain, in addition to the active ingredient, sucrose as a sweetening agent, methyl and propylparabens as preservatives, a dye and a flavoring such as cherry or orange flavor.
- Compounds of formula I may also be administered parenterally. Solutions or suspensions of these active compounds can be prepared in water suitably mixed with a surfactant such as hydroxypropylcellulose. Dispersions can also be prepared in glycerol, liquid polyethylene glycols and mixtures thereof in oils. Under ordinary conditions of storage and use, these preparations contain a preservative to prevent the growth of microorganisms.
- the pharmaceutical forms suitable for injectable use include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersions.
- the form must be sterile and must be fluid to the extent that easy sy ⁇ ngabihty exists. It must be stable under the conditions of manufacture and storage and must be preserved against the contaminating action of microorganisms such as bacteria and fungi.
- the earner can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (e.g. glycerol, propylene glycol and liquid polyethylene glycol), suitable mixtures thereof, and vegetable oils.
- Compounds of Formula I may be used in combination with other drugs that may also be useful in the treatment or amelioration of the diseases or conditions for which compounds of Formula I are useful.
- Such other drugs may be administered, by a route and in an amount commonly used therefor, contemporaneously or sequentially with a compound of Formula I.
- more than one drug is commonly administered.
- the compounds of this invention may generally be administered to a patient who is already taking one or more other drugs for these conditions.
- a pharmaceutical composition in unit dosage form containing such other drugs and the compound of Formula I is preferred.
- the combination therapy also includes therapies in which the compound of Formula I and one or more other drugs are administered on different overlapping schedules
- the pharmaceutical compositions of the present invention include those that contain one or more other active ingredients, in addition to a compound of Formula I.
- Examples of other active ingredients that may be administered in combination with a compound of Formula I, and either administered separately or in the same pharmaceutical composition include, but are not limited to: (a) PPAR gamma agonists and selective PPAR gamma partial agonists (SPPARM's), including both ghtazones and non-ghtazones (e.g. troghtazone, pioghtazone, enghtazone, MCC-555, rosightazone, balaghtazone, netoghtazone, T-131, LY-300512, and LY-818, and SPPARM's described in US Patent 6,525,083, WO 2004/020409, and WO 2004/020408);
- SPPARM's selective PPAR gamma partial agonists
- PTP-IB protein tyrosine phosphatase- IB
- D-FV dipeptidyl peptidase IV
- sulfonylureas such as tolbutamide, glimepi ⁇ de, glipizide, and related materials
- ⁇ -glucosidase inhibitors such as acarbose
- agents which improve a patient's lipid profile such as (i) HMG-CoA reductase inhibitors (lovastatin, simvastatin, rosuvastatm, pravastatin, fluvastatin, atorvastatin, nvastatin, itavastatin, ZD-4522 and other statins), (n) bile acid sequestrants (cholestyramine, colestipol, and dialkylaminoalkyl derivatives of a cross-linked dextran), (in) niacin receptor agonists, nicotinyl alcohol, nicotinic acid, or a salt thereof, (iv) PP ARa agonists such as fenofib ⁇ c acid derivatives (gemfibrozil, clof ⁇ brate, fenofibrate and bezafibrate), (v) cholesterol absorption inhibitors
- PPAR ⁇ / ⁇ dual agonists such as muraghtazar, tesaghtazar, farghtazar, and JT-501;
- PPAR ⁇ agonists such as those disclosed in WO97/28149;
- antiobesity compounds such as fenfluramine, dexfenfluramme, phentiramine, subitramine, orhstat, neuropeptide Y5 inhibitors, Mc4r agonists, cannabmoid receptor 1 (CB-I) antagonists/inverse agonists, and ⁇ adrenergic receptor agonists;
- ileal bile acid transporter inhibitors (m) agents intended for use in inflammatory conditions such as aspirin, non-steroidal anti-inflammatory drugs, glucocorticoids, azulfidine, and cyclo-oxygenase 2 selective inhibitors; (n) glucagon receptor antagonists; (o) GLP-I, (P) GIP-I, (q) GLP-I analogs, such as exendins, for example exenatide (Byetta),
- Hydroxysterol dehydrogenase- 1 (HSD-I) inhibitors include combinations of a compound of the present invention not only with one other active compound, but also with two or more other active compounds.
- Non- limitmg examples include combinations of compounds having Formula I with two or more active compounds selected from biguanides, sulfonylureas, HMG-CoA reductase inhibitors, other PPAR agonists, PTP-IB inhibitors, DP-IV inhibitors, and anti-obesity compounds.
- Human and mouse GPR40 stable cell-lines were generated in CHO cells stably expressing NFAT BLA (Beta-lactamase).
- a human GPR40 stable cell-line was generated in HEK cells stably expressing the aequo ⁇ n expressing reporter.
- the expression plasmids were transfected using hpofectamme (Life Technologies) following manufacturer's instructions. Stable cell-lines were generated following drug selection.
- FLIPR Fluorescence-induced calcium mobilization of the stable clones.
