WO2005077962A2 - Crowned dithiocarbamate metal complexes and methods for their use - Google Patents
Crowned dithiocarbamate metal complexes and methods for their use Download PDFInfo
- Publication number
- WO2005077962A2 WO2005077962A2 PCT/US2005/004872 US2005004872W WO2005077962A2 WO 2005077962 A2 WO2005077962 A2 WO 2005077962A2 US 2005004872 W US2005004872 W US 2005004872W WO 2005077962 A2 WO2005077962 A2 WO 2005077962A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- composition
- formula
- crowned
- crown ether
- Prior art date
Links
- 0 [O-][N+](c1cc(OCCOCCOCCOCC*2)c2cc1NC([S-])=S)=O Chemical compound [O-][N+](c1cc(OCCOCCOCCOCC*2)c2cc1NC([S-])=S)=O 0.000 description 5
- ITWBWJFEJCHKSN-UHFFFAOYSA-N C1NCCNCCNC1 Chemical compound C1NCCNCCNC1 ITWBWJFEJCHKSN-UHFFFAOYSA-N 0.000 description 1
- JCIKVZBEVWXJEU-QPEQYQDCSA-N CC(/C=C(/C)\NP([n]1nc(C)cc1C)([n]1nc(C)cc1C)=O)=[N-] Chemical compound CC(/C=C(/C)\NP([n]1nc(C)cc1C)([n]1nc(C)cc1C)=O)=[N-] JCIKVZBEVWXJEU-QPEQYQDCSA-N 0.000 description 1
- FFLDIIPYZRAROS-IWRWAGCXSA-O CC(CN/C=C\CN)(CN/C=C\C=N)CN/C=C\C=[NH2+] Chemical compound CC(CN/C=C\CN)(CN/C=C\C=N)CN/C=C\C=[NH2+] FFLDIIPYZRAROS-IWRWAGCXSA-O 0.000 description 1
- VAQOVJVTDCNSLR-UHFFFAOYSA-N CCOC[n]1c(C(c2ncc[n]2C[O](C)CC)c2ncc[n]2C[O](C)(C)CC)ncc1 Chemical compound CCOC[n]1c(C(c2ncc[n]2C[O](C)CC)c2ncc[n]2C[O](C)(C)CC)ncc1 VAQOVJVTDCNSLR-UHFFFAOYSA-N 0.000 description 1
- LRPVVAOGGZFVFO-UHFFFAOYSA-N CN1CCCN(C)CCCN(C)CCC1 Chemical compound CN1CCCN(C)CCCN(C)CCC1 LRPVVAOGGZFVFO-UHFFFAOYSA-N 0.000 description 1
- WLDGDTPNAKWAIR-UHFFFAOYSA-N CN1CCN(C)CCN(C)CC1 Chemical compound CN1CCN(C)CCN(C)CC1 WLDGDTPNAKWAIR-UHFFFAOYSA-N 0.000 description 1
- STVKUTANJZWWHV-UHFFFAOYSA-N COCCN1CCN(CCOC)CCN(CCOC)CC1 Chemical compound COCCN1CCN(CCOC)CCN(CCOC)CC1 STVKUTANJZWWHV-UHFFFAOYSA-N 0.000 description 1
- LIOWQFNCPPQNFF-UHFFFAOYSA-N CSC(NCC(COCCOCCO)OCCOCCO)=S Chemical compound CSC(NCC(COCCOCCO)OCCOCCO)=S LIOWQFNCPPQNFF-UHFFFAOYSA-N 0.000 description 1
- XRMOVMMASOEWBW-UHFFFAOYSA-N CSC(Nc(c([N+]([O-])=O)c1)cc2c1OCCOCCOCCOCCO2)=S Chemical compound CSC(Nc(c([N+]([O-])=O)c1)cc2c1OCCOCCOCCOCCO2)=S XRMOVMMASOEWBW-UHFFFAOYSA-N 0.000 description 1
- AXQNHMCRTMJGSX-UHFFFAOYSA-N C[n]1c(CC(Cc2ncc[n]2C)Cc2ncc[n]2C)ncc1 Chemical compound C[n]1c(CC(Cc2ncc[n]2C)Cc2ncc[n]2C)ncc1 AXQNHMCRTMJGSX-UHFFFAOYSA-N 0.000 description 1
- JEOJOJBAMQXQII-UHFFFAOYSA-N C[n]1c(P(c2ncc[n]2C)(c2ncc[n]2C)=O)ncc1 Chemical compound C[n]1c(P(c2ncc[n]2C)(c2ncc[n]2C)=O)ncc1 JEOJOJBAMQXQII-UHFFFAOYSA-N 0.000 description 1
- OPHYGOJQLJVSKW-WWAOEQMPSA-O N=C/C=C\NC(N/C=C\C=[NH2+])[n]1nccc1 Chemical compound N=C/C=C\NC(N/C=C\C=[NH2+])[n]1nccc1 OPHYGOJQLJVSKW-WWAOEQMPSA-O 0.000 description 1
- KBHGBWZFBPCXJP-UHFFFAOYSA-N O=P(c1ccccn1)(c1ncccc1)c1ncccc1 Chemical compound O=P(c1ccccn1)(c1ncccc1)c1ncccc1 KBHGBWZFBPCXJP-UHFFFAOYSA-N 0.000 description 1
- MUYUVVLNROOQHQ-UHFFFAOYSA-N S=C(NCC1OCCOCCOCCOCCOCCOC1)I Chemical compound S=C(NCC1OCCOCCOCCOCCOCCOC1)I MUYUVVLNROOQHQ-UHFFFAOYSA-N 0.000 description 1
- XOWULGQEQXFGNO-ARJAWSKDSA-M [S-]C(NCC1OCCO/C=C\OCCOCCOC1)=S Chemical compound [S-]C(NCC1OCCO/C=C\OCCOCCOC1)=S XOWULGQEQXFGNO-ARJAWSKDSA-M 0.000 description 1
- BOAUNQLCKZCCQZ-UHFFFAOYSA-M [S-]C(NCC1OCCOCCOCCOCCOCCOC1)=S Chemical compound [S-]C(NCC1OCCOCCOCCOCCOCCOC1)=S BOAUNQLCKZCCQZ-UHFFFAOYSA-M 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/0474—Organic compounds complexes or complex-forming compounds, i.e. wherein a radioactive metal (e.g. 111In3+) is complexed or chelated by, e.g. a N2S2, N3S, NS3, N4 chelating group
- A61K51/0478—Organic compounds complexes or complex-forming compounds, i.e. wherein a radioactive metal (e.g. 111In3+) is complexed or chelated by, e.g. a N2S2, N3S, NS3, N4 chelating group complexes from non-cyclic ligands, e.g. EDTA, MAG3
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/041—Heterocyclic compounds
- A61K51/0412—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K51/0423—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having two or more oxygen atoms in the same ring, e.g. crown ethers, guanadrel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/041—Heterocyclic compounds
- A61K51/044—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine, rifamycins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/0474—Organic compounds complexes or complex-forming compounds, i.e. wherein a radioactive metal (e.g. 111In3+) is complexed or chelated by, e.g. a N2S2, N3S, NS3, N4 chelating group
- A61K51/0476—Organic compounds complexes or complex-forming compounds, i.e. wherein a radioactive metal (e.g. 111In3+) is complexed or chelated by, e.g. a N2S2, N3S, NS3, N4 chelating group complexes from monodendate ligands, e.g. sestamibi
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F13/00—Compounds containing elements of Groups 7 or 17 of the Periodic System
- C07F13/005—Compounds without a metal-carbon linkage
Definitions
- the present invention relates to compositions containing crowned dithiocarbamate metal complexes and to methods for their use.
- Perfusion refers to blood flow at the cellular level, such as the delivery of nutrients and removal of waste products to maintain cellular function (Dilsizian, V. J. Nucl. Cardiol. 2000, 7, 180; Marmion M. and Deutsch, E. Quart. J. Nucl. Med. 2000, 7, 701).
- An ideal myocardial perfusion agent has a high first-pass extraction with stable myocardial retention, which linearly tracks myocardial blood flow over a wide range.
- Hepatic and gastrointestinal uptake should be minimal with exercise as well as with pharmacological stress and rest studies.
- the agent may redistribute; but should do so in a predictable and reliable manner (Saha, G. B. et al. Nucl. Med. Biol. 1992, 19, 1; Jain, D. Semin. Nucl. Med. 1999, 29, 221; Banerjee, S. et al. Semin. Nucl. Med. 2001, 31, 260).
- the first crowned dithiocarbamate includes a first crown ether-containing group of formula [(CH 2 )a-O] b -(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2.
- the first crowned dithiocarbamate includes a first crown ether-containing group of formula [(CH 2 )a-O] b -(CH 2 ) c , wherein a is at least 2, b is at
- M is a transition metal selected from the group consisting of Fe(ll), Fe(lll), Mn(ll), Mn(lll), Co(ll), Co(lll), Ni(ll), Cu(ll), Zn(ll), Ru(ll), Ru(lll), Pd(ll), and Pt(ll);
- p and p' are integers and are independently selected from 0-2;
- R 1 and R 2 contain a crown ether-containing group of formula [(CH 2 )a-O] -(CH 2 )c, wherein a is at least 2, b is at least 3, and c is at least 2, or wherein R and R 2 together contain the crown ether-containing group;
- L 6 is a tripodal chelator with a formula selected from the group consisting of:
- a 1 , A 2 and A 3 are imine-N containing heterocycles.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- R 10 , R 11 and R 12 are selected from a group of formula -(CH 2 ) g -, wherein g is 2-5.
- R 13 is selected from the group consisting of H, alkyl and aryl.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- a method for radioimaging a subject embodying features of the present invention includes: (a) providing (i) a subject and (ii) a composition containing a compound having a formula (M ⁇ N)L 1 and pharmaceutically acceptable salts thereof; (b) administering the composition to the subject; and c) scanning at least a portion of the subject using a radioimaging device.
- N is nitrogen
- M is a radioactive transition metal
- L 1 is a first crowned dithiocarbamate.
- the first crowned dithiocarbamate contains a first crown ether-containing group of formula [(CH 2 )a-O] b -(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2.
- a method of treating a disease resulting from overproduction of nitric oxide or reactive oxygen species embodying features of the present invention includes: (a) providing (i) a subject with a disease and (ii) a composition containing a compound having a formula:
- M is a transition metal selected from the group consisting of Fe(ll), Fe(lll), Mn(ll), Mn(lll), Co(ll), Co(lll), Ni(ll), Cu(ll), Zn(ll), Ru(ll), Ru(lll), Pd(ll), and Pt(ll).
- the subscripts p and p' are integers and are independently selected from 0-2.
- R 1 and R 2 contains a crown ether-containing group of formula [(CH 2 ) a -O] b -(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2, or wherein R 1 and R 2 together contain the crown ether-containing group.
- L 6 is a tripodal chelator with a formula selected from the group consisting of:
- a 1 , A 2 and A 3 are imine-N containing heterocycles.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- R 10 , R 11 and R 12 are selected from a group of formula -(CH 2 ) g -, wherein g is 2-5.
- R 13 is selected from the group consisting of H, alkyl and aryl.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- a method of treating metal poisoning embodying features of the present invention includes (a) providing (i) a subject with metal poisoning, and (ii) a composition containing a crowned dithiocarbamate; and (b) administering the composition to the subject.
- the crowned dithiocarbamate includes a crown ether-containing group of formula [(CH 2 ) a -O] b -(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2.
- compositions embodying features of the present invention contain crowned dithiocarbamate metal complexes. Representative compositions and methods for their use are described hereinbelow. Compositions embodying features of the present invention provide neutral and cationic radioactive metal-nitrido complexes of crowned dithiocarbamates (DTCs). Methods embodying features of the present invention include using these complexes as radiopharmaceuticals for diagnosis and treatment of cardiovascular disorders, infectious disease, and cancer. The present invention also provides tripodal chelator-metal complexes of crowned DTCs and methods of using these complexes for treating diseases such as those characterized by nitric oxide overproduction. The present invention further provides methods of using crowned DTCs for heavy metal detoxification.
- DTCs dithiocarbamate metal complexes
- subject and “patient” refer to any animal, such as a mammal. Representative examples include but are not limited to humans, dogs, cats, birds, livestock, and the like.
- substituted refers to the replacement of any one or more hydrogens on the designated atom with a selection from the indicated group, provided that the designated atom's normal valency is not exceeded and that the substitution results in a stable compound.
- two hydrogens on the atom are replaced.
- Keto substituents are not present on aromatic moieties.
