WO2003095509A1 - Sulfonated copolymer - Google Patents
Sulfonated copolymer Download PDFInfo
- Publication number
- WO2003095509A1 WO2003095509A1 PCT/US2003/015178 US0315178W WO03095509A1 WO 2003095509 A1 WO2003095509 A1 WO 2003095509A1 US 0315178 W US0315178 W US 0315178W WO 03095509 A1 WO03095509 A1 WO 03095509A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- membrane
- comonomer
- sulfonated
- mol
- polymer
- Prior art date
Links
- 229920001577 copolymer Polymers 0.000 title claims abstract description 26
- 239000012528 membrane Substances 0.000 claims abstract description 68
- 239000000446 fuel Substances 0.000 claims abstract description 29
- 229920000642 polymer Polymers 0.000 claims description 43
- 239000003054 catalyst Substances 0.000 claims description 23
- 239000000178 monomer Substances 0.000 claims description 22
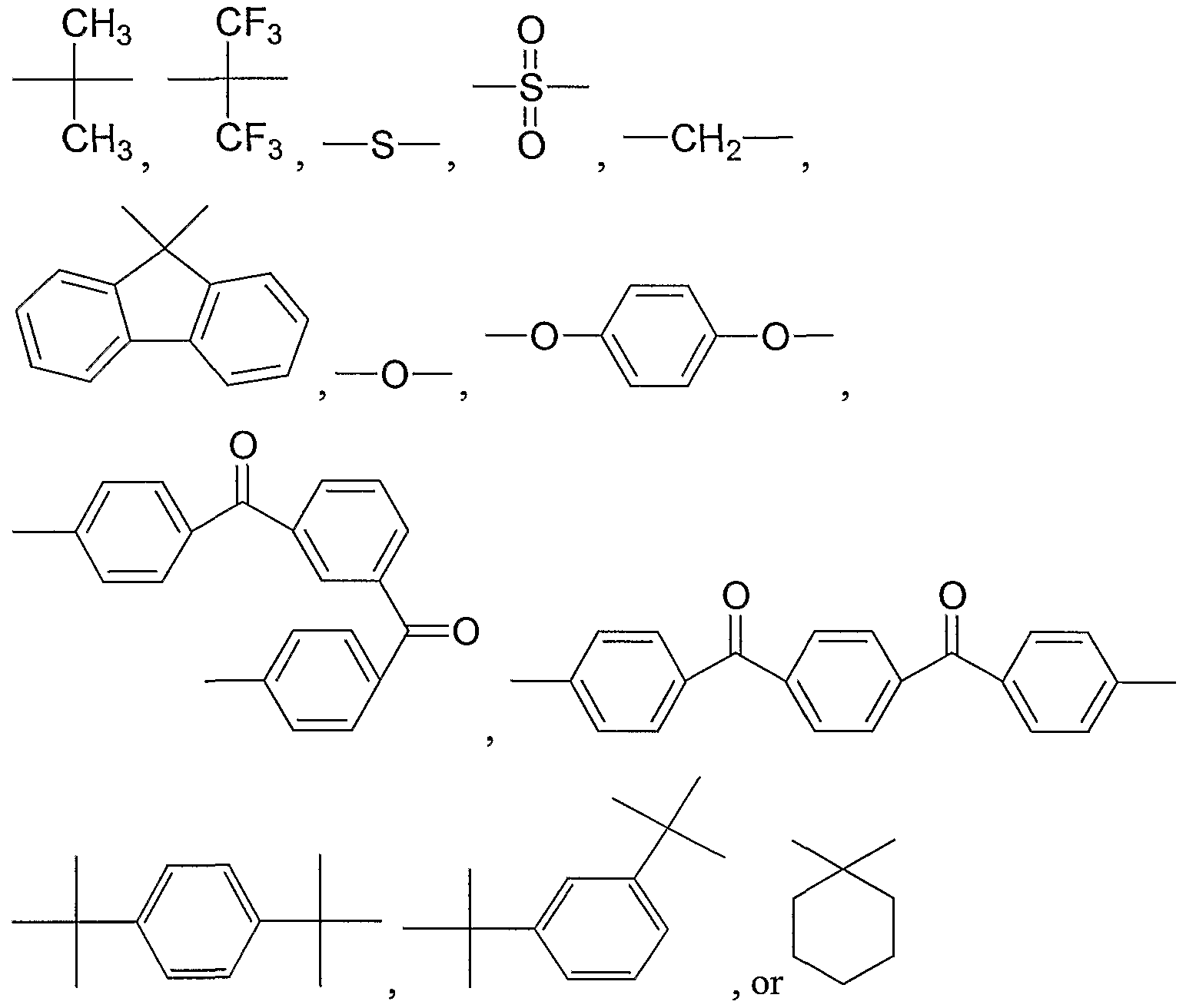
- -1 cyclohexylidene group Chemical group 0.000 claims description 16
- 150000001768 cations Chemical group 0.000 claims description 7
- 238000000034 method Methods 0.000 claims description 6
- 125000005362 aryl sulfone group Chemical group 0.000 claims description 4
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 4
- 238000002360 preparation method Methods 0.000 claims description 3
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 claims description 2
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 claims description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims 1
- 239000005518 polymer electrolyte Substances 0.000 abstract description 7
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 154
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 123
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 84
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 56
- 239000000203 mixture Substances 0.000 description 51
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 30
- 229910000027 potassium carbonate Inorganic materials 0.000 description 28
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 27
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 22
- LSQARZALBDFYQZ-UHFFFAOYSA-N 4,4'-difluorobenzophenone Chemical compound C1=CC(F)=CC=C1C(=O)C1=CC=C(F)C=C1 LSQARZALBDFYQZ-UHFFFAOYSA-N 0.000 description 20
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 18
- 229910052757 nitrogen Inorganic materials 0.000 description 15
- 238000003756 stirring Methods 0.000 description 15
- 230000008961 swelling Effects 0.000 description 15
- 239000011541 reaction mixture Substances 0.000 description 14
- 239000007787 solid Substances 0.000 description 13
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 12
- 239000012043 crude product Substances 0.000 description 11
- 238000010992 reflux Methods 0.000 description 11
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 10
- SDDLEVPIDBLVHC-UHFFFAOYSA-N Bisphenol Z Chemical compound C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)CCCCC1 SDDLEVPIDBLVHC-UHFFFAOYSA-N 0.000 description 9
- YWFPGFJLYRKYJZ-UHFFFAOYSA-N 9,9-bis(4-hydroxyphenyl)fluorene Chemical compound C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)C2=CC=CC=C2C2=CC=CC=C21 YWFPGFJLYRKYJZ-UHFFFAOYSA-N 0.000 description 8
- 229920000557 Nafion® Polymers 0.000 description 8
- JUPQTSLXMOCDHR-UHFFFAOYSA-N benzene-1,4-diol;bis(4-fluorophenyl)methanone Chemical class OC1=CC=C(O)C=C1.C1=CC(F)=CC=C1C(=O)C1=CC=C(F)C=C1 JUPQTSLXMOCDHR-UHFFFAOYSA-N 0.000 description 8
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 7
- 229920005597 polymer membrane Polymers 0.000 description 7
- 229920005604 random copolymer Polymers 0.000 description 7
- 150000002500 ions Chemical class 0.000 description 6
- BWQOPMJTQPWHOZ-UHFFFAOYSA-N (2,3-difluorophenyl)-phenylmethanone Chemical compound FC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1F BWQOPMJTQPWHOZ-UHFFFAOYSA-N 0.000 description 5
- MHABMANUFPZXEB-UHFFFAOYSA-N O-demethyl-aloesaponarin I Natural products O=C1C2=CC=CC(O)=C2C(=O)C2=C1C=C(O)C(C(O)=O)=C2C MHABMANUFPZXEB-UHFFFAOYSA-N 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 238000000576 coating method Methods 0.000 description 5
- XKZQKPRCPNGNFR-UHFFFAOYSA-N 2-(3-hydroxyphenyl)phenol Chemical compound OC1=CC=CC(C=2C(=CC=CC=2)O)=C1 XKZQKPRCPNGNFR-UHFFFAOYSA-N 0.000 description 4
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 229920001940 conductive polymer Polymers 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 229910052697 platinum Inorganic materials 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- SUTQSIHGGHVXFK-UHFFFAOYSA-N 1,2,2-trifluoroethenylbenzene Chemical compound FC(F)=C(F)C1=CC=CC=C1 SUTQSIHGGHVXFK-UHFFFAOYSA-N 0.000 description 3
- NZGQHKSLKRFZFL-UHFFFAOYSA-N 4-(4-hydroxyphenoxy)phenol Chemical compound C1=CC(O)=CC=C1OC1=CC=C(O)C=C1 NZGQHKSLKRFZFL-UHFFFAOYSA-N 0.000 description 3
- 125000003118 aryl group Chemical group 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000002322 conducting polymer Substances 0.000 description 3
- 239000006185 dispersion Substances 0.000 description 3
- 230000004907 flux Effects 0.000 description 3
- 229920000554 ionomer Polymers 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 229920006393 polyether sulfone Polymers 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 238000006277 sulfonation reaction Methods 0.000 description 3
- PLVUIVUKKJTSDM-UHFFFAOYSA-N 1-fluoro-4-(4-fluorophenyl)sulfonylbenzene Chemical compound C1=CC(F)=CC=C1S(=O)(=O)C1=CC=C(F)C=C1 PLVUIVUKKJTSDM-UHFFFAOYSA-N 0.000 description 2
- VWGKEVWFBOUAND-UHFFFAOYSA-N 4,4'-thiodiphenol Chemical compound C1=CC(O)=CC=C1SC1=CC=C(O)C=C1 VWGKEVWFBOUAND-UHFFFAOYSA-N 0.000 description 2
- 229930185605 Bisphenol Natural products 0.000 description 2