- GPR40/CHO NFAT BLA cells were seeded into black-wall-clear-bottom 384-well plates (Costar) at 1.4 x 10e4 cells / 20 ⁇ l medium / well. The cells were incubated with 20 ⁇ l / well of the assay buffer (HBSS, 0.1 % BSA, 20 mM HEPES, 2.5 mM probenecid, pH 7.4) containing 8 ⁇ M fluo-4,AM, 0.08 % pluronic acid at room temperature for 100 minutes. Fluorescence output was measured using FLIPR. Compounds were dissolved in DMSO and diluted to desired concentrations with assay buffer. 13.3 ⁇ l/well of compound solution was added. EC50 activities were measured for the compounds using both the human and mouse
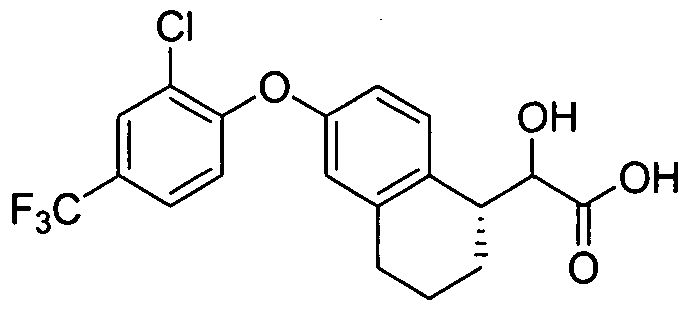
- GPR40 cell lines The activities for the compounds herein are as follows: human FLIPR EC50. 0.012 to 40 ⁇ M; mouse FLIPR EC50: 0.39 to 40 ⁇ M.
- the preferred compounds have an EC50 ⁇ 10 ⁇ M.
- Inositol Phosphate Turnover Assay The assay is performed m 96-well format. HEK cells stably expressing human GPR40 are plated to be 60-80% confluent within 72 hours. After 72 hours, the plates are aspirated and the cells washed with inositol-free DMEM (ICN).
- ICN inositol-free DMEM
- the wash media is replaced with 15OuL of 3H-inositol labeling media (inositol-free media containing 0.4% human albumin or 0.4% mouse albumin, IX pen/strep antibiotics, glutamme, 25mM HEPES to which is added 3H-myo-inositol NEN #NET114A lmCi/mL, 25Ci/mmol diluted 1 : 150 in loading media with a final specific radioactivity of 1UCI/150UL).
- 3H-inositol labeling media inositol-free media containing 0.4% human albumin or 0.4% mouse albumin, IX pen/strep antibiotics, glutamme, 25mM HEPES to which is added 3H-myo-inositol NEN #NET114A lmCi/mL, 25Ci/mmol diluted 1 : 150 in loading media with a final specific radioactivity of 1UCI/150UL).
- the human and mouse albumin can be added after the overnight labeling step before the addition of LiCl.
- the assay is typically run the next day after 18 hours labeling. On the day of the assay, 5uL of 30OmM LiCl is added to all wells and incubated at 37 degrees for 20 mins. 0.75uL of 200X compounds are added and incubated with the cells for 60 minutes at 37 degrees. The media is then aspirated off and the assay terminated with the addition of 6OuL 1OmM formic acid.
- the cells are lysed for 60 mms at room temperature. 15-3OuL of lysate is mixed with 70uL/lmg YSi SPA beads (Amersham) in clear bottom Isoplates. The plates are shaken for 2 hours at room temperature. Beads are allowed to settle and the plates are counted in the Wallac Microbeta.
- Step A To a solution of the material obtained in Step A (3.99 g, 17.79 mmol) in THF (120 mL) were added Et3N (7.5 mL, 53.37 mmol) and tert-butylacetyl chloride (2.79 g, 23.13 mmol), and the mixture was stirred for 1 h at room temperature. LiCl (1.13 g, 26.69 mmol) was added to the mixture, followed by addition of (S)-4-benzyl-2-oxazolidinone (4.73 g, 26.69 mmol), and the mixture was stirred for 3 days at room temperature.
- INTERMEDIATE 5a was prepared from material obtained in the above Step C using a procedure similar to that used in Step D of the preparation of INTERMEDIATE 5.
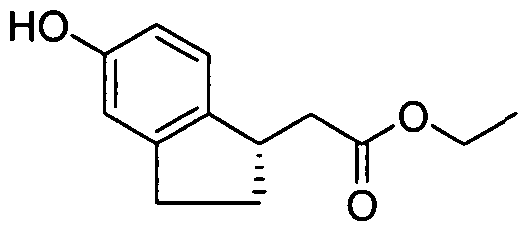
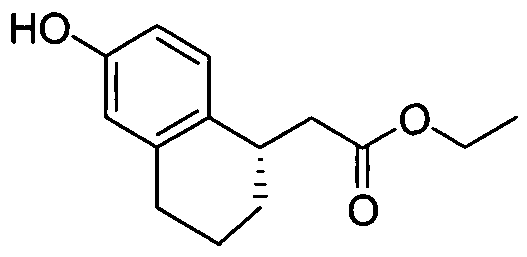
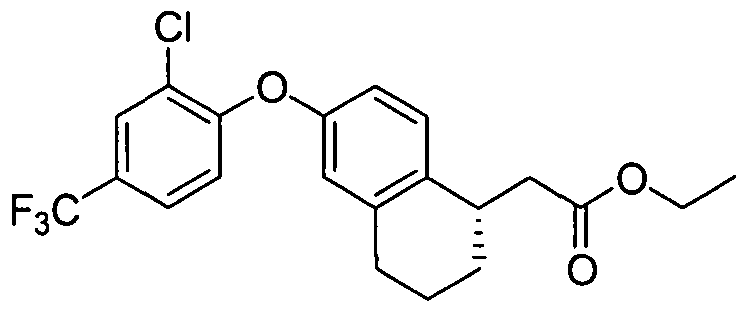
- INTERMEDIATE 1 25 mg, 0.11 mmol was combined with 1 -iodo-2-methyl-4- t ⁇ fluoromethoxybenzene (51 mg, 0.17 mmol), copper (I) iodide (5.3 mg, 0.028 mmol), N,N- dimethylglycine hydrochloride salt (11.9 mg, 0.085 mmol), and Cs 2 CO 3 (111 mg, 0.34 mmol) in 1.2 mL of anhydrous N,N-dimethylformarnide/dioxane (3/1). The reaction tube was sealed, degassed, and backfilled with nitrogen twice, and the mixture was stirred at 110 0 C for 24 hours.