- a ring system e.g., carbocyclic or heterocyclic
- Isotopes include those atoms having the same atomic number but different mass numbers.
- isotopes of hydrogen include tritium and deuterium.
- isotopes of carbon include C-13 and C-14.
- R 6 may optionally be substituted with up toitwo R 6 groups and R 6 at each occurrence is selected independently from the definition of R 6 .
- combinations of substituents and/or variables are preferably stable compounds. When a bond to a substituent is shown to cross a bond connecting two atoms in a ring, then such substituent may be bonded to any atom on the ring. When a substituent is listed without indicating the atom via which such substituent is bonded to the rest of the compound of a given formula, then such substituent may be bonded via any atom in such substituent.
- alkyl refers to both branched and straight-chain saturated aliphatic hydrocarbon groups having the specified number of carbon atoms.
- Representative examples of alkyl include but are not limited to methyl, ethyl, n-propyl, /-propyl, n-butyl, s-butyl, f-butyl, n-pentyl, s-pentyl, and the like.
- Representative examples of haloalkyls include but are not limited to trifluoromethyl, trichloromethyl, pentafluoroethyl, pentachloroethyl, and the like.
- alkoxy refers to an alkyl group as defined above with the indicated number of carbon atoms attached through an oxygen bridge. Representative examples of alkoxys include but are not limited to methoxy, ethoxy, n-propoxy, /-propoxy, n-butoxy, s-butoxy, -butoxy, n-pentoxy, s- pentoxy, and the like.
- cycloalkyl refers to saturated ring groups, including but not limited to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and the like.
- alkenyl refers to hydrocarbon chains of either a straight or branched configuration containing one or more unsaturated carbon-carbon bonds which may occur in any stable point along the chain. Representative examples include but are not limited to ethenyl, propenyl, and the like.
- alkynyl refers to hydrocarbon chains of either a straight or branched configuration containing one or more triple carbon-carbon bonds which may occur in any stable point along the chain. Representative examples include but are not limited to ethynyl, propynyl, and the like.
- heterocycle and “heterocyclic system” refer to a stable 5- to 7-membered monocyclic or bicyclic ring or a 7-to 10-membered bicyclic heterocyclic ring which is saturated, partially unsaturated or unsaturated (e.g., aromatic), and which contains carbon atoms and from 1 to 4 heteroatoms independently selected from the group consisting of N, O and S.
- These terms include any bicyclic group in which any of the above-defined heterocyclic rings is fused to a benzene ring.
- the nitrogen and sulfur heteroatoms may optionally be partially or completely oxidized.
- the heterocyclic ring may be attached to its pendant group at any heteroatom or carbon atom (preferably resulting in a stable structure).
- heterocyclic rings described herein may be substituted on carbon or on a nitrogen atom.
- a nitrogen in the heterocycle may optionally be quatemized. It is preferred that when the total number of S and O atoms in the heterocycle exceeds 1 that the heteroatoms are not adjacent to one another. In some embodiments, it is preferred that the total number of S and O atoms in the heterocycle is not more than 1.
- aromatic heterocyclic system and “heteroaryl” refer to a stable 5- to 7-membered monocyclic or bicyclic ring or a 7-to 10-membered bicyclic heterocyclic aromatic ring which contains carbon atoms and from 1 to 4 heteroatoms independently selected from the group consisting of N, O and S. In some embodiments, it is preferred that the total number of S and O atoms in the aromatic heterocycle is not more than 1.
- heteroatom-containing rings for use in accordance with the present invention include but are not limited to: acridinyl, azocinyl, benzimidazolyl, benzofuranyl, benzothiofuranyl, benzothiophenyl, benzoxazolyl, benzthiazolyl, benztriazolyl, benztetrazolyl, benzisoxazolyl, benzisothiazolyl, benzimidazolinyl, carbazolyl, 4aH-carbazolyl, carbolinyl, chromanyl, chromenyl, cinnolinyl, decahydroquinolinyl, 2H,6H-1 ,5,2- dithiazinyl, dihydrofuro[2,3-b]tetrahydrofuran, furanyl, furazanyl, imidazolidinyl, imidazolinyl, imidazolyl, 1 H-indazolyl, in
- preferred heterocycles include but are not limited to: pyridinyl, furanyl, thienyl, pyrrolyl, pyrazolyl, pyrrolidinyl, imidazolyl, indolyl, benzimidazolyl, 1 H- indazolyl, oxazolidinyl, benzotriazolyl, benzisoxazolyl, oxindolyl, benzoxazolinyl, isatinoyl, and the like. Fused ring and spiro compounds containing, for example, one or more of the above-described representative heterocycles are also contemplated for use in accordance with the present invention.
- phrases "pharmaceutically acceptable” refers to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use with the tissues of human beings and animals without resulting in excessive toxicity, irritation, allergic response, or other problems or complications, commensurate with a reasonable benefit/risk ratio.
- pharmaceutically acceptable salts refers to derivatives of the disclosed compounds wherein the parent compound is modified by making acid or base salts thereof.
- Representative examples of pharmaceutically acceptable salts include but are not limited to: mineral or organic acid salts of basic residues, such as amines; and alkali or organic salts of acidic residues, such as carboxylic acids.
- Pharmaceutically acceptable salts in accordance with the present invention include conventional non-toxic salts or the quaternary ammonium salts of the parent compound formed, for example, from non-toxic inorganic or organic acids.
- such conventional non-toxic salts include but are not limited to those derived from inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, and nitric acids; and the salts prepared from organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic, malic, tartaric, citric, ascorbic, pamoic, maleic, hydroxymaleic, phenylacetic, glutamic, benzoic, salicylic, sulfanilic, 2-acetoxybenzoic, fumaric, toluenesulfonic, methanesulfonic, ethane disulfonic, oxalic, and isethionic acids.
- inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, and nitric acids
- organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic
- salts in accordance with the present invention may be synthesized from a parent compound containing a basic or acidic moiety by conventional chemical methods. Typically, such salts may be prepared by reacting the free acid or base forms of these compounds with a substantially stoichiometric amount and/or a slight excess of the appropriate base or acid in water or in an organic solvent, or in a mixture of the two. In some embodiments, nonaqueous media such as ether, ethyl acetate, ethanol, isopropanol, and acetonitrile are preferred. Lists of suitable salts are found in Remington's Pharmaceutical Sciences, 17th ed., Mack Publishing Company, l
- prodrugs are known to enhance numerous desirable qualities of pharmaceuticals (e.g., solubility, bioavailability, manufacturing, etc.) the compounds of the present invention may be delivered in prodrug forms.
- the present invention includes prodrugs of compounds embodying features of the present invention, methods of delivering the same, and compositions containing the same.
- prodrugs refers to any covalently bonded carriers which release an active parent drug of the present invention in vivo when such prodrug is administered to a mammalian subject.
- Prodrugs in accordance with the present invention may be prepared by modifying functional groups present in the compound in such a way that the modifications are cleaved, either in routine manipulation or in vivo, to the parent compound.
- Representative prodrugs include compounds embodying features of the present invention, wherein a hydroxy, amino, or sulfhydryl group is bonded to any group such that when the prodrug is administered to a mammalian subject, it cleaves to form a free hydroxyl, free amino, or free sulfhydryl group, respectively.
- Representative examples of prodrugs include but are not limited to acetate, formate and benzoate derivatives of alcohol and amine functional groups in compounds embodying features of the present invention.
- the phrases “stable compound” and “stable structure” refer to compounds and structures that are sufficiently robust to survive isolation to a useful degree of purity from a reaction mixture, and formulation into an efficacious therapeutic agent.
- the coordination sphere of the radionuclide includes all the ligands or groups bound to the radionuclide.
- a transition metal radionuclide typically has a coordination number (number of donor atoms) comprised of an integer greater than or equal to 4 and less than or equal to 7 (i.e., there are 4 to 7 atoms bound to the metal and it is said to have a complete coordination sphere).
- the requisite coordination number for a stable radionuclide complex is determined by the identity of the radionuclide, its oxidation state, and the type of donor atoms.
- the present invention provides compositions comprising a compound comprising a formula (M ⁇ N)L 1 and pharmaceutically acceptable salts thereof, wherein N is nitrogen, M is a transition metal, and L 1 is a first crowned dithiocarbamate.
- the first crowned dithiocarbamate comprises a first crown ether-containing group of formula [(CH 2 )a-O]b-(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2.
- the transition metal is covalently bound to the first crowned dithiocarbamate.
- the first crowned dithiocarbamate comprises a formula: R! .N- > V -" R 2
- R 1 or R 2 comprises the first crown ether-containing group, or R and R 2 together comprise the first crown ether-containing group.
- the compound further comprises L 2 and comprises a formula (M ⁇ N)L 1 L 2 and pharmaceutically acceptable salts thereof, wherein L 2 is a second crowned dithiocarbamate.
- the second crowned dithiocarbamate comprises a second crown ether-containing group of formula [(CH 2 ) a -O]b-(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2.
- the transition metal is covalently bound to the first and second crowned dithiocarbamates.
- the second crowned dithiocarbamate comprises a formula:
- the compound further comprises L 3 , L 4 , and L 5 and comprises a formula:
- L 3 , L 4 , and L 5 each comprises an isonitrile of formula: R 5 R 3 — Z-(CH 2 ) q — N ⁇ C R 4
- R 3 , R 4 and R 5 are the same or different, and are selected from the group consisting of H, C1-C10 alkyl substituted with 0-5 R 6 , aryl substituted with 0-5 R 6 , heteroaryl substituted with 0-5 R 6 , and macrocyclic crown ether containing 2-8 ether- oxygen atoms.
- R 7 , R 8 , and R 9 are the same or different, and are selected from the group consisting of H, alkyl, aryl, and heteroaryl, or R 8 and R 9 together form a macrocyclic crown ether containing 2-8 ether-oxygen atoms.
- the compound further comprises L 3 , L 4 , and L 5 and comprises a formula: '
- a 1 , A 2 and A 3 are imine-N containing heterocycles;
- a 4 , A 5 and A 6 are selected from the group consisting of NR 14 , PR 14 , S, and O;
- R 10 , R 11 and R 12 are selected from a group of formula: -(CH 2 ) g -, [0048] wherein g is 2-5;
- R 13 is selected from the group consisting of H, alkyl and aryl group; and
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- the transition metal is a radioactive metal. In some embodiments, the transition metal is 99m Tc or 94m Tc. In other embodiments, the transition metal is 186 Re or 188 Re.
- subscript a in the formula of the first and/or second crown ether-containing group is 2, 3, 4, or 5. In other embodiments, subscript a in the formula of the first and/or second crown ether-containing group is 2 or 3. In some embodiments, subscript a in the formula of the first and/or crown ether- containing group is 2. In other embodiments, subscript b in the formula of the first and/or second crown ether-containing group is 3, 4, 5, 6, 7, or 8.
- subscript b in the formula of the first and/or second crown ether-containing group is 3, 4, 5 or 6.
- subscript c in the formula of the first and/or second crown ether-containing group is 2, 3, 4, or 5.
- subscript c in the formula of the first and/or second crown ether-containing group is 2 or 3.
- subscript c in the formula of the first and/or second crown ether-containing group is 2.
- M is 99m Tc; a is 2 or 3; b is 3-6; c is 2 or 3; q is 1 ; Z is carbon; R 3 , R 4 and R 5 are the same or different, and are selected from the group consisting of H, Cr C 5 alkyl substituted with a R 6 , aryl substituted with a R 6 , heteroaryl substituted with a R 6 , and macrocyclic crown ether containing 3-6 ether-oxygen atoms.
- R 7 , R 8 and R 9 are the same or different, and are selected from the group consisting of H, alkyl, aryl, and heteroaryl, or R 8 and R 9 may be taken together to form a macrocyclic crown ether containing 3-6 ether-oxygen atoms.
- a 1 , A 2 and A 3 are selected from the group consisting of imidazolyl, pyrazolyl, oxazolinyl, methimazolyl, and pyridyl.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- the subscript g is 2 or 3.
- R 13 is selected from the group consisting of H, methyl, and phenyl.
- R 14 is selected from the group consisting of ethyloxyethyl, ethoxylpropyl, methyoxyethyl, and methoxypropyl.
