WO2000020535A1 - Process for producing middle distillates and middle distillates produced by that process - Google Patents
Process for producing middle distillates and middle distillates produced by that process Download PDFInfo
- Publication number
- WO2000020535A1 WO2000020535A1 PCT/ZA1999/000096 ZA9900096W WO0020535A1 WO 2000020535 A1 WO2000020535 A1 WO 2000020535A1 ZA 9900096 W ZA9900096 W ZA 9900096W WO 0020535 A1 WO0020535 A1 WO 0020535A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- middle distillate
- fraction
- isoparaffins
- synthetic middle
- synthetic
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/04—Liquid carbonaceous fuels essentially based on blends of hydrocarbons
- C10L1/08—Liquid carbonaceous fuels essentially based on blends of hydrocarbons for compression ignition
Definitions
- This invention relates to middle distillates having good cold flow properties, such as the Cold Filter Plugging Point (CFPP) measured in accordance with the IP method 309, and a high Cetane number, as well as to a process for production of such distillates. More particularly, this invention relates to middle distillates produced from a mainly paraffinic synthetic crude which is produced by the reaction of CO and H 2 , typically by the Fischer-Tropsch (FT) process.
- CFPP Cold Filter Plugging Point
- Waxy products of a FT hydrocarbon synthesis process particularly the products of a cobalt and/or iron based catalytic process, contain a high proportion of normal paraffins.
- Primary FT products provide notoriously poor cold flow properties, making such products difficult to use where cold flow properties are vital, e.g. diesel fuels, lube oil bases and jet fuel.
- cold flow properties of a middle distillate such as jet fuel
- Hydrocracking produces smaller amounts of gases and light products, which reduce the yield of valuable distillates.
- the middle distillate fuel described in this invention is produced from a highly paraffinic synthetic crude (syncrude) obtained from synthesis gas (syngas) through a reaction like the FT reaction.
- the FT primary products cover a broad range of hydrocarbons from methane to species with molecular masses above 1400; including mainly paraffinic hydrocarbons and much smaller quantities of other species such as olefins, and oxygenates.
- a process for the production of a middle distillate or distillate blend such as diesel, having a high Cetane number as well as good cold flow properties.
- the synthetic middle distillate cut may comprise more than 50% isoparaffins. wherein the isoparaffins are predominantly methyl and/or ethyl and/or propyl branched.
- the gradient of an isoparaffins to n-paraffins mass ratio profile of the synthetic middle distillate cut may increase from about 1: 1 for C s to 8.54: 1 for C 1 5 and decrease again to about 3: 1 for C ]8 .
- a fraction of the synthetic middle distillate cut in the Cjo to Cj 8 carbon number range has a higher ratio of isoparaffins to n-paraffins than a Cs to Co fraction of the synthetic middle distillate cut.
- the isoparaffins to n-paraffins mass ratio of the do to C 18 fraction may be between 1: 1 and 9: 1.
- the isoparaffins to n-paraffins mass ratio may be 8.54:1 for a C 1 5 fraction of the synthetic middle distillate cut.
- a Cjo to C 2 fraction of the middle distillate cut may have a narrow mass ratio range of isoparaffins to n-paraffins of between 3.3: 1 and 5: 1, generally between 4: 1 and 4.9: 1.
- the mass ratio of isoparaffins to n-paraffins may be adjusted by controlling the blend ratio of hydrocracked to straight run components of the synthetic middle distillate cut.
- the isoparaffins to n-paraffins mass ratio of the do to 8 fraction having 30% straight run component may be between 1 : 1 and 2:5: 1.
- the isoparaffins to n-paraffins mass ratio of the do to d s fraction having 20% straight run component mav be between 1.5: 1 and 3:5: 1.
- the isoparaffins to n-paraffins mass ratio of the Cio to Cj S fraction having 10% straight run component may be between 2 3 1 and 4 3 1
- the isoparaffins to n-paraffins mass ratio of the do to Cj g fraction having substantially only a hydrocracked component may be between 4 1 and 9 1
- At least some of the isoparaffins may be methyl branched At least some of the isoparaffins may be dimethyl branched At least 30% (mass) of the isoparaffins are typically mono-methyl branched
- isoparaffins may however be ethyl branched
- a synthetic middle distillate cut having a Cetane number above 70 and a CFPP. in accordance with IP 309. of below -20°C. said distillate having an isoparaffinic content substantially as described above
- the synthetic middle distillate cut is a FT product
- the invention extends to a diesel fuel composition including from 10% to 100% of a middle distillate cut as desc ⁇ bed above
- the diesel fuel composition may include from 0 to 90% of one or more other diesel fuel
- the diesel fuel composition may include at least 20% of the middle distillate cut, the composition having a Cetane number greater than 47 and a CFPP, in accordance with IP 309, below -22°C
- the diesel fuel composition may include at least 30% of the middle distillate cut. the composition having a Cetane number greater than 50 and a CFPP, in accordance with IP 309, below -22°C
- the diesel fuel composition may mlcude at least 50% of the middle distillate cut, the composition having a Cetane number greater than 52 and a CFPP, in accordance with IP 309, below -25°C
- the diesel fuel composition may include at least 70% of the middle distillate cut, the composition having a Cetane number greater than 60 and a cold flow plug point, in accordance with IP 309, below -30°C
- the diesel fuel composition may further include from 0 to 10% additives
- the additives may include a lubricity improver
- the lubricity improver may comprise from 0 to 0 5% of the composition, typically from 0 00001% to 0 05% of the composition In some embodiments, the lubricity improver comp ⁇ ses from 0 008% to 0 02% of the composition
- the diesel fuel composition may include, as the other diesel, a crude oil derived diesel, such as US 2-D grade (low sulphur No 2-D grade for diesel fuel oil as specified in ASTM D 975-94) and/or CARB (California Air Resources Board 1993 specification) diesel fuel
- a process for producing a synthetic middle distillate having a Cetane number higher than 70 including (a) separating the products obtained from synthesis gas via the FT synthesis reaction into one or more heavier fraction and one or more lighter fraction, (b) catalytically processing the heavier fraction under conditions which yield mainly middle distillates,
- step (c) separatmg the middle distillate product of step (b) from a light product fraction and a heavier product fraction which are also produced in step (b), and
- step (d) blending the middle distillate fraction obtained in step (c) with at least a portion of the one or more lighter fraction of step (a), or products thereof
- step (b) may be a hydroprocessing step, for example, hydrocracking