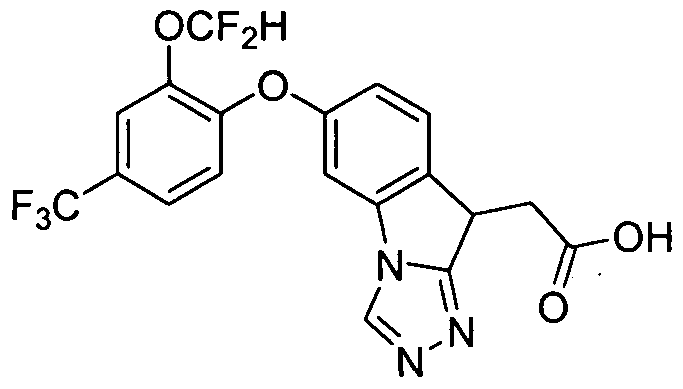
- Example 1 The following examples were prepared following procedures similar to those used in the preparation of Example 1 and Example 2, from their corresponding phenol and aryl hahde precursors. These examples are shown in Table 1.
- Example 20 (46 mg, 0.139 mmol) was dissolved in EtOH, followed by the addition of NaOMe (11 3 mg, 0.209 mmol). Acetophenone (16.7 mg, 0 139 mmol) was then added dropwise, and the reaction was stirred at room temperature for 4 hours. The reaction mixture was acidified with IN HCl and the organic solvent was then removed in vacuo. The aqueous phase was extracted with ethyl acetate (2 X), and the combined extracts were dried with anhydrous Na 2 SO 4 , filtered, and concentrated. The residue was purified by silica gel column chromatography using ethyl acetate/hexanes (1/1) as the eluant to afford the desired product. LCMS for C26H21C1O4: calc. 432, observed 431 [M-H].
- Example 31 (20 mg, 0.05 mmol) was dissolved in MeOH (1 mL) and the reactiom mixture was cooled to 0 0 C. NaBH4 (5.8 mg, 0.15 mmol) was slowly added. The reaction mixture was allowed to slowly warm to room temperature and stirred for another 2 hours. The reaction was quenched by addition of H 2 O followed by the removal of MeOH in vacuo. The aqueous phase was acidified with IN HCl and extracted with ethyl acetate. The organic layer was dried with anhydrous Na 2 SO 4 , filtered, and concentrated to afford the desired compound. LCMS for C20H19F3O5: calc. 396, observed 395 [M-H].
- Example 52 (13 mg, 0.03 mmol) was mixed with TFA (0.2 mL) and triethylsilane (0.1 mL) in a capped tube, and the reaction mixture was heated at 75 0 C for 1 hour in a microwave reactor. Volatiles were removed and the residue was purified by silica gel column chromatography using methanol/dichloromethane (10%) as the eluant to afford the desired product.
- Example 32 (23 mg, 0.06 mmol) was mixed with a solution of trifluoromethyltrimethylsilane (0.18 mL, 0.5 M, 0.09 mmol), and the mixture was cooled to 0 0 C. A solution of tetrabutylammonium fluoride (0.18 mL, 1.0M in THF, 0.18 mmol) was gradually added to the mixture. The temperature was allowed to rise back to room temperature and the mixture was stirred for 1 hour at the same temperature. Excess IN HCl was added, and the mixture was stirred for another 1 hour. Volatiles were removed in vacuo, and the residue was extracted with EtOAc. The organic extract was washed with brine, dried with MgS ⁇ 4, filtered, and concentrated.
- Example 55 To a stirred solution of Example 55 (45 mg, 0.1 mmol) in tetrahydrofuran (2 ml) was added m- tolylboronic acid (15 mg, 0.1 mmol), followed by dppf (diphenylphosphino ferrocene, 10 mg, 0.01 mmol) and an aqueous solution of LiOH (0.5 mL, 2N, 1.0 mmol). The reaction tube was flushed with nitrogen, sealed, and stirred at 40 0 C overnight. After cooling to room temperature, the reaction was quenched with aq. sat. ammonium chloride.
- Example 55 The compound was prepared from Example 55 following procedures similar to the one used in the preparation of Example 56.