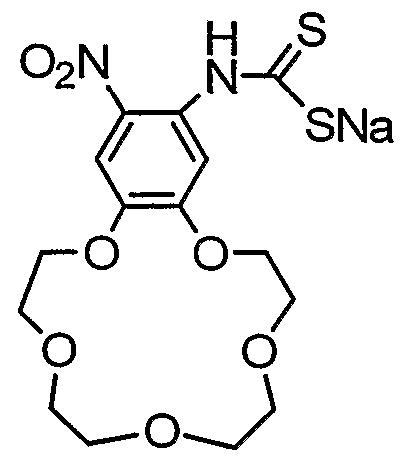
- the first and/or second crowned dithiocarbamate comprises
- the first and/or second crowned dithiocarbamate comprises
- the first and/or second crowned dithiocarbamate comprises
- the first and/or second crowned dithiocarbamate comprises
- the first and/or second crowned dithiocarbamate comprises
- the first and/or second crowned dithiocarbamate comprises
- the first and/or second crowned dithiocarbamate is selected from the group consisting of
- the compound is selected from the group consisting of:
- the compound is selected from the group consisting of: In embodiments including a tripodal chelator, the tripodal chelatord from the group consisting of
- compositions comprising a formula:
- M is a transition metal selected from the group consisting of Fe(ll), Fe(lll), Mn(ll), Mn(lll), Co(ll), Co(lll), Ni(ll), Cu(ll), Zn(II), Ru(ll), Ru(lll), Pd(ll), and Pt(ll);
- p and p' are integers and are independently selected from the group consisting of 0-2.
- R 1 and R 2 comprise a crown ether-containing group of formula [(CH 2 ) a -O] b -(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2, or wherein R 1 and R 2 together comprise the crown ether-containing group.
- L 6 is a tripodal chelator with a formula selected from the group consisting of:
- a 1 , A 2 and A 3 are imine-N containing heterocycles.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- R 10 , R 11 and R 12 are selected from a group of formula: -(CH 2 ) g -, [0064] wherein g is 2-5.
- R 13 is selected from the group consisting of H, alkyl and aryl group.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- M is Ru(lll), p is 1 , p' is 2, R 1 and R 2 are the same or different and are selected from a macrocyclic crown ether- containing group, or R 1 and R 2 are taken together to form a macrocycle of formula [(CH 2 ) a -O] b -(CH 2 ) c , wherein a is 2 or 3, b is 3-6, and c is 2 or 3.
- a 1 , A 2 and A 3 are selected from the group consisting of imidazolyl, pyrazolyl, oxazolinyl, methimazolyl, and pyridyl.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- the subscript g is 2 or 3.
- R 13 is selected from the group consisting of H, methyl, and phenyl.
- R 14 is selected from the group consisting of ethyloxyethyl, ethoxylpropyl, methyoxyethyl, and methoxypropyl.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- the present invention provides methods for radioimaging a subject (e.g., a human) comprising: a) providing a subject, and a composition comprising a compound of formula (M ⁇ N)L 1 and pharmaceutically acceptable salts thereof; b) administering the composition to the subject, and c) scanning at least a portion of the subject using a radioimaging device.
- N is nitrogen
- M is a radioactive transition metal
- L 1 is a first crowned dithiocarbamate, wherein the first crowned dithiocarbamate comprises a first crown ether-containing group of formula [(CH 2 ) a -O] b -(CH 2 )c, wherein a is at least 2, b is at least 3, and c is at least 2;
- at least a portion of the subject is tissue suspected of being diseased.
- the at least a portion of the subject is myocardial tissue.
- the subject is a mammal (e.g., a human, cat, dog, pig, horse or the like).
- the present invention provides methods of treating a disease resulting from overproduction of nitric oxide or reactive oxygen species, comprising; a) providing a subject with a disease, and a composition comprising a compound of formula:
- M is a transition metal selected from the group consisting of Fe(ll), Fe(lll), Mn(ll), Mn(lll), Co(ll), Co(lll), Ni(ll), Cu(ll), Zn(ll), Ru(ll), Ru(lll), Pd(ll), and Pt(ll).
- the subscripts p and p' are integers and are independently selected from 0-2.
- R 1 and R 2 comprise a crown ether-containing group of formula [(CH 2 ) a -O]b- (CH 2 ) C , wherein a is at least 2, b is at least 3, and c is at least 2, or wherein R 1 and R 2 together comprise the crown ether-containing group.
- L 6 is a tripodal chelator with a formula selected from:
- a 1 , A 2 and A 3 are imine-N containing heterocycles.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- R 10 , R 11 and R 12 are selected from a group of formula: -(CH 2 ) g -, [0070] wherein g is 2 - 5.
- R 13 is selected from the group consisting of H, alkyl and aryl.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- the present invention provides methods of treating metal poisoning (metal detoxification), comprising: a) providing a subject with metal poisoning, and a composition comprising a crowned dithiocarbamate, and b) administering the composition (or any other suitable composition described herein) to the subject.
- the crowned dithiocarbamate comprises a crown ether-containing group of formula [(CH 2 ) a -O]b-(CH 2 ) c , wherein a is at least 2, b is at least 3, and c is at least 2.
- the present invention provides methods of making the compositions embodying features of the present invention comprising reacting pertechnetate with (1) a nitrido donor; (2) a reducing agent; (3) and a crowned DTC chelator.
- the nitrido donor is succinyl dihydride
- the reducing agent is stannous chloride.
- kits for the preparation of a radiopharmaceutical embodying features of the present invention comprising: a) a first container (e.g., a bottle) containing a nitrido donor, b) a second container (e.g., a bottle) containing a stannous chloride and a chelating agent able to stabilize the tin cation, and c) a third container (e.g., a bottle) containing a crowned DTC chelator as described herein.
- kits for the preparation of a radiopharmaceutical embodying features of the present invention comprising: a) a first container containing succinyl dihydride, a stannous chloride and a chelating agent able to stabilize the tin cation, and b) a second container containing a crowned DTC chelator as described herein.
- the kits comprise: a) a first container containing succinyl dihydride, stannous chloride and 1,2-diaminopropane-N,N,N',N'-tetraacetic acid or a salt thereof, and b) a second container containing a crowned DTC chelator as described herein.
- kits comprising: a) one or more of compounds or compositions in accordance with the present invention; and b) instructions for using the compounds or compositions for a medical application (e.g. tissue imaging, treating a nitrous oxide related disease, or for metal detoxification).
- a medical application e.g. tissue imaging, treating a nitrous oxide related disease, or for metal detoxification.
- the compound is in a container (e.g., a vial or bottle).
- the instructions are written (e.g., on paper).
- the present invention provides a crowned DTC chelator having a formula (I):
- R 1 and R 2 are the same or different, and are selected from a macrocyclic crown ether- containing group, or R 1 and R 2 may be taken together to form a macrocycle of formula [(CH 2 ) a -O] -(CH 2 ) c , wherein a is 2-5, b is 3-8, and c is 2-5.
- a is 2 or 3
- b is 3-6
- c is 2 or 3 in crowned DTC chelator (I).
- a is 2 and c is 2 in crowned DTC chelator (I).
- crowned DTC chelator (I) comprises
- crowned DTC chelator (I) comprises
- crowned DTC chelator (I) comprises
- crowned DTC chelator (I) comprises
- crowned DTC chelator (I) comprises
- crowned DTC chelator (I) comprises
- methods for preparing DTC chelators in accordance with the first through ninth series of embodiments described above include reacting an amino crown ether with carbon disulfide in the presence of a base.
- the present invention provides a radiopharmaceutical of formula (M ⁇ N)L 1 L 2 and pharmaceutically acceptable salt thereof, wherein M is a radionuclide selected from the group consisting of 99m Tc, 186 Re, and 188 Re.
- L 1 and L 2 are the same or different and comprise a formula (II):
- R 1 and R 2 are the same or different, and are selected from a macrocyclic crown ether-containing group, or R 1 and R 2 may be taken together to form a macrocycle of the formula [(CH 2 ) a -O] b -(CH 2 ) c , wherein a is 2-5, b is 3-8, and c is 2-5.
- M is 99m Tc
- a is 2 or 3
- b is 3-6
- c is 2 or 3 in radiopharmaceutical (II).
- a is 2 and c is 2 in radiopharmaceutical (II).
- radiopharmaceutical (II) comprises: [0091] In a fifteenth series of embodiments, radiopharmaceutical (II) comprises:
- radiopharmaceutical (II) comprises:
- radiopharmaceutical (II) comprises:
- radiopharmaceutical (II) comprises:
- radiopharmaceutical (II) comprises:
- novel radiopharmaceutical compositions contain a metal chelate according to one of the eleventh through nineteenth series of embodiments described above.
- methods for preparing radiopharmaceuticals according to the eleventh through nineteenth series of embodiments described above include reacting pertechnetate with (1) a nitrido donor; (2) a reducing agent; (3) and a crowned DTC chelator according to the first through nineteenth series of embodiments described above.
- the nitrido donor is succinyl dihydride
- the reducing agent is stannous chloride.
- kits for the preparation of a radiopharmaceutical include a nitrido donor (e.g., contained in a first container), stannous chloride and a chelating agent able to stabilize the tin cation (e.g., contained in a second container), and a crowned DTC chelator according to the first through ninth series of embodiments described above (e.g., contained in a third container).
- a nitrido donor e.g., contained in a first container
- stannous chloride and a chelating agent able to stabilize the tin cation e.g., contained in a second container
- a crowned DTC chelator according to the first through ninth series of embodiments described above (e.g., contained in a third container).
- kits for the preparation of a radiopharmaceutical include a first container containing succinyl dihydride, stannous chloride and a chelating agent able to stabilize the tin cation, and a second container containing a crowned DTC chelator according to the first through ninth series of embodiments described above.
- kits for the preparation of a radiopharmaceutical include a first container containing succinyl dihydride, stannous chloride and 1,2-diaminopropane-N,N,N',N'-tetraacetic acid or a salt thereof, and a second container containing a crowned DTC chelator according to the fourth through ninth series of embodiments described above.
- methods for radioimaging a mammal embodying features of the present invention include (i) administering to the mammal an effective amount of a radiopharmaceutical according to the eleventh through twenties series of embodiments described above, and (ii) scanning the mammal using a radioimaging device.
- methods for visualizing sites of myocardial disease in a mammal embodying features of the present invention include (i) administering to the mammal an effective amount of a radiopharmaceutical according to the eleventh through twentieth series of embodiments described above, and (ii) scanning the mammal using a radioimaging device.
- methods of diagnosing a myocardial disease in a mammal embodying features of the present invention include (i) administering to the mammal a radiopharmaceutical according to the eleventh through twentieth series of embodiments described above, and (ii) imaging the mammal.
- the present invention provides a novel radiopharmaceutical of formula (III)
- R 1 and R 2 are the same or different, and are selected from a macrocyclic crown ether-containing group, or R 1 and R 2 may be taken together to form a macrocycle of formula [(CH 2 ) a -O] b -(CH 2 ) c , wherein a is 2-5, b is 3-8, and c is 2-5.
- L 3 , L 4 and L 5 are the same or different, and are selected from an isonitrile of formula: [00107] wherein q is 0-3 and Z is carbon or silicon.
- R 3 , R 4 and R 5 are the same or different, and are selected from the group consisting of H, C1-C10 alkyl substituted with 0 - 5 R 6 , aryl substituted with 0-5 R 6 , heteroaryl substituted with 0-5 R 6 , and macrocyclic crown ether containing 2-8 ether- oxygen atoms.
- R 7 , R 8 and R 9 are the same or different, and are selected from the group ' consisting of H, alkyl, aryl, and heteroaryl, or R 8 and R 9 may be taken together to form a macrocyclic crown ether containing 2-8 ether-oxygen atoms.
- L 3 , L 4 and L 5 may be taken together to form a tripodal chelator of formula:
- a 1 , A 2 and A 3 are the same or different, and are imine-N containing heterocycles.
- a 4 , A 5 and A 6 are the same or different, and are selected from the group consisting of NR 14 , PR 14 , S, and O.
- R 10 , R 11 and R 12 are the same or different, and are selected from a group of formula: -(CH 2 ) g -, [00109] wherein g is 2-5.
- R 13 is selected from the group consisting of H, alkyl and aryl group.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- M is 99 Tc
- a is 2 or 3
- b is 3- 6
- c is 2 or 3
- q is 1
- Z is carbon in radiopharmaceutical (III).
- R 3 , R 4 and R 5 are the same or different, and are selected from the group consisting of H, C 1 -C 5 alkyl substituted with a R 6 , aryl substituted with a R 6 , heteroaryl substituted with a R 6 , and macrocyclic crown ether containing 3-6 ether- oxygen atoms.
- R 7 , R 8 and R 9 are the same or different, and are selected from the group consisting of H, alkyl, aryl, and heteroaryl, or R 8 and R 9 may be taken together to form a macrocyclic crown ether containing 3- 6 ether-oxygen atoms.