- 206010004966 Bite Diseases 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 239000003570 air Substances 0.000 description 2
- 230000000712 assembly Effects 0.000 description 2
- 238000000429 assembly Methods 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 239000001569 carbon dioxide Substances 0.000 description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 2
- 238000005266 casting Methods 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- BNBRIFIJRKJGEI-UHFFFAOYSA-N 2,6-difluorobenzonitrile Chemical compound FC1=CC=CC(F)=C1C#N BNBRIFIJRKJGEI-UHFFFAOYSA-N 0.000 description 1
- BATCUENAARTUKW-UHFFFAOYSA-N 4-[(4-hydroxyphenyl)-diphenylmethyl]phenol Chemical compound C1=CC(O)=CC=C1C(C=1C=CC(O)=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 BATCUENAARTUKW-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical group F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical group C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 229910002848 Pt–Ru Inorganic materials 0.000 description 1
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 1
- 239000012080 ambient air Substances 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- SFTCAQYXDUJAHL-UHFFFAOYSA-N benzene-1,4-diol;4-[1-(4-hydroxyphenyl)cyclohexyl]phenol Chemical compound OC1=CC=C(O)C=C1.C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)CCCCC1 SFTCAQYXDUJAHL-UHFFFAOYSA-N 0.000 description 1
- ZFVMWEVVKGLCIJ-UHFFFAOYSA-N bisphenol AF Chemical compound C1=CC(O)=CC=C1C(C(F)(F)F)(C(F)(F)F)C1=CC=C(O)C=C1 ZFVMWEVVKGLCIJ-UHFFFAOYSA-N 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- FNIATMYXUPOJRW-UHFFFAOYSA-N cyclohexylidene Chemical group [C]1CCCCC1 FNIATMYXUPOJRW-UHFFFAOYSA-N 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000032798 delamination Effects 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 230000033001 locomotion Effects 0.000 description 1
- 230000003137 locomotive effect Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- VUZPPFZMUPKLLV-UHFFFAOYSA-N methane;hydrate Chemical compound C.O VUZPPFZMUPKLLV-UHFFFAOYSA-N 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- 239000012038 nucleophile Substances 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-M phenolate Chemical compound [O-]C1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-M 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002959 polymer blend Polymers 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1025—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having only carbon and oxygen, e.g. polyethers, sulfonated polyetheretherketones [S-PEEK], sulfonated polysaccharides, sulfonated celluloses or sulfonated polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D67/00—Processes specially adapted for manufacturing semi-permeable membranes for separation processes or apparatus
- B01D67/0081—After-treatment of organic or inorganic membranes
- B01D67/0088—Physical treatment with compounds, e.g. swelling, coating or impregnation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/76—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74
- B01D71/82—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74 characterised by the presence of specified groups, e.g. introduced by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/34—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives
- C08G65/38—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives derived from phenols
- C08G65/40—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives derived from phenols from phenols (I) and other compounds (II), e.g. OH-Ar-OH + X-Ar-X, where X is halogen atom, i.e. leaving group
- C08G65/4012—Other compound (II) containing a ketone group, e.g. X-Ar-C(=O)-Ar-X for polyetherketones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/34—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from hydroxy compounds or their metallic derivatives
- C08G65/48—Polymers modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L81/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing sulfur with or without nitrogen, oxygen or carbon only; Compositions of polysulfones; Compositions of derivatives of such polymers
- C08L81/06—Polysulfones; Polyethersulfones
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1027—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having carbon, oxygen and other atoms, e.g. sulfonated polyethersulfones [S-PES]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1032—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having sulfur, e.g. sulfonated-polyethersulfones [S-PES]
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2650/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G2650/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterized by the type of post-polymerisation functionalisation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2650/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G2650/28—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterised by the polymer type
- C08G2650/38—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterised by the polymer type containing oxygen in addition to the ether group
- C08G2650/40—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterised by the polymer type containing oxygen in addition to the ether group containing ketone groups, e.g. polyarylethylketones, PEEK or PEK
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2650/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G2650/62—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterised by the nature of monomer used
- C08G2650/64—Monomer containing functional groups not involved in polymerisation
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0065—Solid electrolytes
