WO1993002151A1 - Methods of drilling well boreholes and compositions used therein - Google Patents
Methods of drilling well boreholes and compositions used therein Download PDFInfo
- Publication number
- WO1993002151A1 WO1993002151A1 PCT/US1992/005879 US9205879W WO9302151A1 WO 1993002151 A1 WO1993002151 A1 WO 1993002151A1 US 9205879 W US9205879 W US 9205879W WO 9302151 A1 WO9302151 A1 WO 9302151A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyoxyalkylene
- acylating agent
- amine
- salt
- composition
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 63
- 238000005553 drilling Methods 0.000 title claims abstract description 57
- 238000000034 method Methods 0.000 title claims abstract description 45
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 84
- 239000007795 chemical reaction product Substances 0.000 claims abstract description 37
- 150000003839 salts Chemical class 0.000 claims abstract description 33
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 24
- 239000012530 fluid Substances 0.000 claims description 52
- 150000001412 amines Chemical class 0.000 claims description 40
- 125000004432 carbon atom Chemical group C* 0.000 claims description 34
- 239000004094 surface-active agent Substances 0.000 claims description 27
- 239000002253 acid Substances 0.000 claims description 26
- -1 alkaline earth metal salt Chemical class 0.000 claims description 25
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 22
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 17
- 239000000194 fatty acid Substances 0.000 claims description 17
- 229930195729 fatty acid Natural products 0.000 claims description 17
- 239000000539 dimer Substances 0.000 claims description 15
- 150000004665 fatty acids Chemical class 0.000 claims description 15
- 229920005862 polyol Polymers 0.000 claims description 15
- 150000003077 polyols Chemical class 0.000 claims description 15
- 238000005698 Diels-Alder reaction Methods 0.000 claims description 13
- 150000002148 esters Chemical class 0.000 claims description 13
- 125000000217 alkyl group Chemical group 0.000 claims description 9
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 8
- 125000003342 alkenyl group Chemical group 0.000 claims description 8
- 150000008064 anhydrides Chemical class 0.000 claims description 8
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical group O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 claims description 8
- 239000000440 bentonite Substances 0.000 claims description 7
- 229910000278 bentonite Inorganic materials 0.000 claims description 7
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims description 6
- 229910052751 metal Inorganic materials 0.000 claims description 6
- 239000002184 metal Substances 0.000 claims description 6
- CFQZKFWQLAHGSL-FNTYJUCDSA-N (3e,5e,7e,9e,11e,13e,15e,17e)-18-[(3e,5e,7e,9e,11e,13e,15e,17e)-18-[(3e,5e,7e,9e,11e,13e,15e)-octadeca-3,5,7,9,11,13,15,17-octaenoyl]oxyoctadeca-3,5,7,9,11,13,15,17-octaenoyl]oxyoctadeca-3,5,7,9,11,13,15,17-octaenoic acid Chemical compound OC(=O)C\C=C\C=C\C=C\C=C\C=C\C=C\C=C\C=C\OC(=O)C\C=C\C=C\C=C\C=C\C=C\C=C\C=C\C=C\OC(=O)C\C=C\C=C\C=C\C=C\C=C\C=C\C=C\C=C CFQZKFWQLAHGSL-FNTYJUCDSA-N 0.000 claims description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 5
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 claims description 5
- 229910052784 alkaline earth metal Inorganic materials 0.000 claims description 5
- 239000004927 clay Substances 0.000 claims description 5
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical group C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims description 4
- 150000001408 amides Chemical class 0.000 claims description 4
- 239000002585 base Substances 0.000 claims description 4
- 150000001875 compounds Chemical class 0.000 claims description 4
- 239000011734 sodium Substances 0.000 claims description 4
- 229910052708 sodium Inorganic materials 0.000 claims description 4
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 claims description 3
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 claims description 3
- 229910021529 ammonia Inorganic materials 0.000 claims description 3
- 229940014800 succinic anhydride Drugs 0.000 claims description 3
- 239000001384 succinic acid Substances 0.000 claims description 2
- 239000003513 alkali Substances 0.000 claims 2
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 claims 2
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 32
- 239000000047 product Substances 0.000 description 30
- 235000002639 sodium chloride Nutrition 0.000 description 24
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 21
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 21
- 239000004711 α-olefin Substances 0.000 description 19
- 150000007513 acids Chemical class 0.000 description 17
- 238000006243 chemical reaction Methods 0.000 description 16
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 15
- 239000012141 concentrate Substances 0.000 description 15