- the process for producing a synthetic middle distillate may include one or more additional step of fractionating at least some of the one or more lighter fraction of step (a), or products thereof, p ⁇ or to step (d)
- the process for producing a synthetic middle distillate may mclude the additional step of hydrotreatmg at least some of the one or more light fraction of step (a), or products thereof, p ⁇ or to step (d)
- the one or more heavier fraction of step (a) may have a boiling point above about 270°C. however, it may be above 300°C
- the one or more lighter fraction may have a boiling point in the range C 5 to the boiling point of the heavier fraction, typically in the range 160°C to 270°C
- step (d) may boil in the range 100°C to 400°C
- the product of step (d) may boil in the range 160°C to 370°C
- the product of step (d) may be a diesel fuel
- the product of step (d) may have a CFPP below -20°C. typically below -30°C. and even below -
- the product of step (d) may be obtained by mixing the middle distillate fraction obtained in step (c) with at least a portion of the one or more lighter fraction of step (a), or products thereof, m a volume ratio of between 1 1 and 9 1, typically 2 1 and 6 1, and in one embodiment, in a volume ratio of 84 16
- the invention extends further to a process for the production of middle distillate fuels from FT pnmary products, comprising predominantly long chain linear paraffins
- the waxy product from the FT process is separated into at least two fractions, a heavier and at least one lighter fraction
- the lighter fraction may be subjected to mild catalytic hydrogenation to remove hetero-atomic compounds such as oxygen and to saturate olefins, thereby producing matenal useful as naphtha, solvents, diesel and/or blending components therefor
- the heavier fraction may be catalytically hydroprocessed without p ⁇ or hydrotreatmg to produce products with good cold flow characteristics
- This hydroprocessed heavier fraction could be blended with all or part of the hydrogenated and/or unhydrogenated light fraction to obtain, after fractionation, naphtha and a diesel fuel characterised by a high Cetane number
- the catalysts suitable for the hydroprocessmg steps are commercially available and can be selected towards an improved quality of the desired final product
- This invention desc ⁇ bes the conversion of primary FT products mto naphtha and middle distillates, for example, diesel having a high Cetane number in excess of 70, while also having good cold flow properties, as described above
- the FT process is used indust ⁇ ally to convert synthesis gas, derived from coal, natural gas, biomass or heavy oil streams, mto hydrocarbons ranging from methane to species with molecular masses above 1400
- Preferred reactors for the production of heavier hydrocarbons are slurry bed or tubular fixed bed reactors, while operating conditions are preferably in the range of 160°C - 280°C. in some cases 210- 260°C. and 18 - 50 Bar, in some cases 20-30 bar
- FT products can be converted into a range of final products, such as middle distillates, gasoline, solvents, lube oil bases, etc
- Such conversion which usually consists of a range of processes such as hydrocracking, hydrotreatment and distillation, can be termed a FT work-up process
- the FT work-up process of this invention uses a feed stream consisting of C 5 and higher hydrocarbons derived from a FT process This feed is separated mto at least two individual fractions, a heavier and at least one lighter fraction The cut point between the two fractions is preferably less than 300°C and typically around 270°C
- the >270°C fraction also referred to as wax, contains a considerable amount of hydrocarbon material, which boils higher than the normal diesel range. If we consider a typical diesel boiling range of 160- 370°C, it means that all material heavier than 370°C needs to be converted into lighter materials by means of a catalytic process often refe ⁇ ed to as hydroprocessing, for example, hydrocracking. Catalysts for this step are of the bifunctional type; i.e. they contain sites active for cracking and for hydrogenation. Catalytic metals active for hydrogenation include group VIII noble metals, such as platinum or palladium, or a sulphided Group VIII base metals, e.g.
- nickel, cobalt which may or may not include a sulphided Group NI metal, e.g. molybdenum.
- the support for the metals can be any refractory oxide, such as silica, alumina, titania, zirconia, vanadia and other Group III, IN, NA and VI oxides, alone or in combination with other refractory oxides.
- the support can partly or totally consist of zeolite.
- the preferred support is amorphous silica- alumina.
- Process conditions for hydrocracking can be varied over a wide range and are usually laboriously chosen after extensive experimentation to optimize the yield of middle distillates. In this regard, it is important to note that, as in many chemical reactions, there is a trade-off between conversion and selectivity. A very high conversion will result in a high yield of gases and low yield of distillate fuels. It is therefore important to painstakingly tune the process conditions in order to limit the conversion of >370°C hydrocarbons. Table 2 gives a list of the preferred conditions. Table 2: Process conditions for hydrocracking
- hydrotreating is a well-known industrial process, catalyzed by any catalyst having a hydrogenation function, e.g. Group VIII noble metal or sulphided base metal or Group VI metals, or combinations thereof.
- Preferred supports are alumina and silica.
- Table 3 gives typical operating conditions for the hydrotreating process.
- hydrotreated fraction may be fractionated into paraffinic materials useful as solvents
- the applicant has now surprisingly found that the hydrotreated fraction may be directly blended with the products obtained from hydrocracking the wax.
- hydroisomerise the material contained in the condensate stream
- isomerisation leads to the formation of branched isomers, which leads to Cetane ratings less than that of the co ⁇ esponding normal paraffins.
- Important parameters for a FT work-up process are maximization of product yield, product quality and cost. While the proposed process scheme is simple and therefore cost-effective, it produces High Performance Diesel, having a Cetane number >70, and naphtha in good yield. In fact, the process of this invention is able to produce a diesel of hitherto unmatched quality, which is characterized by a unique combination of both high Cetane number and excellent cold flow properties. This is believed to be related to a low degree of isomerisation in the 160-270°C fraction of the diesel and contrary to this, a high degree of isomerisation in the 270-370°C fraction of the diesel.
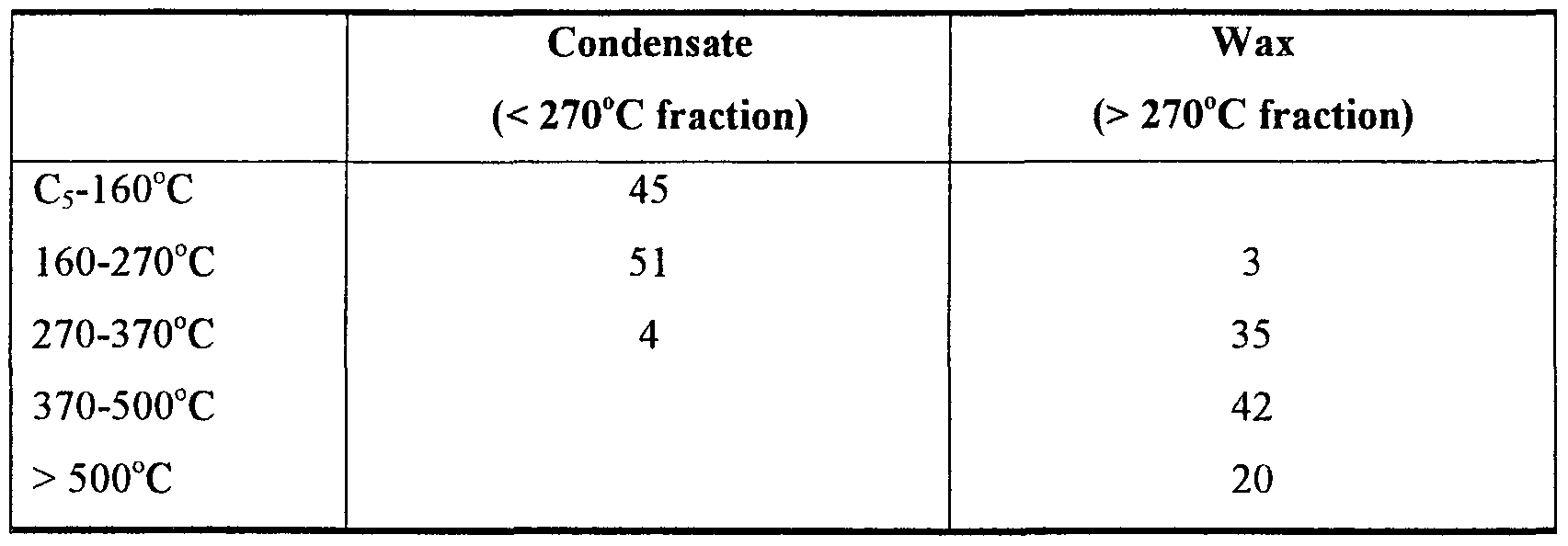
- the total amount of isomers in the light boiling range of the diesel (160-270°C fraction) and the heavier range of the diesel (270°C-370°C) are shown in the following table 4.