- a 1 , A 2 and A 3 are selected from the group consisting of imidazolyl, pyrazolyl, oxazolinyl, methimazolyl, and pyridyl.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- the subscript g is 2 or 3.
- R 13 is selected from the group consisting of H, methyl, and phenyl.
- R 14 is selected from the group consisting of ethyloxyethyl, ethoxylpropyl, methyoxyethyl, and methoxypropyl.
- a is 2 and c is 2 in radiopharmaceutical (III).
- R 3 , R 4 and R 5 are the same or different and are selected from the group consisting of H, C 1 -C 5 alkyl, phenyl, and macrocyclic crown ether containing 3-6 ether-oxygen atoms.
- R 7 , R 8 and R 9 can be the same or different, and are selected from the group consisting of H, alkyl, and phenyl, or R 8 and R 9 may be taken together to form a macrocyclic crown ether containing 3-6 ether-oxygen atoms.
- L 3 , L 4 and L 5 are taken together to form a tripodal chelator having a formula selected from the group consisting of:
- L 3 , L 4 and L 5 are the same in radiopharmaceutical (III) and are selected from any one of following macrocyclic crown ether-containing isonitriles:
- the dithiocarbamate chelator in radiopharmaceutical (III) is selected from any one of the following crowned DTCs:
- novel radiopharmaceutical compositions embodying features of the present invention include a metal chelate according to the twenty-ninth through thirty- third series of embodiments described above.
- methods for the preparation of a radiopharmaceutical according to the twenty-ninth through thirty-third series of embodiments described above include reacting pertechnetate with (1) a nitrido donor; (2) a reducing agent; (3) an organic isonitrile ligand or tripodal chelator according to the twenty-ninth through thirty-third series of embodiments described above, and a crowned DTC chelator according to the first through ninth series of embodiments described above.
- the nitrido donor is succinyl dihydride
- the reducing agent is stannous chloride
- kits for the preparation of a radiopharmaceutical according to twenty-ninth through thirty-third series of embodiments described above includes: a first container containing a nitrido donor, a stannous chloride and a chelating agent able to stabilize the tin cation; a second container containing an organic isonitrile ligand or a tripodal chelator according to the twenty-ninth through thirty-third series of embodiments described above; and a third container containing a crowned DTC chelator according to the first through ninth series of embodiments described above.
- kits for the preparation of a radiopharmaceutical include: a first container containing a succinyl dihydride, a stannous chloride and 1 ,2-diaminopropane-N,N,N',N'-tetraacetic acid or a salt thereof, and a second container containing an organic isonitrile ligand or a tripodal chelator according to the twenty-ninth through thirty-third series of embodiments described above, and a crowned DTC chelator according to the first through ninth series of embodiments described above.
- kits for the preparation of a radiopharmaceutical product according to the thirty-sixth series of embodiments described above includes: a first container containing a succinyl dihydride, a stannous chloride and 1 ,2-diaminopropane-N,N,N',N'-tetraacetic acid or a salt thereof, and a second container containing an organic isonitrile ligand according to the twenty-ninth through thirty-third series of embodiments described above, and a crowned DTC chelator according to fourth through ninth series of embodiments described above.
- methods for radioimaging a mammal include (i) administering to the mammal an effective amount of a radiopharmaceutical according to the twenty-ninth through thirty-third series of embodiments described above, and (ii) scanning the mammal using a radioimaging device.
- methods for visualizing sites of myocardial disease in a mammal by radioimaging include (i) administering to the mammal an effective amount of a radiopharmaceutical according to the twenty-ninth through thirty-third series of embodiments described above, and (ii) scanning the mammal using a radioimaging device.
- methods of diagnosing a myocardial disease in a mammal include (i) administering to the mammal a radiopharmaceutical composition according to embodiments according to the twenty-ninth through thirty-third series of embodiments described above, and (ii) imaging the mammal.
- the present invention provides a pharmaceutical of formula (IV):
- M is a transition metal selected from the group consisting of Fe(ll), Fe(III), Mn(ll), Mn(lll), Co(ll), Co(lll), Ni(ll), Cu(ll), Zn(ll), Ru(ll), Ru(lll), Pd(ll), and Pt(ll).
- the subscripts p and p' are integers and are independently selected from 0-2.
- R 1 and R 2 are the same or different, and are selected from a macrocyclic crown ether-containing group, or R 1 and R 2 may be taken together to form a macrocycle of formula [(CH 2 ) a -O] -(CH 2 ) c , wherein a is 2-5, b is 3-8, and c is 2-5.
- L 6 is a tripodal chelator to complete the coordination sphere of the transition metal, and is selected from a compound of formula:
- a 1 , A 2 and A 3 are the same or different, and are imine-N containing heterocycles.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- R 10 , R 11 and R 12 are the same or different, and are selected from a group of formula: -(CH 2 ) g " > [00126] wherein g is 2-5.
- R 13 is selected from the group consisting of H, alkyl, and aryl.
- R 14 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- M is Ru(lll), p is 1, and p' is 2.
- R 1 and R 2 are the same or different, and are selected from a macrocyclic crown ether-containing group, or R and R 2 may be taken together to form a macrocycle of formula [(CH )a-O] b -(CH 2 ) c , wherein a is 2 or 3, b is 3-6, and c is 2 or 3.
- a 1 , A 2 and A 3 are selected from the group consisting of imidazolyl, pyrazolyl, oxazolinyl, methimazolyl, and pyridyl.
- a 4 , A 5 and A 6 are selected from the group consisting of NR 10 , PR 10 , and S.
- the subscript g is 2 or 3.
- R 13 is selected from the group consisting of H, methyl, and phenyl.
- R 14 is selected from the group consisting of ethyloxyethyl, ethoxylpropyl, methyoxyethyl, and methoxypropyl.
- R 4 is selected from the group consisting of H, alkyl, aryl, and alkoxyalkyl.
- a is 2
- c is 2
- L 6 is selected from any one of the following tripodal chelators:
- the dithiocarbamate chelator in pharmaceutical (IV) is selected from any one of the following crowned DTCs:
- Crowned dithiocarbamate metal complexes embodying features of the present invention may be used as pharmaceutical agents to diagnose or treat disease.
- Radiopharmaceutical Compositions are neutral and cationic radioactive metal-nitrido complexes of crowned dithiocarbamates and are used as radiopharmaceuticals.
- Radiopharmaceuticals are drugs containing a radionuclide, and are used routinely in nuclear medicine for the diagnosis or therapeutic treatment of various diseases. They are mostly small organic or inorganic compounds with definite compositions. Radiopharmaceuticals form the chemical basis for nuclear medicine, a group of techniques used for diagnosis and therapeutic treatment of various diseases.
- the in vivo diagnostic information is obtained by intravenously injecting the radiopharmaceutical and determining its biodistribution using a gamma camera.
- the biodistribution of the radiopharmaceutical depends on the physical and chemical properties of the radiopharmaceutical and may be used to obtain information about the presence, progression, and state of disease.
- the radioactive metal-nitrido complexes of crowned dithiocarbamates embodying features of the present invention may be used as radiopharmaceuticals.
- the radionuclide for a diagnostic radiopharmaceutical is often a gamma-emitting isotope for scintigraphic imaging or a positron-emitting isotope for positron emission tomography (PET).
- PET positron emission tomography
- the choice of radionuclide depends largely on the physical and nuclear properties (e.g., half-life and y- energy), availability, and cost. Nearly 80% of all radiopharmaceuticals used in nuclear medicine are 99m Tc-labeled compounds. The 6 hour half-life is long enough to allow a radiochemist to carry out radiopharmaceutical synthesis and for nuclear medicine practitioners to collect useful images.
- the metallic radionuclide is selected from the group consisting of 99 Tc, 86 Re and 188 Re.
- 99m Tc is a preferred isotope. Its 6 hour half-life and 140 keV gamma ray emission energy are almost ideal for gamma scintigraphy using equipment and procedures well established in the art.
- the rhenium isotopes also have gamma ray emission energies that are compatible with gamma scintigraphy; however, they also emit high energy beta particles that are more damaging to living tissues. These beta particle emissions can be utilized for therapeutic purposes, for example, cancer radiotherapy.
- the related chemistry, medical applications, and radiolabeling with 86/ 88 Re by direct and indirect methods have been reviewed (Fritzberg et al. Pharmaceutical Res. 1988, 5, 325; Liu et al. Bioconjugate Chem. 1997, 8, 621; Dilworth, J. R. and Parrott, S. J. Chem. Soc. Rev. 1998, 27, 43).
- 99 Tc is produced from a parent radionuclide, 99 Mo, a fission product with a half-life of 2.78 days.
- 99 Mo- 99 Tc generator [ 99 Mo]molybdate is absorbed to an alumina column and 99m Tc is formed by decay of 99 Mo.
- the 99m Tc in the form of [ 99m Tc]pertechnetate is eluted from the column with saline.
- detection in accordance with the present invention is performed by PET imaging (e.g., using a SPECT camera or similar type camera).
- PET Compared to other imaging modalities, PET has three important technological features, which enable clinicians to measure biochemical or physiological process in vivo.
- the first feature is its ability to accurately measure the actual 3-D radiotracer distribution, which makes PET similar to autoradiography.
- the second feature is its ability to rapidly acquire a dynamic set of tomographic images through a volume of tissue. This is unique for PET imaging because no other imaging modality except MRI has such ability.
- the third feature is the ability to acquire whole body images. It is the combination of these three features with the high specificity of receptor binding of biomolecules that makes PET imaging using radiolabeled biomolecules extremely attractive for nuclear medicine.
- 94m Tc is a cyclotron-produced isotope with a half-life of 52 min (0.9 h) and a ⁇ + energy of 2.47 MeV (72%). It can be obtained from a number of production methods, including 94 Mo(p, n)/ 94m Tc (13.5 - 11 MeV), nat Nb( 3 He,
- Rhenium shares similar coordination chemistry with technetium due to their periodic relationship. Rhenium has two isotopes ( 186 Re and 188 Re) that are useful for radiotherapy.
- 186 Re is a reactor-produced radionuclide, and is obtained by the irradiation of 185 Re with neutrons ( 185 Re(n, ⁇ ) 186 Re).
- the yield of 186 Re depends on the amount of Re target, the energy of the neutrons available, and the neutron reflux. The specific activity is low or medium, but a carrier-free product is not possible.
- the radioactive metal-nitrido complexes of crowned dithiocarbamates in accordance with the present invention employ 186 Re and 188 Re as the radioactive metal.
- 188 Re can be prepared either from the nuclear reaction ( 187 Re(n, ⁇ ) 188 Re) or from the 188 W- 186 Re generator.
- the generator-produced 188 Re is carrier-free and has very high specific activity.
- the major advantage of using 188 Re in therapeutic nuclear medicine is the inexpensive and readily available 188 W- 186 Re generator, which has a very long and useful shelf-life.
- Tc cores for the routine synthesis of 99m Tc radiopharmaceuticals.
- the nitrido ligand is a powerful ⁇ -electron donor and shows a high capacity to stabilize the Tc(V) oxidation state.
- the [Tc ⁇ N] 2+ core forms Tc(V) nitrido complexes with a variety of chelators.
- Various chelators have been used for the preparation of 99m Tc radiopharmaceuticals.
- 99m Tc-labeling techniques have been extensively reviewed (See, e.g., Horn, R. K. and Katzenellenbogen, J. A. Nucl. Med. Biol. 1997, 24, 485; Dewanjee, M. K. Semin. Nucl. Med. 1990, 20, 5; and Jurisson et al Chem. Rev. 1993, 93, 1137).
- International PCT application no. WO 90/06137 describes a series of technetium-nitrido chelates of dithiocarbamates, including dimethyldithiocarbamate, di-n-propyl dithiocarbamate, N-ethyl-N-(2- ethyoxyethyl)dithiocarbamate.
- International PCT application nos. WO 89/08657, WO 92/00982, and WO 93/01839 describe processes for producing technetium nitrido complexes, which include reacting a polyphosphine as a reducing agent for the technetium oxide, then reacting with a nitride salt of a metal or ammonium ion. Since Tc-nitrido core has four to five coordination sites for various ligands or chelators, the choice of chelator is important for the solution stability and the number of radioactive species formed during ligand exchange reaction.
- Substituents on the phosphine P, secondary amine N, and dithiocarbamate N atoms are selected from alkyl, aryl, alkoxy, or alkoxyalkyl groups. This reference does not teach or suggest the use of crowned DTCs for the preparation of 99m Tc-nitrido complexes.