- Example 58 (219 mg, 0.54 mmol) was dissolved in anhydrous methanol (5 mL), and a catalytic amount of 4.0M HCl in dioxane was then added, and the reaction mixture was heated in a capped tube for 1 hour.
- the crude methyl ester was obtained by removing the solvents and was purified by silica gel column chromatography (20% ethyl acetate/hexane as the eluant).
- Step A To the material obtained in Step A (1.9 g, 5.85 mmol) in dry THF (30 mL) was added sodium hexamethyldisilazide (NaHMDS) (6.43 mL, IM solution, 6.4 mmol) at -78 0 C. After 30 minutes, t ⁇ methylchlorosilane (TMSCl) (6.4 mL, IM solution in THF, 6.43 mmol) was added. The reaction was allowed to warm to room temperature slowly overnight. THF was then removed in vacuo and the residue was suspended in hexane. This hexane suspension was filtered.
- NaHMDS sodium hexamethyldisilazide
- TMSCl t ⁇ methylchlorosilane
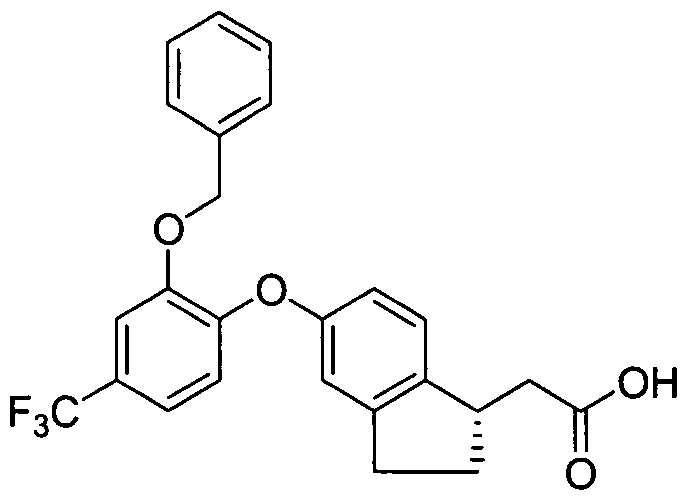
- Example 70 A solution of Example 70 (10 mg) in 30%TFA in CH2Q2 (5 mL) was stirred at room temperature for 1 hour. The reaction was then concentrated in vacuo and the residue was purified by reverse phase HPLC (YMC-Pack Pro Cl 8 5 micron, 20% to 80% CH 3 CN/H 2 O with 0.1%TFA) to give the desired compound. LC-MS for C21H18C1F3O6: calc 458, found 457 [M-H].
- Ethylene glycol 43ml was added, and the mix was stirred at room temp for 1 hour.
- the mix was filtered through a plug of celite plus a layer of silica gel.
- the filter cake was washed with 10%MeOH/CH2Cl2 (750 ml), the filtrate was transferred to a separatory funnel, and the layers were separated.
- the aqueous phase was brought to pH 2 and extracted 3x with
- Example 73 was obtained from corresponding materials following a procedure similar to the one described for Examples 72A and 72B.
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/441,733 US8039484B2 (en) | 2006-10-31 | 2007-10-26 | Antidiabetic bicyclic compounds |
| CA002667249A CA2667249A1 (en) | 2006-10-31 | 2007-10-26 | Antidiabetic bicyclic compounds |
| AU2007314405A AU2007314405A1 (en) | 2006-10-31 | 2007-10-26 | Antidiabetic bicyclic compounds |
| JP2009534659A JP2010508268A (en) | 2006-10-31 | 2007-10-26 | Antidiabetic bicyclic compound |
| EP07839789.0A EP2079467B1 (en) | 2006-10-31 | 2007-10-26 | Antidiabetic bicyclic compounds |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US85572706P | 2006-10-31 | 2006-10-31 | |
| US60/855,727 | 2006-10-31 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008054675A2 true WO2008054675A2 (en) | 2008-05-08 |
| WO2008054675A3 WO2008054675A3 (en) | 2008-11-06 |
Family
ID=39344855
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/022650 WO2008054675A2 (en) | 2006-10-31 | 2007-10-26 | Antidiabetic bicyclic compounds |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8039484B2 (en) |
| EP (1) | EP2079467B1 (en) |
| JP (1) | JP2010508268A (en) |
| AU (1) | AU2007314405A1 (en) |
| CA (1) | CA2667249A1 (en) |
| WO (1) | WO2008054675A2 (en) |
Cited By (48)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2009158426A1 (en) * | 2008-06-25 | 2009-12-30 | Array Biopharma Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| WO2010012650A1 (en) | 2008-07-28 | 2010-02-04 | Syddansk Universitet | Compounds for the treatment of metabolic diseases |
| WO2010143733A1 (en) | 2009-06-09 | 2010-12-16 | Takeda Pharmaceutical Company Limited | Novel fused cyclic compound and use thereof |
| WO2011107494A1 (en) | 2010-03-03 | 2011-09-09 | Sanofi | Novel aromatic glycoside derivatives, medicaments containing said compounds, and the use thereof |
| US8030354B2 (en) | 2007-10-10 | 2011-10-04 | Amgen Inc. | Substituted biphenyl GPR40 modulators |
| WO2011157827A1 (en) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-one derivatives as inhibitors of lipases and phospholipases |
| WO2011161030A1 (en) | 2010-06-21 | 2011-12-29 | Sanofi | Heterocyclic substituted methoxyphenyl derivatives having an oxo group, method for producing same, and use thereof as gpr40 receptor modulators |
| WO2012004269A1 (en) | 2010-07-05 | 2012-01-12 | Sanofi | (2-aryloxy-acetylamino)-phenyl-propionic acid derivatives, method for producing same and use thereof as pharmaceuticals |
| WO2012004270A1 (en) | 2010-07-05 | 2012-01-12 | Sanofi | Spirocyclically substituted 1,3-propane dioxide derivatives, methods for the production thereof and use of the same as medicament |