- the relatively high percentage of normal paraffins in the light boiling range contributes to the high Cetane number of the diesel fuel, without affecting the cold flow properties.
- branching is of utmost importance because the linear hydrocarbons in this range provide very poor cold flow properties and in some cases, may even crystallize. Therefore, the amount of iso-paraffins in this range is maximised during hydroprocessing under the process conditions described herein.
- the basic process is outlined m the attached Figure 1
- the synthesis gas (syngas), a mixture of Hydrogen and Carbon monoxide, enters the FT reactor 1 where the synthesis gas is converted to hydrocarbons by the FT reaction
- a lighter FT fraction is recovered in line 7, and may or may not pass through fractionator 2 and hydrotreater 3
- the product 9 from the hydrotreater may be separated in fractionator 4 or, alternatively, mixed ith hydrocracker products 16 sent to a common fractionator 6
- a waxy FT fraction is recovered in line 13 and sent to hydrocracker 5 If fractionation 2 is considered the bottoms cut 12 are be sent to hydrocracker 5
- the products 16, on their own or mixed with the lighter fraction 9a. are separated in fractionator 6
- a light product fraction, naphtha 19 is obtained from fractionator 6 or by blending equivalent fractions 10 and 17. This is a C 5 -160°C fraction useful as naphtha.
- a somewhat heavier cut, synthetic diesel 20 is obtainable in a similar way from fractionator 6 or by blending equivalent fractions 11 and 18. This cut is recovered as a 160-370°C fraction useful as diesel.
- the heavy unconverted material 21 from fractionator 6 is recycled to extinction to hydrocracker 5.
- the residue may be used for production of synthetic lube oil bases.
- a small amount of Cj-C gases are also separated in fractionator 6.
- a commercially available hydrocracking catalyst was used for hydrocracking of a non-hydrotreated FT hydrocarbon fraction with an initial boiling point of about 280°C.
- the active metals on the catalyst comprised cobalt and molybdenum, while the support was amorphous silica-alumina.
- Operating conditions were temperatures between 375 and 385°C, pressure of 70 bar and hydrogen flow rate of 1500 m 3 n /m 3 feed.
- the experiment was carried out in a pilot plant reactor. The conversion of >370°C material to lighter material ranged between 65 and 80%. Diesel component A is obtained after fractionation of the reactor products. The properties of this diesel component are given in table 1.
- the active metals on the catalyst comprised molybdenum and cobalt, while the support was alumina.
- the process conditions were temperatures around 250°C, pressure of 68 bar and hydrogen flow rate of 1070 mVm 3 feed.
- the test was carried in a commercial scale fixed bed reactor. Diesel components B and C were obtained after fractionation of respectively the reactor feed and reactor product. The properties of these diesel components are given in table 6. Table 6: Diesel Blending Components
- Blend I The diesel fraction obtained from hydrocracking a heavy FT material (component A) was blended with a hydrogenated lighter FT material (component B) in a volume ratio of 84: 16.
- the properties of the final blend, called Blend I, are given in table 7.
- Blend I may be used on its own. but also as a blending feedstock.
- Blend II The diesel fraction obtained from hydrocracking a heavy FT material (component A) was directly blended with a lighter non-hydrogenated FT material (component C) in a volume ratio of 84: 16.
- the properties of the final blend, called Blend II. are given in table 7 Similar to example 3, Blend II may be used on its own, but also as a blending feedstock. In addition to a high Cetane numbers, above 70, and excellent cold flow properties, with CFPP substantially better than -20°C. Blend II contains alcohols and smaller quantities of other oxygenates, the level of which depend on the blending ratio used to prepare the blend.
- the diesel Blend I of Example 3 was blended with US 2-D grade diesel having desired Cetane number and CFPP properties, as shown in Table 8 and charts 1 and 2 below, were obtained.
- Chart 2 Cetane Number of Applicants Diesel and Blends Thereof.
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU63001/99A AU765274B2 (en) | 1998-10-05 | 1999-09-17 | Process for producing middle distillates and middle distillates produced by that process |
| EP99950311A EP1129155A1 (en) | 1998-10-05 | 1999-09-17 | Process for producing middle distillates and middle distillates produced by that process |
| JP2000574637A JP3824490B2 (en) | 1998-10-05 | 1999-09-17 | Synthetic middle distillate fraction |
| US09/787,668 US7217852B1 (en) | 1998-10-05 | 1999-09-17 | Process for producing middle distillates and middle distillates produced by that process |
| US10/712,169 US7294253B2 (en) | 1998-10-05 | 2003-11-12 | Process for producing middle distillates |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ZA989038 | 1998-10-05 | ||
| ZA98/9038 | 1998-10-05 | ||
| US20976298A | 1998-12-11 | 1998-12-11 | |
| US09/209,762 | 1998-12-11 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/787,668 A-371-Of-International US7217852B1 (en) | 1998-10-05 | 1999-09-17 | Process for producing middle distillates and middle distillates produced by that process |
| US10/712,169 Division US7294253B2 (en) | 1998-10-05 | 2003-11-12 | Process for producing middle distillates |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2000020535A1 true WO2000020535A1 (en) | 2000-04-13 |
Family
ID=26904489
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/ZA1999/000096 WO2000020535A1 (en) | 1998-10-05 | 1999-09-17 | Process for producing middle distillates and middle distillates produced by that process |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1129155A1 (en) |
| JP (2) | JP3824490B2 (en) |
| AU (1) | AU765274B2 (en) |
| MY (1) | MY125273A (en) |
| WO (1) | WO2000020535A1 (en) |
Cited By (77)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000060029A1 (en) * | 1999-04-06 | 2000-10-12 | Sasol Technology (Pty) Ltd | Process for producing synthetic naphtha fuel and synthetic naphtha fuel produced by that process |
| WO2001049812A1 (en) * | 1999-12-29 | 2001-07-12 | Chevron U.S.A. Inc. | A diesel fuel having a very high iso-paraffin to normal paraffin mole ratio |