- H01M2300/0082—Organic polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/88—Processes of manufacture
- H01M4/8803—Supports for the deposition of the catalytic active composition
- H01M4/881—Electrolytic membranes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1009—Fuel cells with solid electrolytes with one of the reactants being liquid, solid or liquid-charged
- H01M8/1011—Direct alcohol fuel cells [DAFC], e.g. direct methanol fuel cells [DMFC]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Manufacturing & Machinery (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Sustainable Development (AREA)
- Life Sciences & Earth Sciences (AREA)
- Sustainable Energy (AREA)
- Electrochemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Polyethers (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
- Conductive Materials (AREA)
- Inert Electrodes (AREA)
- Polymers With Sulfur, Phosphorus Or Metals In The Main Chain (AREA)
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020047018336A KR100977234B1 (en) | 2002-05-13 | 2003-05-13 | Sulfonated copolymer |
| DE60333367T DE60333367D1 (en) | 2002-05-13 | 2003-05-13 | SULFONED COPOLYMER |
| EP03736609A EP1517929B1 (en) | 2002-05-13 | 2003-05-13 | Sulfonated copolymer |
| AT03736609T ATE474005T1 (en) | 2002-05-13 | 2003-05-13 | SULFONATED COPOLYMER |
| AU2003237849A AU2003237849B2 (en) | 2002-05-13 | 2003-05-13 | Sulfonated copolymer |
| CA002485727A CA2485727A1 (en) | 2002-05-13 | 2003-05-13 | Sulfonated copolymer |
| JP2004503520A JP2006506472A (en) | 2002-05-13 | 2003-05-13 | Sulfonated copolymer |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US38113602P | 2002-05-13 | 2002-05-13 | |
| US60/381,136 | 2002-05-13 | ||
| US42654002P | 2002-11-15 | 2002-11-15 | |
| US60/426,540 | 2002-11-15 | ||
| US44639503P | 2003-02-10 | 2003-02-10 | |
| US60/446,395 | 2003-02-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2003095509A1 true WO2003095509A1 (en) | 2003-11-20 |
Family
ID=29424538
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2003/015178 WO2003095509A1 (en) | 2002-05-13 | 2003-05-13 | Sulfonated copolymer |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US7202001B2 (en) |
| EP (1) | EP1517929B1 (en) |
| JP (1) | JP2006506472A (en) |
| CN (1) | CN100509875C (en) |
| AT (1) | ATE474005T1 (en) |
| AU (1) | AU2003237849B2 (en) |
| CA (1) | CA2485727A1 (en) |
| DE (1) | DE60333367D1 (en) |
| WO (1) | WO2003095509A1 (en) |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005197235A (en) * | 2003-12-09 | 2005-07-21 | Jsr Corp | Proton conduction film |
| JP2005268145A (en) * | 2004-03-22 | 2005-09-29 | Toyobo Co Ltd | Electrolyte film and its manufacturing method |
| WO2005090439A2 (en) * | 2004-03-12 | 2005-09-29 | Polyfuel, Inc. | Ion-conductive copolymers containing one or more ion-conducting oligomers |
| JP2005276441A (en) * | 2004-02-27 | 2005-10-06 | Toyobo Co Ltd | Ion exchange membrane |
| EP1612232A1 (en) * | 2003-04-07 | 2006-01-04 | Mitsui Chemicals, Inc. | Crosslinkable ionically conducting resin, and ionically conducting polymer membranes, binders and fuel cells, made by using the resin |
| JP2006070126A (en) * | 2004-09-01 | 2006-03-16 | Toray Ind Inc | Heat-resistant resin and resin composition and molding each using the same |
| EP1674498A1 (en) * | 2003-10-17 | 2006-06-28 | Sumitomo Chemical Company, Limited | Block copolymer and use thereof |
| JP2006202598A (en) * | 2005-01-20 | 2006-08-03 | Toray Ind Inc | Fuel cell electrode and fuel cell |
| WO2006087995A1 (en) * | 2005-02-15 | 2006-08-24 | Toray Industries, Inc. | Process for producing polymer electrolyte molded product, polymer electrolyte material, polymer electrolyte membrane, and solid polymer electrolyte fuel cell |
| WO2006132207A1 (en) * | 2005-06-09 | 2006-12-14 | Toyo Boseki Kabushiki Kaisha | Sulfonic acid group-containing polymer, method for producing same, resin composition containing such sulfonic acid group-containing polymer, polymer electrolyte membrane, polymer electrolyte membrane/electrode assembly, and fuel cell |
| JP2007524973A (en) * | 2004-01-22 | 2007-08-30 | ゼネラル・モーターズ・コーポレーション | Membrane electrode assembly prepared by spraying catalyst directly onto membrane |
| EP1862489A1 (en) * | 2006-05-31 | 2007-12-05 | Sumitomo Chemical Company, Limited | Block Copolymer and Use Thereof |
| EP1886373A2 (en) * | 2005-06-01 | 2008-02-13 | Polyfuel, Inc. | Polymer blend comprising ion-conducting copolymer and non-ionic polymer |
| US7507771B2 (en) | 2003-11-13 | 2009-03-24 | Polyfuel, Inc. | Ion conductive copolymers containing one or more hydrophobic oligomers |
| WO2009079625A2 (en) * | 2007-12-18 | 2009-06-25 | Polyfuel, Inc. | Ion conducting copolymers with elastomeric and polyarylene segments |
| US7572535B2 (en) | 2002-05-13 | 2009-08-11 | Polyfuel, Inc. | Ion conductive copolymers containing one or more hydrophobic oligomers |
| CN101218281B (en) * | 2005-06-09 | 2012-03-28 | 东洋纺织株式会社 | Sulfonic acid group-containing polymer, method for producing same, resin composition containing such sulfonic acid group-containing polymer, polymer electrolyte membrane, polymer electrolyte membrane/electrode adapter, fuel cell |
| US8216727B2 (en) | 2004-11-10 | 2012-07-10 | Toyo Boseki Kabushiki Kaisha | Aromatic hydrocarbon based proton exchange membrane and direct methanol fuel cell using same |
| JP2012256601A (en) * | 2005-07-29 | 2012-12-27 | Toray Ind Inc | Polymer electrolyte material, polyelectrolyte component, membrane electrode assembly, and polymer electrolyte fuel cell |
| JP5181474B2 (en) * | 2004-09-03 | 2013-04-10 | 東レ株式会社 | Polymer electrolyte material, polymer electrolyte component, membrane electrode composite, and polymer electrolyte fuel cell |