- 150000004985 diamines Chemical class 0.000 description 14
- 239000000126 substance Substances 0.000 description 12
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- 229920001732 Lignosulfonate Polymers 0.000 description 9
- 150000001336 alkenes Chemical class 0.000 description 9
- 150000001298 alcohols Chemical class 0.000 description 8
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 7
- 239000000463 material Substances 0.000 description 7
- 229910052757 nitrogen Inorganic materials 0.000 description 7
- 229920001223 polyethylene glycol Polymers 0.000 description 7
- 229920001451 polypropylene glycol Polymers 0.000 description 7
- 239000013535 sea water Substances 0.000 description 7
- 235000011121 sodium hydroxide Nutrition 0.000 description 7
- 239000004215 Carbon black (E152) Substances 0.000 description 6
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 6
- 239000007859 condensation product Substances 0.000 description 6
- 229930195733 hydrocarbon Natural products 0.000 description 6
- 150000002430 hydrocarbons Chemical class 0.000 description 6
- 238000006386 neutralization reaction Methods 0.000 description 6
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 239000010428 baryte Substances 0.000 description 5
- 229910052601 baryte Inorganic materials 0.000 description 5
- 150000002194 fatty esters Chemical class 0.000 description 5
- 239000013505 freshwater Substances 0.000 description 5
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 5
- 150000002989 phenols Chemical class 0.000 description 5
- 125000001424 substituent group Chemical group 0.000 description 5
- 239000003784 tall oil Chemical class 0.000 description 5
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 4
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 4
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 4
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 4
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 4
- 125000002947 alkylene group Chemical group 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 150000002193 fatty amides Chemical class 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 150000002334 glycols Chemical class 0.000 description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 4
- 229920000768 polyamine Polymers 0.000 description 4
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 4
- GPRLSGONYQIRFK-MNYXATJNSA-N triton Chemical compound [3H+] GPRLSGONYQIRFK-MNYXATJNSA-N 0.000 description 4
- QGLWBTPVKHMVHM-KTKRTIGZSA-N (z)-octadec-9-en-1-amine Chemical compound CCCCCCCC\C=C/CCCCCCCCN QGLWBTPVKHMVHM-KTKRTIGZSA-N 0.000 description 3
- RFVNOJDQRGSOEL-UHFFFAOYSA-N 2-hydroxyethyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCO RFVNOJDQRGSOEL-UHFFFAOYSA-N 0.000 description 3
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 3
- 241000143060 Americamysis bahia Species 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 239000004952 Polyamide Substances 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 230000002411 adverse Effects 0.000 description 3
- 229910052783 alkali metal Inorganic materials 0.000 description 3
- 150000001340 alkali metals Chemical class 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 150000003863 ammonium salts Chemical class 0.000 description 3
- 239000001913 cellulose Substances 0.000 description 3
- 229920002678 cellulose Polymers 0.000 description 3
- JRBPAEWTRLWTQC-UHFFFAOYSA-N dodecylamine Chemical compound CCCCCCCCCCCCN JRBPAEWTRLWTQC-UHFFFAOYSA-N 0.000 description 3
- SZVJSHCCFOBDDC-UHFFFAOYSA-N iron(II,III) oxide Inorganic materials O=[Fe]O[Fe]O[Fe]=O SZVJSHCCFOBDDC-UHFFFAOYSA-N 0.000 description 3
- 239000000314 lubricant Substances 0.000 description 3
- 230000001050 lubricating effect Effects 0.000 description 3
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid group Chemical group C(CCCCCCC\C=C/CCCCCCCC)(=O)O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- 229920001592 potato starch Polymers 0.000 description 3
- 235000017550 sodium carbonate Nutrition 0.000 description 3
- 229910000029 sodium carbonate Inorganic materials 0.000 description 3
- 239000003760 tallow Substances 0.000 description 3
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 3
- 239000013638 trimer Substances 0.000 description 3
- GGQQNYXPYWCUHG-RMTFUQJTSA-N (3e,6e)-deca-3,6-diene Chemical compound CCC\C=C\C\C=C\CC GGQQNYXPYWCUHG-RMTFUQJTSA-N 0.000 description 2
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 2
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 2
- VQOXUMQBYILCKR-UHFFFAOYSA-N 1-Tridecene Chemical compound CCCCCCCCCCCC=C VQOXUMQBYILCKR-UHFFFAOYSA-N 0.000 description 2
- SPURMHFLEKVAAS-UHFFFAOYSA-N 1-docosene Chemical compound CCCCCCCCCCCCCCCCCCCCC=C SPURMHFLEKVAAS-UHFFFAOYSA-N 0.000 description 2
- CRSBERNSMYQZNG-UHFFFAOYSA-N 1-dodecene Chemical compound CCCCCCCCCCC=C CRSBERNSMYQZNG-UHFFFAOYSA-N 0.000 description 2