| WO2001083406A2 (en) * | 2000-05-02 | 2001-11-08 | Exxonmobil Research And Engineering Company | Low sulfur, low emission blends of fischer-tropsch and conventional diesel fuels |
| WO2001083641A2 (en) * | 2000-05-02 | 2001-11-08 | Exxonmobil Research And Engineering Company | Winter diesel fuel production from a fischer-tropsch wax |
| WO2001083647A2 (en) * | 2000-05-02 | 2001-11-08 | Exxonmobil Research And Engineering Company | Wide cut fischer-tropsch diesel fuels |
| DE10038428A1 (en) * | 2000-08-07 | 2002-02-21 | Volkswagen Ag | Low-emission diesel fuels with high-boiling fraction having high cetane number and/or n-alkane content |
| US6583186B2 (en) | 2001-04-04 | 2003-06-24 | Chevron U.S.A. Inc. | Method for upgrading Fischer-Tropsch wax using split-feed hydrocracking/hydrotreating |
| US6589415B2 (en) | 2001-04-04 | 2003-07-08 | Chevron U.S.A., Inc. | Liquid or two-phase quenching fluid for multi-bed hydroprocessing reactor |
| GB2383798A (en) * | 2001-10-18 | 2003-07-09 | Chevron Usa Inc | Fuel blend having biocide activity |
| EP1365007A1 (en) * | 2002-05-24 | 2003-11-26 | ENI S.p.A. | Hydrocarbon compositions to be used as fuels |
| US6656342B2 (en) | 2001-04-04 | 2003-12-02 | Chevron U.S.A. Inc. | Graded catalyst bed for split-feed hydrocracking/hydrotreating |
| WO2003104361A3 (en) * | 2002-06-07 | 2004-04-08 | Sasol Tech Pty Ltd | Synthetic fuel with reduced particulate matter emissions and a method of operating a compression ignition engine using said fuel in conjunction with oxidation catalysts |
| WO2004035713A1 (en) * | 2002-10-18 | 2004-04-29 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| GB2396160A (en) * | 2002-10-04 | 2004-06-16 | Chevron Usa Inc | Fischer-Tropsch fuels having good ignition and low-temperature flow properties |
| JP2005502764A (en) * | 2001-09-07 | 2005-01-27 | シエル・インターナシヨネイル・リサーチ・マーチヤツピイ・ベー・ウイ | Diesel fuel, its production and use |
| JP2005507025A (en) * | 2001-10-19 | 2005-03-10 | シェブロン ユー.エス.エー. インコーポレイテッド | Thermally stable blends of highly paraffinic distillate fuel components and conventional distillate fuel components |
| WO2005021688A1 (en) * | 2003-09-03 | 2005-03-10 | Shell Internationale Research Maatschappij B.V. | Fuel compositions comprising fischer-tropsch derived fuel |
| WO2005026297A1 (en) * | 2003-09-17 | 2005-03-24 | Shell Internationale Research Maatschappij B.V. | Petroleum- and fischer-tropsch- derived kerosene blend |
| EP1689829A2 (en) * | 2003-11-14 | 2006-08-16 | Chevron U.S.A. Inc. | Process for the upgrading of the products of fischer-tropsch processes |
| WO2007020234A1 (en) * | 2005-08-12 | 2007-02-22 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US7229481B2 (en) | 2002-11-13 | 2007-06-12 | Shell Oil Company | Diesel fuel compositions |
| US7244350B2 (en) | 2001-08-08 | 2007-07-17 | Shell Oil Company | Process to prepare a hydrocarbon product having a sulphur content below 0.05 wt |
| WO2008012320A1 (en) * | 2006-07-27 | 2008-01-31 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| EP1885824A2 (en) * | 2005-04-05 | 2008-02-13 | Exxonmobil Research And Engineering Company | Paraffinic hydroisomerate as a wax crystal modifier |
| US7354507B2 (en) | 2004-03-17 | 2008-04-08 | Conocophillips Company | Hydroprocessing methods and apparatus for use in the preparation of liquid hydrocarbons |
| US7393877B2 (en) | 2003-12-31 | 2008-07-01 | Total France | Process for the conversion of a synthesis gas to hydrocarbons in the presence of beta-SiC and effluent from this process |
| EP2078744A1 (en) | 2008-01-10 | 2009-07-15 | Shell Internationale Researchmaatschappij B.V. | Fuel compositions |
| EP2083061A1 (en) * | 2006-10-06 | 2009-07-29 | Toyota Jidosha Kabushiki Kaisha | Light oil composition |
| US7667086B2 (en) | 2005-01-31 | 2010-02-23 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization and biodegradable compositions therefrom |
| US20100050504A1 (en) * | 2006-12-06 | 2010-03-04 | Landschof Joerg | Normal and iso parafines with low content of aromatics, sulphur and nitrogen as collector for froth flotation |
| US7678954B2 (en) | 2005-01-31 | 2010-03-16 | Exxonmobil Chemical Patents, Inc. | Olefin oligomerization to produce hydrocarbon compositions useful as fuels |
| US7678953B2 (en) | 2005-01-31 | 2010-03-16 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization |
| US7692049B2 (en) | 2005-01-31 | 2010-04-06 | Exxonmobil Chemical Patents Inc. | Hydrocarbon compositions useful for producing fuels and methods of producing the same |
| US7704375B2 (en) | 2002-07-19 | 2010-04-27 | Shell Oil Company | Process for reducing corrosion in a condensing boiler burning liquid fuel |
| US7737311B2 (en) | 2003-09-03 | 2010-06-15 | Shell Oil Company | Fuel compositions |
| US7741526B2 (en) | 2006-07-19 | 2010-06-22 | Exxonmobil Chemical Patents Inc. | Feedstock preparation of olefins for oligomerization to produce fuels |
| WO2010076304A1 (en) | 2008-12-29 | 2010-07-08 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2010076303A1 (en) | 2008-12-29 | 2010-07-08 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2010072838A3 (en) * | 2008-12-26 | 2010-08-26 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| WO2010072839A3 (en) * | 2008-12-26 | 2010-09-16 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| US7837853B2 (en) | 2005-04-11 | 2010-11-23 | Shell Oil Company | Process to blend a mineral and a Fischer-Tropsch derived product onboard a marine vessel |
| WO2011076948A1 (en) | 2009-12-24 | 2011-06-30 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2011080250A1 (en) | 2009-12-29 | 2011-07-07 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2011110551A1 (en) | 2010-03-10 | 2011-09-15 | Shell Internationale Research Maatschappij B.V. | Method of reducing the toxicity of used lubricating compositions |
| EP2371931A1 (en) | 2010-03-23 | 2011-10-05 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US8173439B2 (en) | 2006-12-20 | 2012-05-08 | Roche Diagnostics Operations, Inc. | Measurement system with optical referencing |
| WO2012074924A1 (en) * | 2010-11-30 | 2012-06-07 | Conocophillips Company | High cetane petroleum fuels |
| WO2012163935A2 (en) | 2011-05-30 | 2012-12-06 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2013034617A1 (en) | 2011-09-06 | 2013-03-14 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| US8475647B2 (en) | 2005-08-22 | 2013-07-02 | Shell Oil Company | Diesel fuel and a method of operating a diesel engine |