| WO2012010413A1 (en) | 2010-07-05 | 2012-01-26 | Sanofi | Aryloxy-alkylene substituted hydroxyphenyl hexynoic acids, methods for the production thereof and use of the same as medicament |
| WO2012025811A1 (en) | 2010-08-23 | 2012-03-01 | Lupin Limited | Indolylpyrimidines as modulators of gpr119 |
| WO2012069917A1 (en) | 2010-11-26 | 2012-05-31 | Lupin Limited | Bicyclic gpr119 modulators |
| US8299117B2 (en) | 2010-06-16 | 2012-10-30 | Metabolex Inc. | GPR120 receptor agonists and uses thereof |
| US8309600B2 (en) | 2008-12-18 | 2012-11-13 | Metabolex Inc. | GPR120 receptor agonists and uses thereof |
| WO2013037390A1 (en) | 2011-09-12 | 2013-03-21 | Sanofi | 6-(4-hydroxy-phenyl)-3-styryl-1h-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
| WO2013045413A1 (en) | 2011-09-27 | 2013-04-04 | Sanofi | 6-(4-hydroxy-phenyl)-3-alkyl-1h-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
| GB2498968A (en) * | 2012-02-01 | 2013-08-07 | Prosidion Ltd | Pharmaceutical combination of a GPR119 agonist and a GPR40 agonist |
| WO2014011926A1 (en) | 2012-07-11 | 2014-01-16 | Elcelyx Therapeutics, Inc. | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk |
| WO2014022528A1 (en) | 2012-08-02 | 2014-02-06 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| WO2014064215A1 (en) | 2012-10-24 | 2014-05-01 | INSERM (Institut National de la Santé et de la Recherche Médicale) | TPL2 KINASE INHIBITORS FOR PREVENTING OR TREATING DIABETES AND FOR PROMOTING β-CELL SURVIVAL |
| US8748462B2 (en) | 2008-10-15 | 2014-06-10 | Amgen Inc. | Spirocyclic GPR40 modulators |
| US8785445B2 (en) | 2008-12-22 | 2014-07-22 | Array Biopharma Inc. | 7-phenoxychroman carboxylic acid derivatives |
| WO2014130608A1 (en) | 2013-02-22 | 2014-08-28 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2015051725A1 (en) | 2013-10-08 | 2015-04-16 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| WO2015095256A1 (en) | 2013-12-19 | 2015-06-25 | Merck Sharp & Dohme Corp. | Antidiabetic substituted heteroaryl compounds |
| WO2015097713A1 (en) | 2013-11-14 | 2015-07-02 | Cadila Healthcare Limited | Novel heterocyclic compounds |
| WO2016019863A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | [7,6]-fused bicyclic antidiabetic compounds |
| WO2016022446A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | [5,6]-fused bicyclic antidiabetic compounds |
| WO2016022448A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2016022742A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2016151018A1 (en) | 2015-03-24 | 2016-09-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Method and pharmaceutical composition for use in the treatment of diabetes |
| US9499467B2 (en) | 2011-04-08 | 2016-11-22 | Caldan Therapeutics Limited | Ortho-fluoro substituted compounds for the treatment of metabolic diseases |
| US9957219B2 (en) | 2013-12-04 | 2018-05-01 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2018106518A1 (en) | 2016-12-06 | 2018-06-14 | Merck Sharp & Dohme Corp. | Antidiabetic heterocyclic compounds |
| US10000454B2 (en) | 2014-05-22 | 2018-06-19 | Merck Sharp & Dohme | Antidiabetic tricyclic compounds |
| WO2018118670A1 (en) | 2016-12-20 | 2018-06-28 | Merck Sharp & Dohme Corp. | Antidiabetic spirochroman compounds |
| US10059667B2 (en) | 2014-02-06 | 2018-08-28 | Merck Sharp & Dohme Corp. | Antidiabetic compounds |
| US10519115B2 (en) | 2013-11-15 | 2019-12-31 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| US10676458B2 (en) | 2016-03-29 | 2020-06-09 | Merch Sharp & Dohne Corp. Rahway | Antidiabetic bicyclic compounds |
| US10710986B2 (en) | 2018-02-13 | 2020-07-14 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US10774071B2 (en) | 2018-07-13 | 2020-09-15 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US10899735B2 (en) | 2018-04-19 | 2021-01-26 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US11225471B2 (en) | 2017-11-16 | 2022-01-18 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| US11236085B2 (en) | 2018-10-24 | 2022-02-01 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US11279702B2 (en) | 2020-05-19 | 2022-03-22 | Kallyope, Inc. | AMPK activators |
| US11407768B2 (en) | 2020-06-26 | 2022-08-09 | Kallyope, Inc. | AMPK activators |
| US11512065B2 (en) | 2019-10-07 | 2022-11-29 | Kallyope, Inc. | GPR119 agonists |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DK2582674T3 (en) * | 2010-06-16 | 2014-12-15 | Cymabay Therapeutics Inc | GPR120 receptor agonists and uses thereof. |