- ADOBXTDBFNCOBN-UHFFFAOYSA-N 1-heptadecene Chemical compound CCCCCCCCCCCCCCCC=C ADOBXTDBFNCOBN-UHFFFAOYSA-N 0.000 description 2
- GQEZCXVZFLOKMC-UHFFFAOYSA-N 1-hexadecene Chemical compound CCCCCCCCCCCCCCC=C GQEZCXVZFLOKMC-UHFFFAOYSA-N 0.000 description 2
- JRZJOMJEPLMPRA-UHFFFAOYSA-N 1-nonene Chemical compound CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 2
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 2
- PJLHTVIBELQURV-UHFFFAOYSA-N 1-pentadecene Chemical compound CCCCCCCCCCCCCC=C PJLHTVIBELQURV-UHFFFAOYSA-N 0.000 description 2
- HFDVRLIODXPAHB-UHFFFAOYSA-N 1-tetradecene Chemical compound CCCCCCCCCCCCC=C HFDVRLIODXPAHB-UHFFFAOYSA-N 0.000 description 2
- MCIKGVLBLIZYRY-UHFFFAOYSA-N 2-(hexylamino)ethanol Chemical compound CCCCCCNCCO MCIKGVLBLIZYRY-UHFFFAOYSA-N 0.000 description 2
- RSPWVGZWUBNLQU-FOCLMDBBSA-N 3-[(e)-hexadec-1-enyl]oxolane-2,5-dione Chemical compound CCCCCCCCCCCCCC\C=C\C1CC(=O)OC1=O RSPWVGZWUBNLQU-FOCLMDBBSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- 239000005977 Ethylene Substances 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- REYJJPSVUYRZGE-UHFFFAOYSA-N Octadecylamine Chemical compound CCCCCCCCCCCCCCCCCCN REYJJPSVUYRZGE-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- 235000011941 Tilia x europaea Nutrition 0.000 description 2
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 2
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 2
- 125000002015 acyclic group Chemical group 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 125000002723 alicyclic group Chemical group 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 229960000892 attapulgite Drugs 0.000 description 2
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 239000003729 cation exchange resin Substances 0.000 description 2
- 238000005520 cutting process Methods 0.000 description 2
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 2
- 150000001470 diamides Chemical class 0.000 description 2
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 2
- 239000000428 dust Substances 0.000 description 2
- 229910001651 emery Inorganic materials 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 235000011087 fumaric acid Nutrition 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 150000004820 halides Chemical class 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 2
- 150000002429 hydrazines Chemical class 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- 239000004571 lime Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- 150000002736 metal compounds Chemical class 0.000 description 2
- 238000005555 metalworking Methods 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 150000002762 monocarboxylic acid derivatives Chemical class 0.000 description 2
- 150000002763 monocarboxylic acids Chemical class 0.000 description 2
- VAMFXQBUQXONLZ-UHFFFAOYSA-N n-alpha-eicosene Natural products CCCCCCCCCCCCCCCCCCC=C VAMFXQBUQXONLZ-UHFFFAOYSA-N 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- NHLUYCJZUXOUBX-UHFFFAOYSA-N nonadec-1-ene Chemical compound CCCCCCCCCCCCCCCCCC=C NHLUYCJZUXOUBX-UHFFFAOYSA-N 0.000 description 2
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 2
- CCCMONHAUSKTEQ-UHFFFAOYSA-N octadec-1-ene Chemical compound CCCCCCCCCCCCCCCCC=C CCCMONHAUSKTEQ-UHFFFAOYSA-N 0.000 description 2
- IOQPZZOEVPZRBK-UHFFFAOYSA-N octan-1-amine Chemical compound CCCCCCCCN IOQPZZOEVPZRBK-UHFFFAOYSA-N 0.000 description 2
- FATBGEAMYMYZAF-KTKRTIGZSA-N oleamide Chemical compound CCCCCCCC\C=C/CCCCCCCC(N)=O FATBGEAMYMYZAF-KTKRTIGZSA-N 0.000 description 2
- 235000021313 oleic acid Nutrition 0.000 description 2
- 150000002889 oleic acids Chemical class 0.000 description 2
- 150000002924 oxiranes Chemical class 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052625 palygorskite Inorganic materials 0.000 description 2
- 229920001983 poloxamer Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920000098 polyolefin Polymers 0.000 description 2
- 229920002503 polyoxyethylene-polyoxypropylene Polymers 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 235000007686 potassium Nutrition 0.000 description 2
- 229960003975 potassium Drugs 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 239000001103 potassium chloride Substances 0.000 description 2
- 235000011164 potassium chloride Nutrition 0.000 description 2
- 150000003138 primary alcohols Chemical class 0.000 description 2
- 150000003141 primary amines Chemical class 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- WGYKZJWCGVVSQN-UHFFFAOYSA-N propylamine Chemical compound CCCN WGYKZJWCGVVSQN-UHFFFAOYSA-N 0.000 description 2
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- JHJLBTNAGRQEKS-UHFFFAOYSA-M sodium bromide Chemical compound [Na+].[Br-] JHJLBTNAGRQEKS-UHFFFAOYSA-M 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- VLDHWMAJBNWALQ-UHFFFAOYSA-M sodium;1,3-benzothiazol-3-ide-2-thione Chemical compound [Na+].C1=CC=C2SC([S-])=NC2=C1 VLDHWMAJBNWALQ-UHFFFAOYSA-M 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- ZDLBWMYNYNATIW-UHFFFAOYSA-N tetracos-1-ene Chemical compound CCCCCCCCCCCCCCCCCCCCCCC=C ZDLBWMYNYNATIW-UHFFFAOYSA-N 0.000 description 2