- Ischemia-related diseases particularly coronary artery disease (CAD)
- CAD coronary artery disease
- Myocardial ischemia is a serious condition and the delay in reperfusion of the ischemic tissues can be life threatening. This is particularly true in the aged population. Rapid and accurate early detection of myocardial ischemia is highly desirable so that various therapeutic regiments can be given before irreversible myocardial damage occurs.
- the compositions of the present invention are preferably used for myocardial perfusion imaging.
- Myocardial perfusion imaging with radiotracers is an integral component of the clinical evaluation of patients with known or suspected coronary artery disease (CAD) in current clinical practice (See, e.g., Acmpa, W. et al.
- 201 TI images should be taken soon after injection mainly due to the dynamic nature of its distribution and redistribution dynamics, and may not be suitable for situations where immediate imaging may not be possible (for example, patients with acute myocardial infarction).
- 99m Tc yields relatively high-energy photons and can be used at much higher doses.
- the use of 99m Tc also allows the simultaneous assessment of myocardial perfusion and cardiac function in a single study (Kapur, A. et al. Eur. J. Nucl. Med. 2002, 29, 1608). Because of its ideal nuclear properties (short half-life and ⁇ -energy) and its diverse coordination chemistry, 99m Tc has been the isotope of choice for the development of myocardial perfusion imaging agents.
- 99m TcN-Noet is a member of the neutral 99m Tc-nitrido complexes, which are characterized by the presence of the 99 Tc ⁇ N triple bond and two N-alkyl dithiocarbamate ligands.
- Duatti and coworkers first reported the synthesis of 99 Tc-nitrido complexes with various chelators and their use as heart imaging agents (Marchi, A. et al. J. Chem. Soc. Dalton Trans. 1990, 1743; Duatti, A. et al. J. Chem. Soc, Dalton Trans. 1990, 3729; Marchi, A et al. Inorg. Chem. 1990, 29, 2091).
- One aspect of the present invention relates to neutral 99m Tc-nitrido complexes as new radiopharmaceuticals for myocardial imaging.
- the " m Tc- nitrido complexes described herein are expected to have a first-pass extraction comparable to or better than that of 99m Tc-N-Noet due to their structural similarity.
- the presence of crown ether groups in compounds embodying features of the present invention should allow a faster clearance of 99m Tc-nitrido complexes from the liver and lungs, and better heart/liver and heart/lung ratios.
- Another aspect of the present invention relates to cationic """Tc- nitrido complexes containing two different chelators, one of which is a crowned DTC, and to their use as new radiopharmaceuticals for imaging (e.g., myocardial perfusion imaging).
- These cationic " m Tc-nitrido complexes are expected to have a higher heart uptake and longer myocardial retention than " m Tc-N-Noet due to the cationic character and possible interactions between the crown ether moiety and intracellular K + .
- crown ether groups also results in a faster renal clearance with less hepatobiliary uptake and gastrointestinal retention than 99m Tc-N-Noet, " m Tc-Sestamibi and 99m Tc- Tetrofosmin due to increased hydrophilicity.
- the heart liver ratios were ⁇ 10 times higher than those of " m Tc-Sestamibi and 99 Tc-Tetrofosmin in the same animal model.
- the heart uptake of lipophilic cations is not just limited to " m Tc complexes. It has been reported that cationic 64 Cu complexes show high heart uptake (Packard, A. B. Nucl. Med. Biol. 1998, 25, 531; Packard, A. B. Nucl. Med. Biol. 2002, 29, 289). Lipophilic 68 Ga complex cations have also been found to show a high heart uptake and are useful for evaluation of myocardial perfusion using PET (Tsang, B. W. et al. J.
- the radioactive metal-nitrido complexes of crowned dithiocarbamates embodying features of the present invention may also be used as radiopharmaceuticals for non-invasive imaging of any type of tissue, including but not limited to tumor MDR1 (multidrug resistance) p-glycoprotein (Pgp) transport function (Sharma, V and Piwnica-Worms, D. Chem. Rev. 1999, 99: 2545; and Herman, L. et al. J. Med. Chem. 1995, 38, 2955).
- Various cationic 99m Tc complex radiopharmaceuticals, originally developed for myocardial perfusion imaging, have been shown to be substrates for transport by MDR1
- a crown ether containing dithiodicarbamate and its Co(ll), Ni(ll), Cu(ll) and Zn(ll) complexes have been synthesized and characterized (Wang, J. H. and Wang, Y. L. Yingyong Huaxue 2002, 19, 295-297; Wang, J.-H. and Zhang, Z. Yingyong Huaxue 1994, 11, 101). It was found that all metal complexes were stable, and that the dithiocarbamate group is bidentate. The crystal structure of cobalt tris[(aza-15-crown-5)dithiocarbamate has also been reported (Granell, G. et al J. Chem. Soc, Dalton Trans. 1990, 605); but no specific applications were described.
- Ru(lll) metal complex pharmaceuticals embodying features of the present invention may be beneficial for the treatment of diseases such as septic shock, rheumatoid arthritis, diabetes, asthma, and cancer.
- DTCs Dithiocarbamates
- DTCs are known heavy metal chelators (Sunderman, F. W. Ann. Clin. Lab. Sci. 1978, 8, 259; Jones, M. M. and Cherian, M. G. Toxicology 1990, 62, 1).
- DTCs such as diethyl- dithiocarbamate have been clinically used for the treatment of nickel poisoning, and were used in clinical trials for the treatment of AIDS patients (Reisinger, E. et al. Lancet 1990, 335, 679).
- DTCs such as pyrrolidine dithiocarbamate are potent inhibitors of nuclear factor kappa B in intact cells (Schreck, R. et al. J. Exp. Med. 1992, 175, 1181).
- nuclear factor kappa B has been shown to up-regulate the expression of cell adhesive molecules, including the vascular cell adhesive molecule 1 (VCAM-1) (Lademarco et al. J. Biol. Chem. 1992, 267, 16323).
- VCAM-1 vascular cell adhesive molecule 1
- Endothelial expression of VCAM-1 causes the adherence of neutrophils to the endothelium, an early event leading to inflammation and subsequent vascular damage and reduction of blood flow (Oppenheimer, M. N. et al. J. Immunol. 1991, 147, 42207). Therefore, DTCs and their metal complexes would block VCAM-1 expression, thereby avoiding the vascular problems associated with neutrophil adherence to the endothelium.
- DTC chelators act either as a direct scavenger of hydroxy radicals (due to their thiol group) or as an iron chelator that inhibits hydroxyl radical production by binding to the iron ions or by both mechanisms (Liu et al. Free Rad. Res. 1996, 24, 461).
- compositions of e.g., neutral or cationic metal chelates
- nitric oxide (NO) scavengers e.g., neutral or cationic metal chelates
- the presence of crown ether groups may be used to increase water solubility.
- Metal chelate-based NO scavengers may be useful as therapeutic pharmaceuticals for the treatment of diseases including but not limited to septic shock, rheumatoid arthritis, diabetes, asthma, and cancer.
- nitric oxide has been widely studied because of its essential role in many physiological processes. The overproduction of NO has been implicated to play a significant role in many disease states, such as septic shock (Evans, T. et al.
- compositions embodying features of the present invention may be used, based on their NO attenuation or scavenging properties, as compounds for the treatment of diseases such as septic shock, rheumatoid arthritis, diabetes, asthma, and cancer.
- Nitric oxide is an excellent ligand, especially with iron and ruthenium.
- Ruthenium(lll) complexes are of particular interest as NO scavengers, and have been investigated as immunosuppressive agents (Bastos, C. M. et al. Bioorg. Med. Chem. Lett. 1998, 8, 147), antitumor and anti-metastatic agents (Sava, G. et al. Top. Biol. Inorg. Chem. 1999, 1, 143; Sava, G. et al. Chem.-Biol. Interact. 1995, 95, 109; Sava, G. et al. Top. Biol. Mel-Based Drugs 1995, 2, 221), and nitric oxide (NO) scavengers (Cameron, B. R. Inorg. Chem.
- Another aspect of the present invention provides the use of compounds embodying features of the present 3+ invention as sequestering agents for the treatment of heavy metal (e.g., Fe 2+ and Pb ) intoxication.
- heavy metal e.g., Fe 2+ and Pb
- Dithiocarbamates are sulfur-containing small molecules with extremely useful redox capabilities.
- DTCs have been used as heavy metal chelators.
- Synthesis of crowned DTC chelators was achieved by reacting the amino crown ether with carbon disulfide in the presence of a base, such as sodium hydroxide (Wang, J. H. and Wang, Y. L. Yingyong Huaxue 2002, 19, 295-297; and Wang, J.-H. and Zhang, Z. Yingyong Huaxue 1994, 11, 101). Since these crowned DTCs are slightly air-sensitive, they should be dried and stored under nitrogen. The size and the number of ether-oxygen donors can be systematically varied using synthetic techniques well known in the art.
- DTC chelators form metal complexes with a variety of transition metal ions, including Fe 2+ , Fe 3+ , Co 2+ , Co 3+ , Cu 2+ , Mn 2+ , Zn 2+ , and Ru 3+ . It has been well-documented that DTC chelators form neutral " m Tc-nitrido complexes. Synthesis of neutral " m Tc-nitrido complexes of crowned DTCs may be achieved by following the literature methods (e.g., Marchi, A. et al. J. Chem. Soc. Dalton Trans. 1990, 1743; Duatti, A. et al. J. Chem. Soc, Dalton Trans.
- Macrocyclic crown ether containing groups have been the subject of intensive research for their ability to bind metal ions such as K + and Na + (Valeur, B. and Leray, I. Coord. Chem. Rev. 2000, 205, 3; Gunnlaugsson, T. and Leonard, J. P. J. Chem. Soc, Perkin Trans. 2002, 2, 1980).
- the extracellular Na + concentration is 133 - 145 mM as compared to 3.5-4.8 mM for K + .
- the cytosolic Na + concentration is only 10-40 mM as compared to 120 mM (upper limit) for K + (Gunnlaugsson, T. and Leonard, J. P. J. Chem.
- K + binding capability may serve as a driving force for accumulation and retention of " Tc complexes in myocardium.
- the selectivity for K + may be tuned by changing the size and number of oxygen donor atoms of the macrocycle.
- the tripodal chelators of particular interest include but are not limited to the following examples:
- a bidentate dithiocarbamate chelator may be used to replace the two chlorides and form cationic 99m Tc-nitrido complexes [ 99m TcN(lsonitrile) 3 (L)] + .
- the advantage of this new chelating system (three isonitriles and a dithiocarbamate) is the versatility of both isonitrile and dithiocarbamate ligands, which may be used for modification of physical and biological properties of their cationic 99 Tc- nitrido complexes [ 99m TcN(lsonitrile) 3 (L)] + .
- the technetium and rhenium radionuclides are preferably in the chemical forms of [ 99 Tc]pertechnetate or [ 186 188 Re]perrhenate and a pharmaceutically acceptable cation.
- the [ 99 Tc]pertechnetate salt form is preferably sodium [ 99m Tc]pertechnetate, such as may be obtained from commercial " m Tc generators.
- the amount of [ 99m Tc]pertechnetate used to prepare metal complexes embodying features of the present invention may range from 1 mCi to 1000 mCi, or more preferably from 1 mCi to 50 mCi.
- the [ 99m Tc]pertechnetate Since there is no effective chemistry that can be used to attach the [ 99m Tc]pertechnetate anion to an organic chelator, the [ 99m Tc]pertechnetate is reduced by a reducing agent to a lower oxidation state in order to produce a stable 99m Tc complex or to a reactive intermediate complex from which " m Tc can be easily transferred to the new chelator to form the expected 99m Tc complex.
- Rhenium chemistry is very similar to technetium chemistry due to their periodic relationship. Therefore, the methods used for molecules labeled with " m Tc should apply to those labeled with 186 188 R ⁇ .
- Suitable reducing agents for the synthesis of radiopharmaceuticals in accordance with the present invention include stannous salts, dithionite or bisulfite salts, borohydride salts, and formamidinesulfinic acid, wherein the salts are of any pharmaceutically acceptable form.
- a presently preferred reducing agent is a stannous salt.
- the amount of a reducing agent used may range from 0.001 mg to 10 mg, or more preferably from 0.005 mg to 1 mg.
- the total time of preparation will vary depending on the metallic radionuclide, the identities and amounts of the reactants, and the procedure used for the preparation.
- the preparations may be complete and result in greater than 80% yield of metal complex in 1 minute or may require more time.