| US8455141B2 (en) | 2003-06-25 | 2013-06-04 | Toray Industries, Inc. | Polymer electrolyte as well as polymer electrolyte membrane, membrane electrode assembly and polymer electrolyte fuel cell using the same |
| WO2014117110A1 (en) | 2013-01-28 | 2014-07-31 | Greene, Tweed Technologies, Inc. | Anti-extrusion compositions for sealing and wear components |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE602004021402D1 (en) * | 2003-09-30 | 2009-07-16 | Sumitomo Chemical Co | BLOCK COPOLYMERS AND USE THEREOF |
| WO2005082024A2 (en) * | 2004-02-24 | 2005-09-09 | Ini Power Systems, Inc. | Fuel cell apparatus and method of fabrication |
| JP2005281568A (en) * | 2004-03-30 | 2005-10-13 | Hitachi Chem Co Ltd | Acid group-containing polymer compound, polymer electrolyte membrane and fuel cell using it |
| US20060088744A1 (en) * | 2004-09-15 | 2006-04-27 | Markoski Larry J | Electrochemical cells |
| CN101375457A (en) * | 2005-05-24 | 2009-02-25 | 复合燃料公司 | Ion conductive copolymers containing ion-conducting oligomers |
| EP1883671A2 (en) * | 2005-05-27 | 2008-02-06 | Polyfuel, Inc. | End-capped ion-conductive polymers |
| JP2007002016A (en) * | 2005-06-21 | 2007-01-11 | Toyobo Co Ltd | Sulfonic acid group-containing polyarylene ether compound, application thereof, and method for producing the same |
| US7901817B2 (en) * | 2006-02-14 | 2011-03-08 | Ini Power Systems, Inc. | System for flexible in situ control of water in fuel cells |
| US20080004443A1 (en) * | 2006-07-03 | 2008-01-03 | General Electric Company | Sulfonated polyaryletherketone-block-polyethersulfone copolymers |
| US8158300B2 (en) * | 2006-09-19 | 2012-04-17 | Ini Power Systems, Inc. | Permselective composite membrane for electrochemical cells |
| US8551667B2 (en) * | 2007-04-17 | 2013-10-08 | Ini Power Systems, Inc. | Hydrogel barrier for fuel cells |
| US20090035644A1 (en) * | 2007-07-31 | 2009-02-05 | Markoski Larry J | Microfluidic Fuel Cell Electrode System |
| JP5412718B2 (en) * | 2007-09-05 | 2014-02-12 | 東レ株式会社 | POLYMER ELECTROLYTE MOLDED MANUFACTURING METHOD, POLYMER ELECTROLYTE MATERIAL, POLYMER ELECTROLYTE COMPONENT, MEMBRANE ELECTRODE COMPOSITE AND POLYMER ELECTROLYTE FUEL CELL |
| US8362195B2 (en) * | 2007-10-26 | 2013-01-29 | Lalgudi Ramanathan S | Ionically conductive polymer for use in electrochemical devices |
| JP5315878B2 (en) * | 2008-01-22 | 2013-10-16 | 東レ株式会社 | Polymer electrolyte material and polymer electrolyte fuel cell using the same |
| KR101063215B1 (en) * | 2008-02-20 | 2011-09-07 | 한국과학기술원 | Reinforced composite membrane for polymer electrolyte fuel cell |
| US8268195B2 (en) * | 2008-09-29 | 2012-09-18 | Air Products And Chemicals, Inc. | Electrically conductive films formed from dispersions comprising polythiophenes and ether containing polymers |
| US8163429B2 (en) * | 2009-02-05 | 2012-04-24 | Ini Power Systems, Inc. | High efficiency fuel cell system |
| US8177884B2 (en) * | 2009-05-20 | 2012-05-15 | United Technologies Corporation | Fuel deoxygenator with porous support plate |
| JP5181004B2 (en) * | 2010-08-27 | 2013-04-10 | Jsr株式会社 | Polyarylene block copolymer having sulfonic acid group and use thereof |
| US8783304B2 (en) | 2010-12-03 | 2014-07-22 | Ini Power Systems, Inc. | Liquid containers and apparatus for use with power producing devices |
| US9065095B2 (en) | 2011-01-05 | 2015-06-23 | Ini Power Systems, Inc. | Method and apparatus for enhancing power density of direct liquid fuel cells |
| CN104220152A (en) | 2012-03-15 | 2014-12-17 | 科腾聚合物美国有限责任公司 | Blends of sulfonated block copolymers and particulate carbon and membranes, films and coatings comprising them |
| CN103709379B (en) * | 2013-10-25 | 2015-12-02 | 武汉工程大学 | aromatic sulfonated polyketone and preparation method thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020091225A1 (en) * | 2000-09-20 | 2002-07-11 | Mcgrath James E. | Ion-conducting sulfonated polymeric materials |
Family Cites Families (103)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3134697A (en) | 1959-11-03 | 1964-05-26 | Gen Electric | Fuel cell |
| US3113049A (en) | 1961-01-03 | 1963-12-03 | Exxon Research Engineering Co | Direct production of electrical energy from liquid fuels |
| BE617375A (en) | 1961-05-08 | 1900-01-01 | ||
| US3301893A (en) | 1963-08-05 | 1967-01-31 | Du Pont | Fluorocarbon ethers containing sulfonyl groups |
| US3282875A (en) | 1964-07-22 | 1966-11-01 | Du Pont | Fluorocarbon vinyl ether polymers |
| US3692569A (en) | 1970-02-12 | 1972-09-19 | Du Pont | Surface-activated fluorocarbon objects |
| US3684747A (en) | 1970-04-22 | 1972-08-15 | Du Pont | Method for increasing the liquid absorptive capacity of linear fluorocarbon sulfonic acid polymer |
| US4036714A (en) | 1972-10-19 | 1977-07-19 | E. I. Du Pont De Nemours And Company, Inc. | Electrolytic cells and processes |
| US4303551A (en) | 1976-02-23 | 1981-12-01 | Varen Technology | Supported catalyst |
| US4038213A (en) | 1976-03-04 | 1977-07-26 | Shell Oil Company | Hydrocarbon conversion process using a supported perfluorinated polymer catalyst |
| US4176215A (en) | 1978-03-13 | 1979-11-27 | E. I. Du Pont De Nemours And Company | Ion-exchange structures of copolymer blends useful in electrolytic cells |
| JPS54154048A (en) | 1978-05-26 | 1979-12-04 | Hitachi Ltd | Disolving fuel battery |
| JPS551818A (en) | 1978-06-19 | 1980-01-09 | Hitachi Ltd | Electrode catalyst |
| EP0041780A1 (en) | 1980-06-10 | 1981-12-16 | Imperial Chemical Industries Plc | Sulphonated polyaryletherketones |
| US4417969A (en) | 1980-06-11 | 1983-11-29 | The Dow Chemical Co. | Sulfonic acid electrolytic cell membranes |
| JPS5768142A (en) | 1980-10-14 | 1982-04-26 | Hitachi Ltd | Electrode catalyst for fuel cell and its production |