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 2
- 150000003628 tricarboxylic acids Chemical class 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- XFNJVJPLKCPIBV-UHFFFAOYSA-N trimethylenediamine Chemical compound NCCCN XFNJVJPLKCPIBV-UHFFFAOYSA-N 0.000 description 2
- 229960004418 trolamine Drugs 0.000 description 2
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 2
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 2
- VNDYJBBGRKZCSX-UHFFFAOYSA-L zinc bromide Chemical compound Br[Zn]Br VNDYJBBGRKZCSX-UHFFFAOYSA-L 0.000 description 2
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 2
- ZBUAAIKWLHWSPO-UHFFFAOYSA-N (Z)-20-aminoicos-11-en-1-ol Chemical compound OCCCCCCCCCCC=C/CCCCCCCCN ZBUAAIKWLHWSPO-UHFFFAOYSA-N 0.000 description 1
- RBACIKXCRWGCBB-UHFFFAOYSA-N 1,2-Epoxybutane Chemical compound CCC1CO1 RBACIKXCRWGCBB-UHFFFAOYSA-N 0.000 description 1
- FJLUATLTXUNBOT-UHFFFAOYSA-N 1-Hexadecylamine Chemical compound CCCCCCCCCCCCCCCCN FJLUATLTXUNBOT-UHFFFAOYSA-N 0.000 description 1
- 229940106006 1-eicosene Drugs 0.000 description 1
- FIKTURVKRGQNQD-UHFFFAOYSA-N 1-eicosene Natural products CCCCCCCCCCCCCCCCCC=CC(O)=O FIKTURVKRGQNQD-UHFFFAOYSA-N 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- MIJDSYMOBYNHOT-UHFFFAOYSA-N 2-(ethylamino)ethanol Chemical compound CCNCCO MIJDSYMOBYNHOT-UHFFFAOYSA-N 0.000 description 1
- KKFDCBRMNNSAAW-UHFFFAOYSA-N 2-(morpholin-4-yl)ethanol Chemical compound OCCN1CCOCC1 KKFDCBRMNNSAAW-UHFFFAOYSA-N 0.000 description 1
- UVYBWDBLVDZIOX-UHFFFAOYSA-N 2-(octylamino)ethanol Chemical compound CCCCCCCCNCCO UVYBWDBLVDZIOX-UHFFFAOYSA-N 0.000 description 1
- QJXHPFHKGPEMAM-UHFFFAOYSA-N 2-(pentadecylamino)ethanol Chemical compound CCCCCCCCCCCCCCCNCCO QJXHPFHKGPEMAM-UHFFFAOYSA-N 0.000 description 1
- BITAPBDLHJQAID-KTKRTIGZSA-N 2-[2-hydroxyethyl-[(z)-octadec-9-enyl]amino]ethanol Chemical compound CCCCCCCC\C=C/CCCCCCCCN(CCO)CCO BITAPBDLHJQAID-KTKRTIGZSA-N 0.000 description 1
- VARKIGWTYBUWNT-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanol Chemical compound OCCN1CCN(CCO)CC1 VARKIGWTYBUWNT-UHFFFAOYSA-N 0.000 description 1
- ZXZYUWIRFFYDHL-UHFFFAOYSA-N 2-[ethyl(hexyl)amino]oxyethanol Chemical compound CCCCCCN(CC)OCCO ZXZYUWIRFFYDHL-UHFFFAOYSA-N 0.000 description 1
- WFCSWCVEJLETKA-UHFFFAOYSA-N 2-piperazin-1-ylethanol Chemical compound OCCN1CCNCC1 WFCSWCVEJLETKA-UHFFFAOYSA-N 0.000 description 1
- CXMYWOCYTPKBPP-UHFFFAOYSA-N 3-(3-hydroxypropylamino)propan-1-ol Chemical compound OCCCNCCCO CXMYWOCYTPKBPP-UHFFFAOYSA-N 0.000 description 1
- MEULKYFXJVYKER-UHFFFAOYSA-N 3-methyloxirane-2,2-diamine Chemical compound CC1OC1(N)N MEULKYFXJVYKER-UHFFFAOYSA-N 0.000 description 1
- JIGUICYYOYEXFS-UHFFFAOYSA-N 3-tert-butylbenzene-1,2-diol Chemical compound CC(C)(C)C1=CC=CC(O)=C1O JIGUICYYOYEXFS-UHFFFAOYSA-N 0.000 description 1
- BLFRQYKZFKYQLO-UHFFFAOYSA-N 4-aminobutan-1-ol Chemical compound NCCCCO BLFRQYKZFKYQLO-UHFFFAOYSA-N 0.000 description 1
- RSWGJHLUYNHPMX-UHFFFAOYSA-N Abietic-Saeure Chemical class C12CCC(C(C)C)=CC2=CCC2C1(C)CCCC2(C)C(O)=O RSWGJHLUYNHPMX-UHFFFAOYSA-N 0.000 description 1
- 238000006596 Alder-ene reaction Methods 0.000 description 1
- 241000238426 Anostraca Species 0.000 description 1
- 241000238582 Artemia Species 0.000 description 1
- 229910021532 Calcite Inorganic materials 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 239000009261 D 400 Substances 0.000 description 1
- MHZGKXUYDGKKIU-UHFFFAOYSA-N Decylamine Chemical compound CCCCCCCCCCN MHZGKXUYDGKKIU-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 1
- 229920002166 Pluracol® V 10 Polymers 0.000 description 1
- 229920002556 Polyethylene Glycol 300 Polymers 0.000 description 1
- NPYPAHLBTDXSSS-UHFFFAOYSA-N Potassium ion Chemical compound [K+] NPYPAHLBTDXSSS-UHFFFAOYSA-N 0.000 description 1
- KHPCPRHQVVSZAH-HUOMCSJISA-N Rosin Chemical class O(C/C=C/c1ccccc1)[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 KHPCPRHQVVSZAH-HUOMCSJISA-N 0.000 description 1
- 239000004113 Sepiolite Substances 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- AWMVMTVKBNGEAK-UHFFFAOYSA-N Styrene oxide Chemical compound C1OC1C1=CC=CC=C1 AWMVMTVKBNGEAK-UHFFFAOYSA-N 0.000 description 1
- PLZVEHJLHYMBBY-UHFFFAOYSA-N Tetradecylamine Chemical compound CCCCCCCCCCCCCCN PLZVEHJLHYMBBY-UHFFFAOYSA-N 0.000 description 1
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 1
- 229920002359 Tetronic® Polymers 0.000 description 1
- XSTXAVWGXDQKEL-UHFFFAOYSA-N Trichloroethylene Chemical compound ClC=C(Cl)Cl XSTXAVWGXDQKEL-UHFFFAOYSA-N 0.000 description 1
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001339 alkali metal compounds Chemical class 0.000 description 1
- 150000001341 alkaline earth metal compounds Chemical class 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 125000005263 alkylenediamine group Chemical group 0.000 description 1