- the resulting reaction mixture may optionally be purified using one or more chromatographic methods, such as Sep-Pack or high performance liquid chromatography (HPLC).
- HPLC high performance liquid chromatography
- the preferred methods are those in which the " m Tc complex is prepared in high yield and high radiochemical purity without post-labeling purification.
- the amounts of DTC chelator used for the preparation of radiometal chelates may range, for example, from 1 mg to 1000 mg, or more preferably from 1 mg to 10 mg.
- the exact amount of the DTC chelator is a function of the identity of a specific metal chelate, the procedure used for preparation of the metal chelate, and the amount and identities of the reactants used for the radiolabeling.
- the compounds described herein may have one or more asymmetric centers.
- Compounds embodying features of the present invention containing an asymmetrically substituted atom may be isolated in optically active or racemic forms. It is well known in the art how to prepare optically active forms, such as by the resolution of racemic forms or by synthesis from optically active starting materials. All chiral, diastereomeric, racemic forms and all geometric isomeric forms of a structure are contemplated for use, unless the specific stereochemistry or isomeric form is specifically indicated. All processes used to prepare compounds of the present invention and intermediates made therein are considered to be part of the present invention.
- Kits and therapeutic compositions embodying features of the present invention, as well as various routes of therapeutically administering compositions embodying features of the present invention, will now be described.
- the present invention provides diagnostic kits for the preparation of radiopharmaceuticals useful as imaging agents for the diagnosis of cardiovascular disorders, infectious diseases, inflammatory diseases, and cancer.
- Diagnostic kits in accordance with the present invention may include one or more vials containing the sterile, non-pyrogenic, formulation comprised of a predetermined amount of a chelator in accordance with the present invention, a stabilizing coligand if needed, a reducing agent, and optionally other components, such as buffers, lyophilization aids, stabilization aids, solubilization aids and bacteriostats.
- a radiopharmaceutical composition may contain, for example, the metal complex radiopharmaceutical, a buffer, a stabilization aid to prevent autoradiolysis, and a bacteriostat. If a radiopharmaceutical is prepared using the kit formulation, the radiopharmaceutical composition may contain the metal complex radiopharmaceutical and kit components, including unlabeled chelator, excess stabilizing coligand, a reducing agent, buffer, lyophilization aid, stabilization aid, solubilizing aids, and bacteriostats.
- Buffers useful in the preparation of radiopharmaceuticals and in diagnostic kits useful for the preparation of the radiopharmaceuticals include but are not limited to phosphate, citrate, sulfosalicylate, and acetate. A more complete list may be found in the United States Pharmacopeia. Lyophilization aids useful in the preparation of diagnostic kits useful for the preparation of radiopharmaceuticals include but are not limited to mannitol, lactose, sorbitol, dextran, Ficoll, and polyvinylpyrrolidine (PVP).
- PVP polyvinylpyrrolidine
- Stabilization aids useful in the preparation of radiopharmaceuticals and in diagnostic kits useful for the preparation of the radiopharmaceuticals include but are not limited to ascorbic acid, cysteine, monothioglycerol, sodium bisulfite, sodium metabisulfite, gentisic acid, ascorbic acid, and inositol.
- Solubilization aids useful in the preparation of radiopharmaceuticals and in diagnostic kits useful for the preparation of the radiopharmaceuticals include but are not limited to ethanol, glycerin, polyethylene glycol, propylene glycol, polyoxyethylene sorbitan monooleate, sorbitan monoloeate, polysorbates, poly(oxyethylene)poly(oxypropylene), poly(oxyethylene) block copolymers (Pluronics) and lecithin.
- Presently preferred solubilizing aids are polyethylene glycol and Pluronics.
- Bacteriostats useful in the preparation of radiopharmaceuticals and in diagnostic kits useful for the preparation of the radiopharmaceuticals include but are not limited to benzyl alcohol, benzalkonium chloride, chlorbutanol, and methyl, propyl or butyl paraben.
- a component in a diagnostic kit may also serve more than one function.
- a reducing agent may also serve as a stabilization aid
- a buffer may also serve as a transfer ligand
- a lyophilization aid may also serve as a transfer, ancillary or coligand, and so forth.
- the predetermined amounts of each component in the formulation are determined by a variety of considerations that are in some cases specific for that component and in other cases dependent on the amount of another component or the presence and amount of an optional component. In general, the minimal amount of each component is used that will give the desired effect of the formulation.
- the desired effect of the formulation is that the practicing end user may synthesize the radiopharmaceutical and have a high degree of certainty that the radiopharmaceutical may be safely injected into a patient and will provide diagnostic information about the disease state of that patient.
- kits and “reagent kit” refer to an assembly of materials that are used in performing a method embodying features of the present invention.
- the reagents may be provided in packaged combination in the same or in separate containers, depending on their cross- reactivities and stabilities, and in liquid or in lyophilized form.
- the amounts and proportions of reagents provided in the kit may be selected so as to provide optimum results for a particular application.
- Reagents included in kits embodying features of the present invention may be supplied in all manner of containers such that the activities of the different components are substantially preserved, while the components themselves are not substantially adsorbed or altered by the materials of the container.
- Suitable containers include but are not limited to ampoules, bottles, test tubes, vials, flasks, syringes, bags and envelopes (e.g., foil-lined), and the like.
- the containers may be comprised of any suitable material including but not limited to glass, organic polymers (e.g., polycarbonate, polystyrene, polyethylene, etc.), ceramic, metal (e.g., aluminum), metal alloys (e.g., steel), cork, and the like.
- the containers may contain one or more sterile access ports (e.g., for access via a needle), such as may be provided by a septum.
- Preferred materials for septa include rubber and polymers including but not limited to, for example, polytetrafluoroethylene of the type sold under the trade name TEFLON by DuPont (Wilmington, DE).
- the containers may contain two or more compartments separated by partitions or membranes that can be removed to allow mixing of the components. '
- Kits embodying features of the present invention may also be supplied with other items known in the art and/or which may be desirable from a commercial and user standpoint, such as instructions for treating a tissue, a container for the tissue, diluents, preservation agents, antibiotics, antifungal drugs, antiviral drugs, anti-inflammatory drugs, surfactants, buffers, empty syringes, tubing, gauze, pads, disinfectant solution, etc.
- the diagnostic kits of the present invention may also contain instructions for the practicing end user to follow to synthesize radiopharmaceuticals embodying features of the present invention.
- the instructions may be affixed to one or more of the containers (e.g., vials) or to a larger container in which one or more smaller containers are packaged for shipping.
- the instructions may also be provided as a separate insert, termed the package insert.
- the instructional materials may be printed (e.g., on paper) and/or supplied in an electronic-readable medium (e.g., floppy disc, CD-ROM, DVD- ROM, zip disc, videotape, audio tape, etc.).
- instructions may be provided by directing a user to an Internet web site (e.g., specified by the manufacturer or distributor of the kit) and/or via electronic mail.
- compositions embodying features of the present invention comprise pharmaceutical carriers including but not limited to any sterile biocompatible pharmaceutical carrier such as saline, buffered saline, dextrose, water, and the like. Accordingly, in some embodiments, methods embodying features of the present invention comprise administering to a subject a pharmaceutical composition embodying features of the present invention in a suitable pharmaceutical carrier. In some embodiments, particular pharmaceutical compositions or therapies comprise a mixture of two or more different species of pharmaceutical compositions. [00191] In some embodiments, the pharmaceutical compositions comprise a plurality of compositions administered to a subject under one or more of the following conditions: at different periodicities, different durations, different concentrations, or by different administration routes or the like.
- the pharmaceutical compositions and methods embodying features of the present invention may find use in treating diseases or altered physiological states characterized by pathogenic infection.
- the present invention is not limited to ameliorating (e.g., treating) any particular disease or infection.
- various embodiments of the present invention are provided for treating (including prophylaxis) a range of physiological symptoms and disease etiologies in subjects including but limited to those characterized by aberrant cellular growth or proliferation (e.g., cancer), autoimmunity (e.g., rheumatoid arthritis), and other aberrant biochemical, genetic, and physiological symptoms.
- the pharmaceutical compositions may be formulated and administered systemically or locally.
- compositions are administered to a subject (e.g., a patient) alone or in combination with one or more other drugs or therapies (e.g., antibiotics and antiviral agents, etc.) or in compositions where they are mixed with excipients or other pharmaceutically acceptable carriers.
- a subject e.g., a patient
- drugs or therapies e.g., antibiotics and antiviral agents, etc.
- compositions in accordance with the present invention may be delivered via any suitable method, including but not limited to orally, intravenously, subcutaneously, intratumorally, intraperitoneally, or topically (e.g., to mucosal surfaces).
- pharmaceutical compositions embodying features of the present invention are formulated for parenteral administration, including intravenous, subcutaneous, intramuscular, and intraperitoneal administration.
- a pharmaceutically acceptable carrier such as physiological saline.
- the pharmaceutical compositions are typically formulated in aqueous solution, preferably in physiologically compatible buffers (e.g., Hanks' solution, Ringer's solution, or physiologically buffered saline).
- penetrants appropriate to the particular barrier to be permeated are also preferable. Such penetrants are well known in the art. Other embodiments use standard intracellular delivery (e.g., delivery via liposomes) techniques. Intracellular delivery methods are well known in the art. Administration of some agents to a patient's bone marrow may necessitate delivery in a manner different from intravenous injections. [00196] In some embodiments, active pharmaceutical compositions are prepared as oily injection suspensions. Suitable lipophilic solvents or vehicles include but are not limited to fatty oils such as sesame oil, synthetic fatty acid esters, such as ethyl oleate or triglycerides, or liposomes.
- Aqueous injectable suspensions may additionally comprise substances that increase the viscosity of the suspension, such as sodium carboxymethyl cellulose, sorbitol, and dextran.
- the injectable suspension may also comprise suitable stabilizers and agents that increase or prolong the solubility of the compounds, thus allowing preparation of highly concentrated solutions.
- pharmaceutical compositions in accordance with the present invention are formulated using pharmaceutically acceptable carriers in suitable dosages for oral administration. Suitable carriers enable the compositions to be formulated as tablets, pills, capsules, dragees, liquids, gels, syrups, slurries, suspensions and the like, for oral or nasal ingestion by a subject.
- compositions for oral use are made by combining the active compounds (e.g., chemical address tag- therapeutic agent conjugates) with a solid excipient, optionally grinding the resulting mixture, and processing the mixture of granules, after adding suitable auxiliaries, if desired, so as to obtain tablets or dragee cores.
- suitable excipients include but are not limited: carbohydrate fillers such as sugars, including, lactose, sucrose, mannitol, or sorbitol; starch from corn, wheat, rice, potato; cellulose such as methyl cellulose, hydroxypropylmethyl- cellulose, and sodium carboxymethylcellulose; gums including arabic and tragacanth; and proteins such as gelatin and collagen.
- disintegrating or solubilizing agents may be added, such as cross-linked polyvinyl pyrrolidone, agar, alginic acid or a salt thereof, such as sodium alginate.
- Ingestible formulations of pharmaceutical compositions in accordance with the present invention may further comprise any material approved by the United States Department of Agriculture (or other similar international agency) for inclusion in foodstuffs and substances that are generally recognized as safe (GRAS), such as food additives, flavorings, colorings, vitamins, minerals, and phytonutrients.
- GRAS United States Department of Agriculture
- the term refers to organic compounds isolated from plants that have a biological effect, including but not limited to compounds in the following classes: isoflavonoids, oligomeric proanthcyanidins, indol-3-carbinol, sulforaph ⁇ ne, fibrous ligands, plant phytosterols, ferulic acid, anthocyanocides, triterpenes, omega 3/6 fatty acids, polyacetylene, quinones, terpenes, cathechins, gallates, quercitin, and the like.
- dragee cores are provided with suitable coatings such as concentrated sugar solutions, which may contain gum arabic, talc, polyvinylpyrrolidone, carbopol gel, polyethylene glycol, titanium dioxide, lacquer solutions, and suitable organic solvents or solvent mixtures.
- Dyestuffs or pigments may be added to tablets or dragee coatings for product identification or to characterize the quantity of active compound, (i.e., dosage).
- compositions embodying ifeatures of the present invention may include but are not limited to push-fit capsules (e.g., those made of gelatin) and soft sealed capsules (e.g., those made of gelatin), optionally having a coating such as glycerol or sorbitol.
- Push-fit capsules may contain active ingredients mixed with fillers or binders such as lactose or starches, lubricants such as talc or magnesium stearate, and, optionally, stabilizers.