| UY33758A (en) * | 2010-12-01 | 2012-06-29 | Boehringer Ingelheim Int | INDANILOXIDIHIDROBENZOFURANILACETIC ACIDS |
| BR112014005104A2 (en) | 2011-09-02 | 2017-07-04 | Salk Inst For Biological Studi | camkii, ip3r, calcineurin, p38 and mk2 / 3 inhibitors to treat bind metabolic disorders of obesity |
| CN104812748B (en) | 2012-11-16 | 2017-10-24 | 百时美施贵宝公司 | Pyrazoline GPR40 conditioning agents |
| WO2014174745A1 (en) * | 2013-04-26 | 2014-10-30 | 国立大学法人京都大学 | Eg5 INHIBITOR |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7407978B2 (en) * | 1999-04-06 | 2008-08-05 | Theracos, Inc. | Heterocyclic analogs of diphenylethylene compounds |
| GB0208384D0 (en) * | 2002-04-11 | 2002-05-22 | Karobio Ab | Novel compounds |
| WO2004106276A1 (en) * | 2003-05-30 | 2004-12-09 | Takeda Pharmaceutical Company Limited | Condensed ring compound |
| US7759493B2 (en) * | 2005-01-31 | 2010-07-20 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
-
2007
- 2007-10-26 WO PCT/US2007/022650 patent/WO2008054675A2/en active Application Filing
- 2007-10-26 EP EP07839789.0A patent/EP2079467B1/en active Active
- 2007-10-26 CA CA002667249A patent/CA2667249A1/en not_active Abandoned
- 2007-10-26 JP JP2009534659A patent/JP2010508268A/en not_active Withdrawn
- 2007-10-26 US US12/441,733 patent/US8039484B2/en active Active
- 2007-10-26 AU AU2007314405A patent/AU2007314405A1/en not_active Abandoned
Non-Patent Citations (2)
| Title |
|---|
| None |
| See also references of EP2079467A4 |
Cited By (81)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| US8030354B2 (en) | 2007-10-10 | 2011-10-04 | Amgen Inc. | Substituted biphenyl GPR40 modulators |
| US9096567B2 (en) | 2008-06-25 | 2015-08-04 | Array Biopharma Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| US8569522B2 (en) | 2008-06-25 | 2013-10-29 | Array Biopharma Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| US8716498B2 (en) | 2008-06-25 | 2014-05-06 | Array Biopharma, Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| CN103450138A (en) * | 2008-06-25 | 2013-12-18 | 阵列生物制药公司 | Preparation of intermidiates of 6-substituted phenoxychroman carboxylic acid derivatives |
| CN102131794B (en) * | 2008-06-25 | 2013-09-25 | 阵列生物制药公司 | 6-substituted phenoxychroman carboxylic acid derivatives |
| EP2826774A1 (en) * | 2008-06-25 | 2015-01-21 | Array Biopharma, Inc. | Intermediates for 6-substituted phenoxychroman carboxylic acid derivatives |
| US9556139B2 (en) | 2008-06-25 | 2017-01-31 | Array Biopharma Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| JP2011526290A (en) * | 2008-06-25 | 2011-10-06 | アレイ バイオファーマ、インコーポレイテッド | 6-substituted phenoxycyclomancarboxylic acid derivatives |
| EP3144303A1 (en) * | 2008-06-25 | 2017-03-22 | Array Biopharma, Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| KR101728175B1 (en) | 2008-06-25 | 2017-04-18 | 어레이 바이오파마 인크. | 6-substituted phenoxychroman carboxylic acid derivatives |
| RU2507200C2 (en) * | 2008-06-25 | 2014-02-20 | Эррэй Биофарма Инк. | Position six-substituted phenoxychromanecarboxylic acid derivatives |
| WO2009158426A1 (en) * | 2008-06-25 | 2009-12-30 | Array Biopharma Inc. | 6-substituted phenoxychroman carboxylic acid derivatives |
| WO2010012650A1 (en) | 2008-07-28 | 2010-02-04 | Syddansk Universitet | Compounds for the treatment of metabolic diseases |
| US8586607B2 (en) | 2008-07-28 | 2013-11-19 | Syddansk Universitet | Compounds for the treatment of metabolic diseases |
| US8748462B2 (en) | 2008-10-15 | 2014-06-10 | Amgen Inc. | Spirocyclic GPR40 modulators |
| US8598374B2 (en) | 2008-12-18 | 2013-12-03 | Metabolex, Inc. | GPR120 receptor agonists and uses thereof |
| US8309600B2 (en) | 2008-12-18 | 2012-11-13 | Metabolex Inc. | GPR120 receptor agonists and uses thereof |
| TWI469977B (en) * | 2008-12-22 | 2015-01-21 | Array Biopharma Inc | 7-phenoxychroman carboxylic acid derivatives |
| US8785445B2 (en) | 2008-12-22 | 2014-07-22 | Array Biopharma Inc. | 7-phenoxychroman carboxylic acid derivatives |
| WO2010143733A1 (en) | 2009-06-09 | 2010-12-16 | Takeda Pharmaceutical Company Limited | Novel fused cyclic compound and use thereof |
| WO2011107494A1 (en) | 2010-03-03 | 2011-09-09 | Sanofi | Novel aromatic glycoside derivatives, medicaments containing said compounds, and the use thereof |
| US8476308B2 (en) | 2010-06-16 | 2013-07-02 | Metabolex, Inc. | GPR120 receptor agonists and uses thereof |
| US8299117B2 (en) | 2010-06-16 | 2012-10-30 | Metabolex Inc. | GPR120 receptor agonists and uses thereof |