- KVLCHQHEQROXGN-UHFFFAOYSA-N aluminium(1+) Chemical compound [Al+] KVLCHQHEQROXGN-UHFFFAOYSA-N 0.000 description 1
- 229940007076 aluminum cation Drugs 0.000 description 1
- 238000005576 amination reaction Methods 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 238000009360 aquaculture Methods 0.000 description 1
- 244000144974 aquaculture Species 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 244000144987 brood Species 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229960005069 calcium Drugs 0.000 description 1
- 229910001622 calcium bromide Inorganic materials 0.000 description 1
- WGEFECGEFUFIQW-UHFFFAOYSA-L calcium dibromide Chemical compound [Ca+2].[Br-].[Br-] WGEFECGEFUFIQW-UHFFFAOYSA-L 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 229910052923 celestite Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000013530 defoamer Substances 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- 239000002283 diesel fuel Substances 0.000 description 1
- 229940043237 diethanolamine Drugs 0.000 description 1
- 238000006471 dimerization reaction Methods 0.000 description 1
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N dimethylmethane Natural products CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- UKMSUNONTOPOIO-UHFFFAOYSA-N docosanoic acid Chemical class CCCCCCCCCCCCCCCCCCCCCC(O)=O UKMSUNONTOPOIO-UHFFFAOYSA-N 0.000 description 1
- ILRSCQWREDREME-UHFFFAOYSA-N dodecanamide Chemical compound CCCCCCCCCCCC(N)=O ILRSCQWREDREME-UHFFFAOYSA-N 0.000 description 1
- 229940069096 dodecene Drugs 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000010459 dolomite Substances 0.000 description 1
- 229910000514 dolomite Inorganic materials 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 150000002171 ethylene diamines Chemical class 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 239000010685 fatty oil Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 150000002238 fumaric acids Chemical class 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 238000005227 gel permeation chromatography Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- JTOGFHAZQVDOAO-UHFFFAOYSA-N henicos-1-ene Chemical compound CCCCCCCCCCCCCCCCCCCC=C JTOGFHAZQVDOAO-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- XPXMKIXDFWLRAA-UHFFFAOYSA-N hydrazinide Chemical compound [NH-]N XPXMKIXDFWLRAA-UHFFFAOYSA-N 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 229940082005 hydrogenated tallow acid Drugs 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 239000003999 initiator Substances 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- LIKBJVNGSGBSGK-UHFFFAOYSA-N iron(3+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[Fe+3].[Fe+3] LIKBJVNGSGBSGK-UHFFFAOYSA-N 0.000 description 1
- 229910000015 iron(II) carbonate Inorganic materials 0.000 description 1
- YDZQQRWRVYGNER-UHFFFAOYSA-N iron;titanium;trihydrate Chemical compound O.O.O.[Ti].[Fe] YDZQQRWRVYGNER-UHFFFAOYSA-N 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- XCAUINMIESBTBL-UHFFFAOYSA-N lead(ii) sulfide Chemical compound [Pb]=S XCAUINMIESBTBL-UHFFFAOYSA-N 0.000 description 1
- 239000003077 lignite Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 229940096405 magnesium cation Drugs 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N n-Octanol Natural products CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- BBDGYADAMYMJNO-UHFFFAOYSA-N n-butyl-n-ethylbutan-1-amine Chemical compound CCCCN(CC)CCCC BBDGYADAMYMJNO-UHFFFAOYSA-N 0.000 description 1
- AFFLGGQVNFXPEV-UHFFFAOYSA-N n-decene Natural products CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 1
- GNVRJGIVDSQCOP-UHFFFAOYSA-N n-ethyl-n-methylethanamine Chemical compound CCN(C)CC GNVRJGIVDSQCOP-UHFFFAOYSA-N 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- LYRFLYHAGKPMFH-UHFFFAOYSA-N octadecanamide Chemical compound CCCCCCCCCCCCCCCCCC(N)=O LYRFLYHAGKPMFH-UHFFFAOYSA-N 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940113162 oleylamide Drugs 0.000 description 1
- 125000005702 oxyalkylene group Chemical class 0.000 description 1
- IPCSVZSSVZVIGE-UHFFFAOYSA-N palmitic acid group Chemical group C(CCCCCCCCCCCCCCC)(=O)O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 229920001083 polybutene Polymers 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000011181 potassium carbonates Nutrition 0.000 description 1
- 235000011118 potassium hydroxide Nutrition 0.000 description 1
- 229940093932 potassium hydroxide Drugs 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- AOHJOMMDDJHIJH-UHFFFAOYSA-N propylenediamine Chemical compound CC(N)CN AOHJOMMDDJHIJH-UHFFFAOYSA-N 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 229910052624 sepiolite Inorganic materials 0.000 description 1
- 235000019355 sepiolite Nutrition 0.000 description 1
- 229910021646 siderite Inorganic materials 0.000 description 1
- 229940083542 sodium Drugs 0.000 description 1