| US8481796B2 (en) | 2005-01-31 | 2013-07-09 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization and compositions therefrom |
| US8486876B2 (en) | 2007-10-19 | 2013-07-16 | Shell Oil Company | Functional fluids for internal combustion engines |
| US8541635B2 (en) | 2006-03-10 | 2013-09-24 | Shell Oil Company | Diesel fuel compositions |
| US8715371B2 (en) | 2007-05-11 | 2014-05-06 | Shell Oil Company | Fuel composition |
| EP2738240A1 (en) | 2012-11-30 | 2014-06-04 | Schepers Handels- en domeinnamen B.V. | Use of a Gas-to-Liquids gas oil in a lamp oil composition or fire lighter |
| WO2014096234A1 (en) | 2012-12-21 | 2014-06-26 | Shell Internationale Research Maatschappij B.V. | Liquid diesel fuel compositions containing organic sunscreen compounds |
| US8926716B2 (en) | 2006-10-20 | 2015-01-06 | Shell Oil Company | Method of formulating a fuel composition |
| US20150052803A1 (en) * | 2012-03-30 | 2015-02-26 | Jx Nippon Oil & Energy Corporation | Gas oil composition |
| WO2015091458A1 (en) | 2013-12-16 | 2015-06-25 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| EP2889361A1 (en) | 2013-12-31 | 2015-07-01 | Shell Internationale Research Maatschappij B.V. | Diesel fuel formulation and use thereof |
| WO2015157207A1 (en) | 2014-04-08 | 2015-10-15 | Shell Oil Company | Diesel fuel with improved ignition characteristics |
| EP2949732A1 (en) | 2014-05-28 | 2015-12-02 | Shell Internationale Research Maatschappij B.V. | Use of an oxanilide compound in a diesel fuel composition for the purpose of modifying the ignition delay and/or the burn period |
| US9447356B2 (en) | 2013-02-20 | 2016-09-20 | Shell Oil Company | Diesel fuel with improved ignition characteristics |
| WO2016019403A3 (en) * | 2014-07-28 | 2016-10-06 | Sasol Technology Proprietary Limited | Production of oilfield hydrocarbons |
| WO2017050777A1 (en) | 2015-09-22 | 2017-03-30 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2017081199A1 (en) | 2015-11-11 | 2017-05-18 | Shell Internationale Research Maatschappij B.V. | Process for preparing a diesel fuel composition |
| US9663735B2 (en) | 2013-10-24 | 2017-05-30 | Shell Oil Company | Liquid fuel compositions |
| WO2017093203A1 (en) | 2015-11-30 | 2017-06-08 | Shell Internationale Research Maatschappij B.V. | Fuel composition |
| EP3184612A1 (en) | 2015-12-21 | 2017-06-28 | Shell Internationale Research Maatschappij B.V. | Process for preparing a diesel fuel composition |
| WO2018077976A1 (en) | 2016-10-27 | 2018-05-03 | Shell Internationale Research Maatschappij B.V. | Process for preparing an automotive gasoil |
| WO2018206729A1 (en) | 2017-05-11 | 2018-11-15 | Shell Internationale Research Maatschappij B.V. | Process for preparing an automotive gas oil fraction |
| US10294436B2 (en) | 2014-11-12 | 2019-05-21 | Shell Oil Company | Fuel composition |
| WO2019201630A1 (en) | 2018-04-20 | 2019-10-24 | Shell Internationale Research Maatschappij B.V. | Diesel fuel with improved ignition characteristics |
| WO2020007790A1 (en) | 2018-07-02 | 2020-01-09 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2022228989A1 (en) | 2021-04-26 | 2022-11-03 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2022228990A1 (en) | 2021-04-26 | 2022-11-03 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2023235201A1 (en) * | 2022-05-31 | 2023-12-07 | ExxonMobil Technology and Engineering Company | Heavy distillate composition |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6949180B2 (en) * | 2002-10-09 | 2005-09-27 | Chevron U.S.A. Inc. | Low toxicity Fischer-Tropsch derived fuel and process for making same |
| CN100587043C (en) * | 2003-04-11 | 2010-02-03 | Sasol技术股份有限公司 | Low sulphur diesel fuel and aviation turbine fuel |
| JP4580152B2 (en) * | 2003-06-12 | 2010-11-10 | 出光興産株式会社 | Fuel oil for diesel engines |
| US20060016722A1 (en) * | 2004-07-08 | 2006-01-26 | Conocophillips Company | Synthetic hydrocarbon products |
| JP4617862B2 (en) * | 2004-12-13 | 2011-01-26 | 日油株式会社 | Fuel oil composition for diesel engines |
| EP1927645A4 (en) * | 2005-09-22 | 2009-05-27 | Japan Oil Gas & Metals Jogmec | Method for producing hydrocarbon fuel oil |
| JP4829659B2 (en) * | 2006-03-30 | 2011-12-07 | Jx日鉱日石エネルギー株式会社 | A heavy oil composition |
| JP4885627B2 (en) * | 2006-06-29 | 2012-02-29 | Jx日鉱日石エネルギー株式会社 | Method for producing A heavy oil composition |
| JP4885628B2 (en) * | 2006-06-29 | 2012-02-29 | Jx日鉱日石エネルギー株式会社 | Method for producing A heavy oil composition |
| WO2009015400A1 (en) * | 2007-05-31 | 2009-01-29 | Sasol Technology (Pty) Ltd | Cold flow response of diesel fuels |
| DK2288676T3 (en) * | 2008-05-20 | 2013-07-29 | Shell Int Research | USE OF FUEL COMPOSITIONS |
| JP5043754B2 (en) * | 2008-06-04 | 2012-10-10 | コスモ石油株式会社 | Fuel oil composition for diesel engines |
| JP5147550B2 (en) * | 2008-06-04 | 2013-02-20 | コスモ石油株式会社 | Fuel oil composition for diesel engines |
| JP5147549B2 (en) * | 2008-06-04 | 2013-02-20 | コスモ石油株式会社 | Fuel oil composition for diesel engines |
| JP5341452B2 (en) * | 2008-09-30 | 2013-11-13 | 独立行政法人石油天然ガス・金属鉱物資源機構 | Operation analysis device |
| JP2011127086A (en) * | 2009-12-21 | 2011-06-30 | Showa Shell Sekiyu Kk | Gas oil fuel composition |
| JP2011127083A (en) * | 2009-12-21 | 2011-06-30 | Showa Shell Sekiyu Kk | Multi-grade gas oil fuel composition |
| JP5460297B2 (en) * | 2009-12-21 | 2014-04-02 | 昭和シェル石油株式会社 | Light oil fuel composition |
| JP5312646B2 (en) * | 2012-07-11 | 2013-10-09 | コスモ石油株式会社 | Fuel oil composition for diesel engines |
| JP5328974B2 (en) * | 2012-11-26 | 2013-10-30 | コスモ石油株式会社 | Fuel oil composition for diesel engines |
| JP5328973B2 (en) * | 2012-11-26 | 2013-10-30 | コスモ石油株式会社 | Fuel oil composition for diesel engines |
| JP6228943B2 (en) * | 2015-01-15 | 2017-11-08 | 東燃ゼネラル石油株式会社 | Light oil composition |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997014769A1 (en) * | 1995-10-17 | 1997-04-24 | Exxon Research And Engineering Company | Synthetic diesel fuel and process for its production |
| WO1998034998A1 (en) * | 1997-02-07 | 1998-08-13 | Exxon Research And Engineering Company | Diesel additive for improving cetane, lubricity, and stability |
-
1999
- 1999-09-17 EP EP99950311A patent/EP1129155A1/en not_active Withdrawn
- 1999-09-17 AU AU63001/99A patent/AU765274B2/en not_active Expired
- 1999-09-17 WO PCT/ZA1999/000096 patent/WO2000020535A1/en active IP Right Grant
- 1999-09-17 JP JP2000574637A patent/JP3824490B2/en not_active Expired - Fee Related
- 1999-10-01 MY MYPI99004459A patent/MY125273A/en unknown
-
2006
- 2006-02-07 JP JP2006029766A patent/JP4261552B2/en not_active Expired - Lifetime
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997014769A1 (en) * | 1995-10-17 | 1997-04-24 | Exxon Research And Engineering Company | Synthetic diesel fuel and process for its production |
| WO1998034998A1 (en) * | 1997-02-07 | 1998-08-13 | Exxon Research And Engineering Company | Diesel additive for improving cetane, lubricity, and stability |
Cited By (128)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2364066A (en) * | 1999-04-06 | 2002-01-16 | Sasol Technology | Process for producing synthetic naphtha fuel and synthetic naphtha fuel produced by that process |
| US6656343B2 (en) | 1999-04-06 | 2003-12-02 | Sasol Technology (Pty) Ltd. | Process for producing synthetic naphtha fuel and synthetic naphtha fuel produced by that process |
| WO2000060029A1 (en) * | 1999-04-06 | 2000-10-12 | Sasol Technology (Pty) Ltd | Process for producing synthetic naphtha fuel and synthetic naphtha fuel produced by that process |
| WO2001049812A1 (en) * | 1999-12-29 | 2001-07-12 | Chevron U.S.A. Inc. | A diesel fuel having a very high iso-paraffin to normal paraffin mole ratio |
| KR100754582B1 (en) * | 2000-05-02 | 2007-09-05 | 엑손모빌 리서치 앤드 엔지니어링 컴퍼니 | Wide cut fischer-tropsch diesel fuels |
| JP2003531948A (en) * | 2000-05-02 | 2003-10-28 | エクソンモービル リサーチ アンド エンジニアリング カンパニー | Production of winter diesel fuel from Fischer-Tropsch wax |
| JP2004515562A (en) * | 2000-05-02 | 2004-05-27 | エクソンモービル リサーチ アンド エンジニアリング カンパニー | Low sulfur low emission blends of Fischer-Tropsch diesel fuel and conventional diesel fuel |
| WO2001083647A3 (en) * | 2000-05-02 | 2002-03-07 | Exxonmobil Res & Eng Co | Wide cut fischer-tropsch diesel fuels |
| WO2001083641A3 (en) * | 2000-05-02 | 2002-09-06 | Exxonmobil Res & Eng Co | Winter diesel fuel production from a fischer-tropsch wax |
| WO2001083406A3 (en) * | 2000-05-02 | 2003-01-30 | Exxonmobil Res & Eng Co | Low sulfur, low emission blends of fischer-tropsch and conventional diesel fuels |
| US6833064B2 (en) * | 2000-05-02 | 2004-12-21 | Exxonmobil Research And Engineering Company | Wide cut Fischer Tropsch diesel fuels |
| AU2001255280B2 (en) * | 2000-05-02 | 2005-12-08 | Exxonmobil Research And Engineering Company | Wide cut fischer-tropsch diesel fuels |
| WO2001083641A2 (en) * | 2000-05-02 | 2001-11-08 | Exxonmobil Research And Engineering Company | Winter diesel fuel production from a fischer-tropsch wax |
| WO2001083647A2 (en) * | 2000-05-02 | 2001-11-08 | Exxonmobil Research And Engineering Company | Wide cut fischer-tropsch diesel fuels |
| JP2003531949A (en) * | 2000-05-02 | 2003-10-28 | エクソンモービル リサーチ アンド エンジニアリング カンパニー | Wide Cut Fisher-Tropsch Diesel Fuel Oil |
| US6787022B1 (en) | 2000-05-02 | 2004-09-07 | Exxonmobil Research And Engineering Company | Winter diesel fuel production from a fischer-tropsch wax |
| WO2001083406A2 (en) * | 2000-05-02 | 2001-11-08 | Exxonmobil Research And Engineering Company | Low sulfur, low emission blends of fischer-tropsch and conventional diesel fuels |
| US6663767B1 (en) | 2000-05-02 | 2003-12-16 | Exxonmobil Research And Engineering Company | Low sulfur, low emission blends of fischer-tropsch and conventional diesel fuels |
| DE10038428A1 (en) * | 2000-08-07 | 2002-02-21 | Volkswagen Ag | Low-emission diesel fuels with high-boiling fraction having high cetane number and/or n-alkane content |
| US6656342B2 (en) | 2001-04-04 | 2003-12-02 | Chevron U.S.A. Inc. | Graded catalyst bed for split-feed hydrocracking/hydrotreating |
| US6589415B2 (en) | 2001-04-04 | 2003-07-08 | Chevron U.S.A., Inc. | Liquid or two-phase quenching fluid for multi-bed hydroprocessing reactor |
| US6583186B2 (en) | 2001-04-04 | 2003-06-24 | Chevron U.S.A. Inc. | Method for upgrading Fischer-Tropsch wax using split-feed hydrocracking/hydrotreating |
| US7244350B2 (en) | 2001-08-08 | 2007-07-17 | Shell Oil Company | Process to prepare a hydrocarbon product having a sulphur content below 0.05 wt |
| JP2005502764A (en) * | 2001-09-07 | 2005-01-27 | シエル・インターナシヨネイル・リサーチ・マーチヤツピイ・ベー・ウイ | Diesel fuel, its production and use |
| GB2383798A (en) * | 2001-10-18 | 2003-07-09 | Chevron Usa Inc | Fuel blend having biocide activity |
| GB2383798B (en) * | 2001-10-18 | 2004-07-21 | Chevron Usa Inc | Inhibition of biological degradation in fischer-tropsch products |
| US6924404B2 (en) | 2001-10-18 | 2005-08-02 | Chevron U.S.A. Inc. | Inhibition of biological degradation of Fischer-Tropsch products |
| JP2005507025A (en) * | 2001-10-19 | 2005-03-10 | シェブロン ユー.エス.エー. インコーポレイテッド | Thermally stable blends of highly paraffinic distillate fuel components and conventional distillate fuel components |
| JP4796277B2 (en) * | 2001-10-19 | 2011-10-19 | シェブロン ユー.エス.エー. インコーポレイテッド | Thermally stable blends of highly paraffinic distillate fuel components and conventional distillate fuel components |
| EP1365007A1 (en) * | 2002-05-24 | 2003-11-26 | ENI S.p.A. | Hydrocarbon compositions to be used as fuels |
| WO2003104361A3 (en) * | 2002-06-07 | 2004-04-08 | Sasol Tech Pty Ltd | Synthetic fuel with reduced particulate matter emissions and a method of operating a compression ignition engine using said fuel in conjunction with oxidation catalysts |
| JP2005529213A (en) * | 2002-06-07 | 2005-09-29 | セイソル テクノロジー (プロプライエタリー) リミテッド | Synthetic fuel with reduced particulate emissions and method of operating a compression ignition engine using said fuel in conjunction with an oxidation catalyst |
| US7704375B2 (en) | 2002-07-19 | 2010-04-27 | Shell Oil Company | Process for reducing corrosion in a condensing boiler burning liquid fuel |
| US7909894B2 (en) | 2002-10-04 | 2011-03-22 | Chevron U.S.A. Inc. | Systems and methods of improving diesel fuel performance in cold climates |
| GB2396160B (en) * | 2002-10-04 | 2005-05-11 | Chevron Usa Inc | Systems and methods of improving diesel fuel performance in cold climates |
| GB2396160A (en) * | 2002-10-04 | 2004-06-16 | Chevron Usa Inc | Fischer-Tropsch fuels having good ignition and low-temperature flow properties |
| US7354462B2 (en) | 2002-10-04 | 2008-04-08 | Chevron U.S.A. Inc. | Systems and methods of improving diesel fuel performance in cold climates |
| AU2003301273B2 (en) * | 2002-10-18 | 2007-07-19 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US7189269B2 (en) | 2002-10-18 | 2007-03-13 | Shell Oil Company | Fuel composition comprising a base fuel, a fischer tropsch derived gas oil, and an oxygenate |
| WO2004035713A1 (en) * | 2002-10-18 | 2004-04-29 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US7229481B2 (en) | 2002-11-13 | 2007-06-12 | Shell Oil Company | Diesel fuel compositions |
| CN100413946C (en) * | 2003-09-03 | 2008-08-27 | 国际壳牌研究有限公司 | Fuel compositions comprising fischer-tropsch derived fuel |
| WO2005021688A1 (en) * | 2003-09-03 | 2005-03-10 | Shell Internationale Research Maatschappij B.V. | Fuel compositions comprising fischer-tropsch derived fuel |
| US7737311B2 (en) | 2003-09-03 | 2010-06-15 | Shell Oil Company | Fuel compositions |
| WO2005026297A1 (en) * | 2003-09-17 | 2005-03-24 | Shell Internationale Research Maatschappij B.V. | Petroleum- and fischer-tropsch- derived kerosene blend |
| US7666294B2 (en) | 2003-09-17 | 2010-02-23 | Shell Oil Company | Depressed freeze point kerosene fuel compositions and methods of making and using same |
| EP1689829A4 (en) * | 2003-11-14 | 2012-02-29 | Chevron Usa Inc | Process for the upgrading of the products of fischer-tropsch processes |
| EP1689829A2 (en) * | 2003-11-14 | 2006-08-16 | Chevron U.S.A. Inc. | Process for the upgrading of the products of fischer-tropsch processes |
| US7393877B2 (en) | 2003-12-31 | 2008-07-01 | Total France | Process for the conversion of a synthesis gas to hydrocarbons in the presence of beta-SiC and effluent from this process |
| US7354507B2 (en) | 2004-03-17 | 2008-04-08 | Conocophillips Company | Hydroprocessing methods and apparatus for use in the preparation of liquid hydrocarbons |
| US8481796B2 (en) | 2005-01-31 | 2013-07-09 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization and compositions therefrom |
| US7678953B2 (en) | 2005-01-31 | 2010-03-16 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization |
| US7667086B2 (en) | 2005-01-31 | 2010-02-23 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization and biodegradable compositions therefrom |
| US7678954B2 (en) | 2005-01-31 | 2010-03-16 | Exxonmobil Chemical Patents, Inc. | Olefin oligomerization to produce hydrocarbon compositions useful as fuels |
| US7692049B2 (en) | 2005-01-31 | 2010-04-06 | Exxonmobil Chemical Patents Inc. | Hydrocarbon compositions useful for producing fuels and methods of producing the same |
| AU2006252955B2 (en) * | 2005-04-05 | 2011-06-16 | Exxonmobil Research And Engineering Company | Paraffinic hydroisomerate as a wax crystal modifier |
| EP1885824A4 (en) * | 2005-04-05 | 2009-09-02 | Exxonmobil Res & Eng Co | Paraffinic hydroisomerate as a wax crystal modifier |
| EP1885824A2 (en) * | 2005-04-05 | 2008-02-13 | Exxonmobil Research And Engineering Company | Paraffinic hydroisomerate as a wax crystal modifier |
| US7837853B2 (en) | 2005-04-11 | 2010-11-23 | Shell Oil Company | Process to blend a mineral and a Fischer-Tropsch derived product onboard a marine vessel |
| US8076522B2 (en) | 2005-08-12 | 2011-12-13 | Shell Oil Company | Fuel compositions |
| CN101273116B (en) * | 2005-08-12 | 2012-11-07 | 国际壳牌研究有限公司 | Fuel compositions |
| WO2007020234A1 (en) * | 2005-08-12 | 2007-02-22 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US8475647B2 (en) | 2005-08-22 | 2013-07-02 | Shell Oil Company | Diesel fuel and a method of operating a diesel engine |
| US8541635B2 (en) | 2006-03-10 | 2013-09-24 | Shell Oil Company | Diesel fuel compositions |
| US7741526B2 (en) | 2006-07-19 | 2010-06-22 | Exxonmobil Chemical Patents Inc. | Feedstock preparation of olefins for oligomerization to produce fuels |
| WO2008012320A1 (en) * | 2006-07-27 | 2008-01-31 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| EP2083061A1 (en) * | 2006-10-06 | 2009-07-29 | Toyota Jidosha Kabushiki Kaisha | Light oil composition |
| EP2083061A4 (en) * | 2006-10-06 | 2013-03-20 | Toyota Motor Co Ltd | Light oil composition |
| US8926716B2 (en) | 2006-10-20 | 2015-01-06 | Shell Oil Company | Method of formulating a fuel composition |
| US20100050504A1 (en) * | 2006-12-06 | 2010-03-04 | Landschof Joerg | Normal and iso parafines with low content of aromatics, sulphur and nitrogen as collector for froth flotation |
| US8173439B2 (en) | 2006-12-20 | 2012-05-08 | Roche Diagnostics Operations, Inc. | Measurement system with optical referencing |
| US8715371B2 (en) | 2007-05-11 | 2014-05-06 | Shell Oil Company | Fuel composition |
| US8486876B2 (en) | 2007-10-19 | 2013-07-16 | Shell Oil Company | Functional fluids for internal combustion engines |
| EP2078744A1 (en) | 2008-01-10 | 2009-07-15 | Shell Internationale Researchmaatschappij B.V. | Fuel compositions |
| CN102307973A (en) * | 2008-12-26 | 2012-01-04 | 国际壳牌研究有限公司 | Diesel fuel composition |
| US20120046506A1 (en) * | 2008-12-26 | 2012-02-23 | Nobuhiro Okabe | Diesel fuel composition |
| WO2010072839A3 (en) * | 2008-12-26 | 2010-09-16 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| WO2010072838A3 (en) * | 2008-12-26 | 2010-08-26 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| US9017429B2 (en) | 2008-12-29 | 2015-04-28 | Shell Oil Company | Fuel compositions |
| US8771385B2 (en) | 2008-12-29 | 2014-07-08 | Shell Oil Company | Fuel compositions |
| WO2010076304A1 (en) | 2008-12-29 | 2010-07-08 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2010076303A1 (en) | 2008-12-29 | 2010-07-08 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2011076948A1 (en) | 2009-12-24 | 2011-06-30 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2011080250A1 (en) | 2009-12-29 | 2011-07-07 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2011110551A1 (en) | 2010-03-10 | 2011-09-15 | Shell Internationale Research Maatschappij B.V. | Method of reducing the toxicity of used lubricating compositions |
| EP2371931A1 (en) | 2010-03-23 | 2011-10-05 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US8876923B2 (en) | 2010-03-23 | 2014-11-04 | Shell Oil Company | Fuel compositions |
| US8757106B2 (en) | 2010-11-30 | 2014-06-24 | Phillips 66 Company | High cetane petroleum fuels |
| WO2012074924A1 (en) * | 2010-11-30 | 2012-06-07 | Conocophillips Company | High cetane petroleum fuels |
| WO2012163935A2 (en) | 2011-05-30 | 2012-12-06 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| WO2013034617A1 (en) | 2011-09-06 | 2013-03-14 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| EP2832829A4 (en) * | 2012-03-30 | 2015-09-02 | Jx Nippon Oil & Energy Corp | Gas oil composition |
| RU2630225C1 (en) * | 2012-03-30 | 2017-09-06 | ДжиЭкс НИППОН ОЙЛ ЭНД ЭНЕРДЖИ КОРПОРЕЙШЕН | Gas-oil composition |
| US20150052803A1 (en) * | 2012-03-30 | 2015-02-26 | Jx Nippon Oil & Energy Corporation | Gas oil composition |
| EP2738240A1 (en) | 2012-11-30 | 2014-06-04 | Schepers Handels- en domeinnamen B.V. | Use of a Gas-to-Liquids gas oil in a lamp oil composition or fire lighter |
| WO2014096234A1 (en) | 2012-12-21 | 2014-06-26 | Shell Internationale Research Maatschappij B.V. | Liquid diesel fuel compositions containing organic sunscreen compounds |
| US9222047B2 (en) | 2012-12-21 | 2015-12-29 | Shell Oil Company | Liquid fuel compositions |
| US9447356B2 (en) | 2013-02-20 | 2016-09-20 | Shell Oil Company | Diesel fuel with improved ignition characteristics |
| US9663735B2 (en) | 2013-10-24 | 2017-05-30 | Shell Oil Company | Liquid fuel compositions |
| WO2015091458A1 (en) | 2013-12-16 | 2015-06-25 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| US9587195B2 (en) | 2013-12-16 | 2017-03-07 | Shell Oil Company | Liquid composition |
| EP2889361A1 (en) | 2013-12-31 | 2015-07-01 | Shell Internationale Research Maatschappij B.V. | Diesel fuel formulation and use thereof |
| WO2015157207A1 (en) | 2014-04-08 | 2015-10-15 | Shell Oil Company | Diesel fuel with improved ignition characteristics |
| US9862905B2 (en) | 2014-04-08 | 2018-01-09 | Shell Oil Company | Diesel fuel with improved ignition characteristics |
| EP2949732A1 (en) | 2014-05-28 | 2015-12-02 | Shell Internationale Research Maatschappij B.V. | Use of an oxanilide compound in a diesel fuel composition for the purpose of modifying the ignition delay and/or the burn period |
| CN110305693A (en) * | 2014-07-28 | 2019-10-08 | 沙索技术有限公司 | The production of oil field hydrocarbon |
| WO2016019403A3 (en) * | 2014-07-28 | 2016-10-06 | Sasol Technology Proprietary Limited | Production of oilfield hydrocarbons |
| CN110305693B (en) * | 2014-07-28 | 2022-05-10 | 沙索技术有限公司 | Production of oilfield hydrocarbons |
| US10487273B2 (en) | 2014-07-28 | 2019-11-26 | Sasol Technology Proprietary Limited | Production of oilfield hydrocarbons |
| EP3495452A1 (en) * | 2014-07-28 | 2019-06-12 | Sasol Technology Proprietary Limited | Production of oilfield hydrocarbons and lubricant base oils |
| US10190063B2 (en) | 2014-07-28 | 2019-01-29 | Sasol Technology Proprietary Limited | Production of oilfield hydrocarbons |
| US10294436B2 (en) | 2014-11-12 | 2019-05-21 | Shell Oil Company | Fuel composition |
| WO2017050777A1 (en) | 2015-09-22 | 2017-03-30 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| US10808195B2 (en) | 2015-09-22 | 2020-10-20 | Shell Oil Company | Fuel compositions |
| US11084997B2 (en) | 2015-11-11 | 2021-08-10 | Shell Oil Company | Process for preparing a diesel fuel composition |
| WO2017081199A1 (en) | 2015-11-11 | 2017-05-18 | Shell Internationale Research Maatschappij B.V. | Process for preparing a diesel fuel composition |
| WO2017093203A1 (en) | 2015-11-30 | 2017-06-08 | Shell Internationale Research Maatschappij B.V. | Fuel composition |
| US11959033B2 (en) | 2015-11-30 | 2024-04-16 | Shell Usa, Inc. | Fuel composition |
| EP3184612A1 (en) | 2015-12-21 | 2017-06-28 | Shell Internationale Research Maatschappij B.V. | Process for preparing a diesel fuel composition |
| WO2018077976A1 (en) | 2016-10-27 | 2018-05-03 | Shell Internationale Research Maatschappij B.V. | Process for preparing an automotive gasoil |
| WO2018206729A1 (en) | 2017-05-11 | 2018-11-15 | Shell Internationale Research Maatschappij B.V. | Process for preparing an automotive gas oil fraction |
| US11512261B2 (en) | 2018-04-20 | 2022-11-29 | Shell Usa, Inc. | Diesel fuel with improved ignition characteristics |
| WO2019201630A1 (en) | 2018-04-20 | 2019-10-24 | Shell Internationale Research Maatschappij B.V. | Diesel fuel with improved ignition characteristics |
| WO2020007790A1 (en) | 2018-07-02 | 2020-01-09 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| US11499107B2 (en) | 2018-07-02 | 2022-11-15 | Shell Usa, Inc. | Liquid fuel compositions |
| WO2022228989A1 (en) | 2021-04-26 | 2022-11-03 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2022228990A1 (en) | 2021-04-26 | 2022-11-03 | Shell Internationale Research Maatschappij B.V. | Fuel compositions |
| WO2023235201A1 (en) * | 2022-05-31 | 2023-12-07 | ExxonMobil Technology and Engineering Company | Heavy distillate composition |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2002526637A (en) | 2002-08-20 |
| JP4261552B2 (en) | 2009-04-30 |
| EP1129155A1 (en) | 2001-09-05 |
| JP2006161056A (en) | 2006-06-22 |
| MY125273A (en) | 2006-07-31 |
| AU765274B2 (en) | 2003-09-11 |
| JP3824490B2 (en) | 2006-09-20 |
| AU6300199A (en) | 2000-04-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU765274B2 (en) | Process for producing middle distillates and middle distillates produced by that process | |
| EP1171551B1 (en) | Process for producing synthetic naphtha fuel | |
| US7294253B2 (en) | Process for producing middle distillates | |
| US7252754B2 (en) | Production of biodegradable middle distillates | |
| GB2385861A (en) | Removal of carbon oxides from Fischer-Tropsch products prior to hydroprocessing | |
| ZA200505976B (en) | Process for the preparation of and composition of a feedstock usable for the preparation of lower olefins | |
| AU2003252879B2 (en) | Process for producing synthetic naphtha fuel and synthetic naphtha fuel produced by that process | |
| ZA200102751B (en) | Process for producing middle distillates and middle distillates produced by that process. | |
| AU2004207852B2 (en) | Process for the preparation of and composition of a feedstock usable for the preparation of lower olefins | |
| CN1821362B (en) | Synthetic naphtha fuel produced by that process for producing synthetic naphtha fuel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AL AM AT AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ CZ DE DE DK DK DM EE EE ES FI FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) |
Free format text: (EXCEPT US) |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 63001/99 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1999950311 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2000 574637 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2001/02751 Country of ref document: ZA Ref document number: 200102751 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09787668 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 63001/99 Country of ref document: AU |