| WO2011157827A1 (en) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-one derivatives as inhibitors of lipases and phospholipases |
| WO2011161030A1 (en) | 2010-06-21 | 2011-12-29 | Sanofi | Heterocyclic substituted methoxyphenyl derivatives having an oxo group, method for producing same, and use thereof as gpr40 receptor modulators |
| WO2012010413A1 (en) | 2010-07-05 | 2012-01-26 | Sanofi | Aryloxy-alkylene substituted hydroxyphenyl hexynoic acids, methods for the production thereof and use of the same as medicament |
| US8648038B2 (en) | 2010-07-05 | 2014-02-11 | Sanofi | (2-aryloxyacetylamino)phenylpropionic acid derivatives, processes for preparation thereof and use thereof as medicaments |
| WO2012004269A1 (en) | 2010-07-05 | 2012-01-12 | Sanofi | (2-aryloxy-acetylamino)-phenyl-propionic acid derivatives, method for producing same and use thereof as pharmaceuticals |
| WO2012004270A1 (en) | 2010-07-05 | 2012-01-12 | Sanofi | Spirocyclically substituted 1,3-propane dioxide derivatives, methods for the production thereof and use of the same as medicament |
| US8859494B2 (en) | 2010-07-05 | 2014-10-14 | Sanofi | Spirocyclically substituted 1,3-propane dioxide derivatives, processes for preparation thereof and use thereof as a medicament |
| WO2012025811A1 (en) | 2010-08-23 | 2012-03-01 | Lupin Limited | Indolylpyrimidines as modulators of gpr119 |
| WO2012069917A1 (en) | 2010-11-26 | 2012-05-31 | Lupin Limited | Bicyclic gpr119 modulators |
| US9000175B2 (en) | 2010-11-26 | 2015-04-07 | Lupin Limited | Bicyclic GPR119 modulators |
| US9499467B2 (en) | 2011-04-08 | 2016-11-22 | Caldan Therapeutics Limited | Ortho-fluoro substituted compounds for the treatment of metabolic diseases |
| WO2013037390A1 (en) | 2011-09-12 | 2013-03-21 | Sanofi | 6-(4-hydroxy-phenyl)-3-styryl-1h-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
| WO2013045413A1 (en) | 2011-09-27 | 2013-04-04 | Sanofi | 6-(4-hydroxy-phenyl)-3-alkyl-1h-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
| GB2498968A (en) * | 2012-02-01 | 2013-08-07 | Prosidion Ltd | Pharmaceutical combination of a GPR119 agonist and a GPR40 agonist |
| WO2014011926A1 (en) | 2012-07-11 | 2014-01-16 | Elcelyx Therapeutics, Inc. | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk |
| WO2014022528A1 (en) | 2012-08-02 | 2014-02-06 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| US9527875B2 (en) | 2012-08-02 | 2016-12-27 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| WO2014064215A1 (en) | 2012-10-24 | 2014-05-01 | INSERM (Institut National de la Santé et de la Recherche Médicale) | TPL2 KINASE INHIBITORS FOR PREVENTING OR TREATING DIABETES AND FOR PROMOTING β-CELL SURVIVAL |
| WO2014130608A1 (en) | 2013-02-22 | 2014-08-28 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| US9840512B2 (en) | 2013-02-22 | 2017-12-12 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2015051725A1 (en) | 2013-10-08 | 2015-04-16 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| US9932311B2 (en) | 2013-10-08 | 2018-04-03 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| WO2015097713A1 (en) | 2013-11-14 | 2015-07-02 | Cadila Healthcare Limited | Novel heterocyclic compounds |
| US10246470B2 (en) | 2013-11-14 | 2019-04-02 | Cadila Healthcare Limited | Heterocyclic compounds |
| US10011609B2 (en) | 2013-11-14 | 2018-07-03 | Cadila Healthcare Limited | Heterocyclic compounds |
| US10519115B2 (en) | 2013-11-15 | 2019-12-31 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| US9957219B2 (en) | 2013-12-04 | 2018-05-01 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| US9834563B2 (en) | 2013-12-19 | 2017-12-05 | Merck Sharp & Dohme Corp. | Antidiabetic substituted heteroaryl compounds |
| EP3974413A1 (en) | 2013-12-19 | 2022-03-30 | Merck Sharp & Dohme Corp. | Antidiabetic substituted heteroaryl compounds |
| WO2015095256A1 (en) | 2013-12-19 | 2015-06-25 | Merck Sharp & Dohme Corp. | Antidiabetic substituted heteroaryl compounds |
| US10059667B2 (en) | 2014-02-06 | 2018-08-28 | Merck Sharp & Dohme Corp. | Antidiabetic compounds |
| US10000454B2 (en) | 2014-05-22 | 2018-06-19 | Merck Sharp & Dohme | Antidiabetic tricyclic compounds |
| WO2016019863A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | [7,6]-fused bicyclic antidiabetic compounds |
| WO2016022446A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | [5,6]-fused bicyclic antidiabetic compounds |