| US4453991A (en) | 1981-05-01 | 1984-06-12 | E. I. Du Pont De Nemours And Company | Process for making articles coated with a liquid composition of perfluorinated ion exchange resin |
| US4390603A (en) | 1981-06-30 | 1983-06-28 | Hitachi, Ltd. | Methanol fuel cell |
| US4612261A (en) | 1981-08-21 | 1986-09-16 | Hitachi, Ltd. | Fuel cell battery using acidic electrolyte |
| JPS58131667A (en) | 1982-01-29 | 1983-08-05 | Hitachi Ltd | Fuel cell |
| JPS58165266A (en) | 1982-03-26 | 1983-09-30 | Hitachi Ltd | Fuel cell |
| JPS5923473A (en) | 1982-07-30 | 1984-02-06 | Hitachi Ltd | Fuel cell, electrolyte structure for fuel cell and electrolyte composite for fuel cell |
| DE3402471A1 (en) * | 1984-01-25 | 1985-08-01 | Forschungsinstitut Berghof Gmbh, 7412 Eningen | Cation exchanger membrane, and a process for the production thereof |
| US4673624A (en) | 1984-02-08 | 1987-06-16 | Hockaday Robert G | Fuel cell |
| US4598137A (en) * | 1984-11-30 | 1986-07-01 | Canadian Patents And Development Limited | Polyarylene polyethersulfone ionomers |
| DE3602090A1 (en) | 1986-01-24 | 1987-07-30 | Basf Ag | METHOD FOR PRODUCING AROMATIC BLOCK COPOLYETHERS |
| US4755272A (en) | 1986-05-02 | 1988-07-05 | The Dow Chemical Company | Bipolar electrochemical cell having novel means for electrically connecting anode and cathode of adjacent cell units |
| DE3618840A1 (en) | 1986-06-04 | 1987-12-10 | Basf Ag | METHANOL / AIR FUEL CELLS |
| US4855193A (en) | 1986-06-20 | 1989-08-08 | United Technologies Corporation | Bipolar fuel cell |
| US4797190A (en) | 1986-10-06 | 1989-01-10 | T And G. Corporation | Ionic semiconductor materials and applications thereof |
| DE3640209A1 (en) | 1986-11-25 | 1988-06-01 | Basf Ag | METHANOL / AIR FUEL CELL BATTERIES WITH POLYMERIC ELECTROLYTE HIGH ENERGY AND PERFORMANCE DENSITY AND TUBULAR ARRANGEMENT |
| US4876115A (en) | 1987-01-30 | 1989-10-24 | United States Department Of Energy | Electrode assembly for use in a solid polymer electrolyte fuel cell |
| US4865925A (en) | 1987-12-14 | 1989-09-12 | Hughes Aircraft Company | Gas permeable electrode for electrochemical system |
| ES2058427T3 (en) | 1988-10-01 | 1994-11-01 | Bayer Ag | AROMATIC POLYETERCETONES. |
| US5102751A (en) | 1990-02-07 | 1992-04-07 | Sri International | Plasticizers useful for enhancing ionic conductivity of solid polymer electrolytes |
| US5061581A (en) | 1990-02-07 | 1991-10-29 | Sri International | Novel solid polymer electrolytes |
| US5480735A (en) | 1990-06-25 | 1996-01-02 | International Fuel Cells Corporation | High current alkaline fuel cell electrodes |
| JPH04162365A (en) | 1990-10-25 | 1992-06-05 | Tanaka Kikinzoku Kogyo Kk | Method for preparing electrode of fuel cell |
| JP3107229B2 (en) | 1991-02-14 | 2000-11-06 | 田中貴金属工業株式会社 | Diaphragm humidification structure of solid polymer electrolyte fuel cell and electrolytic cell |
| US5234777A (en) | 1991-02-19 | 1993-08-10 | The Regents Of The University Of California | Membrane catalyst layer for fuel cells |
| US5211984A (en) | 1991-02-19 | 1993-05-18 | The Regents Of The University Of California | Membrane catalyst layer for fuel cells |
| US5252410A (en) | 1991-09-13 | 1993-10-12 | Ballard Power Systems Inc. | Lightweight fuel cell membrane electrode assembly with integral reactant flow passages |
| JPH05144444A (en) | 1991-11-25 | 1993-06-11 | Toshiba Corp | Fuel cell and electrode manufacturing method |
| TW256843B (en) | 1992-06-11 | 1995-09-11 | Hoechst Ag | |
| EP0574791B1 (en) | 1992-06-13 | 1999-12-22 | Aventis Research & Technologies GmbH & Co. KG | Polymer electrolyte membrane and process for its manufacture |
| JP3271801B2 (en) | 1992-09-22 | 2002-04-08 | 田中貴金属工業株式会社 | Polymer solid electrolyte fuel cell, humidifying method of the fuel cell, and manufacturing method |
| DE4241150C1 (en) | 1992-12-07 | 1994-06-01 | Fraunhofer Ges Forschung | Electrode membrane composite, process for its production and its use |
| EP0604882B1 (en) | 1992-12-30 | 1998-08-26 | Hoechst Aktiengesellschaft | Semipermeable membranes made of homogeneous mixable polymeric alloys |
| US5312876A (en) | 1993-03-12 | 1994-05-17 | The United States Of America As Represented By The Secretary Of The Air Force | Water soluble rigid-rod polymer |
| US5312895A (en) | 1993-03-12 | 1994-05-17 | The United States Of America As Represented By The Secretary Of The Air Force | Benzobisazole copolymer system soluble in aprotic solvents |
| US5403675A (en) | 1993-04-09 | 1995-04-04 | Maxdem, Incorporated | Sulfonated polymers for solid polymer electrolytes |
| US5330860A (en) | 1993-04-26 | 1994-07-19 | E. I. Du Pont De Nemours And Company | Membrane and electrode structure |
| US5635039A (en) | 1993-07-13 | 1997-06-03 | Lynntech, Inc. | Membrane with internal passages to permit fluid flow and an electrochemical cell containing the same |
| US5367051A (en) | 1993-07-22 | 1994-11-22 | Sri International | Amine-containing polymerizable monomers and polymers functionalized with fullerenes to provide polymers with high temperature stability |
| US5773480A (en) | 1993-09-21 | 1998-06-30 | Ballard Power Systems Inc. | Trifluorostyrene and substituted trifluorostyrene copolymeric compositions and ion-exchange membranes formed therefrom |
| US5834523A (en) | 1993-09-21 | 1998-11-10 | Ballard Power Systems, Inc. | Substituted α,β,β-trifluorostyrene-based composite membranes |
| US5773162A (en) | 1993-10-12 | 1998-06-30 | California Institute Of Technology | Direct methanol feed fuel cell and system |
| US5599638A (en) | 1993-10-12 | 1997-02-04 | California Institute Of Technology | Aqueous liquid feed organic fuel cell using solid polymer electrolyte membrane |