- FVEFRICMTUKAML-UHFFFAOYSA-M sodium tetradecyl sulfate Chemical compound [Na+].CCCCC(CC)CCC(CC(C)C)OS([O-])(=O)=O FVEFRICMTUKAML-UHFFFAOYSA-M 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- PXQLVRUNWNTZOS-UHFFFAOYSA-N sulfanyl Chemical class [SH] PXQLVRUNWNTZOS-UHFFFAOYSA-N 0.000 description 1
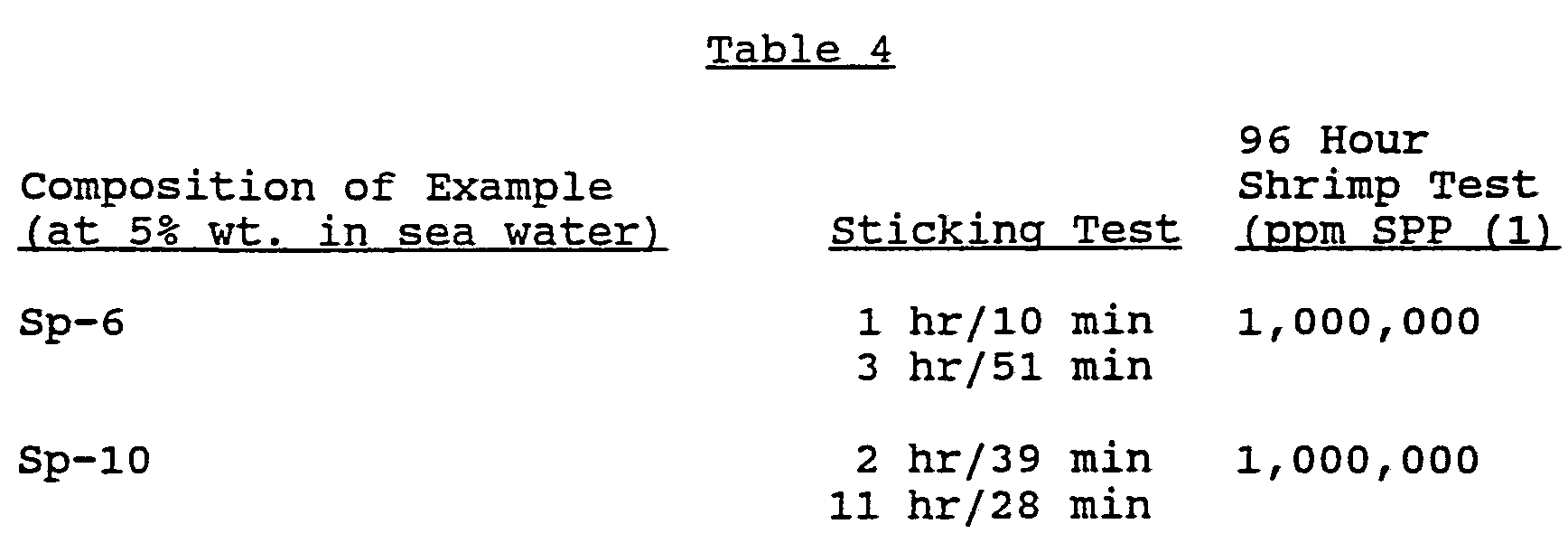
- 230000004083 survival effect Effects 0.000 description 1
- 239000012085 test solution Substances 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 231100000167 toxic agent Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 239000003440 toxic substance Substances 0.000 description 1
- KHPCPRHQVVSZAH-UHFFFAOYSA-N trans-cinnamyl beta-D-glucopyranoside Chemical class OC1C(O)C(O)C(CO)OC1OCC=CC1=CC=CC=C1 KHPCPRHQVVSZAH-UHFFFAOYSA-N 0.000 description 1
- 150000003623 transition metal compounds Chemical class 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000005829 trimerization reaction Methods 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229940102001 zinc bromide Drugs 0.000 description 1
- 229940006486 zinc cation Drugs 0.000 description 1
- 239000011592 zinc chloride Substances 0.000 description 1
- 235000005074 zinc chloride Nutrition 0.000 description 1
- 150000007934 α,β-unsaturated carboxylic acids Chemical class 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/04—Aqueous well-drilling compositions
- C09K8/14—Clay-containing compositions
- C09K8/18—Clay-containing compositions characterised by the organic compounds
- C09K8/22—Synthetic organic compounds
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH DRILLING; MINING
- E21B—EARTH DRILLING, e.g. DEEP DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B31/00—Fishing for or freeing objects in boreholes or wells
- E21B31/035—Fishing for or freeing objects in boreholes or wells controlling differential pipe sticking
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/04—Aqueous well-drilling compositions
- C09K8/06—Clay-free compositions
- C09K8/12—Clay-free compositions containing synthetic organic macromolecular compounds or their precursors
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2208/00—Aspects relating to compositions of drilling or well treatment fluids
- C09K2208/02—Spotting, i.e. using additives for releasing a stuck drill
Definitions
- the invention relates to a method of drilling a well borehole which encompasses using water-base drilling fluids and spotting fluids.
- the fluids contain the reaction products of a polycarboxylic acylating agent and an amine-terminated polyoxyalkylene.
- the primary functions of a drilling fluid or mud are: to carry chips and cuttings produced by drilling to the surface; to lubricate and cool the drill bit and drill string; to form a filter cake which obstructs filtrate invasion in the formation; to maintain the walls of the borehole; to control formation pressures and prevent lost returns; to suspend cuttings during rig shutdowns; and to protect the formation for later successful completion and production.
- Sticking of the drill pipe during the drilling is a common problem. This condition is commonly referred to as being a stuck pipe. Spotting fluids or stuck pipe fluids are generally used to free the drill pipe so that drilling may continue. Oil-base drilling fluids are commonly used to prevent stuck pipe. However, these fluids contain refined mineral oils or diesel fuels which adversely effect marine life. It is desirable to have drilling fluids and spotting fluids which are effective and yet do not adversely impact the environment.
- Re. 30,885 which is a reissue of U.S. Patent 4,239,635 relates to diamides and lubricants containing the same.
- the carboxylic acid terminated acid diamides and alkali metal, ammonium or amine salts thereof have lubrieating properties and are especially useful in aqueous metal-working fluids.
- Re. 31,522 which is a reissue of U.S. Patent 4,374,741, relates to polyamides and functional fluids containing the same.
- the polyamides are polyoxylene polyamides having a terminal carboxylic acid group and terminal amine group in the same molecule and the degree of polymerization of 210 and salts thereof are useful in preparing stable aqueous based metal-working and hydraulic fluids.
- U.S. Patent 4,107,061 relates to amino-amide lubricants derived from polymeric fatty acids and poly(oxy- ethylene) diamines. These materials are readily compatible with water in all proportions and form clear, aqueous solutions which have lubricating properties.
- U.S. Patents 4,661,275 and 4,664,834 relate to hydrocarbyl-substituted succinic acid and/or anhydride/amine-terminated poly(oxyalkylene) reaction products and aqueous systems containing the same. These reaction products are useful as shear-stable thickeners for functional fluids.
- U.S. Patent 4,795,581 relates to aqueous fluids thickened with fatty acid modified polyoxyalkylene diamines. These materials are useful for water/glycol based hydraulic fluids, cosmetics and surfactants.
- the present invention relates to a method, comprising the steps of:
- compositions comprising water and an amount, sufficient to reduce drilling torque, prevent sticking or twisting of a pipe during drilling, or release a stuck pipe in a well borehole, of (A) at least one reaction product of (A-1) at least one carboxylic acylating agent and (A-2) at least one amine- terminated polyoxyalkylene having a number average molecular weight from about 600 to about 10,000, or at least one salt of the reaction product, and drilling the well borehole.
- Drilling torque, twisting and sticking of the pipe during drilling and releasing of stuck pipe in a well borehole are all effectively controlled by the methods and compositions of the present invention.
- hydrocarbyl includes hydrocarbon, as well as substantially hydrocarbon, groups.
- substantially hydrocarbon describes groups which contain non-hydrocarbon substituents which do not alter the predominately hydrocarbon nature of the group.
- hydrocarbyl groups include the following:
- hydrocarbon substituents that is, aliphatic (e. g. , alkyl or alkenyl), alicyclic (e.g., cycloalkyl, cycloalkenyl) substituents, aromatic-, aliphatic- and alicyclic-substituted aromatic substituents and the like as well as cyclic substituents wherein the ring is completed through another portion of the molecule (that is, for example, any two indicated substituents may together form an alicyclic radical);
- aliphatic e. g. , alkyl or alkenyl
- alicyclic e.g., cycloalkyl, cycloalkenyl
- substituted hydrocarbon substituents that is, those substituents containing non-hydrocarbon groups which, in the context of this invention, do not alter the predominantly hydrocarbon substituent; those skilled in the art will be aware of such groups (e.g., halo (especially chloro and fluoro), hydroxy, alkoxy, mercapto, alkylmercapto, nitro, nitroso, sulfoxy, etc.);
- hetero substituents that is, substituents which will, while having a predominantly hydrocarbon character within the context of this invention, contain other than carbon present in a ring or chain otherwise composed of carbon atoms.
- Suitable heteroatoms will be apparent to those of ordinary skill in the art and include, for example, sulfur, oxygen, nitrogen and such substituents as, e.g., pyridyl, furyl, thienyl, imidazolyl, etc.
- no more than about 2, preferably no more than one, non-hydrocarbon substituent will be present for every ten carbon atoms in the hydrocarbyl group.
- the methods and fluids of the present invention use (A) the reaction product of (A-1) a hydrocarbyl-substi- tuted carboxylic acylating agent and (A-2) an amine-terminated polyoxyalkylene having a number average molecular weight of about 600 to about 10,000.
- the carboxylic acylating agents (A-1) are carbox- ylic acylating agents having from about 1 to about 4 carboxylic groups, preferably 2 or 3.
- the term acylating agents encompasses acids, anhydrides, lower esters (C 1-7 esters), halides, etc.
- the acylating agents are acids or anhydrides.
- Carboxylic acylating agents may be monocarboxylic or polycarboxylic acylating agents.
- Monocarboxylic acylating agents include fatty carboxylic acylating agents including fatty acids and Diels-Alder monocarboxylic reaction products.
- Fatty acids generally contain from about 8, preferably from about 10, more preferably from about 12 to about 30, more preferably to about 24 carbon atoms.
- Examples of fatty acids include stearic, oleic, lauric, linoleic, abietic, palmitic, sebacic, linolenic, behenic, tall oil and rosin acids.
- the monocarboxylic acylating agents may also be the reaction product of an ⁇ , ⁇ -unsaturated carboxylic acid (e.g., acrylic or methacrylic acid) with one or more olefins.
- olefins are preferably alpha-olefins (sometimes referred to as mono-1-olefins) or isomerized alpha-olefins.
- alpha-olefins examples include 1- octene, 1-nonene, l-decene, 1-dodecene, 1-tridecene, 1- tetradecene, 1-pentadecene, 1-hexadecene, 1-heptadecene, 1- octadecene, 1-nonadecene, 1-eicosene, 1-henicosene, 1- docosene, 1-tetracosene, etc.
- alpha-olefin fractions that can be used include the C 15-18 alpha-olefins, C 12-16 alpha-olefins, C 14-16 alpha-olefins, C 14-18 alpha-olefins, C 16-18 alpha-olefins, C 16-20 alpha-olefins, C 22-28 alpha-olefins, etc.
- the C 16 and C 16-18 alpha-olefins are particularly preferred.
- Isomerized alpha-olefins may also be used to form Diels-Alder reaction products. These olefins are alphaolefins that have been converted to internal olefins.
- the isomerized alpha-olefins suitable for use herein are usually in the form of mixtures of internal olefins with some alpha-olefins present.
- the procedures for isomerizing alpha-olefins are well known to those in the art. Briefly these procedures involve contacting alpha-olefin with a cation exchange resin at a temperature in a range of about 80° to about 130°C until the desired degree of isomeriza- tion is achieved. These procedures are described for example in U.S. 4,108,889 which is incorporated herein by reference.
- the polycarboxylic acylating agents of the present invention include dicarboxylic acylating agents such as succinic acylating agents, dimer acylating agents, and Diels-Alder dicarboxylic acylating agents.
- dicarboxylic acylating agents such as succinic acylating agents, dimer acylating agents, and Diels-Alder dicarboxylic acylating agents.
- Tricarboxylic acylating agents include trimer acylating agents and Diels-Alder tricarboxylic acylating agents.
- the dimer acylating agents include products resulting from the dimerization of unsaturated fatty acids, e.g., the above-described fatty acids.
- the dimer acids have an average from about 18 , preferably from about 28 to about 44, prefereably to about 40 carbon atoms. In one embodiment, the dimer acids have preferably about 36 carbon atoms.
- the dimer acids are preferably prepared from c 18 fatty acids, such as oleic acids.