- the active compounds may be dissolved or suspended in suitable liquids, such as fatty oils, liquid paraffin, or liquid polyethylene glycol, with or without stabilizers.
- the pharmaceutically acceptable carriers are pharmaceutically inert.
- compositions used in methods embodying features of the present invention are manufactured according to well-known and standard pharmaceutical manufacturing techniques (e.g., by means of conventional mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping or lyophilizing processes).
- compositions suitable for use in accordance with the present invention may further include compositions wherein the active ingredient(s) is/are contained in an effective amount to achieve the intended purpose.
- a therapeutically effective dose refers to that amount of a composition(s) that reduces symptoms or eliminates at least one symptom of the disease state.
- an effective amount of a therapeutic compound(s) may be that amount that destroys or disables pathogens as compared to a control.
- Dosing is dependent on severity and responsiveness of the disease state to be treated, with the course of treatment lasting from several days to several months, or until a cure is effected or a diminution of the disease state is achieved.
- Guidance as to particular dosing considerations and methods of delivery are provided in the literature (See, e.g., U.S. Patent Nos. 4,657,760; 5,206,344; and 5,225,212).
- Optimal dosing schedules are calculated from measurements of composition accumulation in the subject's body. The administering physician may easily determine optimum dosages, dosing methodologies and repetition rates.
- Optimum dosages may vary depending on the relative potency of compositions and may generally be estimated based on the EC 50 values found to be effective in in vitro and in vivo animal models. Additional factors that may be taken into account include but are not limited to the severity of the disease state; the subject's age, weight, and gender; the subject's diet; the time and frequency of administration; combination(s) of agents or compositions; possible reaction sensitivities or allergies; and the subject's tolerance/response to prior treatments. In general, dosage is from 0.001 ⁇ g to 100 g per kg of body weight, and may be given once or more daily, weekly, monthly or yearly.
- the treating physician preferably estimates dosing repetition rates based on measured residence times and concentrations of the agents/drugs in the subject's fluids or tissues. Following successful treatment, it may be desirable to have the subject undergo maintenance therapy to prevent the recurrence of the disease state, wherein the therapeutic agent is administered in maintenance doses, ranging from 0.001 ⁇ g to 100 g per kg of body weight, once or more daily, weekly, or the like.
- the therapeutically effective dose may be estimated initially from cell culture assays. Then, preferably, dosages may be formulated in animal models (e.g., murine or rat models) to achieve a desirable circulating concentration range.
- animal models e.g., murine or rat models
- Toxicity and therapeutic efficacy of administered pharmaceutical compositions may be determined by standard pharmaceutical procedures in cell cultures or experimental animals, e.g., for determining the LD 0 (the dose lethal to 50% of the population) and the ED 50 (the dose therapeutically effective in 50% of the population).
- the dose ratio between toxic and therapeutic effects is the therapeutic index, which may be expressed as the ratio LD 50 / ED 50 .
- Compounds that exhibit large therapeutic indices are preferred.
- the data obtained from cell culture assays and additional animal studies may be used in formulating a range of dosages, for example, for mammalian use (e.g., in humans).
- the dosage of such compounds preferably lies within a range of circulating concentrations that include the ED5 0 with little or no toxicity.
- the present invention is not limited to this range.
- the solution containing succinic dihydrazide (SDH) and propylenediaminetetraacetic acid (PDTA) was prepared according to the literature procedure (Zhang, J. et al. J. Labelled Compounds & Radiopharm. 2000, 43, 693-700). To a 5-mL vial was added the solution containing PDTA (5 mg/mL) and SDH (5 mg/mL), followed by addition of 1.0 mL saline containing 2.0 mCi of " m Tc-pertechnetate, and 10 ⁇ L SnCI 2 solution in 1.0 N HCI. The reaction mixture was kept at room temperature for 15 min to give the 99m Tc-nitrido intermediate.
- SDH succinic dihydrazide
- PDTA propylenediaminetetraacetic acid
- the compounds of the present invention may be tested in vivo to determine the therapeutic characteristics of these compounds and to identify particular compounds with desired characteristics.
- animal studies may be performed following the literature procedures (e.g., Boschi, A. et al. Nucl. Med. Commun. 2002, 23, 689).
- Biodistribution studies may be carried out using Sprague-Dawley rats in compliance with NIH animal experiment guidelines (Principles of Laboratory Animal Care, NIH Publication No. 86-23, revised 1985).
- One objective of these studies is to determine the characteristics of the " m Tc complexes (or other compounds embodying features of the present invention) as radiopharmaceuticals.
- These studies may be used as a preliminary screening tool to determine the biodistribution characteristics, excretion kinetics, and metabolism of 99m Tc complexes of crowned DTC chelators.
- Sprague-Dawley rats (200-250 g) may be anesthetized with an intramuscle injection of a mixture of ketamine (80 mg/kg) and xylazine (19 mg/kg). These rats may then receive the 99m Tc radiopharmaceutical (50-100 ⁇ Ci in 100 ⁇ L solution) via intravenous injection into the tail vein.
- the amount of activity administered to each animal may be quantified, ultimately allowing the biodistribution of each radiopharmaceutical to be calculated as both a percentage of the injected dose per organ (% ID/organ) and a percentage of the injected dose per gram of tissue wet mass (% ID/g).
- the animals may then be sacrificed via exsanguinations and opening of the thoracic cavity at selected time points. Relevant tissues and organs may then be excised, weighed, and counted to determine the tissue uptake of the " m Tc complex (or other compounds in accordance with the present invention that are tested).
- the organs that may be examined include, for example, heart, brain, blood, lung, liver, spleen, kidneys, muscle, intestines, and bone. Five rats, for example, may be used at each selected time point to ensure acquisition of reliable biological data.
- Statistical analysis of the biodistribution data from these experiments may employ the Student r test for comparison of radiotracer biodistribution between different 99m Tc radiopharmaceuticals (or other compounds in accordance with the present invention).
- m Tc radiopharmaceuticals would be those that have high heart uptake, long heart retention time, and rapid blood clearance, preferably via renal system.
- This model may also be used to assess the effectiveness of radiopharmaceuticals embodying features of the present invention comprised of a positron emitting isotope such as 94m Tc.
- the radiopharmaceuticals may be administered in appropriate amounts and the uptake in the heart may be quantified non-invasively by imaging for those isotopes with a coincident imageable gamma emission.
- the diagnostic radiopharmaceuticals may be administered by intravenous injection, usually in saline solution, at a dose of 1 to 100 mCi per 70 kg body weight, or preferably at a dose of 5 to 30 mCi. Imaging is performed using known procedures.
- the therapeutic radiopharmaceuticals may be administered by intravenous injection, usually in saline solution, at a dose of 0.1 to 100 mCi per 70 kg body weight, or preferably at a dose of 0.5 to 50 mCi per 70 kg body weight.
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/588,343 US20070166227A1 (en) | 2004-02-10 | 2005-02-10 | Crowned dithiocarbamate metal complexes and methods for their use |
| EP05726497A EP1718656A2 (en) | 2004-02-10 | 2005-02-10 | Crowned dithiocarbamate metal complexes and methods for their use |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US54317604P | 2004-02-10 | 2004-02-10 | |
| US60/543,176 | 2004-02-10 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005077962A2 true WO2005077962A2 (en) | 2005-08-25 |
| WO2005077962A3 WO2005077962A3 (en) | 2005-12-15 |
Family
ID=34860386
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2005/004872 WO2005077962A2 (en) | 2004-02-10 | 2005-02-10 | Crowned dithiocarbamate metal complexes and methods for their use |
Country Status (3)
| Country | Link |
|---|---|
| US (1) | US20070166227A1 (en) |
| EP (1) | EP1718656A2 (en) |
| WO (1) | WO2005077962A2 (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008061792A2 (en) * | 2006-11-24 | 2008-05-29 | Mallinckrodt Inc. | Tricarbonyl complexes with tridentate chelators for myocardium imaging |
| US7708961B2 (en) | 2007-03-31 | 2010-05-04 | Advanced Applied Physics Solutions, Inc. | Method and apparatus for isolating the radioisotope 186Rhenium |
| US8026370B2 (en) | 2004-11-04 | 2011-09-27 | Mitsubishi Tanabe Pharma Corporation | Carboxylic acid derivative containing thiazole ring and pharmaceutical use thereof |
| US8282967B2 (en) | 2005-05-27 | 2012-10-09 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US8591876B2 (en) | 2010-12-15 | 2013-11-26 | Novan, Inc. | Methods of decreasing sebum production in the skin |
| US8981139B2 (en) | 2011-02-28 | 2015-03-17 | The University Of North Carolina At Chapel Hill | Tertiary S-nitrosothiol-modified nitric—oxide-releasing xerogels and methods of using the same |
| US9526738B2 (en) | 2009-08-21 | 2016-12-27 | Novan, Inc. | Topical gels and methods of using the same |
| US9587292B2 (en) | 2009-10-01 | 2017-03-07 | Advanced Applied Physics Solutions, Inc. | Method and apparatus for isolating the radioisotope molybdenum-99 |
| CN106588729A (en) * | 2016-12-06 | 2017-04-26 | 青岛柏森环保工程有限公司 | Preparation method of multi-sulfo hydrazide type heavy metal chelating agent |
| US9919072B2 (en) | 2009-08-21 | 2018-03-20 | Novan, Inc. | Wound dressings, methods of using the same and methods of forming the same |
Families Citing this family (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8909325B2 (en) | 2000-08-21 | 2014-12-09 | Biosensors International Group, Ltd. | Radioactive emission detector equipped with a position tracking system and utilization thereof with medical systems and in medical procedures |
| US8489176B1 (en) | 2000-08-21 | 2013-07-16 | Spectrum Dynamics Llc | Radioactive emission detector equipped with a position tracking system and utilization thereof with medical systems and in medical procedures |
| US8565860B2 (en) | 2000-08-21 | 2013-10-22 | Biosensors International Group, Ltd. | Radioactive emission detector equipped with a position tracking system |
| WO2005119025A2 (en) | 2004-06-01 | 2005-12-15 | Spectrum Dynamics Llc | Radioactive-emission-measurement optimization to specific body structures |
| US8036731B2 (en) | 2001-01-22 | 2011-10-11 | Spectrum Dynamics Llc | Ingestible pill for diagnosing a gastrointestinal tract |
| IL157007A0 (en) | 2001-01-22 | 2004-02-08 | Target Technologies Ltd V | Ingestible device |
| WO2007054935A2 (en) | 2005-11-09 | 2007-05-18 | Spectrum Dynamics Llc | Dynamic spect camera |
| WO2007010534A2 (en) | 2005-07-19 | 2007-01-25 | Spectrum Dynamics Llc | Imaging protocols |
| US9040016B2 (en) | 2004-01-13 | 2015-05-26 | Biosensors International Group, Ltd. | Diagnostic kit and methods for radioimaging myocardial perfusion |
| US9470801B2 (en) | 2004-01-13 | 2016-10-18 | Spectrum Dynamics Llc | Gating with anatomically varying durations |
| US7968851B2 (en) | 2004-01-13 | 2011-06-28 | Spectrum Dynamics Llc | Dynamic spect camera |
| US8586932B2 (en) | 2004-11-09 | 2013-11-19 | Spectrum Dynamics Llc | System and method for radioactive emission measurement |
| US8571881B2 (en) | 2004-11-09 | 2013-10-29 | Spectrum Dynamics, Llc | Radiopharmaceutical dispensing, administration, and imaging |