| US5470448A (en) | 1994-01-28 | 1995-11-28 | United Technologies Corporation | High performance electrolytic cell electrode/membrane structures and a process for preparing such electrode structures |
| US5973025A (en) | 1994-04-12 | 1999-10-26 | Sri International | Aqueous ink compositions containing a binder of a neutralized acidic resin |
| US5624965A (en) | 1994-04-26 | 1997-04-29 | The Graver Company | Arylenedialkylidene polymers for high performance ion exchange membranes |
| DE4415678A1 (en) | 1994-05-04 | 1995-11-09 | Hoechst Ag | Electrochemical cell |
| US5468574A (en) | 1994-05-23 | 1995-11-21 | Dais Corporation | Fuel cell incorporating novel ion-conducting membrane |
| US5540981A (en) | 1994-05-31 | 1996-07-30 | Rohm And Haas Company | Inorganic-containing composites |
| US5488087A (en) | 1994-06-16 | 1996-01-30 | Dow Corning Corporation | Preparation of polydiorganosiloxane and sulfonated polyolefin blends |
| DE4422158A1 (en) | 1994-06-24 | 1996-01-04 | Hoechst Ag | Homogeneous polymer alloys based on sulfonated, aromatic polyether ketones |
| US5482568A (en) | 1994-06-28 | 1996-01-09 | Hockaday; Robert G. | Micro mirror photovoltaic cells |
| US5547911A (en) | 1994-10-11 | 1996-08-20 | E. I. Du Pont De Nemours And Company | Process of imprinting catalytically active particles on membrane |
| US5523177A (en) | 1994-10-12 | 1996-06-04 | Giner, Inc. | Membrane-electrode assembly for a direct methanol fuel cell |
| WO1996013073A1 (en) | 1994-10-20 | 1996-05-02 | Hoechst Aktiengesellschaft | Metallized cation exchanger membrane |
| US5525436A (en) | 1994-11-01 | 1996-06-11 | Case Western Reserve University | Proton conducting polymers used as membranes |
| US5795668A (en) | 1994-11-10 | 1998-08-18 | E. I. Du Pont De Nemours And Company | Fuel cell incorporating a reinforced membrane |
| US5548055A (en) | 1995-01-13 | 1996-08-20 | Sri International | Single-ion conducting solid polymer electrolytes |
| WO1996021639A1 (en) | 1995-01-13 | 1996-07-18 | Sri International | Organic liquid electrolyte plasticizer |
| US5804325A (en) | 1995-01-13 | 1998-09-08 | Westfield Trading Corporation | Non poisoning fuel cell and method |
| US5573866A (en) | 1995-05-08 | 1996-11-12 | International Fuel Cells Corp. | Direct methanol oxidation polymer electrolyte membrane power system |
| JP3203150B2 (en) | 1995-05-18 | 2001-08-27 | 三洋電機株式会社 | Polymer electrolyte fuel cell and polymer electrolyte fuel cell system |
| US5702838A (en) | 1995-08-18 | 1997-12-30 | Matsushita Electric Industrial Co., Ltd. | Fuel cell device equipped with catalyst material for removing carbon monoxide and method for removing carbon monoxide |
| US5631099A (en) | 1995-09-21 | 1997-05-20 | Hockaday; Robert G. | Surface replica fuel cell |
| US5702755A (en) | 1995-11-06 | 1997-12-30 | The Dow Chemical Company | Process for preparing a membrane/electrode assembly |
| US5795496A (en) | 1995-11-22 | 1998-08-18 | California Institute Of Technology | Polymer material for electrolytic membranes in fuel cells |
| US5672439A (en) | 1995-12-18 | 1997-09-30 | Ballard Power Systems, Inc. | Method and apparatus for reducing reactant crossover in an electrochemical fuel cell |
| US5573867A (en) | 1996-01-31 | 1996-11-12 | Westinghouse Electric Corporation | Purge gas protected transportable pressurized fuel cell modules and their operation in a power plant |
| US5882810A (en) | 1996-03-08 | 1999-03-16 | The Dow Chemicalcompany | Active layer for membrane electrode assembly |
| US5945231A (en) | 1996-03-26 | 1999-08-31 | California Institute Of Technology | Direct liquid-feed fuel cell with membrane electrolyte and manufacturing thereof |
| US5716727A (en) | 1996-04-01 | 1998-02-10 | Case Western Reserve University | Proton conducting polymers prepared by direct acid casting |
| JP3724064B2 (en) | 1996-06-28 | 2005-12-07 | 住友化学株式会社 | Polymer electrolyte for fuel cell and fuel cell |
| US5783325A (en) | 1996-08-27 | 1998-07-21 | The Research Foundation Of State Of New York | Gas diffusion electrodes based on poly(vinylidene fluoride) carbon blends |
| US5759712A (en) | 1997-01-06 | 1998-06-02 | Hockaday; Robert G. | Surface replica fuel cell for micro fuel cell electrical power pack |
| US5952119A (en) | 1997-02-24 | 1999-09-14 | Regents Of The University Of California | Fuel cell membrane humidification |
| DE19754305A1 (en) * | 1997-12-08 | 1999-06-10 | Hoechst Ag | Process for producing a membrane for operating fuel cells and electrolysers |
| US5958616A (en) | 1998-02-06 | 1999-09-28 | Lynntech, Inc. | Membrane and electrode structure for methanol fuel cell |
| US5992008A (en) | 1998-02-10 | 1999-11-30 | California Institute Of Technology | Direct methanol feed fuel cell with reduced catalyst loading |
| DE19813613A1 (en) * | 1998-03-27 | 1999-09-30 | Jochen Kerres | Modified polymer and modified polymer membrane |
| US6024848A (en) | 1998-04-15 | 2000-02-15 | International Fuel Cells, Corporation | Electrochemical cell with a porous support plate |
| DE19817376A1 (en) * | 1998-04-18 | 1999-10-21 | Univ Stuttgart Lehrstuhl Und I | Ion exchange membranes and polymer blends, useful for fuel cells, gas separation, pervaporation and reverse osmosis |
| DE19909028A1 (en) * | 1999-03-02 | 2000-09-07 | Aventis Res & Tech Gmbh & Co | Sulfonated aromatic polymers, membrane containing these polymers, process for their preparation and their use |
| AU4475000A (en) * | 1999-04-30 | 2000-11-17 | E.I. Du Pont De Nemours And Company | Electrochemical uses of amorphous fluoropolymers |
| DE10010001A1 (en) * | 2000-03-02 | 2001-09-06 | Celanese Ventures Gmbh | Membranes useful in fuel cells comprise a blend of sulfonated aryl polymer, aminated or nitrated polyether(ether)sulfone and plasticizer |