| US10968193B2 (en) | 2014-08-08 | 2021-04-06 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| US10100042B2 (en) | 2014-08-08 | 2018-10-16 | Merck Sharp & Dohme Corp. | [5,6]—fused bicyclic antidiabetic compounds |
| US10131651B2 (en) | 2014-08-08 | 2018-11-20 | Merck Sharp & Dohme Corp. | [7,6]-fused bicyclic antidiabetic compounds |
| WO2016022448A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2016022742A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| US10662171B2 (en) | 2014-08-08 | 2020-05-26 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2016151018A1 (en) | 2015-03-24 | 2016-09-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Method and pharmaceutical composition for use in the treatment of diabetes |
| US10676458B2 (en) | 2016-03-29 | 2020-06-09 | Merch Sharp & Dohne Corp. Rahway | Antidiabetic bicyclic compounds |
| WO2018106518A1 (en) | 2016-12-06 | 2018-06-14 | Merck Sharp & Dohme Corp. | Antidiabetic heterocyclic compounds |
| US11072602B2 (en) | 2016-12-06 | 2021-07-27 | Merck Sharp & Dohme Corp. | Antidiabetic heterocyclic compounds |
| WO2018118670A1 (en) | 2016-12-20 | 2018-06-28 | Merck Sharp & Dohme Corp. | Antidiabetic spirochroman compounds |
| US10968232B2 (en) | 2016-12-20 | 2021-04-06 | Merck Sharp & Dohme Corp. | Antidiabetic spirochroman compounds |
| US11225471B2 (en) | 2017-11-16 | 2022-01-18 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| US10710986B2 (en) | 2018-02-13 | 2020-07-14 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US11555029B2 (en) | 2018-02-13 | 2023-01-17 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US10899735B2 (en) | 2018-04-19 | 2021-01-26 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US10774071B2 (en) | 2018-07-13 | 2020-09-15 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US11236085B2 (en) | 2018-10-24 | 2022-02-01 | Gilead Sciences, Inc. | PD-1/PD-L1 inhibitors |
| US11512065B2 (en) | 2019-10-07 | 2022-11-29 | Kallyope, Inc. | GPR119 agonists |
| US11279702B2 (en) | 2020-05-19 | 2022-03-22 | Kallyope, Inc. | AMPK activators |
| US11851429B2 (en) | 2020-05-19 | 2023-12-26 | Kallyope, Inc. | AMPK activators |
| US11407768B2 (en) | 2020-06-26 | 2022-08-09 | Kallyope, Inc. | AMPK activators |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2079467A4 (en) | 2010-12-15 |
| CA2667249A1 (en) | 2008-05-08 |
| EP2079467B1 (en) | 2015-11-18 |
| US20100105725A1 (en) | 2010-04-29 |
| JP2010508268A (en) | 2010-03-18 |
| US8039484B2 (en) | 2011-10-18 |
| AU2007314405A1 (en) | 2008-05-08 |
| WO2008054675A3 (en) | 2008-11-06 |
| EP2079467A2 (en) | 2009-07-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2008054675A2 (en) | Antidiabetic bicyclic compounds | |
| EP2021327B1 (en) | Antidiabetic bicyclic compounds | |
| EP1846379B1 (en) | Antidiabetic bicyclic compounds | |
| WO2008054674A2 (en) | Antidiabetic bicyclic compounds | |
| US20080090865A1 (en) | Antidiabetic Bicyclic Compounds | |
| JP6330011B2 (en) | Kynurenin-3-monooxygenase inhibitor, pharmaceutical composition thereof, and method of use thereof | |
| JP4866901B2 (en) | Tricyclic compounds | |
| JP4759517B2 (en) | Production and use of arylalkyl acid derivatives for the treatment of obesity | |
| EP1861382B1 (en) | Fused aromatic compounds having anti-diabetic activity | |
| AU2010206783A1 (en) | Pentafluorosulpholane-containing antidiabetic compounds | |
| AU2010206786A1 (en) | Bridged and fused heterocyclic antidiabetic compounds | |
| EP1569915A2 (en) | Spirocyclic ureas, compositions containing such compounds and methods of use | |
| CA3115297A1 (en) | Benzoxazole and related compounds useful as chaperone-mediated autophagy modulators | |
| JP2012526136A (en) | Substituted spirocyclic amines useful as antidiabetic compounds | |
| WO2004011446A1 (en) | Indane, dihydrobenzofuran, and tetrahydronaphthalene carboxylic acid derivatives and their use as antidiabetics | |
| EP3704122B1 (en) | Fused triazole agonists of the apj receptor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07839789 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12441733 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007314405 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2009534659 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2667249 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007839789 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2007314405 Country of ref document: AU Date of ref document: 20071026 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |