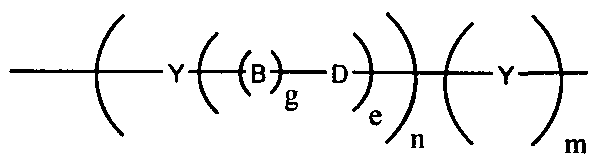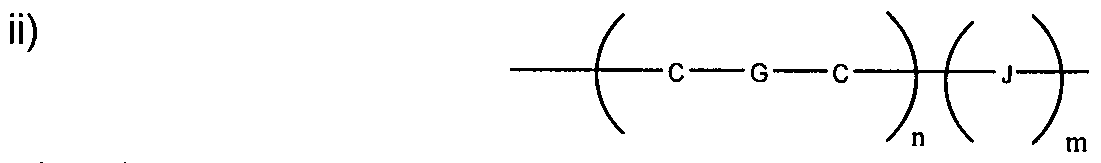WO1998020162A2 - Electrodes linked via conductive oligomers to nucleic acids - Google Patents
Electrodes linked via conductive oligomers to nucleic acids Download PDFInfo
- Publication number
- WO1998020162A2 WO1998020162A2 PCT/US1997/020014 US9720014W WO9820162A2 WO 1998020162 A2 WO1998020162 A2 WO 1998020162A2 US 9720014 W US9720014 W US 9720014W WO 9820162 A2 WO9820162 A2 WO 9820162A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- nucleic acid
- electron transfer
- electrode
- zero
- conductive oligomer
- Prior art date
Links
- 0 CC(C(C)OC1*)C1NC(*C(C)(C)*B*C(C)(C)C)=O Chemical compound CC(C(C)OC1*)C1NC(*C(C)(C)*B*C(C)(C)C)=O 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H21/00—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6813—Hybridisation assays
- C12Q1/6816—Hybridisation assays characterised by the detection means
- C12Q1/6825—Nucleic acid detection involving sensors
Definitions
- Gene probe assays currently play roles in identifying infectious organisms such as bacteria and viruses, in probing the expression of normal genes and identifying mutant genes such as oncogenes, in typing tissue for compatibility preceding tissue transplantation, in matching tissue or blood samples for forensic medicine, and for exploring homology among genes from different species.
- a gene probe assay should be sensitive, specific and easily automatable (for a review, see Nickerson, Current Opinion in Biotechnology 4:48-51 (1993)).
- the requirement for sensitivity i.e. low detection limits
- PCR polymerase chain reaction
- other amplification technologies which allow researchers to amplify exponentially a specific nucleic acid sequence before analysis (for a review, see Abramson et al., Current Opinion in Biotechnology, 4:41-47 (1993)).
- New experimental techniques for mismatch detection with standard probes include DNA ligation assays where single point mismatches prevent ligation and probe digestion assays in which mismatches create sites for probe cleavage.
- PCT applications WO 95/15971 , PCT/US96/09769 and PCT/US97/09739 describe novel compositions comprising nucleic acids containing electron transfer moieties, including electrodes, which allow for novel detection methods of nucleic acid hybridization.
- compositions comprising (a) a first electron transfer moiety comprising an electrode; (b) a first single stranded nucleic acid; (c) a conductive oligomer covalently attached to both the electrode and the first nucleic acid; and (d) a second electron transfer moiety covalently attached to a second single stranded nucleic acid.
- the conductive oligomer has the formula: wherein
- n is an integer from 1 to 50;
- m is 0 or 1 ;
- the invention provides methods of detecting a target sequence in a nucleic acid sample.
- the method comprises applying a first input signal to a hybridization complex and detecting electron transfer.
- the hybridization complex comprises the target sequence, if present, and at least a first probe nucleic acid.
- the probe nucleic acid comprises a a covalently attached conductive oligomer.
- the conductive oligomer is also covalently attached to a first electron transfer moiety comprising an electrode.
- the hybridization complex has a covalently attached second electron transfer moiety.
- the conductive oligomer has the formula:
- the first input signal comprises an AC component and a non-zero DC component.
- the first input signal comprises an AC component at a first frequency and a non-zero DC component
- the method further comprises applying a second input signal comprising an AC component at at least a second frequency and a non-zero DC component.
- the first input signal comprises an AC component and a first non-zero DC component
- the method further comprises applying a second input signal comprising an AC component and a second non-zero DC component.
- the invention provides methods of making the compositions of the invention.
- the methods comprise attaching a conductive oligomer to a nucleic acid, and attaching the conductive oligomer to said electrode. These steps may be done in any order.
- compositions comprising a conductive oligomer covalently attached to a nucleoside, wherein said conductive oligomer has the formula: is selected from the group consisting of:
- the invention provides peptide nucleic acids with at least one chemical substituent covalently attached to the ⁇ -carbon of a subunit of the peptide nucleic acid.
- Figure 3 depicts the synthetic scheme for a conductive oligomer covalently attached to a uridine nucleoside via the base.
- Figure 4 depicts the synthetic scheme for a conductive oligomer covalently attached to a nucleoside via a phosphate of the ribose-phosphate backbone.
- the conductive oligomer is a phenyl-acetylene Structure 5 oligomer, although other oiigomers may be used, and terminates in an ethyl pyridine protecting group, as described herein, for attachment to gold electrodes.
- Figure 5 depicts the synthetic scheme for a conductive oligomer covalently attached to a nucleoside via a phosphate of the ribose-phosphate backbone, using an amide linkage and an ethylene linker, although other linkers may be used.
- the conductive oligomer is a phenyl-acetylene Structure 5 oligomer, although other oiigomers may be used, and terminates in an ethyl pyridine protecting group, as described herein, for attachment to gold electrodes.
- Figure 6 depicts the synthetic scheme for a conductive polymer containing an aromatic group with a substitution group.
- the conductive oligomer is a phenyl-acetylene Structure 5 oligomer with a single methyl R group on each phenyl ring, although other oiigomers may be used, and terminates in an ethyl pyridine protecting group, as described herein, for attachment to gold electrodes.
- Figure 8 depicts a model compound, ferrocene attached to a C 16 alkane molecule (insulator-1), at 200 mV AC amplitude and frequencies of 1 , 5 and 100 Hz. The sample responds at all three frequencies, with higher currents resulting from higher frequencies.
- Figures 9A and 9B depict the response with varying frequency.
- Figure 9A shows overlaid voltammograms of an electrode coated with a ferrocene-conductive oligomer model complex (wire-2). Four excitation frequencies were applied, 10 Hz, 100 Hz, 1 kHz and 10 kHz, all at 25 mV overpotential. Again, current increases with frequency.
- Figure 9B shows overlaid voltammograms of electrodes coated with either ssDNA or dsDNA.
- ssDNA was run at 1 Hz and 10 Hz at 100 mV overpotential (bottom two lines).
- dsDNA was run at 1 , 10, 50 and 100 Hz at 10 mV overpotential (top four lines). Note that the scales between Figure 8 and Figures 9A and 9B are different.
- Figure 11 depicts the frequency responses of ssDNA (open circles; sample 5) and dsDNA (filled circles; sample 6) at 25 mV overpotential.
- the current has been normalized.
- the curves are not a fit to the data; rather, these are models of RC circuits, illustrating that the data can be fit to such curves, and that the system is in fact mimic standard RC circuits.
- the top curve was modeled using a 500 ohm resistor and a 0.001 farad capacitor.
- the bottom curve was modeled using a 20 ohm resistor and a 0.002 farad capacitor.
- Figure 12 shows that increasing the overpotential will increase the output current.
- Figures 13A and 13B illustrate that the overpotential and frequency can be tuned to increase the selectivity and sensitivity, using Sample 1.
- Figure 14 shows that ferrocene added to the solution (Sample 7; open circles) has a frequency response related to diffusion that is easily distinguishable from attached ferrocene (Sample 3; filled circles).
- Figures 15A and 15B shows the phase shift that results with different samples.
- Figure 15A uses two experiments of Sample 1 , Sample 3 and Sample 4.
- Figure 15B uses Sample 5 and Sample 6.
- Figure 16 depicts the synthetic scheme for a conductive oligomer covalently attached to a uridine nucleoside via an amine bond, with a CH2 group as a Z linker.
- Compound C4 can be extended as outlined herein and in Figure 1.
- Figures 17A, 17B, 17C, 17D, 17E, 17F and 17G depict other conductive oiigomers, attached either through the base (A-D) or through the ribose of the backbone (E-G), which have been synthesized using the techniques outlined herein.
- Figure 17H depicts a conductive oligomer attached to a ferrocene.
- the compounds are shown as containing CPG groups, phosphoramidite groups, or neither; however, they may all be made as any of these.
- Figure 18 depicts a synthetic scheme for a four unit conductive oligomer attached to the base.
- Figure 19 depicts a synthetic scheme for a four unit conductive oligomer attached to the base.
- Figure 20 depicts the use of a trimethylsilylethyl protecting group in synthesizing a five unit wire attached via the base.
- Figure 21 depicts the use of a trimethylsilylethyl protecting group in synthesizing a five unit wire attached via the ribose.
- Figures 22A and 22B depict simulations based on traditional electrochemical theory (Figure 22B) and the simulation model developed herein ( Figure 22A).
- Figures 23A and 23B depict experimental data plotted with theoretical model, showing good correlation.
- Fc-wire of Example 7 was used as 10 Hz ( Figure 23A) and 100 Hz ( Figure 23B).
- Figure 24 depicts the synthetic scheme for protecting and derivatizing adenine for incorporation into PNA.
- Figure 25 depicts the synthetic scheme for protecting and derivatizing cytosine for incorporation into PNA.
- Figure 26 depicts the synthetic scheme for protecting and derivatizing guanine for incorporation into PNA.
- Figures 28A, 28B, 28C, 28D and 28E depict the synthetic scheme for making PNA monomeric subunits.
- Figures 28B-28E depict the PNA monomers.
- Figure 29 depicts the synthetic scheme for a PNA monomeric subunit with a ferrocene covalently attached to a uracil base, for incorporation into a growing PNA.
- Figure 30 depicts the synthetic scheme for a three unit conductive oligomer covalently attached to a base of a PNA monomeric subunit.
- Figure 32 depicts the synthetic scheme for a ferrocene covalently attached to the backbone of a PNA monomeric subunit.
- the present invention capitalizes on the previous discovery that electron transfer apparently proceeds through the stacked ⁇ -orbitals of the heterocyclic bases of double stranded (hybridized) nucleic acid ("the ⁇ -way").
- This finding allows the use of nucleic acids containing electron transfer moieties to be used as nucleic acid probes. See PCT publication WO 95/15971 , hereby incorporated by reference in its entirety, and cited references.
- This publication describes the site-selective modification of nucleic acids with redox active moieties, i.e. electron donor and acceptor moieties, which allow the longdistance electron transfer through a double stranded nucleic acid.
- the present invention provides for novel gene probes, which are useful in molecular biology and diagnostic medicine.
- single stranded nucleic acids having a predetermined sequence and covalently attached electron transfer moieties, including an electrode are synthesized.
- the sequence is selected based upon a known target sequence, such that if hybridization to a complementary target sequence occurs in the region between the electron donor and the electron acceptor, electron transfer proceeds at an appreciable and detectable rate.
- the invention has broad general use, as a new form of labelled gene probe.
- the probes of the present invention allow detection of target sequences without the removal of unhybridized probe.
- the invention is uniquely suited to automated gene probe assays or field testing.
- compositions comprising nucleic acids covalently attached via conductive oiigomers to an electrode, of a general structure depicted below in Structure 1 :
- X is a conductive oligomer as defined herein.
- F is a linkage that allows the covalent attachment of the electrode and the conductive oligomer, including bonds, atoms or linkers such as is described herein, for example as "A", defined below.
- F 2 is a linkage that allows the covalent attachment of the conductive oligomer to the nucleic acid, and may be a bond, an atom or a linkage as is herein described.
- F 2 may be part of the conductive oligomer, part of the nucleic acid, or exogeneous to both, for example, as defined herein for "Z".
- nucleic acid or "oligonucleotide” or grammatical equivalents herein means at least two nucleotides covalently linked together.
- a nucleic acid of the present invention will generally contain phosphodiester bonds, although in some cases, as outlined below, nucleic acid analogs are included that may have alternate backbones, comprising, for example, phosphoramide (Beaucage et al., Tetrahedron 49(10): 1925 (1993) and references therein; Letsinger, J. Org. Chem. 35:3800 (1970); Sblul et al., Eur. J. Biochem. 81:579 (1977); Letsinger et al., Nucl. Acids Res.
- nucleic acids containing one or more carbocyclic sugars are also included within the definition of nucleic acids (see Jenkins et al., Chem. Soc. Rev. (1995) pp169- 176).
- nucleic acid analogs are described in Rawls, C & E News June 2, 1997 page 35. All of these references are hereby expressly incorporated by reference. These modifications of the ribose- phosphate backbone may be done to facilitate the addition of electron transfer moieties, or to increase the stability and half-life of such molecules in physiological environments.
- nucleic acid analogs may find use in the present invention.
- mixtures of naturally occurring nucleic acids and analogs can be made; for example, at the site of conductive oligomer or electron transfer moiety attachment, an analog structure may be used.
- mixtures of different nucleic acid analogs, and mixtures of naturally occuring nucleic acids and analogs may be made.
- PNA peptide nucleic acids
- These backbones are substantially non-ionic under neutral conditions, in contrast to the highly charged phosphodiester backbone of naturally occurring nucleic acids. This results in two advantages.
- the PNA backbone exhibits improved hybridization kinetics. PNAs have larger changes in the melting temperature (Tm) for mismatched versus perfectly matched basepairs. DNA and RNA typically exhibit a 2-4°C drop in Tm for an internal mismatch. With the non-ionic PNA backbone, the drop is closer to 7-9°C. This allows for better detection of mismatches.
- hybridization of the bases attached to these backbones is relatively insensitive to salt concentration. This is particularly advantageous in the systems of the present invention, as a reduced salt hybridization solution has a lower Faradaic current than a physiological salt solution (in the range of 150 mM).
- the nucleic acids may be single stranded or double stranded, as specified, or contain portions of both double stranded or single stranded sequence.
- the nucleic acid may be DNA, both genomic and cDNA, RNA or a hybrid, where the nucleic acid contains any combination of deoxyribo- and ribo- nucleotides, and any combination of bases, including uracil, adenine, thymine, cytosine, guanine, inosine, xathanine hypoxathanine, isocytosine, isoguanine, etc.
- nucleoside includes nucleotides and nucleoside and nucleotide analogs, and modified nucleosides such as amino modified nucleosides.
- nucleoside includes non-naturally occuring analog structures.
- the individual units of a peptide nucleic acid, each containing a base, are referred to herein as a nucleoside.
- conductive oligomer herein is meant a substantially conducting oligomer, preferably linear, some embodiments of which are referred to in the literature as “molecular wires”.
- substantially conducting herein is meant that the rate of electron transfer through the conductive oligomer is faster than the rate of electron transfer through single stranded nucleic acid, such that the conductive oligomer is not the rate limiting step in the detection of hybridization, although as noted below, systems which use spacers that are the rate limiting step are also acceptable. Stated differently, the resistance of the conductive oligomer is less than that of the nucleic acid.
- the rate of electron transfer through the conductive oligomer is faster than the rate of electron transfer through double stranded nucleic acid, i.e. through the stacked ⁇ -orbitals of the double helix.
- the conductive oligomer has substantially overlapping ⁇ -orbitals, i.e. conjugated ⁇ -orbitals, as between the monomeric units of the conductive oligomer, although the conductive oligomer may also contain one or more sigma ( ⁇ ) bonds.
- a conductive oligomer may be defined functionally by its ability to inject or receive electrons into or from an attached nucleic acid.
- the conductive oligomer is more conductive than the insulators as defined herein.
- the conductive oiigomers have a conductivity, S, of from between about 10 "6 to about 10 4 ⁇ "1 cr ⁇ r 1 , with from about 10 5 to about 10 3 ⁇ "1 cm “1 being preferred, with these S values being calculated for molecules ranging from about 20A to about 200A.
- insulators have a conductivity S of about 10 "7 ⁇ "1 cn ⁇ 1 or lower, with less than about 10 "8 ⁇ ⁇ 1 cn ⁇ 1 being preferred. See generally Gardner et al., Sensors and Actuators A 51 (1995) 57-66, incorporated herein by reference.
- Desired characteristics of a conductive oligomer include high conductivity, sufficient solubility in organic solvents and/or water for synthesis and use of the compositions of the invention, and preferably chemical resistance to reactions that occur i) during nucleic acid synthesis (such that nucleosides containing the conductive oiigomers may be added to a nucleic acid synthesizer during the synthesis of the compositions of the invention), ii) during the attachment of the conductive oligomer to an electrode, or iii) during hybridization assays.
- oiigomers of the invention comprise at least two monomeric subunits, as described herein.
- oiigomers include homo- and hetero-oligomers, and include polymers.
- the conductive oligomer has the structure depicted in Structure 2:
- the conductive oligomer of Structure 2 may be attached to electron transfer moieties, such as electrodes, transition metal complexes, organic electron transfer moieties, and metallocenes, and to nucleic acids, or to several of these.
- electron transfer moieties such as electrodes, transition metal complexes, organic electron transfer moieties, and metallocenes
- nucleic acids or to several of these.
- the conductive oiigomers depicted herein will be attached at the left side to an electrode; that is, as depicted in
- Y is an aromatic group
- n is an integer from 1 to 50
- g is either 1 or zero
- e is an integer from zero to 10
- m is zero or 1.
- D is preferably carbonyl, or a heteroatom moiety, wherein the heteroatom is selected from oxygen, sulfur, nitrogen, silicon or phosphorus.
- suitable heteroatom moieties include, but are not limited to, -NH and -NR, wherein R is as defined herein; substituted sulfur; sulfonyl (-S0 2 -) sulfoxide (-SO-); phosphine oxide (-PO- and -RPO-); and thiophosphine (-PS- and -RPS-).
- sulfur derivatives are not preferred.
- aromatic group or grammatical equivalents herein is meant an aromatic monocyclic or polycyclic hydrocarbon moiety generally containing 5 to 14 carbon atoms (although larger polycyclic rings structures may be made) and any carbocylic ketone or thioketone derivative thereof, wherein the carbon atom with the free valence is a member of an aromatic ring.
- Aromatic groups include arylene groups and aromatic groups with more than two atoms removed. For the purposes of this application aromatic includes heterocycle.
- Heterocycle or “heteroaryl” means an aromatic group wherein 1 to 5 of the indicated carbon atoms are replaced by a heteroatom chosen from nitrogen, oxygen, sulfur, phosphorus, boron and silicon wherein the atom with the free valence is a member of an aromatic ring, and any heterocyclic ketone and thioketone derivative thereof.
- heterocycle includes thienyl, furyl, pyrrolyl, pyrimidinyl, oxalyl, indolyl, purinyl, quinolyl, isoquinolyl, thiazolyl, imidozyl, etc.
- the Y aromatic groups of the conductive oligomer may be different, i.e.
- the conductive oligomer may be a heterooligomer. That is, a conductive oligomer may comprise a oligomer of a single type of Y groups, or of multiple types of Y groups. Thus, in a preferred embodiment, when a barrier monolayer is used as is described below, one or more types of Y groups are used in the conductive oligomer within the monolayer with a second type(s) of Y group used above the monolayer level.
- the conductive oligomer may comprise Y groups that have good packing efficiency within the monolayer at the electrode surface, and a second type(s) of Y groups with greater flexibility and hydrophilicity above the monolayer level to facilitate nucleic acid hybridization.
- unsubstituted benzyl rings may comprise the Y rings for monolayer packing, and substituted benzyl rings may be used above the monolayer.
- substituted benzyl rings may be used above the monolayer.
- heterocylic rings either substituted or unsubstituted, may be used above the monolayer.
- heterooligomers are used even when the conductive oligomer does not extend out of the monolayer.
- the aromatic group may be substituted with a substitution group, generally depicted herein as R.
- R groups may be added as necessary to affect the packing of the conductive oiigomers, i.e. when the nucleic acids attached to the conductive oiigomers form a monolayer on the electrode, R groups may be used to alter the association of the oiigomers in the monolayer. R groups may also be added to 1) alter the solubility of the oligomer or of compositions containing the oiigomers; 2) alter the conjugation or electrochemical potential of the system; and 3) alter the charge or characteristics at the surface of the monolayer.
- R groups are preferred to increase solubility when solution synthesis is done.
- the R groups, and their positions, are chosen to minimally effect the packing of the conductive oiigomers on a surface, particularly within a monolayer, as described below.
- only small R groups are used within the monolayer, with larger R groups generally above the surface of the monolayer.
- attachment of methyl groups to the portion of the conductive oligomer within the monolayer to increase solubility is preferred, with attachment of longer alkoxy groups, for example, C3 to C10, is preferably done above the monolayer surface.
- this generally means that attachment of sterically significant R groups is not done on any of the first two or three oligomer subunits, depending on the length of the insulator molecules.
- alkyl group or grammatical equivalents herein is meant a straight or branched chain alkyl group, with straight chain alkyl groups being preferred. If branched, it may be branched at one or more positions, and unless specified, at any position.
- the alkyl group may range from about 1 to about 30 carbon atoms (C1 -C30), with a preferred embodiment utilizing from about 1 to about 20 carbon atoms (C1 -C20), with about C1 through about C12 to about C15 being preferred, and C1 to C5 being particularly preferred, although in some embodiments the alkyl group may be much larger.
- the base may contain additional modifications as needed, i.e. the carbonyl or amine groups may be altered or protected, for example as is depicted in Figure 3 or 18.
- Preferred embodiments of Structure 12 include Z is a methylene or ethylene.
- the amide attachment can also be done using an amino group of the base, either a naturally occurring amino group such as in cytidine or adenidine, or from an amino-modified base as are known in the art.
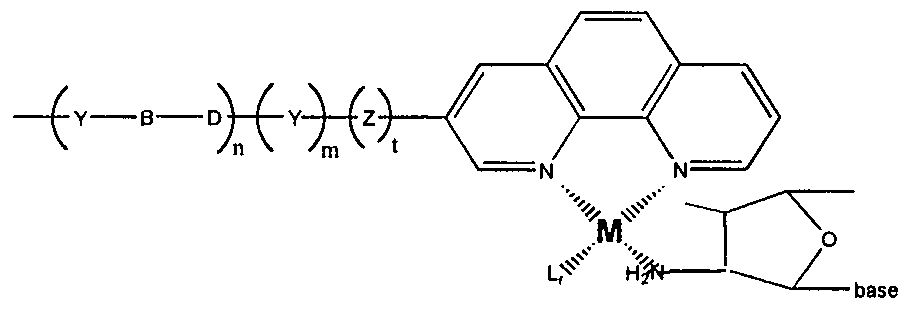
- M is a metal atom, with transition metals being preferred Suitable transition metals for use in the invention include, but are not limited to, cadmium (Cd), copper (Cu), cobalt (Co), palladium (Pd), zinc (Zn), iron (Fe), ruthenium (Ru), rhodium (Rh), osmium (Os), rhenium (Re), platinium (Pt), scandium (Sc), titanium (Ti), Vanadium (V), chromium (Cr), manganese (Mn), nickel (Ni), Molybdenum (Mo), technetium (Tc), tungsten (W), and indium (Ir) That is, the first series of transition metals, the platinum metals (Ru, Rh, Pd, Os, Ir and Pt), along with Fe, Re, W, Mo and Tc, are preferred Particularly preferred are ruthenium, rhenium, osmium, platinium, cobalt
- L are the co-hgands, that provide the coordination atoms for the binding of the metal ion
- the number and nature of the co-hgands will depend on the coordination number of the metal ion.
- Mono-, di- or polydentate co-ligands may be used at any position.
- the metal has a coordination number of six
- the L from the terminus of the conductive oligomer, the L contributed from the nucleic acid, and r, add up to six.
- r may range from zero (when all coordination atoms are provided by the other two ligands) to four, when all the co-ligands are monodentate.
- r will be from 0 to 8, depending on the coordination number of the metal ion and the choice of the other ligands.
- the co-ligands can be the same or different. Suitable ligands fall into two categories: ligands which use nitrogen, oxygen, sulfur, carbon or phosphorus atoms (depending on the metal ion) as the coordination atoms (generally referred to in the literature as sigma ( ⁇ ) donors) and organometailic ligands such as metallocene ligands (generally referred to in the literature as pi ( ⁇ ) donors, and depicted herein as L .
- Suitable sigma donating ligands using carbon, oxygen, sulfur and phosphorus are known in the art.
- suitable sigma carbon donors are found in Cotton and Wilkenson, Advanced Organic Chemistry, 5th Edition, John Wiley & Sons, 1988, hereby incorporated by reference; see page 38, for example.
- suitable oxygen ligands include crown ethers, water and others known in the art.
- Phosphines and substituted phosphines are also suitable; see page 38 of Cotton and Wilkenson.
- the oxygen, sulfur, phosphorus and nitrogen-donating ligands are attached in such a manner as to allow the heteroatoms to serve as coordination atoms.
- organometallic ligands are used.
- transition metal organometallic compounds with ⁇ -bonded organic ligands see Advanced Inorganic Chemistry, 5th Ed., Cotton & Wilkinson, John Wiley & Sons, 1988, chapter 26; Organometallics, A Concise Introduction, Elschenbroich et al., 2nd Ed., 1992, VCH; and Comprehensive Organometallic Chemistry II, A Review of the Literature 1982-1994, Abel et al. Ed., Vol. 7, chapters 7, 8, 10 & 11 ,
- ferrocene [(C 5 H 5 ) 2 Fe] and its derivatives are prototypical examples which have been used in a wide variety of chemical (Connelly et al., Chem. Rev. 96:877- 910 (1996), incorporated by reference) and electrochemical (Geiger et al., Advances in Organometallic Chemistry 23:1-93; and Geiger et al., Advances in Organometallic Chemistry 24:87, incorporated by reference) electron transfer or "redox" reactions.
- Metallocene derivatives of a variety of the first, second and third row transition metals are potential candidates as redox moieties that are covalently attached to either the ribose ring or the nucleoside base of nucleic acid.
- Other potentially suitable organometallic ligands include cyclic arenes such as benzene, to yield bis(arene)metal compounds and their ring substituted and ring fused derivatives, of which bis(benzene)chromium is a prototypical example,
- Other acyclic n-bonded ligands such as the allyl(-1) ion, or butadiene yield potentially suitable organometallic compounds, and all such ligands, in conjuction with other ⁇ -bonded and ⁇ - bonded ligands constitute the general class of organometallic compounds in which there is a metal to carbon bond.
- any combination of ligands may be used.
- Preferred combinations include: a) all ligands are nitrogen donating ligands; b) all ligands are organometallic ligands; and c) the ligand at the terminus of the conductive oligomer is a metallocene ligand and the ligand provided by the nucleic acid is a nitrogen donating ligand, with the other ligands, if needed, are either nitrogen donating ligands or metallocene ligands, or a mixture.
- the ligand attached to the nucleic acid is an amino group attached to the 2' or 3' position of a ribose of the ribose-phosphate backbone. This - 29 -
- ligand may contain a multiplicity of amino groups so as to form a polydentate ligand which binds the metal ion.
- Other preferred ligands include cyclopentadiene and phenanthroline.
- the total length of the nucleic acid will depend on its use. Generally, the nucleic acid compositions of the invention are useful as oligonucleotide probes. As is appreciated by those in the art, the length of the probe will vary with the length of the target sequence and the hybridization and wash conditions.
- probe specificity can be increased by adding oligonucleotides on either side of the electron transfer moieties, thus increasing probe specificity without increasing the distance an electron must travel.
- nucleosides may be replaced with nucleic acids.
- an electrode herein is meant a composition, which, when connected to an electronic device, is able to sense a current or charge and convert it to a signal.
- an electrode is an electron transfer moiety as described herein.
- Preferred electodes include, but are not limited to, certain metals and their oxides, including gold; platinum; palladium; silicon; aluminum; metal oxide electrodes including platinum oxide, titanium oxide, tin oxide, indium tin oxide, palladium oxide, silicon oxide, aluminum oxide, molybdenum oxide (Mo 2 0 6 ), tungsten oxide (W0 3 ) and ruthenium oxides; and carbon (including glassy carbon electrodes, graphite and carbon paste).
- Preferred electrodes include gold, silicon, carbon and metal oxide electrodes.
- the covalent attachment of the conductive oligomer containing the nucleoside may be accomplished in a variety of ways, depending on the electrode and the conductive oligomer used. Generally, some type of linker is used, as depicted below as "A" in Structure 23, where X is the conductive oligomer, and the hatched surface is the electrode:
- A is a linker or atom.
- A may be a sulfur moiety when a gold electrode is used.
- A may be a silicon (silane) moiety attached to the oxygen of the oxide (see for example Chen et al., Langmuir 10:3332-3337 (1994); Lenhard et al., J.
- A may be an amino moiety (preferably a primary amine; see for example Deinhammer et al., Langmuir 10:1306-1313 (1994)).
- preferred A moieties include, but are not limited to, silane moieties, sulfur moieties (including alkyl sulfur moieties), and amino moieties.
- epoxide type linkages with redox polymers such as are known in the art are not used.
- the conductive oligomer may be attached to the electrode with more than one "A" moiety; the "A" moieties may be the same or different.
- the electrode is a gold electrode
- "A" is a sulfur atom or moiety, such as generally depicted below in Structure 27
- multiple sulfur atoms may be used to attach the conductive oligomer to the electrode, such as is generally depicted below in Structures 24, 25 and 26.
- other such structures can be made.
- the A moiety is just a sulfur atom, but substituted sulfur moieties may also be used.
- the electrode is a carbon electrode, i.e. a glassy carbon electrode, and attachment is via a nitrogen of an amine group.
- a representative structure is depicted in Structure 28. Again, additional atoms may be present, i.e. Z type linkers.
- the oxygen atom is from the oxide of the metal oxide electrode.
- the Si atom may also contain other atoms, i.e. be a silicon moiety containing substitution groups.
- electrodes are made that comprise conductive oiigomers attached to nucleic acids for the purposes of hybridization assays, as is more fully described herein.
- electrodes can be made that have a single species of nucleic acid, i.e. a single nucleic acid sequence, or multiple nucleic acid species.
- oligonucleotide arrays are well known in the art.
- techniques are known for "addressing" locations within an electrode and for the surface modification of electrodes.
- arrays of different nucleic acids are laid down on the electrode, each of which are covalently attached to the electrode via a conductive linker.
- the number of different probe species of oligonucleotides may vary widely, from one to thousands, with from about 4 to about 100,000 being preferred, and from about 10 to about 10,000 being particularly preferred.
- the electrode further comprises a passivation agent, preferably in the form of a monolayer on the electrode surface.
- a passivation agent layer facilitates the maintenance of the nucleic acid away from the electrode surface.
- a passivation agent serves to keep charge carriers away from the surface of the electrode. Thus, this layer helps to prevent electrical contact between the electrodes and the electron transfer moieties, or between the electrode and charged species within the solvent. Such contact can result in a direct "short circuit" or an indirect short circuit via charged species which may be present in the sample.
- the monolayer of passivation agents is preferably tightly packed in a uniform layer on the electrode surface, such that a minimum of "holes" exist.
- the passivation agent may not be in the form of a monolayer, but may be present to help the packing of the conductive oiigomers or other characteristics.
- the passivation agents thus serve as a physical barrier to block solvent community to the electrode.
- the passivation agents themselves may in fact be either (1) conducting or (2) nonconducting, i.e. insulating, molecules.
- the passivation agents are conductive oiigomers, as described herein, with or without a terminal group to block or decrease the transfer of charge to the electrode.
- Other passivation agents which may be conductive include oiigomers of -(CF 2 ) n -, -(CHF) n - and -(CFR) n -.
- the passivation agents are insulator moieties.
- an “insulator” is a substantially nonconducting oligomer, preferably linear.
- substantially nonconducting herein is meant that the rate of electron transfer through the insulator is slower than the rate of electron transfer through the stacked ⁇ -orbitals of double stranded nucleic acid.
- the electrical resistance of the insulator is higher than the electrical resistance of the nucleic acid.
- the rate of electron transfer through the insulator is slower than or comparable to the rate through single stranded nucleic acid.
- the rate of electron transfer through the insulator is preferrably slower than the rate through the conductive oiigomers described herein.
- the insulators have a conductivity, S, of about 10 "7 ⁇ "1 c ⁇ r 1 or lower, with less than about 10 "8 ⁇ "1 cn ⁇ 1 being preferred. See generally Gardner et al., supra.
- insulators are alkyl or heteroalkyl oiigomers or moieties with sigma bonds, although any particular insulator molecule may contain aromatic groups or one or more conjugated bonds.
- heteroalkyl herein is meant an alkyl group that has at least one heteroatom, i.e. nitrogen, oxygen, sulfur, phosphorus, silicon or boron included in the chain.
- the insulator may be quite similar to a conductive oligomer with the addition of one or more heteroatoms or bonds that serve to inhibit or slow, preferably substantially, electron transfer.
- the passivation agents may be substituted with R groups as defined herein to alter the packing of the moieties or conductive oiigomers on an electrode, the hydrophilicity or hydrophobicity of the insulator, and the flexibility, i.e. the rotational, torsional or longitudinal flexibility of the insulator.
- R groups as defined herein to alter the packing of the moieties or conductive oiigomers on an electrode, the hydrophilicity or hydrophobicity of the insulator, and the flexibility, i.e. the rotational, torsional or longitudinal flexibility of the insulator.
- branched alkyl groups may be used.
- the terminus of the passivation agent, including insulators may contain an additional group to influence the exposed surface of the monolayer. For example, there may be negatively charged groups on the terminus to form a negatively charged surface such that when the nucleic acid is DNA or RNA the nucleic acid is repelled or prevented from lying down on the surface, to facilitate hybridization.
- Preferred passivation agent terminal groups include -NH 2 , -OH, -COOH, -CH 3 , and (poly)alkyloxides such as (poly)ethylene glycol, with -OCH 2 CH 2 OH, -(OCH2CH20) 2 H and -(OCH 2 CH 2 0) 3 H being preferred and the latter being particularly preferred.
- the length of the passivation agent will vary as needed. As outlined above, it appears that hybridization is more efficient at a distance from the surface. Thus, the length of the passivation agents is similar to the length of the conductive oiigomers, as outlined above. In addition, the conductive oiigomers may be basically the same length as the passivation agents or longer than them, resulting in the nucleic acids being more accessible to the solvent for hybridization.
- the monolayer may comprise a single type of passivation agent, including insulators, or different types.
- Suitable insulators include, but are not limited to, -(CH 2 ) n -, -(CRH) n -, and -(CR 2 ) n -, ethylene glycol or derivatives using other heteroatoms in place of oxygen, i.e. nitrogen or sulfur (sulfur derivatives are not preferred when the electrode is gold).
- the passivation agents are generally attached to the electrode in the same manner as the conductive oligomer, and may use the same "A" linker as defined above.
- the compositions of the present invention find use in standard nucleic acid assays, such as general array-type technologies, i.e. the electrode may serve just as a solid support, with detection proceeding using techniques well known in the art, such as fluoroscence or radioisotope labelling.
- the compositions may comprise a conductive oligomer covalently attached to a nucleoside or nucleic acid. It will be recognized by those in the art that the conductive oiigomers in this embodiment may not be functioning as conductive oiigomers but rather as linkers that can be used to keep the nucleic acids off the surface.
- the composition comprises (a) a solid support comprising a monolayer of passivation agent; (b) a nucleic acid comprising at least one nucleoside, wherein the nucleic acid is covalently attached to the solid support with a linker.
- the solid support is the electrode, which is not necessarily functioning as an electron transfer moiety in this embodiment.
- the monolayer of passivation agent is shown herein to result in excellent hybridization kinetics and can therefore be quite useful in both electron-transfer based and traditional nucleic acid detection schemes.
- the linkers are preferably the conductive oiigomers of the invention, although as outlined above, they may not be functioning as conductive moieties.
- the conductive oligomer, or linker in this case, may have the structure depicted in Structures 2, 3, 4, 9 or 10.
- each nucleic acid be the same, as an "anchor sequence", such that a second sequence can be added which contains the probe sequence and a sequence complementary to the anchor sequence.
- anchor sequence such that a second sequence can be added which contains the probe sequence and a sequence complementary to the anchor sequence.
- compositions are provided comprising a conductive oligomer covalently attached to an electrode and to a first single stranded anchor sequence.
- a second single stranded nucleic acid which contains a probe region and a region substantially complementary to the anchor sequence, such that a first hybridization complex is formed between the two complementary anchor regions, leaving the probe region as a single stranded region.
- a target sequence which is substantially complementary to the probe region is then added to form a second hybridization complex.
- the second hybridization complex is then detected, for example by labelling the target nucleic acid as is well known in the art.
- compositions comprising electrodes with conductive oiigomers attached to probe nucleic acids, without second electron transfer moieties, and soluble second probe sequences with second electron transfer moieties.
- the target sequence which contains a first target domain for the first probe sequence and a second target domain for the second probe sequence, which preferably are adjacent, electron transfer may occur.
- compositions of the present invention comprise a conductive oligomer, covalently attached to both an electrode, which serves as a first electron transfer moiety, and a nucleic acid, which has at least a second covalently attached electron transfer moiety.
- the conductive oligomer and the second electron transfer moiety may be attached at any position of the nucleic acid.
- the electron transfer moieties are transition metal complexes.
- Transition metals are those whose atoms have a partial or complete d shell of electrons. Suitable transition metals for use in the invention are listed above.
- transition metals are complexed with a variety of ligands, L, defined above, to form suitable transition metal complexes, as is well known in the art.
- 10-methylphenoxyazine chloride 10-methylphenoxyazine chloride), methylene blue; Nile blue A (aminoaphthodiethylaminophenoxazine sulfate), indigo-5,5',7,7'-tetrasulfonic acid, indigo-5,5',7-trisulfonic acid; phenosafranine, indigo-5- monosulfonic acid; safranine T; bis(dimethylglyoximato)-iron(ll) chloride; induline scarlet, neutral red, anthracene, coronene, pyrene, 9-phenylanthracene, rubrene, binaphthyl, DPA, phenothiazene, fluoranthene, phenanthrene, chrysene, 1 ,8-diphenyl-1 ,3,5,7-octatetracene, naphthalene, acenaphthalene,
- the electron donors and acceptors are redox proteins as are known in the art. However, redox proteins in many embodiments are not preferred.
- these electron transfer moieties are covalently attached to the nucleic acid in a variety of positions.
- the attachment is via attachment to the base of the nucleoside, or via attachment to the backbone of the nucleic acid, including either to a ribose of the ribose-phosphate backbone or to a phosphate moiety.
- the compositions of the invention are designed such that the electron transfer moieties are as close to the " ⁇ -way" as possible without significantly disturbing the secondary and tertiary structure of the double helical nucleic acid, particularly the Watson-Crick basepairing.
- the attachment can be via a conductive oligomer, which is used as outlined above with a nucleoside and an electrode; that is, an electron transfer moiety may be covalently attached to a conductive oligomer at one end and to a nucleoside at the other, thus forming a general structure depicted in Structure 30:
- ETM is an electron transfer moiety
- X is a conductive oligomer
- q is an integer from zero to about 25, with preferred q being from about 2 to about 10.
- linker moieties for example as are generally described herein as "Z" may also be present between the nucleoside and the conductive oligomer, and/or between the conductive oligomer and the electron transfer moiety.
- the depicted nucleosides may be either terminal or internal nucleosides, and are usually separated by a number of nucleosides.
- the second electron transfer moiety is attached to the base of a nucleoside, as is generally outlined above for attachment of the conductive oligomer.
- this attachment does not perturb the Watson-Crick basepairing of the base to which the electron transfer moiety is attached, as long as the moiety is not too large. In fact, it appears that attachment at this site actually results in less perturbation than attachment at the ribose of the ribose-phosphate backbone, as measured by nucleic acid melting curves.
- the size of the second electron transfer moiety should be such that the structure of double stranded nucleic acid containing the base-attached electron transfer moiety is not significantly disrupted, and will not disrupt the annealing of single stranded nucleic acids.
- ligands and full second electron transfer moieties are generally smaller than the size of the major groove of double stranded nucleic acid.
- the second electron transfer moiety can be attached to the base of a terminal nucleoside
- a probe when the target sequence to be detected is n nucleosides long, a probe can be made which has the second electron transfer moiety attached at the n base.
- the probe may contain an extra terminal nucleoside at an end of the nucleic acid (n + 1 or n + 2), which are used to covalently attach the electron transfer moieties but which do not participate in basepair hybridization.
- the terminal nucleoside containing the electron transfer moiety covalently attached at the base be directly adjacent to Watson-Crick basepaired nucleosides; that is, the electron transfer moiety should be as close as possible to the stacked ⁇ -orbitals of the bases such that an electron travels through a minimum of ⁇ bonds to reach the " ⁇ -way", or alternatively can otherwise electronically contact the ⁇ -way.
- the covalent attachment to the base will depend in part on the second electron transfer moiety chosen, but in general is similar to the attachment of conductive oiigomers to bases, as outlined above.
- the second electron transfer moiety is a transition metal complex, and thus attachment of a suitable metal ligand to the base leads to the covalent attachment of the electron transfer moiety.
- similar types of linkages may be used for the attachment of organic electron transfer moieties, as will be appreciated by those in the art.
- the C4 attached amino group of cytosine, the C6 attached amino group of adenine, or the C2 attached amino group of guanine may be used as a transition metal ligand, although in this embodiment attachment at a terminal base is preferred since attachment at these positions will perturb Watson-Crick basepairing.
- Ligands containing aromatic groups can be attached via acetylene linkages as is known in the art (see Comprehensive Organic Synthesis, Trost et al., Ed., Pergamon Press, Chapter 2.4: Coupling Reactions Between sp 2 and sp Carbon Centers, Sonogashira, pp521-549, and pp950-953, hereby incorporated by reference).
- Structure 31 depicts a representative structure in the presence of the metal ion and any other necessary ligands; Structure 31 depicts uridine, although as for all the structures herein, any other base may also be used.
- L a is a ligand, which may include nitrogen, oxygen, sulfur or phosphorus donating ligands or organometallic ligands such as metallocene ligands.
- Suitable L a ligands include, but not limited to, phenanthroline, imidazole, bpy and terpy.
- L r and M are as defined above.
- a conductive oligomer may be included between the nucleoside and the electron transfer moiety.
- the linkage may be done using a linker, which may utilize an amide linkage (see generally Telser et al., J. Am. Chem. Soc. 111 :7221-7226 (1989); Telser et al., J. Am. Chem. Soc. 111 :7226-7232 (1989), both of which are expressly incorporated by reference).
- a linker which may utilize an amide linkage (see generally Telser et al., J. Am. Chem. Soc. 111 :7221-7226 (1989); Telser et al., J. Am. Chem. Soc. 111 :7226-7232 (1989), both of which are expressly incorporated by reference).
- amide linkage see generally Telser et al., J. Am. Chem. Soc. 111 :7221-7226 (1989); Telser et al., J. Am. Chem. Soc. 111 :7226-7232 (1989), both
- L is a ligand as defined above, with L r and M as defined above as well.
- L is amino, phen, byp and terpy.
- the second electron transfer moiety attached to a nucleoside is a metallocene; i.e. the L and L r of Structure 32 are both metallocene ligands, L m , as described above.
- Structure 33 depicts a preferred embodiment wherein the metallocene is ferrocene, and the base is uridine, although other bases may be used:
- the invention provides metallocenes covalently attached to nucleosides.
- the metallocene is attached to the base of a nucleoside.
- the metallocene is attached to the ribose of a nucleoside.
- modification groups may be used as a transition metal ligand, or as a chemically functional moiety for attachment of other transition metal ligands and organometallic ligands, or organic electron donor moieties as will be appreciated by those in the art.
- a linker such as depicted herein for "Z” may be used as well, or a conductive oligomer between the ribose and the electron transfer moiety.
- Preferred embodiments utilize attachment at the 2' or 3' position of the ribose, with the 2' position being preferred.
- a metallocene serves as the second electron transfer moiety, and is attached via an amide bond as depicted below in Structure 34.
- the examples outline the synthesis of a preferred compound when the metallocene is ferrocene.
- Amine linkages or linkages via other heteroatoms, are also possible.
- the second electron transfer moiety is attached to a phosphate at any position of the ribose-phosphate backbone of the nucleic acid.
- phosphodiester bond analogs such as phosphoramide or phosphoramidite linkages may be incorporated into a nucleic acid as a transition metal ligand (see PCT publication WO 95/15971 , incorporated by reference).
- the conductive oiigomers depicted in Structures 16 and 17 may be replaced by electron transfer moieties; alternatively, the electron transfer moieties may be added to the free terminus of the conductive oligomer.
- Preferred electron transfer moieties for covalent attachment to a single stranded nucleic acid include, but are not limited to, transition metal complexes, including metallocenes and substituted metallocenes such as metalloceneophanes, and complexes of Ru, Os, Re and Pt. Particularly preferred are ferrocene and its derivatives (particularly pentamethylferrocene and ferroceneophane) and complexes of transition metals including Ru, Os, Re and Pt containing one or more amine or polyamine, imidazole, phenathroline, pyridine, bipyridine and or terpyridine and their derivatives.
- additional preferred ligands include the diimine dithiolate complexes such as quinoxaline-2,3-dithiolate complexes.
- the invention provides compositions containing electrodes as a first electron transfer moiety linked via a conductive oligomer to a nucleic acid which has at least a second electron transfer moiety covalently attached.
- Any combination of positions of electron transfer moiety attachment can be made; i.e. an electrode at the 5' terminus, a second electron transfer moiety at an internal position; electrode at the 5' terminus, second moiety at the 3' end; second moiety at the 5' terminus, electrode at an internal position; both electrode and second moiety at internal positions; electrode at an internal position, second moiety at the 3' terminus, etc.
- a preferred embodiment utilizes both the electrode and the second electron transfer moiety attached to internal nucleosides.
- compositions of the invention may additionally contain one or more labels at any position.
- label herein is meant an element (e.g. an isotope) or chemical compound that is attached to enable the detection of the compound.
- Preferred labels are radioactive isotopic labels, and colored or fluorescent dyes.
- the labels may be incorporated into the compound at any position.
- the compositions of the invention may also contain other moieties such as cross-linking agents to facilitate cross-linking of the target-probe complex. See for example, Lukhtanov et al., Nucl. Acids. Res. 24(4):683 (1996) and Tabone et al., Biochem. 33:375 (1994), both of which are expressly incorporated by reference.
- compositions of the invention are generally synthesized as outlined below, generally utilizing techniques well known in the art. As will be appreciated by those in the art, many of the techniques outlined below are directed to nucleic acids containing a ribose-phosphate backbone. However, as outlined above, many alternate nucleic acid analogs may be utilized, some of which may not contain either ribose or phosphate in the backbone. In these embodiments, for attachment at positions other than the base, attachment is done as will be appreciated by those in the art, depending on the backbone. Thus, for example, attachment can be made at the carbon atoms of the PNA backbone, as is described below, or at either terminus of the PNA.
- compositions may be made in several ways.
- a preferred method first synthesizes a conductive oligomer attached to the nucleoside, with addition of additional nucleosides followed by attachment to the electrode.
- a second electron transfer moiety if present, may be added prior to attachment to the electrode or after.
- the whole nucleic acid may be made and then the completed conductive oligomer added, followed by attachment to the electrode.
- the conductive oligomer and monolayer (if present) are attached to the electrode first, followed by attachment of the nucleic acid.
- the latter two methods may be preferred when conductive oiigomers are used which are not stable in the solvents and under the conditions used in traditional nucleic acid synthesis.
- compositions of the invention are made by first forming the conductive oligomer covalently attached to the nucleoside, followed by the addition of additional nucleosides to form a nucleic acid, including, if present, a nucleoside containing a second electron transfer moiety, with the last step comprising the addition of the conductive oligomer to the electrode.
- the attachment of the conductive oligomer to the nucleoside may be done in several ways.
- all or part of the conductive oligomer is synthesized first (generally with a functional group on the end for attachment to the electrode), which is then attached to the nucleoside. Additional nucleosides are then added as required, with the last step generally being attachment to the electrode.
- oligomer units are added one at a time to the nucleoside, with addition of additional nucleosides and attachment to the electrode.
- FIG. 1 A general outline of a preferred embodiment is depicted in Figure 1 , using a phenyl-acetylene oligomer as generally depicted in Structure 5.
- Other conductive oiigomers will be made using similar techniques, such as heterooligomers, or as known in the art.
- conductive oiigomers using alkene or acetylene bonds are made as is known in the art.
- the conductive oligomer is then attached to a nucleoside that may contain one (or more) of the oligomer units, attached as depicted herein.
- attachment is to a ribose of the ribose-phosphate backbone.
- Figure 1 depicts attachment via an amide linkage
- Figures 2 and 16 depict the synthesis of compounds with amine linkages.
- a representative synthesis is shown in Figure 16.
- attachment is via a phosphate of the ribose-phosphate backbone.
- Figure 4 synthesis of Structure 16 type compounds
- Figure 5 synthesis of Structure 16 type compounds
- Z is an ethylene linker, although other linkers may be used as well, as will be appreciated by those in the art.
- attachment is via the base.
- a general scheme is depicted in Figure 3, using uridine as the nucleoside and a phenylene-acetylene conductive oligomer.
- amide linkages are also possible, such as depicted in Structure 12, using techniques well known in the art.
- protecting groups may be added to the base prior to addition of the conductive oiigomers, as is generally outlined in Figures 18 and 19.
- the palladium cross-coupling reactions may be altered to prevent dimerization problems; i.e. two conductive oiigomers dimerizing, rather than coupling to the base.
- attachment to the base may be done by making the nucleoside with one unit of the oligomer, followed by the addition of others.
- terminal deoxynucleotidyltransferase may be used.
- Terminal deoxynucleotidyltransferase Terminal deoxynucleotidyltransferase. In The Enzymes, Vol 14A. P.D. Boyer ed. pp
- the amino nucleoside is converted to the phosphoramidite or H-phosphonate form, which are then used in solid-phase or solution syntheses of oligonucleotides.
- the modified nucleoside either for attachment at the ribose (i.e. amino- or thiol-modified nucleosides) or the base, is incorporated into the oligonucleotide at either an internal position or the 5' terminus. This is generally done in one of two ways.
- the 5' position of the ribose is protected with 4',4-dimethoxytrityl (DMT) followed by reaction with either 2-cyanoethoxy-bis-diisopropylaminophosphine in the presence of diisopropylammonium tetrazolide, or by reaction with chlorodiisopropylamino 2'- cyanoethyoxyphosphine, to give the phosphoramidite as is known in the art; although other techniques may be used as will be appreciated by those in the art. See Gait, supra; Caruthers, Science 230:281
- a preferred method utilizes the attachment of the modified nucleoside to controlled pore glass (CPG) or other oligomeric supports.
- CPG controlled pore glass
- the modified nucleoside is protected at the 5' end with DMT, and then reacted with succinic anhydride with activation.
- the resulting succinyl compound is attached to CPG or other oligomeric supports as is known in the art.
- Further phosphoramidite nucleosides are added, either modified or not, to the 5' end after deprotection.
- a preferred method comprises 1) adding an ethyl pyridine or trimethylsilylethyl protecting group to a sulfur atom attached to a first subunit of a conductive oligomer, generally done by adding a vinyl pyridine or trimethylsilylethyl group to a sulfhydryl; 2) adding additional subunits to form the conductive oligomer; 3) adding at least a first nucleoside to the conductive oligomer; 4) adding additional nucleosides to the first nucleoside to form a nucleic acid; 5) attaching the conductive oligomer to the gold electrode. This may also be done in the absence of nucleosides, as is described in the Examples.
- the above method may also be used to attach passivation molecules to a gold electrode.
- a monolayer of passivation agents is added to the electrode.
- the chemistry of addition is similar to or the same as the addition of conductive oiigomers to the electrode, i.e. using a sulfur atom for attachment to a gold electrode, etc.
- compositions comprising monolayers in addition to the conductive oiigomers covalently attached to nucleic acids (with or without second electron transfer moieties) may be made in at least one of five ways: (1 ) addition of the monolayer, followed by subsequent addition of the conductive oligomer-nucleic acid complex; (2) addition of the conductive oligomer-nucleic acid complex followed by addition of the monolayer; (3) simultaneous addition of the monolayer and conductive oligomer-nucleic acid complex; (4) formation of a monolayer (using any of 1 , 2 or 3) which includes conductive oiigomers which terminate in a functional moiety suitable for attachment of a completed nucleic acid; or (5) formation of a monolayer which includes conductive oiigomers which terminate in a functional moiety suitable for nucleic acid synthesis, i.e.
- nucleic acid is synthesized on the surface of the monolayer as is known in the art.
- suitable functional moieties include, but are not limited to, nucleosides, amino groups, carboxyl groups, protected sulfur moieties, or hydroxyl groups for phosphoramidite additions.
- the examples describe the formation of a monolayer on a gold electrode using the preferred method (1).
- the nucleic acid is a peptide nucleic acid or analog.
- the invention provides peptide nucleic acids with at least one covalently attached chemical substituent.
- chemical substituent herein is meant any chemical or biological moiety.
- Preferred chemical substituents include, but are not limited to, chemical functional moieties such as amino groups, thiol groups, carbon atoms, etc., which can be used to attach other moieties; labels; signaling moieties which can be used for detection; etc.
- chemical substitutents include, but are not limited to, electron transfer moieties, including electrodes, transition metal complexes, and organic electron transfer moieties; other transition metal complexes; other labels including fluoroscent labels, radioisotope labels and chemiluminescent labels; haptens such as biotin, avidin, and digoxigenin; antigens; proteins such as antibodies, ligands, receptors, and enzymes; conductive oiigomers and other polymers; and other components of binding pairs such as nucleic acids.
- the chemical substituents are covalently attached to an monomeric subunit of the PNA.
- monomeric subunit of PNA herein is meant the -NH-CH 2 CH 2 -N(COCH 2 -Base)-CH 2 - CO- monomer, or derivatives (herein included within the definition of "nucleoside") of PNA.
- the number of carbon atoms in the PNA backbone may be altered; see generally Nielsen et al., Chem. Soc. Rev. 1997 page 73, which discloses a number of PNA derivatives, herein expressly incorporated by reference.
- the amide bond linking the base to the backbone may be altered; phosphoramide and sulfuramide bonds may be used.
- the chemical substituents are attached to an internal monomeric subunit.
- internal herein is meant that the monomeric subunit is not either the N-terminal monomeric subunit or the C-terminal monomeric subunit.
- a modified monomeric subunit is synthesized with a chemical substituent, or a functional group for its attachment, and then the base is added and the modified monomer can be incorporated into a growing PNA chain.
- Figure 31 depicts the synthesis of a conductive oligomer covalently attached to the backbone of a PNA monomeric subunit
- Figure 32 depicts the synthesis of a ferrocene attached to the backbone of a monomeric subunit.
- the monomeric subunits with covalently attached chemical substituents are incorporated into a PNA using the techniques outlined in Will et al., Tetrahedron 51 (44): 12069-12082 (1995), and Vanderlaan et al., Tett. Let. 38:2249-2252 (1997), both of which are hereby expressly incorporated in their entirety. These procedures allow the addition of chemical substituents to peptide nucleic acids without destroying the chemical substituents.
- chemical substituents other than electron transfer moieties and transition metal complexes are attached to either or both of the bases of the terminal monomeric subunits.
- preferred chemical substituents include fluoroscent, radioisotope and chemiluminescent labels.
- electrodes may be made that have any combination of nucleic acids, conductive oiigomers and passivation agents.
- conductive oiigomers or passivation agents may be used on a single electrode.
- the probes of the present invention are designed to be complementary to the target sequence, such that hybridization of the target sequence and the probes of the present invention occurs. As outlined below, this complementarity need not be perfect; there may be any number of base pair mismatches which will interfere with hybridization between the target sequence and the single stranded nucleic acids of the present invention. However, if the number of mutations is so great that no hybridization can occur under even the least stringent of hybridization conditions, the sequence is not a complementary target sequence.
- sequence between the nucleosides containing the electron transfer moieties is double stranded, i.e. contains stacked ⁇ -orbitals, such that upon initiation, the complex is capable of transferring at least one electron from one of the electron transfer moieties to the other.
- an electrode may serve as either an electron donor or acceptor, and the choice of the second electron transfer species is made accordingly.
- the first target domain may be directly adjacent to the second target domain, or the first and second target domains may be separated by an intervening target domain. Preferably, there are no gaps between the domains; i.e. they are contiguous.
- the terms "first” and “second” are not meant to confer an orientation of the sequences with respect to the 5'-3' orientation of the target sequence. For example, assuming a 5'-3' orientation of the complementary target sequence, the first target domain may be located either 5' to the second domain, or 3' to the second domain.
- the first single stranded nucleic acid is hybridized to the first target domain, and the second single stranded nucleic acid is hybridized to the second target domain to form a hybridization complex.
- the hybridization complex is then capable of transferring at least one electron between the electron transfer moieties upon initiation.
- compositions comprising a) a single stranded nucleic acid covalently attached to an electrode via a conductive oligomer, and b) a target nucleic acid are made.
- a hybridization indicator is added.
- Hybridization indicators serve as an electron transfer moiety that will preferentially associate with double stranded nucleic acid is added, usually reversibly, similar to the method of Millan et al., Anal. Chem. 65:2317- 2323 (1993); Millan et al., Anal. Chem. 662943-2948 (1994), both of which are hereby expressly incorporated by reference.
- a further embodiment utilizes compositions comprising a) a first single stranded nucleic acid covalently attached to an electrode via a conductive oligomer; b) a second single stranded nucleic acid containing a second electron transfer moiety; and c) an intervening single stranded nucleic acid, which may or may not be labelled or contain an electron transfer moiety.
- the first single stranded nucleic acid hybridizes to the first target domain
- the second single stranded nucleic acid hybridizes to the second target domain
- the intervening nucleic acid hybridizes to the intervening target domain, with electron transfer upon initiation.
- the intervening nucleic acid may be any length, taking into consideration the parameters for the distance between the electron transfer moieties, although it may be a single nucleoside.
- Electron transfer is generally initiated electronically, with voltage being preferred.
- a potential is applied to a sample containing modified nucleic acid probes. Precise control and variations in the applied potential can be via a potentiostat and either a three electrode system (one reference, one sample and one counter electrode) or a two electrode system (one sample and one counter electrode). This allows matching of applied potential to peak electron transfer potential of the system which depends in part on the choice of electron acceptors attached to the nucleic acid and in part on the conductive oligomer used. As described herein, ferrocene is a preferred electron transfer moiety.
- a co-reductant or co-oxidant (collectively, co-redoxant) is used, as an additional electron source or sink.
- co-reductant or co-oxidant (collectively, co-redoxant) is used, as an additional electron source or sink.
- ferrocene as a second ETM attached to the compositions of the invention as described in the examples, has a redox potential of roughly 200 mV in aqueous solution (which changes slightly depending on what the ferrocene is bound to).
- Ferrocyanide an electron source, has a redox potential of roughly 200 mV as well (in aqueous solution). Accordingly, at or above voltages of roughly 200 mV, ferrocene is converted to ferricenium, which then transfers an electron to the nucleic acid. If this nucleic acid is double stranded, transfer proceeds rapidly through the double stranded nucleic acid, through the conductive oligomer, to the electrode.
- the ferricyanide can be oxidized to transfer an electron to the ETM.
- the electron source or co-reductant serves to amplify the signal generated in the system, as the electron source molecules rapidly and repeatedly donate electrons to the second ETM attached to the nucleic acid.
- the rate of electron donation or acceptance will be limited by the rate of diffusion of the co-reductant, which in turn is affected by the concentration and size, etc.
- the input source such as ferrocyanide is unable to be oxided and thus is unable to donate an electron to the ETM; i.e. no electron transfer occurs.
- the use of electron source molecules is only possible when an insulating or passivation layer is present, since otherwise the source molecule will transfer electrons directly to the electrode. Accordingly, in a preferred embodiment, an electron source is used in solution to amplify the signal generated in the presence of hybridized target sequence.
- Iuminol an electron source
- the ferrocene is oxided, and transfers a single electron to the electrode via the conductive oligomer.
- the ETM is unable to accept any electrons from the Iuminol electron source, since the voltages are less than the redox potential of the Iuminol.
- the Iuminol then transfers an electron to the ETM, allowing rapid and repeated electron transfer.
- the electron source (or co-reductant) serves to amplify the signal generated in the system, as the electron source molecules rapidly and repeatedly donate electrons to the second ETM attached to the nucleic acid.
- Luminol has the added benefit of becoming a chemiluminiscent species upon oxidation (see Jirka et al., Analytica Chimica Acta 284:345 (1993)), thus allowing photo-detection of electron transfer through double-stranded nucleic acid.
- luminol can only be oxidized by transferring an electron to the second electron transfer moiety on the nucleic acid (e.g. ferrocene).
- the nucleic acid e.g. ferrocene
- the system when the target sequence is not hybridized to the composition of the invention, the system has a high impedance, resulting in a low photon emission and thus a low (if any) signal from the luminol.
- the second electron transfer moieties In the presence of double stranded nucleic acid, i.e. target sequence hybridization, the second electron transfer moieties have low impedance, thus generating a much larger signal.
- the measure of luminol oxidation by photon emission is an indirect measurement of the ability of the second electron transfer moiety to donate electrons to the electrode.
- photon detection is generally more sensitive than electronic detection, the sensitivity of the system may be increased. Initial results suggest that luminescence may depend on hydrogen peroxide concentration, pH, and luminol concentration, the latter of which appears to be non-linear.
- output electron acceptors or sinks could be used, i.e. the above reactions could be run in reverse, with the ETM such as a metallocene receiving an electron from the electrode, converting it to the metallicenium, with the output electron acceptor then accepting the electron rapidly and repeatedly.
- the ETM such as a metallocene receiving an electron from the electrode, converting it to the metallicenium, with the output electron acceptor then accepting the electron rapidly and repeatedly.
- cobalticenium is the preferred ETM.
- Electron transfer through nucleic acid can be detected in a variety of ways.
- a variety of detection methods may be used, including, but not limited to, optical detection, which includes fluorescence, phosphorescence, luminiscence, chemiluminescence, electrochemiluminescence, and refractive index; and electronic detection, including, but not limited to, amperommetry, voltammetry, capacitance and impedence.
- optical detection which includes fluorescence, phosphorescence, luminiscence, chemiluminescence, electrochemiluminescence, and refractive index
- electronic detection including, but not limited to, amperommetry, voltammetry, capacitance and impedence.
- These methods include time or frequency dependent methods based on AC or DC currents, pulsed methods, lock-in techniques, filtering (high pass, low pass, band pass), and time- resolved techniques including time-resolved fluoroscence.
- all that is required is electron transfer detection; in others, the rate of electron transfer may be determined.
- the efficient transfer of electrons from one end of a nucleic acid double helix to the other results in stereotyped changes in the redox state of both the electron donor and acceptor.
- electron transfer moieties including the complexes of ruthenium containing bipyridine, pyridine and imidazole rings, these changes in redox state are associated with changes in spectral properties.
- possible electron donors and acceptors include all the derivatives listed above for photoactivation or initiation.
- Preferred electron donors and acceptors have characteristically large spectral changes upon oxidation and reduction resulting in highly sensitive monitoring of electron transfer.
- Such examples include Ru(NH 3 ) 4 py and Ru(bpy) 2 im as preferred examples. It should be understood that only the donor or acceptor that is being monitored by absorbance need have ideal spectral characteristics. That is, the electron acceptor can be optically invisible if only the electron donor is monitored for absorbance changes.
- the electron transfer is detected fluorometrically.
- Numerous transition metal complexes, including those of ruthenium, have distinct fluorescence properties. Therefore, the change in redox state of the electron donors and electron acceptors attached to the nucleic acid can be monitored very sensitively using fluorescence, for example with Ru(4,7-biphenyl 2 -phenanthroline) 3 2+
- this compound can be easily measured using standard fluorescence assay techniques. For example, laser induced fluorescence can be recorded in a standard single cell fluorimeter, a flow through "on-line” fluorimeter (such as those attached to a chromatography system) or a multi-sample "plate-reader” similar to those marketed for 96-well immuno assays.
- fluorescence can be measured using fiber optic sensors with nucleic acid probes in solution or attached to the fiber optic. Fluorescence is monitored using a photomuitiplier tube or other light detection instrument attached to the fiber optic.
- the advantage of this system is the extremely small volumes of sample that can be assayed.
- scanning fluorescence detectors such as the Fluorlmager sold by Molecular Dynamics are ideally suited to monitoring the fluorescence of modified nucleic acid molecules arrayed on solid surfaces.
- the advantage of this system is the large number of electron transfer probes that can be scanned at once using chips covered with thousands of distinct nucleic acid probes.
- a reduction in fluorescence associated with hybridization can be measured using these systems.
- An electron transfer "donor” molecule that fluoresces readily when on single stranded nucleic acid (with an “acceptor” on the other end) will undergo a reduction in fluorescent intensity when complementary nucleic acid binds the probe allowing efficient transfer of the excited state electron. This drop in fluorescence can be easily monitored as an indicator of the presence of a target sequence using the same methods as those above.
- electrochemiluminescence is used as the basis of the electron transfer detection.
- electron transfer moieties such as Ru 2+ (bpy) 3
- direct luminescence accompanies excited state decay. Changes in this property are associated with nucleic acid hybridization and can be monitored with a simple photomultiplier tube arrangement (see Blackburn, G. F. Clin. Chem. 37: 1534-1539 (1991); and Juris et al., supra.
- electronic detection is used, including amperommetry, voltammetry, capacitance, and impedence.
- Suitable techniques include, but are not limited to, electrogravimetry; coulometry (including controlled potential coulometry and constant current coulometry); voltametry (cyclic voltametry, pulse voltametry (normal pulse voltametry, square wave voltametry, differential pulse voltametry, Osteryoung square wave voltametry, and coulostatic pulse techniques); stripping analysis (aniodic stripping analysis, cathiodic stripping analysis, square wave stripping voltammetry); conductance measurements (electrolytic conductance, direct analysis); time-dependent electrochemical analyses (chronoamperometry, chronopotentiometry, cyclic chronopotentiometry and amperometry, AC polography, chronogalvametry, and chronocoulometry); AC impedance measurement; capacitance measurement; AC voltametry; and photoelectrochemistry.
- monitoring electron transfer through nucleic acid is via amperometric detection.
- This method of detection involves applying a potential (as compared to a separate reference electrode) between the nucleic acid-conjugated electrode and a reference (counter) electrode in the sample containing target genes of interest. Electron transfer of differing efficiencies is induced in samples in the presence or absence of target nucleic acid; that is, the presence or absence of the target nucleic acid alters the impedance of the nucleic acid (i.e. double stranded versus single stranded) system which can result in different currents.
- the device for measuring electron transfer amperometrically involves sensitive current detection and includes a means of controlling the voltage potential, usually a potentiostat.
- This voltage is optimized with reference to the potential of the electron donating complex on the nucleic acid.
- Possible electron donating complexes include those previously mentioned with complexes of iron, osmium, platinum, cobalt, rhenium and ruthenium being preferred and complexes of iron being most preferred.
- alternative electron detection modes are utilized.
- potentiometric (or voltammetric) measurements involve non-faradaic (no net current flow) processes and are utilized traditionally in pH and other ion detectors. Similar sensors are used to monitor electron transfer through nucleic acid.
- insulators such as resistance
- conductors such as conductivity, impedance and capicitance
- any system that generates a current also generates a small magnetic field, which may be monitored in some embodiments.
- one benefit of the fast rates of electron transfer observed in the compositions of the invention is that time resolution can greatly enhance the signal-to-noise results of monitors based on absorbance, fluorescence and electronic current.
- the fast rates of electron transfer of the present invention result both in high signals and stereotyped delays between electron transfer initiation and completion. By amplifying signals of particular delays, such as through the use of pulsed initiation of electron transfer and "lock-in" amplifiers of detection, between two and four orders of magnitude improvements in signal-to-noise may be achieved.
- electron transfer is initiated using alternating current (AC) methods.
- AC alternating current
- nucleic acids, bound to an electrode generally respond similarly to an AC voltage across a resistor and capacitor in series.
- any methods which enable the determination of the nature of these complexes, which act as a resistor and capacitor can be used as the basis of detection.
- traditional electrochemical theory such as exemplified in Laviron et al., J. Electroanal. Chem. -97:135 (1979) and Laviron et al., J. Electroanal. Chem.
- Equation 3 can be rearranged as follows, using normalization of the concentration to equal 1 for simplicity, as shown in Equations 4, 5 and 6. This requires the subsequent multiplication by the total number of molecules.
- Nernstian curve Specifically, if the number of oxidized molecules at the bottom of the AC cycle is subtracted from the amount at the top of the AC cycle, the total change in a given AC cycle is obtained, as is generally described by Equation 9. Dividing by 2 then gives the AC amplitude.
- a first input electrical signal is then applied to the system, preferably via at least the sample electrode
- the AC amplitude is varied. Without being bound by theory, it appears that increasing the amplitude increases the driving force. Thus, higher amplitudes, which result in higher overpotentials give faster rates of electron transfer. Thus, generally, the same system gives an improved response (i.e. higher output signals) at any single frequency through the use of higher overpotentials at that frequency. Thus, the amplitude may be increased at high frequencies to increase the rate of electron transfer through the system, resulting in greater sensitivity. In addition, this may be used, for example, to induce responses in slower systems such as single stranded nucleic acids for identification, calibration and/or quantification.
- the amount of unhybridized single stranded nucleic acid on an electrode may be compared to the amount of hybridized double stranded nucleic acid to quantify the amount of target sequence in a sample.
- This is quite significant to serve as an internal control of the sensor or system. This allows a measurement either prior to the addition of target or after, on the same molecules that will be used for detection, rather than rely on a similar but different control system.
- the actual molecules that will be used for the detection can be quantified prior to any experiment. For example, a preliminary run at 1 Hz or less, for example, will quantify the actual number of molecules that are on the surface of the electrode. The sample can then be added, an output signal determined, and the ratio of bound/unbound molecules determined. This is a significant advantage over prior methods.
- measurements of the system are taken at at least two separate amplitudes or overpotentials, with measurements at a plurality of amplitudes being preferred.
- changes in response as a result of changes in amplitude may form the basis of identification, calibration and quantification of the system.
- one or more AC frequencies can be used as well.
- the AC frequency is varied.
- different molecules respond in different ways.
- increasing the frequency generally increases the output current.
- higher frequencies result in a loss or decrease of output signal.
- a response may be detected at 1 Hz for both single stranded nucleic acid and double stranded nucleic acid.
- the higher frequencies such as 200 Hz and above, the response of the single stranded nucleic acid is absent, while the response of the double stranded nucleic acid continues to increase.
- the frequency will be greater than the rate of electron transfer through even double-stranded nucleic acid, and then the output signal will also drop.
- the different frequency responses of single stranded and double stranded nucleic acids based on the rate at which electrons may travel through the nucleic acid (i.e. the impedance of the nucleic acid), forms the basis of selective detection of double stranded nucleic acids versus single stranded nucleic acids.
- detection utilizes a single measurement of output signal at a single frequency.
- the frequency response of a single stranded nucleic acid can be previously determined to be very low at a particular high frequency.
- any response at a high frequency for example such as 10 to 100 kHz, where the frequency response of the single stranded nucleic acid is very low or absent, will show the presence of the double stranded hybridization complex. That is, any response at a high frequency is characteristic of the hybridization complex.
- any frequency response is an indication that the hybridization complex is present, and thus that the target sequence is present.
- one or more frequencies can be chosen that prevent a frequency response of one or more charge carriers in solution, whether or not a monolayer is present. This is particularly significant since many biological fluids such as blood contain significant amounts of redox active molecules which can interfere with amperometric detection methods.
- measurements of the system are taken at at least two separate frequencies, with measurements at a plurality of frequencies being preferred.
- a plurality of frequencies includes a scan. For example, measuring the output signal, e.g., the AC current, at a low input frequency such as 1 - 20 Hz, and comparing the response to the output signal at high frequency such as 10 - 100 kHz will show a frequency response difference between double stranded nucleic acids with fast electron transfer rates and single stranded nucleic acids with slow electron transfer rates.
- the frequency response is determined at at least two, preferably at least about five, and more preferably at least about ten frequencies.
- an output signal is received or detected.
- the presence and magnitude of the output signal will depend on the overpotential/amplitude of the input signal; the frequency of the input AC signal; the composition of the intervening medium, i.e. the impedance, between the electron transfer moieties (i.e. single stranded versus double stranded, etc.); the DC offset; the environment of the system; the nature of the second electron transfer moiety; and the solvent.
- the presence and magnitude of the output signal will depend in general on the impedance of the medium between the two electron transfer moieties and the character of the input signal.
- Double stranded nucleic acids, i.e. hybridization complexes have relatively low impedance as compared to single stranded nucleic acids, and thus result in greater output signals.
- the output signal comprises an AC current.
- the magnitude of the output current will depend on a number of parameters. By varying these parameters, the system may be optimized in a number of ways.
- the output signal is phase shifted in the AC component relative to the input signal.
- the systems of the present invention are sufficiently uniform to allow phase-shifting based detection. That is, the complex biomolecules of the invention through which electron transfer occurs react to the AC input in a homogeneous manner, similar to standard electronic components, such that a phase shift can be determined. This may serve as the basis of detection between single-stranded and double stranded nucleic acids, but more importantly, may allow the detection of mismatches, since small changes in impedance, such as would be assumed from a mismatch present in the hybridization complex, may effect the output AC phase in a greater manner than the frequency response.
- nucleic acids in this system have a certain faradaic impedance, that will depend on the distance between the electron transfer moieties, their electronic properties, and the composition of the intervening medium, among other things.
- the faradaic impedance between the electron transfer moieties is signficantly different depending on whether the intervening nucleic acid is single stranded or double stranded.
- the faradaic impedance of the system changes upon the formation of a hybridization complex, and it is this change which is characteristic of the hybridization complex.
- the present invention further provides apparatus for the detection of nucleic acids using
- the apparatus includes a test chamber which has at least a first measuring or sample electrode, and a second measuring or counter electrode. Three electrode systems are also useful.
- the first and second measuring electrodes are in contact with a test sample receiving region, such that in the presence of a liquid test sample, the two electrodes may be in electrical contact.
- the first measuring electrode comprises a single stranded nucleic acid covalently attached via a spacer, and preferably via a conductive oligomer, such as are described herein.
- the second electron transfer moiety may be attached to the probe single stranded nucleic acid, or it may be attached to a second probe nucleic acid, the target nucleic acid, or may be added separately, for example as an intercalator.
- the second electron transfer moiety is covalently attached to the probe single stranded nucleic acid.
- the apparatus further comprises an AC voltage source electrically connected to the test chamber; that is, to the measuring electrodes.
- the AC voltage source is capable of delivering DC offset voltage as well.
- the apparatus further comprises a processor capable of comparing the input signal and the output signal.
- the processor is coupled to the electrodes and configured to receive an output signal, and thus detect the presence of the target nucleic acid.
- compositions of the present invention may be used in a variety of research, clinical, quality control, or field testing settings.
- viral and bacterial detection is done using the complexes of the invention.
- probes are designed to detect target sequences from a variety of bacteria and viruses.
- current blood-screening techniques rely on the detection of anti- HIV antibodies.
- the methods disclosed herein allow for direct screening of clinical samples to detect HIV nucleic acid sequences, particularly highly conserved HIV sequences. In addition, this allows direct monitoring of circulating virus within a patient as an improved method of assessing the efficacy of anti-viral therapies.
- viruses associated with leukemia, HTLV-I and HTLV-II may be detected in this way.
- Bacterial infections such as tuberculosis, clymidia and other sexually transmitted diseases, may also be detected.
- the nucleic acids of the invention find use as probes for toxic bacteria in the screening of water and food samples.
- samples may be treated to lyse the bacteria to release its nucleic acid, and then probes designed to recognize bacterial strains, including, but not limited to, such pathogenic strains as, Salmonella, Campylobacter, Vibrio cholerae, Leishmania, enterotoxic strains of E. coli, and Legionnaire's disease bacteria.
- bioremediation strategies may be evaluated using the compositions of the invention.
- the probes are used for forensic "DNA fingerprinting" to match crime-scene DNA against samples taken from victims and suspects.
- the present invention also finds use as a unique methodology for the detection of mutations or mismatches in target nucleic acid sequences.
- a single stranded nucleic acid containing electron transfer moieties is hybridized to a target sequence with a mutation, the resulting perturbation of the base pairing of the nucleosides will measurably affect the electron transfer rate.
- the mutation is a substitution, insertion or deletion.
- two single stranded nucleic acids each with a covalently attached electron transfer species that hybridize adjacently to a target sequence may be used. Accordingly, the present invention provides for the detection of mutations in target sequences.
- the PCR reaction is done as is known in the art, and then added to a composition of the invention comprising the target nucleic acid with a second ETM, covalently attached to an electrode via a conductive oligomer with subsequent detection of the target sequence.
- PCR is done using nucleotides labelled with a second ETM, either in the presence of, or with subsequent addition to, an electrode with a conductive oligomer and a target nucleic acid. Binding of the PCR product containing second ETMs to the electrode composition will allow detection via electron transfer.
- the nucleic acid attached to the electrode via a conductive polymer may be one PCR primer, with addition of a second primer labelled with an ETM. Elongation results in double stranded nucleic acid with a second ETM and electrode covalently attached. In this way, the present invention is used for PCR detection of target sequences.
- the present invention provides methods which can result in sensitive detection of nucleic acids.
- less than about 10 X 10 6 molecules are detected, with less than about 10 X 10 5 being preferred, less than 10 X 10 4 being particularly preferred, less than about 10 X 10 3 being especially preferred, and less than about 10 X 10 2 being most preferred.
- this assumes a 1 :1 correlation between target sequences and reporter molecules; if more than one reporter molecule (i.e. second electron transfer moeity) is used for each target sequence, the sensitivity will go up.
- the present invention provides novel compositions comprising metallocenes covalently attached via conductive oiigomers to an electrode, such as are generally depicted in Structure 35: Structure 35
- Structure 35 utilizes a Structure 4 conductive oligomer, although as will be appreciated by those in the art, other conductive oiigomers such as Structures 2, 3, 9 or 10 types may be used. Preferred embodiments of Structure 35 are depicted below.
- Compound # 6 To a glass bottle were added 0.23 gm (0.68 mmol) of Compound # 5, 0.5 gm (0.64 mmol) of 2'-deoxy-2'-(4-iodophenylcarbonyl) amino-5'-0-DMT uridine, 60 mg of bis(triphenylphosphine)palladium (II) chloride, 30 mg of cuprous iodide. The bottle was sealed and bubbled with Argon. Pyrrodine(15 mL) and DMF(15 mL) were introduced by a syringe. The reaction mixture was heated at 85 °C overnight. The solvents were removed in vacuo and the residue was dissolved in 300 mL of dichloromethane.
- Reverse-phase silica gel(C-18, 120 gm) was packed with 60 % CH 3 CN/40 % H z O and the sample was dissolved in very small amount of THF and loaded.
- the column was eluted with 100 mL of 60 % CH 3 CN/40 % H 2 0, 100 mL of 70 %
- Conductive oligomer-Uridine-CPG To 1.4 gm of LCAA-CPG(500 _) in 100 mL round bottom flask were added 110 mgm(101 ⁇ mol) of the Compound # 7, 100 mgm (230 ⁇ mol) of BOP reagent, 30 mgm (220 ⁇ mol) of HBT, 70 mL of dichloromethane and 2 mL of TEA. The mixture was shaken for three days. The CPG was filtered off and washed twice with dichloromethane and transferred into another 100 mL flask. Into CPG were added 50 mL of pyridine, 10 mL of acetic anhydride and 2mL of N-methylimidizole. The CPG was filtered off, washed twice with pyridine, methanol, dichloromethane and ether, and dried over a vacuum. The loading of the nucleoside was measured according to the standard procedure to be 7.1 ⁇ mol/gm.
- the column was eluted with 300 mL of 1 % TEA/2 % CH 3 OH/CH 2 CI 2 , and 500 mL of 1 % TEA/4 % CH 3 OH/CH 2 CI 2 .
- the solution was diluted by adding 100 mL of dichloromethane and washed once with 200 mL od 5 % aqueous NaHC0 3 solution, and once with the brine (200 mL) and dried over Na 2 S0 4 and concentrated to dryness.
- Silica gel(47 gm) was packed with 2 % TEA/1 % CH 3 OH/CH 2 CI 2 .
- the residue was dissolved in 10 mL of dichloromethane and loaded.
- the column was eluted with 150 mL of 1 % TEA / 1 % CH 3 OH / CH 2 CI 2 and 250 mL of 1 % TEA 12 % CH 3 OH / CH 2 CI 2 .
- the fractions were pooled and concentrated to give 0.5 gm (30.3 %) of the title compound.
- HS-(CH2)16-OH (herein “insulator-2") was made as follows.
- 16-Mercaptohexadecan-1-ol Under inert atmosphere 365 mg of sodium metal suspension (40% in mineral oil) were added dropwise to 20 ml of dry methanol at 0°C. After completion of addition the reaction mixture was stirred for 10 min at RT followed by addition of 0.45 ml (6.30 mmole) of thioacetic acid. After additional 10 min of stirring 3 ml degassed methanolic solution of 1.0 gr (3.11 mmole) of 16-bromohexadecan-1-ol were added. The resulted mixture was refluxed for 15 hrs, allowed to cool to RT and 20 ml of degassed 1.0 M NaOH aqueous solution were injected.
- reaction completion required additional 3 hr of reflux.
- Hybridization efficiency was determined using 32 P complementary and noncomplementary 15 mers corresponding to the wire-1 sequence.
- the electrodes were incubated with 50 microliters of each of the labelled non-complementary (herein "A5") or complementary (herein “S5") target sequences applied over the entire electrode in 1XSSC as depicted in Table 1.
- the electrodes were then incubated for 1-2 hours at room temperature in a moist chamber, and rinsed as described above.
- the amount of radiolabelled DNA was measured for each electrode in a scintllation counter, and the electrodes were dried and exposed to X-ray film for 4 hours. Table 1
- HS-(CH2)15NHCO-Fc (herein “insulator-1") was made as described in Ward et al., Anal. Chem.
- the electrodes were then rinsed again. This procedure resulted in 1-10% coverage of insulator-1 as compared to calculated values of close packed ferrocene molecules on a surface. More or less coverage could easily be obtained by altering the mixture concentration and/or incubation times.
- Sample 1 labeled herein as "Fc-alkane” contained a mixed monolayer of insulator-2 and insulator-1.
- Sample 2 labeled herein as “Fc-amido-alkane” contained a mixed monolayer of insulator-2 and a derivative of insulator-1 which has an amido attachment of the ferrocene to the alkane.
- Sample 3 labeled herein as “Fc-wire”, contained a mixed monolayer of insulator-2 and wire-2.
- Sample 4 was the same as Sample 3, with the exception that a new in situ deprotection step was used, described below.
- Sample 5 labeled herein as “ssDNA” (AGCTGAGTCCA(UBF)GGU-conductive oligomer), contained a mixed monolayer of insulator-2 and wire-3.
- Sample 7 was a solution of ferrocene in solution. As is shown herein, the rate of electron transfer, from fast to slower, is as follows: Sample 3 > Sample 6 > Sample 1 > Sample 2 >
- Sample 5 Generally, Sample 1 models ssDNA, and Sample 3 models dsDNA.
- the experiments were run as follows. A DC offset voltage between the working (sample) electrode and the reference electrode was swept through the electrochemical potential of the ferrocene, typically from 0 to 500 mV. On top of the DC offset, an AC signal of variable amplitude and frequency was applied. The AC current at the excitation frequency was plotted versus the DC offset.
- Figure 8 depicts an experiment with Sample 1 , at 200 mV AC amplitude and frequencies of 1 , 5 and
- FIG. 9 shows overlaid AC voltammograms of an electrode coated with Sample 3.
- Figure 10 shows the frequency response of samples 1 , 2 and 3 by measuring the peak currents vs. frequency. Sample 3 response to increasing frequencies through 10 kHz (the detector system limit), while Sample 1 lose its responses at between 20 and 200 Hz. Thus, to discriminate between Sample 1 and Sample 3, one could simplify the methods by analyzing it at 1 Hz and 1000 Hz and compare the responses, although as will be appreciated, this is only one method of a variety of possible methods. This should be similar to the dsDNA and ssDNA system.
- Figure 11 shows Sample 5 and Sample 6, plotted as a function of normalized current (with the highest current being 1 for both cases; the actual current of dsDNA is much higher than that of ssDNA, so the graph was normalized to show both).
- the lines are modeled RC circuits, as described above, and not a fit to the data. At 1 Hz, both ssDNA and dsDNA respond; at 200 Hz, the ssDNA signal is gone.
- Figure 12 shows that increasing the overpotential will increase the output signal for slow systems like samples 1 and 2.
- Figures 13A and 13B show that the overpotential and frequency can be tuned to increase the selectivity and sensitivity. For example, a low overpotential and high frequence can be used to minimize the slower species
- Figure 14 shows that the ferrocene added to the solution (Sample 7) has a frequency response related to diffusion that is easily distinguishable from the frequency response of attached ferrocene. This indicates that by varying frequency, signals from bound molecules, particular fast bound molecules such as dsDNA, can be easily distinguished from any signal generated by contaminating redox molecules in the sample.
- Figures 15A and 15B shows the phase shift that results with different samples.
- Figure 15A shows the model compounds, and 15B shows data with dsDNA and ssDNA. While at this frequency, the phase shift is not large, a frequency can always be found that results in a 90° shift in the phase.
- Example 11 The use of trimethylsilylethyl protecting groups The use of an alternate protecting group for protection of the sulfur atom prior to attachment to the gold surface was explored.
- the aqueous solution was extracted once with 100 mL ether and once with 100 of dichloromethane. After decolorization with 5 gm of Norit, the aqueous solution was neutralized by adding solid NaOH to precipitate the crude product, which, after chilling, was filtered and rinsed with 800 mL of water and 300 mL of ethanol. The wet product was recrystallized from 200 mL of acetic acid to produce 37.5 gm of 4-iodo-L-phenylalanine. - 90 -
- Methyl 4-lodophenyl Alaninate Hydrochloride To 10 mL of methanol cooled in an ice-water bath was added dropwise 10.2 gm of thionyl chloride. Into the cold solution was added 5.0 gm of 4- iodophenylalanine and the yellow solution was formed and refluxed for 2 h. After removing the solvent, the white solid was obtained and recrystallized from 10 mL of methanol by addition of 50 mL of ether. The title product(5.4 gm) was prepared.
- Methyl N-Aminoethyl-4-lodophenyl Alaninate To a solution of 3.46 gm (8 mmol) of [l,l- bis(trifloroacetoxy)iodo]benzene in 24 mL of acetonitrile was added 12 mL of the glass-distilled water, followed adding 2.98 gm(8 mmol) of methyl N-amidocarboxylethyl alninate. The mixture was stirred for 6 h at room temperature and diluted by 150 mL of water and 16 mL of concentrated HCI solution. The aqueous solution was extracted once with 150 mL of ether and concentrated to about one third of the original volume.
- Methyl N-(2-MMT-aminoethyl)-N-(2-Nitrobenzenesulfonyl)-4-lodophenyl Alaninate To a solution of 16.5 gm (49.5 mmol) of 2-MMT-amino ethanol, 20.0 gm (40.8 mmol) of methyl N-(2- nitrobenzensulfonyl)-4-iodophenyl alaninate and 13 gm (49.5 mmol) of triphenylphosphine in 250 mL of dry THF cooled in an ice-water bath was added 7.8 mL (49.5 mmol) of diethyl azodicarboxylate under Argon. The solution was warmed up to room temperature and stirred overnight. After removing
- aqueous solution was extracted three times by ether (2x 500 mL) and the combined extracts was washed once with the diluted NaOH solution and dried over sodium sulfate. After the removal of the solvent, the residue was used for column separation.
- Silica Gel (220 gm) was packed with 1% TEA/hexane, upon loading the sample solution, the column was eluted with 500 mL of 1% TEA/hexane, 1000 mL of 1% TEA/25% ether/hexane, and 1000 mL of 1% TEA/50% ether/hexane. The fractions were identified by TLC (Ether: Hexane) and pooled and concentrated to afford 5.6 gm (43.1 %) of the title product.
- Methyl N-(2-MMT-aminoethyl)-N-[(Thymin-1-yl)acetyl]-4-lodophenyl Alaninate To a solution of 3.37 g (5.43 mmol) of methyl N-(2-MMT-aminoethyl)-4-iodophenyl alaninate in DMF (10 mL) was added 3,4- Dihydro-3-hydroxy-4-oxo-1 ,2,3-benzotriazine (.884 g, 5.43 mmol) and 4-ethylmorpholine (1.38 mL, 10.86 mmol).
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP52166898A JP4072206B2 (en) | 1996-11-05 | 1997-11-05 | Electrode linked to nucleic acid via conductive oligomer |
| AU51967/98A AU739375B2 (en) | 1996-11-05 | 1997-11-05 | Electrodes linked via conductive oligomers to nucleic acids |
| CA002270633A CA2270633A1 (en) | 1996-11-05 | 1997-11-05 | Electrodes linked via conductive oligomers to nucleic acids |
| EP97946876A EP0939762A2 (en) | 1996-11-05 | 1997-11-05 | Electrodes linked via conductive oligomers to nucleic acids |
| US09/452,277 US7160678B1 (en) | 1996-11-05 | 1999-11-30 | Compositions for the electronic detection of analytes utilizing monolayers |
| US11/208,384 US7393645B2 (en) | 1996-11-05 | 2005-08-19 | Compositions for the electronic detection of analytes utilizing monolayers |
Applications Claiming Priority (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/743,798 US6096273A (en) | 1996-11-05 | 1996-11-05 | Electrodes linked via conductive oligomers to nucleic acids |
| US4015597P | 1997-03-07 | 1997-03-07 | |
| US08/873,597 US7381525B1 (en) | 1997-03-07 | 1997-06-12 | AC/DC voltage apparatus for detection of nucleic acids |
| US08/873,978 US7014992B1 (en) | 1996-11-05 | 1997-06-12 | Conductive oligomers attached to electrodes and nucleoside analogs |
| US08/899,510 US6221583B1 (en) | 1996-11-05 | 1997-07-24 | Methods of detecting nucleic acids using electrodes |
| US08/911,085 US6090933A (en) | 1996-11-05 | 1997-08-14 | Methods of attaching conductive oligomers to electrodes |
| US08/911,085 | 1997-08-14 | ||
| US08/911,589 | 1997-08-14 | ||
| US60/040,155 | 1997-08-14 | ||
| US08/873,597 | 1997-08-14 | ||
| US08/899,510 | 1997-08-14 | ||
| US08/743,798 | 1997-08-14 | ||
| US08/873,978 | 1997-08-14 | ||
| US08/911,589 US6232062B1 (en) | 1997-03-07 | 1997-08-14 | AC methods for the detection of nucleic acids |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO1998020162A2 true WO1998020162A2 (en) | 1998-05-14 |
| WO1998020162A3 WO1998020162A3 (en) | 1998-11-12 |
Family
ID=27567906
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1997/020014 WO1998020162A2 (en) | 1996-11-05 | 1997-11-05 | Electrodes linked via conductive oligomers to nucleic acids |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP0939762A2 (en) |
| JP (1) | JP4072206B2 (en) |
| AU (1) | AU739375B2 (en) |
| CA (1) | CA2270633A1 (en) |
| WO (1) | WO1998020162A2 (en) |
Cited By (69)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1999037819A2 (en) * | 1998-01-27 | 1999-07-29 | Clinical Micro Sensors, Inc. | Amplification of nucleic acids with electronic detection |
| WO1999057319A1 (en) * | 1998-05-06 | 1999-11-11 | Clinical Micro Sensors | Electronic detection of nucleic acids using monolayers |
| WO1999057317A1 (en) * | 1998-05-06 | 1999-11-11 | Clinical Micro Sensors, Inc. | Electronic methods for the detection of analytes utilizing monolayers |
| WO1999067425A2 (en) * | 1998-06-23 | 1999-12-29 | Clinical Micro Sensors, Inc. | Binding acceleration techniques for the detection of analytes |
| WO1999067628A1 (en) * | 1998-06-24 | 1999-12-29 | Therasense, Inc. | Multi-sensor array for electrochemical recognition of nucleotide sequences and methods |
| US6013170A (en) * | 1997-06-12 | 2000-01-11 | Clinical Micro Sensors, Inc. | Detection of analytes using reorganization energy |
| WO2000024941A1 (en) * | 1998-10-27 | 2000-05-04 | Clinical Micro Sensors, Inc. | Detection of target analytes using particles and electrodes |
| WO2000038836A1 (en) * | 1998-12-30 | 2000-07-06 | Clinical Micro Sensors, Inc. | Tissue collection devices containing biosensors |
| WO2000042217A2 (en) * | 1999-01-18 | 2000-07-20 | Fritz, Biochem Gmbh | Method for electrochemically detecting nucleic acid-oligomer hybridisation events |
| EP1035216A1 (en) * | 1999-03-05 | 2000-09-13 | Azur Environmental Ltd. | Method of and apparatus for the detection of analytes |
| US6127127A (en) * | 1995-06-27 | 2000-10-03 | The University Of North Carolina At Chapel Hill | Monolayer and electrode for detecting a label-bearing target and method of use thereof |
| WO2001006016A2 (en) * | 1999-07-20 | 2001-01-25 | Clinical Micro Sensors, Inc. | Amplification of nucleic acids with electronic detection |
| WO2001007665A2 (en) * | 1999-07-26 | 2001-02-01 | Clinical Micro Sensors, Inc. | Sequence determination of nucleic acids using electronic detection |
| WO2001021635A2 (en) * | 1999-09-22 | 2001-03-29 | Friz Biochem Gmbh | Method for electrochemically detecting sequence-specific nucleic acid-oligomer hybridisation events |
| WO2001031057A2 (en) * | 1999-10-22 | 2001-05-03 | Nanogen Recognomics Gmbh | Double-strand nucleic acid probes and the use thereof |
| US6235484B1 (en) | 1999-05-03 | 2001-05-22 | Gen-Probe Incorporated | Polynucleotide probes for detection and quantitation of actinomycetes |
| WO2001044501A2 (en) * | 1999-12-13 | 2001-06-21 | november Aktiengesellschaft Gesellschaft für Molekulare Medizin | Method and device for detecting and quantifying biomolecules |
| US6291188B1 (en) | 1995-06-07 | 2001-09-18 | California Institute Of Technology | Metallic solid supports modified with nucleic acids |
| US6326486B1 (en) | 1999-05-03 | 2001-12-04 | Gen-Probe Incorporated | Polynucleotide probes for detection and quantitation of bacteria in the family enterobacteriaceae |
| WO2001092277A1 (en) * | 2000-05-26 | 2001-12-06 | Minerva Biotechnologies Corporation | Metallocenes as signal-transducer for detection of chemical or biological events |
| US6376186B1 (en) | 1999-05-03 | 2002-04-23 | Gen-Probe Incorporated | Polynucleotide probes for detection and quantitation of staphylococcus |
| WO2001040511A3 (en) * | 1999-12-01 | 2002-05-10 | Secr Defence | Detection system |
| US6387625B1 (en) | 1995-06-27 | 2002-05-14 | The University Of North Carolina At Chapel Hill | Monolayer and electrode for detecting a label-bearing target and method of use thereof |
| US6432723B1 (en) | 1999-01-22 | 2002-08-13 | Clinical Micro Sensors, Inc. | Biosensors utilizing ligand induced conformation changes |
| EP1231214A2 (en) * | 2001-02-01 | 2002-08-14 | Hitachi Software Engineering Co., Ltd. | Biopolymer detector |
| US6479240B1 (en) | 1996-11-05 | 2002-11-12 | Clinical Micro Sensors, Inc. | Electrodes linked via conductive oligomers to nucleic acids |
| US6518024B2 (en) | 1999-12-13 | 2003-02-11 | Motorola, Inc. | Electrochemical detection of single base extension |
| US6528266B2 (en) | 1993-12-10 | 2003-03-04 | California Institute Of Technology | Nucleic acid mediated electron transfer |
| US6653462B2 (en) | 1998-05-29 | 2003-11-25 | Applera Corporation | Nucleotide compounds including a rigid linker |
| US6686150B1 (en) | 1998-01-27 | 2004-02-03 | Clinical Micro Sensors, Inc. | Amplification of nucleic acids with electronic detection |
| US6753143B2 (en) | 2000-05-01 | 2004-06-22 | Clinical Micro Sensors, Inc. | Target analyte detection using asymmetrical self-assembled monolayers |
| US6787339B1 (en) | 2000-10-02 | 2004-09-07 | Motorola, Inc. | Microfluidic devices having embedded metal conductors and methods of fabricating said devices |
| US6821770B1 (en) | 1999-05-03 | 2004-11-23 | Gen-Probe Incorporated | Polynucleotide matrix-based method of identifying microorganisms |
| US6833267B1 (en) | 1998-12-30 | 2004-12-21 | Clinical Micro Sensors, Inc. | Tissue collection devices containing biosensors |
| EP1542000A1 (en) * | 2002-07-31 | 2005-06-15 | Kabushiki Kaisha Toshiba | Base sequence detecting device and base sequence automatic analyzer |
| US6977151B2 (en) | 1996-11-05 | 2005-12-20 | Clinical Micro Sensors, Inc. | Electrodes linked via conductive oligomers to nucleic acids |
| US7045285B1 (en) | 1996-11-05 | 2006-05-16 | Clinical Micro Sensors, Inc. | Electronic transfer moieties attached to peptide nucleic acids |
| US7056664B1 (en) | 1998-11-23 | 2006-06-06 | Fritz Biochem Gesellschaft für Bioanalytik mbH | Method of the electrochemical detection of nucleic acid oligomer hybrids |
| US7138121B2 (en) | 2003-01-23 | 2006-11-21 | Spangler Brenda D | Biosensors utilizing dendrimer-immobilized ligands and there use thereof |
| US7160678B1 (en) | 1996-11-05 | 2007-01-09 | Clinical Micro Sensors, Inc. | Compositions for the electronic detection of analytes utilizing monolayers |
| WO2007011946A2 (en) * | 2005-07-15 | 2007-01-25 | Applera Corporation | Detection of nucleic acid amplification |
| US7393645B2 (en) | 1996-11-05 | 2008-07-01 | Clinical Micro Sensors, Inc. | Compositions for the electronic detection of analytes utilizing monolayers |
| WO2009061941A2 (en) | 2007-11-06 | 2009-05-14 | Osmetech Molecular Diagnostics | Baseless nucleotide analogues and uses thereof |
| US7615340B2 (en) | 2000-10-03 | 2009-11-10 | Minerva Biotechnologies Corporation | Electronic detection of interaction and detection of interaction based on the interruption of flow |
| WO2010007526A1 (en) | 2008-07-17 | 2010-01-21 | Andreas Pfuetzner | Biomarkers for cardiodiabetes |
| WO2010009438A1 (en) | 2008-07-17 | 2010-01-21 | Andreas Pfuetzner | Biomarkers for insulin resistance and beta-cell dysfunction |
| US7655129B2 (en) | 1998-06-23 | 2010-02-02 | Osmetech Technology Inc. | Binding acceleration techniques for the detection of analytes |
| WO2010064147A2 (en) | 2008-12-04 | 2010-06-10 | Ikfe Gmbh | Biomarkers for atherosclerosis |
| WO2010067207A1 (en) | 2008-12-11 | 2010-06-17 | Ikfe Gmbh | Biomarkers for insulin sensitizer drug response |
| US7745203B2 (en) | 2002-07-31 | 2010-06-29 | Kabushiki Kaisha Toshiba | Base sequence detection apparatus and base sequence automatic analyzing apparatus |
| WO2010076655A1 (en) | 2008-12-30 | 2010-07-08 | Ikfe Gmbh | Biomarkers for adipose tissue activity |
| WO2010079428A2 (en) | 2009-01-07 | 2010-07-15 | Ikfe Gmbh | Biomarkers for appetite regulation |
| EP2286792A1 (en) | 1999-02-26 | 2011-02-23 | Novartis Vaccines and Diagnostics, Inc. | Microemulsions with an adsorbent surface, comprising a microdroplet emulsion |
| US8080380B2 (en) | 1999-05-21 | 2011-12-20 | Illumina, Inc. | Use of microfluidic systems in the detection of target analytes using microsphere arrays |
| WO2012021749A1 (en) | 2010-08-11 | 2012-02-16 | Celula, Inc. | Genotyping dna |
| WO2012158967A1 (en) | 2011-05-17 | 2012-11-22 | Dxterity Diagnostics Incorporated | Methods and compositions for detecting target nucleic acids |
| US8481268B2 (en) | 1999-05-21 | 2013-07-09 | Illumina, Inc. | Use of microfluidic systems in the detection of target analytes using microsphere arrays |
| US8486247B2 (en) | 1999-04-21 | 2013-07-16 | Osmetch Technology, Inc. | Use of microfluidic systems in the electrochemical detection of target analytes |
| EP2677039A2 (en) | 2006-05-10 | 2013-12-25 | Dxterity Diagnostics | Detection of nucleic acid targets using chemically reactive oligonucleotide probes |
| WO2014066704A1 (en) | 2012-10-24 | 2014-05-01 | Genmark Diagnostics, Inc. | Integrated multiplex target analysis |
| EP2765205A1 (en) | 2009-04-01 | 2014-08-13 | DXTerity Diagnostics Incorporated | Chemical ligation dependent probe amplification (CLPA) |
| US9194836B2 (en) | 2009-08-07 | 2015-11-24 | Ohmx Corporation | Enzyme triggered redox altering chemical elimination (E-trace) immunoassay |
| US9250234B2 (en) | 2011-01-19 | 2016-02-02 | Ohmx Corporation | Enzyme triggered redox altering chemical elimination (E-TRACE) immunoassay |
| US9250203B2 (en) | 2012-01-09 | 2016-02-02 | Ohmx Corporation | Enzyme cascade methods for E-TRACE assay signal amplification |
| EP1114317B1 (en) * | 1998-09-17 | 2016-12-14 | Clinical Micro Sensors, Inc. | Signal detection techniques for the detection of analytes |
| US9624522B2 (en) | 2009-08-07 | 2017-04-18 | Ohmx Corporation | Single, direct detection of hemoglobin A1c percentage using enzyme triggered redox altering chemical elimination (e-trace) immunoassay |
| US9757095B2 (en) | 2014-06-10 | 2017-09-12 | Dxterity Diagnostics Incorporated | Devices and methods for collecting and stabilizing biological samples |
| US9874542B2 (en) | 1998-09-17 | 2018-01-23 | Clinical Micro Sensors, Inc. | Signal detection techniques for the detection of analytes |
| US10106847B1 (en) | 2017-08-24 | 2018-10-23 | Clinical Micro Sensors, Inc. | Electrochemical detection of bacterial and/or fungal infections |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7112366B2 (en) * | 2001-01-05 | 2006-09-26 | The Ohio State University | Chemical monolayer and micro-electronic junctions and devices containing same |
| JP5181386B2 (en) * | 2008-02-01 | 2013-04-10 | 国立大学法人 新潟大学 | Chemical biosensor |
| WO2011087130A1 (en) * | 2010-01-18 | 2011-07-21 | 株式会社クラレ | Acetylene compound and organic semiconductor material comprising same |
| US9340567B2 (en) | 2011-11-04 | 2016-05-17 | Ohmx Corporation | Chemistry used in biosensors |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1990005732A1 (en) * | 1988-11-07 | 1990-05-31 | The Trustees Of Columbia University In The City Ofnew York | Mixed ligand complexes and uses thereof as binding agents and probres to dna |
| WO1993010267A1 (en) * | 1991-11-15 | 1993-05-27 | Igen, Inc. | Rapid assays for amplification products |
| WO1994022889A1 (en) * | 1993-03-31 | 1994-10-13 | Cis Bio International | Electronically conductive polymer/nucleotide copolymer, preparation method therefor and use thereof |
| WO1995015971A2 (en) * | 1993-12-10 | 1995-06-15 | California Institute Of Technology | Nucleic acid mediated electron transfer |
| WO1996040712A1 (en) * | 1995-06-07 | 1996-12-19 | California Institute Of Technology | Nucleic acid mediated electron transfer |
-
1997
- 1997-11-05 WO PCT/US1997/020014 patent/WO1998020162A2/en active IP Right Grant
- 1997-11-05 AU AU51967/98A patent/AU739375B2/en not_active Ceased
- 1997-11-05 JP JP52166898A patent/JP4072206B2/en not_active Expired - Lifetime
- 1997-11-05 CA CA002270633A patent/CA2270633A1/en not_active Abandoned
- 1997-11-05 EP EP97946876A patent/EP0939762A2/en not_active Withdrawn
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1990005732A1 (en) * | 1988-11-07 | 1990-05-31 | The Trustees Of Columbia University In The City Ofnew York | Mixed ligand complexes and uses thereof as binding agents and probres to dna |
| WO1993010267A1 (en) * | 1991-11-15 | 1993-05-27 | Igen, Inc. | Rapid assays for amplification products |
| WO1994022889A1 (en) * | 1993-03-31 | 1994-10-13 | Cis Bio International | Electronically conductive polymer/nucleotide copolymer, preparation method therefor and use thereof |
| WO1995015971A2 (en) * | 1993-12-10 | 1995-06-15 | California Institute Of Technology | Nucleic acid mediated electron transfer |
| US5591578A (en) * | 1993-12-10 | 1997-01-07 | California Institute Of Technology | Nucleic acid mediated electron transfer |
| WO1996040712A1 (en) * | 1995-06-07 | 1996-12-19 | California Institute Of Technology | Nucleic acid mediated electron transfer |
Non-Patent Citations (8)
| Title |
|---|
| AIZAWA M ET AL: "INTEGRATED MOLECULAR SYSTEMS FOR BIOSENSORS*" SENSORS AND ACTUATORS B, vol. B24, no. 1/03, PART 01, March 1995, pages 1-5, XP000521315 * |
| J.R.REIMERS ET AL.: "Towards Efficient Molecular Wires and Switches : The Brooker Ions." BIOSYSTEMS, vol. 35, 1995, pages 107-111, XP002077969 * |
| MURPHY C J ET AL: "LONG-RANGE PHOTOINDUCED ELECTRON TRANSFER THROUGH A DNA HELIX" SCIENCE, vol. 262, 12 November 1993, pages 1025-1029, XP002041920 * |
| P.LINCOLN ET AL.: "Short-Circuiting the Molecular Wire." JOURNAL OF THE AMERICAN CHEMICAL SOCIETY., vol. 119, no. 6, 1997, pages 1454-1455, XP002077971 DC US * |
| R.P.HSUNG ET AL.: "Synthesis and CHaracterization of Unsymmetric Ferrocene-Terminated Phenylethynyl Oligomers." ORGANOMETALLICS, vol. 14, no. 10, 1995, pages 4808-4815, XP002077968 cited in the application * |
| See also references of EP0939762A2 * |
| TURRO N J ET AL: "PHOTOELECTRON TRANSFER BETWEEN MOLECULES ADSORBED IN RESTICTED SPACES" PROCEEDINGS OF THE INTERNATIONAL CONFERENCE ON PHOTOCHEMICAL CONVERSION AND STORAGE OF SOLAR ENERGY,1990, pages 121-139, XP002041921 * |
| W.M.ALBERS ET AL.: "Design of Novel Molecular Wires for Realizing Long-Distance Electron Transfer." BIOCHEMISTRY AND BIOENERGETICS, vol. 42, 1997, page 25-33 XP002077970 * |
Cited By (108)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6528266B2 (en) | 1993-12-10 | 2003-03-04 | California Institute Of Technology | Nucleic acid mediated electron transfer |
| US6291188B1 (en) | 1995-06-07 | 2001-09-18 | California Institute Of Technology | Metallic solid supports modified with nucleic acids |
| US6387625B1 (en) | 1995-06-27 | 2002-05-14 | The University Of North Carolina At Chapel Hill | Monolayer and electrode for detecting a label-bearing target and method of use thereof |
| US6127127A (en) * | 1995-06-27 | 2000-10-03 | The University Of North Carolina At Chapel Hill | Monolayer and electrode for detecting a label-bearing target and method of use thereof |
| US7045285B1 (en) | 1996-11-05 | 2006-05-16 | Clinical Micro Sensors, Inc. | Electronic transfer moieties attached to peptide nucleic acids |
| US7393645B2 (en) | 1996-11-05 | 2008-07-01 | Clinical Micro Sensors, Inc. | Compositions for the electronic detection of analytes utilizing monolayers |
| US6977151B2 (en) | 1996-11-05 | 2005-12-20 | Clinical Micro Sensors, Inc. | Electrodes linked via conductive oligomers to nucleic acids |
| US6479240B1 (en) | 1996-11-05 | 2002-11-12 | Clinical Micro Sensors, Inc. | Electrodes linked via conductive oligomers to nucleic acids |
| US7160678B1 (en) | 1996-11-05 | 2007-01-09 | Clinical Micro Sensors, Inc. | Compositions for the electronic detection of analytes utilizing monolayers |
| US7267939B2 (en) | 1997-06-12 | 2007-09-11 | Clinical Micro Sensors, Inc. | Detection of analytes using reorganization energy |
| US6013459A (en) * | 1997-06-12 | 2000-01-11 | Clinical Micro Sensors, Inc. | Detection of analytes using reorganization energy |
| US6013170A (en) * | 1997-06-12 | 2000-01-11 | Clinical Micro Sensors, Inc. | Detection of analytes using reorganization energy |
| US7514228B2 (en) | 1997-06-12 | 2009-04-07 | Clinical Micro Sensors, Inc. | Detection of analytes using reorganization energy |
| US6686150B1 (en) | 1998-01-27 | 2004-02-03 | Clinical Micro Sensors, Inc. | Amplification of nucleic acids with electronic detection |
| WO1999037819A2 (en) * | 1998-01-27 | 1999-07-29 | Clinical Micro Sensors, Inc. | Amplification of nucleic acids with electronic detection |
| WO1999037819A3 (en) * | 1998-01-27 | 1999-10-14 | Clinical Micro Sensors Inc | Amplification of nucleic acids with electronic detection |
| US6600026B1 (en) | 1998-05-06 | 2003-07-29 | Clinical Micro Sensors, Inc. | Electronic methods for the detection of analytes utilizing monolayers |
| WO1999057317A1 (en) * | 1998-05-06 | 1999-11-11 | Clinical Micro Sensors, Inc. | Electronic methods for the detection of analytes utilizing monolayers |
| WO1999057319A1 (en) * | 1998-05-06 | 1999-11-11 | Clinical Micro Sensors | Electronic detection of nucleic acids using monolayers |
| US6653462B2 (en) | 1998-05-29 | 2003-11-25 | Applera Corporation | Nucleotide compounds including a rigid linker |
| US6264825B1 (en) | 1998-06-23 | 2001-07-24 | Clinical Micro Sensors, Inc. | Binding acceleration techniques for the detection of analytes |
| US7655129B2 (en) | 1998-06-23 | 2010-02-02 | Osmetech Technology Inc. | Binding acceleration techniques for the detection of analytes |
| US6290839B1 (en) | 1998-06-23 | 2001-09-18 | Clinical Micro Sensors, Inc. | Systems for electrophoretic transport and detection of analytes |
| WO1999067425A2 (en) * | 1998-06-23 | 1999-12-29 | Clinical Micro Sensors, Inc. | Binding acceleration techniques for the detection of analytes |
| WO1999067425A3 (en) * | 1998-06-23 | 2000-02-03 | Clinical Micro Sensors Inc | Binding acceleration techniques for the detection of analytes |