| CA2421627A1 (en) * | 2000-09-20 | 2002-03-28 | Virginia Tech Intellectual Properties, Inc. | Ion-conducting sulfonated polymeric materials |
| US20020093006A1 (en) * | 2001-01-16 | 2002-07-18 | Vardeny Z. Valy | Performance of organic light-emitting devices using spin-dependent processes |
| US20020161174A1 (en) * | 2001-02-15 | 2002-10-31 | Sumitomo Chemical Company, Limited | Aromatic polymer phosphonic acid derivative and process for production the same |
| US20030013817A1 (en) * | 2001-06-26 | 2003-01-16 | Kelly Lu | High temperature ionic polymers and membranes made therefrom |
-
2003
- 2003-05-13 DE DE60333367T patent/DE60333367D1/en not_active Expired - Lifetime
- 2003-05-13 CN CNB038163497A patent/CN100509875C/en not_active Expired - Fee Related
- 2003-05-13 AU AU2003237849A patent/AU2003237849B2/en not_active Ceased
- 2003-05-13 JP JP2004503520A patent/JP2006506472A/en active Pending
- 2003-05-13 WO PCT/US2003/015178 patent/WO2003095509A1/en active Search and Examination
- 2003-05-13 CA CA002485727A patent/CA2485727A1/en not_active Abandoned
- 2003-05-13 EP EP03736609A patent/EP1517929B1/en not_active Expired - Lifetime
- 2003-05-13 US US10/438,186 patent/US7202001B2/en not_active Expired - Fee Related
- 2003-05-13 AT AT03736609T patent/ATE474005T1/en not_active IP Right Cessation
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020091225A1 (en) * | 2000-09-20 | 2002-07-11 | Mcgrath James E. | Ion-conducting sulfonated polymeric materials |
Non-Patent Citations (5)
| Title |
|---|
| GAN ET AL.: "Synthesis and characterization of poly(ether ketone ketone)(PEEK)/sodium sulfonated poly(arylene ethylene ketone)(S-PAEK) block copolymers", POLYMER INTERNATIONAL, vol. 50, 2001, pages 812 - 816, XP002969654 * |
| LIU ET AL.: "Novel sodium sulfonate-functionalized poly(ether ether ketone)s derived from 4,4'-thiodiphenol", POLYMER, vol. 42, 2001, pages 3293 - 3296, XP004227295 * |
| LIU ET AL.: "Synthesis of poly(ether ether ketone)s with high content of sodium sulfonate groups as gas dehumidification membrane materials", MACROMOL. RAPID COMMUN., vol. 222, 2001, pages 579 - 582, XP002969655 * |
| WANG ET AL.: "Sodium sulfonate-functionalized poly(ether ether ketone)s", MACROMOL. CHEM. PHYS., vol. 199, 1998, pages 1421 - 1426, XP000777518 * |
| XIAO ET AL.: "Synthesis and characterization of novel sulfonated poly(arylene ether ketone)s derived from 4,4'-sulfonyldiphenol", POLYMER BULLETIN, vol. 48, 2002, pages 309 - 315, XP002969656 * |
Cited By (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7572535B2 (en) | 2002-05-13 | 2009-08-11 | Polyfuel, Inc. | Ion conductive copolymers containing one or more hydrophobic oligomers |
| EP1612232A1 (en) * | 2003-04-07 | 2006-01-04 | Mitsui Chemicals, Inc. | Crosslinkable ionically conducting resin, and ionically conducting polymer membranes, binders and fuel cells, made by using the resin |
| EP1612232A4 (en) * | 2003-04-07 | 2006-05-17 | Mitsui Chemicals Inc | Crosslinkable ionically conducting resin, and ionically conducting polymer membranes, binders and fuel cells, made by using the resin |
| US8455141B2 (en) | 2003-06-25 | 2013-06-04 | Toray Industries, Inc. | Polymer electrolyte as well as polymer electrolyte membrane, membrane electrode assembly and polymer electrolyte fuel cell using the same |
| EP1674498A4 (en) * | 2003-10-17 | 2007-07-04 | Sumitomo Chemical Co | Block copolymer and use thereof |
| EP1674498A1 (en) * | 2003-10-17 | 2006-06-28 | Sumitomo Chemical Company, Limited | Block copolymer and use thereof |
| US7803884B2 (en) | 2003-10-17 | 2010-09-28 | Sumitomo Chemical Company, Limited | Block copolymers and use thereof |
| US7507771B2 (en) | 2003-11-13 | 2009-03-24 | Polyfuel, Inc. | Ion conductive copolymers containing one or more hydrophobic oligomers |
| JP2005197235A (en) * | 2003-12-09 | 2005-07-21 | Jsr Corp | Proton conduction film |
| EP1693406A4 (en) * | 2003-12-09 | 2012-09-05 | Jsr Corp | Proton conducting membrane |
| EP1693406A1 (en) * | 2003-12-09 | 2006-08-23 | JSR Corporation | Proton conducting membrane |
| US8039165B2 (en) | 2003-12-09 | 2011-10-18 | Jsr Corporation | Proton conductive membrane comprising a copolymer |
| US7955758B2 (en) | 2004-01-22 | 2011-06-07 | GM Global Technology Operations LLC | Membrane electrode assembly prepared by direct spray of catalyst to membrane |
| JP2007524973A (en) * | 2004-01-22 | 2007-08-30 | ゼネラル・モーターズ・コーポレーション | Membrane electrode assembly prepared by spraying catalyst directly onto membrane |
| JP4833860B2 (en) * | 2004-01-22 | 2011-12-07 | ゼネラル・モーターズ・コーポレーション | Membrane electrode assembly prepared by spraying catalyst directly onto membrane |
| JP4729857B2 (en) * | 2004-02-27 | 2011-07-20 | 東洋紡績株式会社 | Ion exchange membrane |
| JP2005276441A (en) * | 2004-02-27 | 2005-10-06 | Toyobo Co Ltd | Ion exchange membrane |
| JP2007528930A (en) * | 2004-03-12 | 2007-10-18 | ポリフューエル・インコーポレイテッド | Ion conductive copolymers containing one or more ion conductive oligomers |
| WO2005090439A3 (en) * | 2004-03-12 | 2005-11-24 | Polyfuel Inc | Ion-conductive copolymers containing one or more ion-conducting oligomers |
| US7504461B2 (en) | 2004-03-12 | 2009-03-17 | Polyfuel, Inc. | Ion-conductive copolymers containing one or more ion-conducting oligomers |
| WO2005090439A2 (en) * | 2004-03-12 | 2005-09-29 | Polyfuel, Inc. | Ion-conductive copolymers containing one or more ion-conducting oligomers |
| JP2005268145A (en) * | 2004-03-22 | 2005-09-29 | Toyobo Co Ltd | Electrolyte film and its manufacturing method |
| JP2006070126A (en) * | 2004-09-01 | 2006-03-16 | Toray Ind Inc | Heat-resistant resin and resin composition and molding each using the same |