| CN1981210A (en) | 2004-01-13 | 2007-06-13 | 光谱动力学有限责任公司 | Multi-dimensional image reconstruction |
| EP1766550A2 (en) * | 2004-06-01 | 2007-03-28 | Spectrum Dynamics LLC | Methods of view selection for radioactive emission measurements |
| US8000773B2 (en) | 2004-11-09 | 2011-08-16 | Spectrum Dynamics Llc | Radioimaging |
| US9943274B2 (en) | 2004-11-09 | 2018-04-17 | Spectrum Dynamics Medical Limited | Radioimaging using low dose isotope |
| EP1827505A4 (en) | 2004-11-09 | 2017-07-12 | Biosensors International Group, Ltd. | Radioimaging |
| US8615405B2 (en) | 2004-11-09 | 2013-12-24 | Biosensors International Group, Ltd. | Imaging system customization using data from radiopharmaceutical-associated data carrier |
| US9316743B2 (en) | 2004-11-09 | 2016-04-19 | Biosensors International Group, Ltd. | System and method for radioactive emission measurement |
| WO2008059489A2 (en) | 2006-11-13 | 2008-05-22 | Spectrum Dynamics Llc | Radioimaging applications of and novel formulations of teboroxime |
| US7872235B2 (en) * | 2005-01-13 | 2011-01-18 | Spectrum Dynamics Llc | Multi-dimensional image reconstruction and analysis for expert-system diagnosis |
| US8837793B2 (en) | 2005-07-19 | 2014-09-16 | Biosensors International Group, Ltd. | Reconstruction stabilizer and active vision |
| US8111886B2 (en) | 2005-07-19 | 2012-02-07 | Spectrum Dynamics Llc | Reconstruction stabilizer and active vision |
| EP1966984A2 (en) | 2005-12-28 | 2008-09-10 | Starhome GmbH | Optimal voicemail deposit for roaming cellular telephony |
| US8894974B2 (en) | 2006-05-11 | 2014-11-25 | Spectrum Dynamics Llc | Radiopharmaceuticals for diagnosis and therapy |
| US9275451B2 (en) | 2006-12-20 | 2016-03-01 | Biosensors International Group, Ltd. | Method, a system, and an apparatus for using and processing multidimensional data |
| US8521253B2 (en) | 2007-10-29 | 2013-08-27 | Spectrum Dynamics Llc | Prostate imaging |
| CU23844B1 (en) * | 2009-04-17 | 2012-10-15 | Ct De Neurociencias De Cuba | PROCEDURE FOR OBTAINING NEW DERIVATIVES OF NAFTALENE FOR THE LIVE DIAGNOSIS OF ALZHEIMER'S DISEASE |
| US8338788B2 (en) | 2009-07-29 | 2012-12-25 | Spectrum Dynamics Llc | Method and system of optimized volumetric imaging |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6322771B1 (en) * | 1999-06-18 | 2001-11-27 | University Of Virginia Patent Foundation | Induction of pharmacological stress with adenosine receptor agonists |
-
2005
- 2005-02-10 US US10/588,343 patent/US20070166227A1/en not_active Abandoned
- 2005-02-10 EP EP05726497A patent/EP1718656A2/en not_active Withdrawn
- 2005-02-10 WO PCT/US2005/004872 patent/WO2005077962A2/en not_active Application Discontinuation
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6322771B1 (en) * | 1999-06-18 | 2001-11-27 | University Of Virginia Patent Foundation | Induction of pharmacological stress with adenosine receptor agonists |
Non-Patent Citations (6)
| Title |
|---|
| CAMERON, B ET AL.: "Ruthenium (III) Triazacyclononane Dithiocarbamate..." INORGANIC CHEMISTRY, vol. 42, no. 13, 2003, pages 4102-4108, XP002335490 * |
| DATABASE CA [Online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; BERRY, NEIL G. ET AL: "Heteroditopic transition metal dithiocarbamate receptors for binding cation-anion ion pairs" XP002332544 retrieved from STN Database accession no. 2003:732428 & JOURNAL OF SUPRAMOLECULAR CHEMISTRY , VOLUME DATE 2002, 2(1-3), 89-92 CODEN: JSCOC9; ISSN: 1472-7862, 2003, * |
| DATABASE CA [Online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; GRANELL, JAUME ET AL: "Studies on the synthesis and electrochemistry of crown ether dithiocarbamates and the molecular dynamics of bis(aza-15-crown- 5)thiuram disulfide. Crystal structure of cobalt tris[(aza-15-crown-5)dithiocarbamate]" XP002332545 retrieved from STN Database accession no. 1990:490225 & JOURNAL OF THE CHEMICAL SOCIETY, DALTON TRANSACTIONS: INORGANIC CHEMISTRY (1972-1999) , (2), 605-14 CODEN: JCDTBI; ISSN: 0300-9246, 1990, cited in the application -& DATABASE BEILSTEIN [Online] BEILSTEIN INSTITUTE FOR ORGANIC CHEMISTRY, FRANKFURT-MAIN, DE; 23 October 1991 (1991-10-23), XP002332644 Database accession no. BRN 3580252; BRN3582715 * |
| DIAZ ET AL.: "Interactions of nitric ocide with copper (II) dithiocarbamates in aqueous solution" JOURNAL OF INORGANIC BIOCHEMISTRY, no. 95, 2003, pages 283-290, XP002335489 * |
| SCHRAMM D ET AL: "SYNTHESIS OF CATIONIC HYDROTRIS(PYRAZOLYL)BORATE ZIRCONIUM COMPOUNDS" INTERNATIONAL CONFERENCE ON ORGANOMETALLIC CHEMISTRY. BOOK OF ABSTRACTS, 16 August 1998 (1998-08-16), page A150, XP002249590 * |
| VÖGTLE, FRITZ, NEUMANN, PETER: "Stereochemie im Innern cyclischer Verbindungen: Kronenäther, Katapinate und Kryptate" CHEMIKER-ZEITUNG, vol. 97, no. 11, 1973, pages 600-610, XP002335491 * |
Cited By (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8026370B2 (en) | 2004-11-04 | 2011-09-27 | Mitsubishi Tanabe Pharma Corporation | Carboxylic acid derivative containing thiazole ring and pharmaceutical use thereof |
| US8956658B2 (en) | 2005-05-27 | 2015-02-17 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US8282967B2 (en) | 2005-05-27 | 2012-10-09 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US8962029B2 (en) | 2005-05-27 | 2015-02-24 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US11691995B2 (en) | 2005-05-27 | 2023-07-04 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US9403851B2 (en) | 2005-05-27 | 2016-08-02 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US9403852B2 (en) | 2005-05-27 | 2016-08-02 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
| US9023315B2 (en) | 2006-11-24 | 2015-05-05 | Mallinckrodt Llc | Tricarbonyl complexes with tridentate chelators for myocardium imaging |
| WO2008061792A2 (en) * | 2006-11-24 | 2008-05-29 | Mallinckrodt Inc. | Tricarbonyl complexes with tridentate chelators for myocardium imaging |
| US8372379B2 (en) | 2006-11-24 | 2013-02-12 | Mallinckrodt Llc | Technetium-99m (I) tricarbonyl complexes with tridentate chelators for myocardium imaging |
| US8940273B2 (en) | 2006-11-24 | 2015-01-27 | Mallinckrodt Llc | Tricarbonyl complexes with tridentate chelators for myocardium imaging |
| WO2008061792A3 (en) * | 2006-11-24 | 2008-08-07 | Mallinckrodt Inc | Tricarbonyl complexes with tridentate chelators for myocardium imaging |
| WO2008120084A3 (en) * | 2007-03-31 | 2013-07-18 | Advanced Applied Physics Solutions Inc. | Method and apparatus for isolating 186rhenium |
| US8211390B2 (en) | 2007-03-31 | 2012-07-03 | Advanced Applied Physics Solutions, Inc. | Method and apparatus for isolating a radioisotope |
| US7708961B2 (en) | 2007-03-31 | 2010-05-04 | Advanced Applied Physics Solutions, Inc. | Method and apparatus for isolating the radioisotope 186Rhenium |
| US10376538B2 (en) | 2009-08-21 | 2019-08-13 | Novan, Inc. | Topical gels and methods of using the same |
| US9526738B2 (en) | 2009-08-21 | 2016-12-27 | Novan, Inc. | Topical gels and methods of using the same |
| US11583608B2 (en) | 2009-08-21 | 2023-02-21 | Novan, Inc. | Wound dressings, methods of using the same and methods of forming the same |
| US9737561B2 (en) | 2009-08-21 | 2017-08-22 | Novan, Inc. | Topical gels and methods of using the same |
| US9919072B2 (en) | 2009-08-21 | 2018-03-20 | Novan, Inc. | Wound dressings, methods of using the same and methods of forming the same |
| US9587292B2 (en) | 2009-10-01 | 2017-03-07 | Advanced Applied Physics Solutions, Inc. | Method and apparatus for isolating the radioisotope molybdenum-99 |
| US8591876B2 (en) | 2010-12-15 | 2013-11-26 | Novan, Inc. | Methods of decreasing sebum production in the skin |
| US8981139B2 (en) | 2011-02-28 | 2015-03-17 | The University Of North Carolina At Chapel Hill | Tertiary S-nitrosothiol-modified nitric—oxide-releasing xerogels and methods of using the same |
| US9713652B2 (en) | 2011-02-28 | 2017-07-25 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing S-nitrosothiol-modified silica particles and methods of making the same |
| CN106588729B (en) * | 2016-12-06 | 2021-05-28 | 青岛柏森环保工程有限公司 | Preparation method of polythiohydrazide type heavy metal chelating agent |
| CN106588729A (en) * | 2016-12-06 | 2017-04-26 | 青岛柏森环保工程有限公司 | Preparation method of multi-sulfo hydrazide type heavy metal chelating agent |
Also Published As
| Publication number | Publication date |
|---|---|
| US20070166227A1 (en) | 2007-07-19 |
| EP1718656A2 (en) | 2006-11-08 |
| WO2005077962A3 (en) | 2005-12-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20070166227A1 (en) | Crowned dithiocarbamate metal complexes and methods for their use | |
| Lever et al. | Design, preparation, and biodistribution of a technetium-99m triaminedithiol complex to assess regional cerebral blood flow | |
| US20080124273A1 (en) | Novel cationic metal complex radiopharmaceuticals | |
| TW201034690A (en) | Technetium-and rhenium-bis (heteroaryl) complexes and methods of use thereof for inhibiting PSMA | |
| JPS6069090A (en) | Complex of technetium-99m with alkylene amine oxime | |
| Zhang et al. | Synthesis and biodistribution of a novel 99mTcN complex of ciprofloxacin dithiocarbamate as a potential agent for infection imaging | |
| Paparidis et al. | Synthesis and evaluation of 99mTc/Re-tricarbonyl complexes of the triphenylphosphonium cation for mitochondrial targeting | |
| Yang et al. | Formulation of 68Ga BAPEN kit for myocardial positron emission tomography imaging and biodistribution study | |
| Del Rosario et al. | Synthesis and in vivo evaluation of a 99m99Tc-DADT-Benzovesamicol: a potential marker for cholinergic neurons | |
| Kim et al. | Synthesis and characterization of a 68Ga-labeled N-(2-diethylaminoethyl) benzamide derivative as potential PET probe for malignant melanoma | |
| Li et al. | Kit formulation for preparation and biological evaluation of a novel 99mTc-oxo complex with metronidazole xanthate for imaging tumor hypoxia | |
| Zhang et al. | Synthesis and biological evaluation of a novel 99mTc nitrido radiopharmaceutical with deoxyglucose dithiocarbamate, showing tumor uptake | |
| Gniazdowska et al. | Synthesis, physicochemical and biological studies of technetium-99m labeled tacrine derivative as a diagnostic tool for evaluation of cholinesterase level | |
| Giglio et al. | Design and evaluation of “3+ 1” mixed ligand oxorhenium and oxotechnetium complexes bearing a nitroaromatic group with potential application in nuclear medicine oncology | |
| Lin et al. | Synthesis and preliminary biological evaluation of a 99mTc‐chlorambucil derivative as a potential tumor imaging agent | |
| Zhang et al. | Synthesis of a bis-(N-sec-butyl-dithiocarbamato)-nitrido-99mTc complex: a potential new radiopharmaceutical for brain perfusion studies | |
| Chen et al. | Preparation and biological evaluation of 99mTc-CO-MIBI as myocardial perfusion imaging agent | |
| Zhuang et al. | Neutral and stereospecific Tc-99m complexes:[99mTc] N-benzyl-3, 4-di-(N-2-mercaptoethyl)-amino-pyrrolidines (P-BAT) | |
| Sivapackiam et al. | Synthesis, molecular structure, and validation of metalloprobes for assessment of MDR1 P-glycoprotein-mediated functional transport | |
| Tang et al. | Synthesis and application of 188Re-MN-16ET/Lipiodol in a hepatocellular carcinoma animal model | |
| Pillai et al. | Radiochemical studies of 99mTc complexes of modified cysteine ligands and bifunctional chelating agents | |
| Tamagnan et al. | Synthesis of β-CIT-BAT, a potential technetium-99m imaging ligand for dopamine transporter | |
| Satpati et al. | Synthesis, characterization and biological activity of 99mTc‐labeled piperidine analogues targeting sigma receptors | |
| Babich et al. | 6-Mercaptomethylpyridine-3-carboxylic acid (MEMNIC): a new reagent for peptide labeling with Tc-99m | |
| EP2673257B1 (en) | Pet/spect agents for applications in biomedical imaging |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2007166227 Country of ref document: US Ref document number: 10588343 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005726497 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005726497 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10588343 Country of ref document: US |