| WO1999067628A1 (en) * | 1998-06-24 | 1999-12-29 | Therasense, Inc. | Multi-sensor array for electrochemical recognition of nucleotide sequences and methods |
| US9874542B2 (en) | 1998-09-17 | 2018-01-23 | Clinical Micro Sensors, Inc. | Signal detection techniques for the detection of analytes |
| EP1114317B1 (en) * | 1998-09-17 | 2016-12-14 | Clinical Micro Sensors, Inc. | Signal detection techniques for the detection of analytes |
| WO2000024941A1 (en) * | 1998-10-27 | 2000-05-04 | Clinical Micro Sensors, Inc. | Detection of target analytes using particles and electrodes |
| US6541617B1 (en) | 1998-10-27 | 2003-04-01 | Clinical Micro Sensors, Inc. | Detection of target analytes using particles and electrodes |
| US8012743B2 (en) | 1998-10-27 | 2011-09-06 | Osmetech Technology Inc. | Detection of target analytes using particles and electrodes |
| US7056664B1 (en) | 1998-11-23 | 2006-06-06 | Fritz Biochem Gesellschaft für Bioanalytik mbH | Method of the electrochemical detection of nucleic acid oligomer hybrids |
| WO2000038836A1 (en) * | 1998-12-30 | 2000-07-06 | Clinical Micro Sensors, Inc. | Tissue collection devices containing biosensors |
| US6833267B1 (en) | 1998-12-30 | 2004-12-21 | Clinical Micro Sensors, Inc. | Tissue collection devices containing biosensors |
| WO2000042217A2 (en) * | 1999-01-18 | 2000-07-20 | Fritz, Biochem Gmbh | Method for electrochemically detecting nucleic acid-oligomer hybridisation events |
| WO2000042217A3 (en) * | 1999-01-18 | 2000-11-30 | Gerhard Hartwich | Method for electrochemically detecting nucleic acid-oligomer hybridisation events |
| US6432723B1 (en) | 1999-01-22 | 2002-08-13 | Clinical Micro Sensors, Inc. | Biosensors utilizing ligand induced conformation changes |
| EP2286792A1 (en) | 1999-02-26 | 2011-02-23 | Novartis Vaccines and Diagnostics, Inc. | Microemulsions with an adsorbent surface, comprising a microdroplet emulsion |
| EP1035216A1 (en) * | 1999-03-05 | 2000-09-13 | Azur Environmental Ltd. | Method of and apparatus for the detection of analytes |
| US9557295B2 (en) | 1999-04-21 | 2017-01-31 | Osmetech Technology, Inc. | Use of microfluidic systems in the electrochemical detection of target analytes |
| US9151746B2 (en) | 1999-04-21 | 2015-10-06 | Osmetech Technology, Inc. | Use of microfluidic systems in the electrochemical detection of target analytes |
| US8486247B2 (en) | 1999-04-21 | 2013-07-16 | Osmetch Technology, Inc. | Use of microfluidic systems in the electrochemical detection of target analytes |
| US7449328B2 (en) | 1999-05-03 | 2008-11-11 | Gen-Probe Incorporated | Probe matrix-based device for identifying microorganisms |
| US6376186B1 (en) | 1999-05-03 | 2002-04-23 | Gen-Probe Incorporated | Polynucleotide probes for detection and quantitation of staphylococcus |
| US6235484B1 (en) | 1999-05-03 | 2001-05-22 | Gen-Probe Incorporated | Polynucleotide probes for detection and quantitation of actinomycetes |
| US6326486B1 (en) | 1999-05-03 | 2001-12-04 | Gen-Probe Incorporated | Polynucleotide probes for detection and quantitation of bacteria in the family enterobacteriaceae |
| US6821770B1 (en) | 1999-05-03 | 2004-11-23 | Gen-Probe Incorporated | Polynucleotide matrix-based method of identifying microorganisms |
| US8481268B2 (en) | 1999-05-21 | 2013-07-09 | Illumina, Inc. | Use of microfluidic systems in the detection of target analytes using microsphere arrays |
| US9289766B2 (en) | 1999-05-21 | 2016-03-22 | Illumina, Inc. | Use of microfluidic systems in the detection of target analytes using microsphere arrays |
| US8080380B2 (en) | 1999-05-21 | 2011-12-20 | Illumina, Inc. | Use of microfluidic systems in the detection of target analytes using microsphere arrays |
| US8883424B2 (en) | 1999-05-21 | 2014-11-11 | Illumina, Inc. | Use of microfluidic systems in the detection of target analytes using microsphere arrays |
| WO2001006016A2 (en) * | 1999-07-20 | 2001-01-25 | Clinical Micro Sensors, Inc. | Amplification of nucleic acids with electronic detection |
| WO2001006016A3 (en) * | 1999-07-20 | 2001-08-23 | Clinical Micro Sensors Inc | Amplification of nucleic acids with electronic detection |
| AU779220B2 (en) * | 1999-07-26 | 2005-01-13 | Clinical Micro Sensors, Inc. | Sequence determination of nucleic acids using electronic detection |
| US7935481B1 (en) * | 1999-07-26 | 2011-05-03 | Osmetech Technology Inc. | Sequence determination of nucleic acids using electronic detection |
| WO2001007665A2 (en) * | 1999-07-26 | 2001-02-01 | Clinical Micro Sensors, Inc. | Sequence determination of nucleic acids using electronic detection |
| WO2001007665A3 (en) * | 1999-07-26 | 2002-05-02 | Clinical Micro Sensors Inc | Sequence determination of nucleic acids using electronic detection |
| WO2001021635A3 (en) * | 1999-09-22 | 2001-05-17 | Friz Biochem Gmbh | Method for electrochemically detecting sequence-specific nucleic acid-oligomer hybridisation events |
| WO2001021635A2 (en) * | 1999-09-22 | 2001-03-29 | Friz Biochem Gmbh | Method for electrochemically detecting sequence-specific nucleic acid-oligomer hybridisation events |
| WO2001031057A3 (en) * | 1999-10-22 | 2001-12-27 | Aventis Res & Tech Gmbh & Co | Double-strand nucleic acid probes and the use thereof |
| WO2001031057A2 (en) * | 1999-10-22 | 2001-05-03 | Nanogen Recognomics Gmbh | Double-strand nucleic acid probes and the use thereof |
| WO2001040511A3 (en) * | 1999-12-01 | 2002-05-10 | Secr Defence | Detection system |
| WO2001044501A2 (en) * | 1999-12-13 | 2001-06-21 | november Aktiengesellschaft Gesellschaft für Molekulare Medizin | Method and device for detecting and quantifying biomolecules |
| WO2001044501A3 (en) * | 1999-12-13 | 2002-07-18 | November Ag Molekulare Medizin | Method and device for detecting and quantifying biomolecules |
| US6518024B2 (en) | 1999-12-13 | 2003-02-11 | Motorola, Inc. | Electrochemical detection of single base extension |
| US6753143B2 (en) | 2000-05-01 | 2004-06-22 | Clinical Micro Sensors, Inc. | Target analyte detection using asymmetrical self-assembled monolayers |
| WO2001092277A1 (en) * | 2000-05-26 | 2001-12-06 | Minerva Biotechnologies Corporation | Metallocenes as signal-transducer for detection of chemical or biological events |
| US6787339B1 (en) | 2000-10-02 | 2004-09-07 | Motorola, Inc. | Microfluidic devices having embedded metal conductors and methods of fabricating said devices |
| US7615340B2 (en) | 2000-10-03 | 2009-11-10 | Minerva Biotechnologies Corporation | Electronic detection of interaction and detection of interaction based on the interruption of flow |
| EP1231214A3 (en) * | 2001-02-01 | 2002-11-20 | Hitachi Software Engineering Co., Ltd. | Biopolymer detector |
| EP1231214A2 (en) * | 2001-02-01 | 2002-08-14 | Hitachi Software Engineering Co., Ltd. | Biopolymer detector |
| US6582954B2 (en) | 2001-02-01 | 2003-06-24 | Hitachi Software Engineering Co., Ltd. | Biopolymer detector |
| EP1986000A3 (en) * | 2002-07-31 | 2009-07-22 | Kabushiki Kaisha Toshiba | Base sequence detection apparatus and base sequence automatic analyzing apparatus |
| US7745203B2 (en) | 2002-07-31 | 2010-06-29 | Kabushiki Kaisha Toshiba | Base sequence detection apparatus and base sequence automatic analyzing apparatus |
| EP1542000A1 (en) * | 2002-07-31 | 2005-06-15 | Kabushiki Kaisha Toshiba | Base sequence detecting device and base sequence automatic analyzer |
| EP1542000A4 (en) * | 2002-07-31 | 2007-08-15 | Toshiba Kk | Base sequence detecting device and base sequence automatic analyzer |
| US7138121B2 (en) | 2003-01-23 | 2006-11-21 | Spangler Brenda D | Biosensors utilizing dendrimer-immobilized ligands and there use thereof |
| WO2007011946A2 (en) * | 2005-07-15 | 2007-01-25 | Applera Corporation | Detection of nucleic acid amplification |
| WO2007011946A3 (en) * | 2005-07-15 | 2007-04-05 | Applera Corp | Detection of nucleic acid amplification |
| EP2677039A2 (en) | 2006-05-10 | 2013-12-25 | Dxterity Diagnostics | Detection of nucleic acid targets using chemically reactive oligonucleotide probes |
| US10829803B2 (en) | 2006-05-10 | 2020-11-10 | Dxterity Diagnostics Incorporated | Detection of nucleic acid targets using chemically reactive oligonucleotide probes |
| WO2009061941A2 (en) | 2007-11-06 | 2009-05-14 | Osmetech Molecular Diagnostics | Baseless nucleotide analogues and uses thereof |
| WO2010007526A1 (en) | 2008-07-17 | 2010-01-21 | Andreas Pfuetzner | Biomarkers for cardiodiabetes |
| WO2010009438A1 (en) | 2008-07-17 | 2010-01-21 | Andreas Pfuetzner | Biomarkers for insulin resistance and beta-cell dysfunction |
| WO2010064147A2 (en) | 2008-12-04 | 2010-06-10 | Ikfe Gmbh | Biomarkers for atherosclerosis |
| WO2010067207A1 (en) | 2008-12-11 | 2010-06-17 | Ikfe Gmbh | Biomarkers for insulin sensitizer drug response |
| WO2010076655A1 (en) | 2008-12-30 | 2010-07-08 | Ikfe Gmbh | Biomarkers for adipose tissue activity |
| WO2010079428A2 (en) | 2009-01-07 | 2010-07-15 | Ikfe Gmbh | Biomarkers for appetite regulation |
| EP2765205A1 (en) | 2009-04-01 | 2014-08-13 | DXTerity Diagnostics Incorporated | Chemical ligation dependent probe amplification (CLPA) |
| US9976177B2 (en) | 2009-04-01 | 2018-05-22 | Dxterity Diagnostics Incorporated | Chemical ligation dependent probe amplification (CLPA) |
| US9194836B2 (en) | 2009-08-07 | 2015-11-24 | Ohmx Corporation | Enzyme triggered redox altering chemical elimination (E-trace) immunoassay |
| US9624522B2 (en) | 2009-08-07 | 2017-04-18 | Ohmx Corporation | Single, direct detection of hemoglobin A1c percentage using enzyme triggered redox altering chemical elimination (e-trace) immunoassay |
| US10066257B2 (en) | 2010-03-29 | 2018-09-04 | Dxterity Diagnostics Incorporated | Methods and compositions for detecting target nucleic acids |
| WO2012021749A1 (en) | 2010-08-11 | 2012-02-16 | Celula, Inc. | Genotyping dna |
| US9250234B2 (en) | 2011-01-19 | 2016-02-02 | Ohmx Corporation | Enzyme triggered redox altering chemical elimination (E-TRACE) immunoassay |
| US9745616B2 (en) | 2011-05-17 | 2017-08-29 | Dxterity Diagnostics Incorporated | Methods and compositions for detecting target nucleic acids |
| WO2012158967A1 (en) | 2011-05-17 | 2012-11-22 | Dxterity Diagnostics Incorporated | Methods and compositions for detecting target nucleic acids |
| US9250203B2 (en) | 2012-01-09 | 2016-02-02 | Ohmx Corporation | Enzyme cascade methods for E-TRACE assay signal amplification |
| WO2014066704A1 (en) | 2012-10-24 | 2014-05-01 | Genmark Diagnostics, Inc. | Integrated multiplex target analysis |
| EP3427830A1 (en) | 2012-10-24 | 2019-01-16 | Genmark Diagnostics Inc. | Integrated multiplex target analysis |
| EP2965817A1 (en) | 2012-10-24 | 2016-01-13 | Genmark Diagnostics Inc. | Integrated multiplex target analysis |
| EP3919174A2 (en) | 2012-10-24 | 2021-12-08 | Genmark Diagnostics Inc. | Integrated multiplex target analysis |
| US9757095B2 (en) | 2014-06-10 | 2017-09-12 | Dxterity Diagnostics Incorporated | Devices and methods for collecting and stabilizing biological samples |
| US10463347B2 (en) | 2014-06-10 | 2019-11-05 | Dxterity Diagnostics Incorporated | Devices and methods for collecting and stabilizing biological samples |
| US10106847B1 (en) | 2017-08-24 | 2018-10-23 | Clinical Micro Sensors, Inc. | Electrochemical detection of bacterial and/or fungal infections |
| US10273535B2 (en) | 2017-08-24 | 2019-04-30 | Clinical Micro Sensors, Inc. | Electrochemical detection of bacterial and/or fungal infections |
| US10669592B2 (en) | 2017-08-24 | 2020-06-02 | Clinical Micro Sensors, Inc. | Electrochemical detection of bacterial and/or fungal infections |
| US11021759B2 (en) | 2017-08-24 | 2021-06-01 | Clinical Micro Sensors, Inc. | Electrochemical detection of bacterial and/or fungal infections |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4072206B2 (en) | 2008-04-09 |
| WO1998020162A3 (en) | 1998-11-12 |
| CA2270633A1 (en) | 1998-05-14 |
| JP2001507930A (en) | 2001-06-19 |
| AU739375B2 (en) | 2001-10-11 |
| EP0939762A2 (en) | 1999-09-08 |
| AU5196798A (en) | 1998-05-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU739375B2 (en) | Electrodes linked via conductive oligomers to nucleic acids | |
| US6090933A (en) | Methods of attaching conductive oligomers to electrodes | |
| US6495323B1 (en) | AC methods for the detection of nucleic acids | |
| US7384749B2 (en) | Electrodes linked via conductive oligomers to nucleic acids | |
| US6063573A (en) | Cycling probe technology using electron transfer detection | |
| US6096273A (en) | Electrodes linked via conductive oligomers to nucleic acids | |
| AU763494B2 (en) | Electronic methods for the detection of analytes utilizing monolayers | |
| US7090804B2 (en) | Amplification of nucleic acids with electronic detection | |
| US7160678B1 (en) | Compositions for the electronic detection of analytes utilizing monolayers | |
| US7393645B2 (en) | Compositions for the electronic detection of analytes utilizing monolayers | |
| US6290839B1 (en) | Systems for electrophoretic transport and detection of analytes | |
| AU765597B2 (en) | Electronic detection of nucleic acids using monolayers | |
| US20030087228A1 (en) | Electronic detection of nucleic acids using monolayers | |
| AU780739B2 (en) | Electrodes linked via conductive oligomers to nucleic acids | |
| AU2003271352A1 (en) | Electronic detection of nucleic acids using monolayers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH HU ID IL IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZW AM AZ BY KG KZ MD RU TJ TM |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH KE LS MW SD SZ UG ZW AT BE CH DE DK ES FI FR GB GR IE IT LU MC |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 51967/98 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1997946876 Country of ref document: EP |
|
| ENP | Entry into the national phase in: |
Ref country code: JP Ref document number: 1998 521668 Kind code of ref document: A Format of ref document f/p: F |
|
| ENP | Entry into the national phase in: |
Ref country code: CA Ref document number: 2270633 Kind code of ref document: A Format of ref document f/p: F Ref document number: 2270633 Country of ref document: CA |
|
| WWP | Wipo information: published in national office |
Ref document number: 1997946876 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 51967/98 Country of ref document: AU |