| JP5181474B2 (en) * | 2004-09-03 | 2013-04-10 | 東レ株式会社 | Polymer electrolyte material, polymer electrolyte component, membrane electrode composite, and polymer electrolyte fuel cell |
| US8216727B2 (en) | 2004-11-10 | 2012-07-10 | Toyo Boseki Kabushiki Kaisha | Aromatic hydrocarbon based proton exchange membrane and direct methanol fuel cell using same |
| JP2006202598A (en) * | 2005-01-20 | 2006-08-03 | Toray Ind Inc | Fuel cell electrode and fuel cell |
| KR101341274B1 (en) * | 2005-02-15 | 2013-12-12 | 도레이 카부시키가이샤 | Process for producing polymer electrolyte molded product, polymer electrolyte material, polymer electrolyte membrane, and solid polymer electrolyte fuel cell |
| US8986903B2 (en) | 2005-02-15 | 2015-03-24 | Toray Industries, Inc. | Method for producing polymer electrolyte molded article, polymer electrolyte material, polymer electrolyte membrane, and polymer electrolyte fuel cell |
| US11108071B2 (en) | 2005-02-15 | 2021-08-31 | Toray Industries, Inc. | Method for producing polymer electrolyte molded article, polymer electrolyte material, polymer electrolyte membrane, and polymer electrolyte fuel cell |
| WO2006087995A1 (en) * | 2005-02-15 | 2006-08-24 | Toray Industries, Inc. | Process for producing polymer electrolyte molded product, polymer electrolyte material, polymer electrolyte membrane, and solid polymer electrolyte fuel cell |
| EP1886373A4 (en) * | 2005-06-01 | 2009-04-01 | Polyfuel Inc | Polymer blend comprising ion-conducting copolymer and non-ionic polymer |
| EP1886373A2 (en) * | 2005-06-01 | 2008-02-13 | Polyfuel, Inc. | Polymer blend comprising ion-conducting copolymer and non-ionic polymer |
| CN101218281B (en) * | 2005-06-09 | 2012-03-28 | 东洋纺织株式会社 | Sulfonic acid group-containing polymer, method for producing same, resin composition containing such sulfonic acid group-containing polymer, polymer electrolyte membrane, polymer electrolyte membrane/electrode adapter, fuel cell |
| US8445141B2 (en) | 2005-06-09 | 2013-05-21 | Toyo Boseki Kabushiki Kaisha | Sulfonic acid group-containing polymer, method for producing the same, resin composition containing such sulfonic acid group-containing polymer, polymer electrolyte membrane, polymer electrolyte membrane/electrode assembly, and fuel cell |
| WO2006132207A1 (en) * | 2005-06-09 | 2006-12-14 | Toyo Boseki Kabushiki Kaisha | Sulfonic acid group-containing polymer, method for producing same, resin composition containing such sulfonic acid group-containing polymer, polymer electrolyte membrane, polymer electrolyte membrane/electrode assembly, and fuel cell |
| JP2012256601A (en) * | 2005-07-29 | 2012-12-27 | Toray Ind Inc | Polymer electrolyte material, polyelectrolyte component, membrane electrode assembly, and polymer electrolyte fuel cell |
| US8178590B2 (en) | 2006-05-31 | 2012-05-15 | Sumitomo Chemical Company, Limited | Block copolymer and use thereof |
| EP1862489A1 (en) * | 2006-05-31 | 2007-12-05 | Sumitomo Chemical Company, Limited | Block Copolymer and Use Thereof |
| WO2009079625A3 (en) * | 2007-12-18 | 2009-08-27 | Polyfuel, Inc. | Ion conducting copolymers with elastomeric and polyarylene segments |
| WO2009079625A2 (en) * | 2007-12-18 | 2009-06-25 | Polyfuel, Inc. | Ion conducting copolymers with elastomeric and polyarylene segments |
| WO2014117110A1 (en) | 2013-01-28 | 2014-07-31 | Greene, Tweed Technologies, Inc. | Anti-extrusion compositions for sealing and wear components |
| EP2948423A4 (en) * | 2013-01-28 | 2016-08-17 | Delsper LP | Anti-extrusion compositions for sealing and wear components |
| US9475938B2 (en) | 2013-01-28 | 2016-10-25 | Delsper, Lp | Anti-extrusion compositions for sealing and wear components |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1517929B1 (en) | 2010-07-14 |
| CN100509875C (en) | 2009-07-08 |
| AU2003237849B2 (en) | 2009-07-02 |
| US20040039148A1 (en) | 2004-02-26 |
| CN1668656A (en) | 2005-09-14 |
| DE60333367D1 (en) | 2010-08-26 |
| AU2003237849A1 (en) | 2003-11-11 |
| EP1517929A4 (en) | 2005-07-20 |
| ATE474005T1 (en) | 2010-07-15 |
| EP1517929A1 (en) | 2005-03-30 |
| CA2485727A1 (en) | 2003-11-20 |
| US7202001B2 (en) | 2007-04-10 |
| JP2006506472A (en) | 2006-02-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2003237849B2 (en) | Sulfonated copolymer | |
| US20060135657A1 (en) | Ion conductive block copolymers | |
| US7504461B2 (en) | Ion-conductive copolymers containing one or more ion-conducting oligomers | |
| WO2005050671A2 (en) | Ion conductive random copolymers | |
| CA2607966A1 (en) | Ion-conducting polymers containing pendant ion conducting groups | |
| US20060280988A1 (en) | Cross-linked ion-conductive copolymer | |
| US7354679B2 (en) | Ion conductive random copolymers | |
| KR100977234B1 (en) | Sulfonated copolymer | |
| US20060280990A1 (en) | Polymer blend comprising ion-conducting copolymer and non-ionic polymer | |
| US7572535B2 (en) | Ion conductive copolymers containing one or more hydrophobic oligomers | |
| MXPA06005344A (en) | Ion conductive random copolymers | |
| MXPA06005345A (en) | Ion conductive copolymers containing one or more hydrophobic oligomers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SC SD SE SG SK SL TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003736609 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004503520 Country of ref document: JP Ref document number: 2485727 Country of ref document: CA Ref document number: 1020047018336 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003237849 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038163497 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020047018336 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003736609 Country of ref document: EP |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) |