- the dimer acids are described in U.S. Patents 2,482,760, 2,482,761, 2,731,481, 2,793,219, 2,964,545, 2,978,468, 3,157,681, and 3,256,304, the entire disclosures of which are incorporated herein by reference. Examples of dimer acids include Empol® 1014, 1016 and 1018 Dimer Acid, each available from Emery Industries, Inc. and Hystrene® dimer acids 3675, 3680, 3687 and 3695, available from Humko Chemical.
- the polycarboxylic acylating agents are dicarboxylic acylating agents which are the Diels-Alder type reaction products of an unsaturated fatty acid (e.g., the above-described fatty acids, preferably tall oil acids and oleic acids) with alpha,beta-ethyl- enically unsaturated carboxylic acylating agent (e.g., acrylic or methacrylic acylating agents) such as are taught in U.S. Pat. No. 2,444,328, the disclosure of which is incorporated herein by reference.
- These Diels-Alder acylating agents include Westvaco® Diacid H-240, 1525 and 1550, each being commercially available from the Westvaco Corporation.
- the polycarboxylic acids or anhydrides are hydrocarbyl-substituted succinic acylating agents, preferably acids or anhydrides, more preferably anhydrides.
- the hydrocarbyl group generally contains an average from about eight, preferably from about 14, more preferably from about 16 to about 40, preferably to about 30, more preferably to about 24, still more preferably to about 18 carbon atoms.
- the hydrocarbyl group is an alkenyl group.
- the alkenyl group may be derived from one or more of the above-described olefins.
- the succinic acylating agents are prepared by reacting the above-described olefins or isomerized olefins with unsaturated carboxylic acids such as fumaric acids or maleic acid or anhydride at a temperature of about 160° to about 240°C, preferably about 185° to about 210°C.
- Free radical initiators e.g., t-butyl catechol
- the procedures for preparing the acylating agents are well known to those skilled in the art and have been described for example in U.S. Patent 3,412,111; and Ben et al, "The Ene Reaction of Maleic Anhydride With Alkenes", J.C.S. Perkin II (1977), pages 535-537. These references are incorporated by reference for their disclosure of procedures for making the above acylating agents.
- the polycarboxylic acylating agent may also be a tricarboxylic acylating agent.
- tricarboxylic acylating agents include trimer and Diels-Alder tricarboxylic acylating agents. These acylating agents generally contain an average from about 18, preferably from about 30, more preferably from about 36 to about 66, preferably to about 60 carbon atoms. Trimer acids are prepared by the trimerization of the above-described fatty acids.
- the Diels-Alder tricarboxylic acylating agents are prepared by reacting an unsaturated monocarboxylic acid with a alpha,beta-ethlenically unsaturated dicarboxylic acid (e.g., fumaric acid or maleic acid or anhydride).
- a alpha,beta-ethlenically unsaturated dicarboxylic acid e.g., fumaric acid or maleic acid or anhydride
- the Diels-Alder acylating agent contains an average from about 12, preferably from about 18 to about 40, preferably to about 30 carbon atoms.
- these tricarboxylic acids include Empol® 1040 available commer- cially from Emery Industries, Hystrene® 5460 available commercially from Humko Chemical, and Unidyme® 60 available commercially from Union Camp Corporation.
- the carboxylic acylating agent (A-1) is a mixture containing at least 10% by weight of a carboxylic acylating agent having at least three carboxylic groups.
- the mixture preferably contains at least 50% by weight, preferably 80% by weight, preferably 90% by weight tricarboxylic acylating agent.
- the carboxylic acylating agents may be mixtures of the above-identified tricarboxylic acylating agents with monocarboxylic acyl- ating agents and the above-identified dicarboxylic acylating agents.
- the mixture may contain mono-, di-, or tricarboxylic acids.
- the monocarboxylic acids may have from 2, preferably from about 8, more preferably from about 12 to about 30, preferably to about 24 carbon atoms.
- Examples of monocarboxylic acids include acetic, propionic, butyric and fatty carboxylic acids such as oleic, stearic, linoleic, dodecanoic or tall oil acids.
- the amine-terminated polyoxyalkylenes are preferably alpha-omega amine-terminated polyoxyalkylenes. These amines include alpha-omega diamino polyoxyethylene, alpha-omega diamino polyoxypropylene-polyoxyethylene- polyoxypropylene or alpha-omega diamino propyleneoxide capped polyoxyethylene.
- the amine-terminated polyoxyalkylenes may also be a urea condensate of such alpha-omega diamino polyoxyalkylenes described above.
- the amine- terminated polyoxyalkylenes may also be polyamino (e.g., triamino, tetramino, etc.). These compounds generally have a number average molecular weight from about 600, typically from about 2000, preferably from about 3000, more prefera- bly from about 4000 to about 10,000, preferably to about 8000, more preferably to about 7000.
- the diamines are represented by the formula R R R R
- each R is independently hydrogen or an alkyl group having from 1 to about 8 carbon atoms, preferably 1 to about 3, more preferably 1; a is a number in the range of from zero to about 200; b is a number in the range of form about 10 to about 650; and c is a number in the range of from zero to about 200.
- These diamines preferably have number average molecular weights in the range from about 600, preferably from about 1000, more preferably from about 3000, more preferably from about 4000 to about 10,000, preferably to about 7000.
- diamines include Jeffamine® ED-600 wherein a+c is approximately 2.5 and b is approximately 8.5; Jeffamine® ED-900 wherein a+c is approximately 2.5 and b is approximately 15.5; Jeffamine® ED-2001 wherein a+c is approximately 2.5 and b is approximately 40.5; Jeffamine® ED-4000 wherein a+c is approximately 2.5 and b is approximately 86.0; and ED- 6000 wherein a+c is approximately 2.5 and b is approximately 131.5.
- the numerical value of the Jeffamine® approximate number average molecular weight. For instance, Jeffamine® ED-6000 has an approximate number average molecular weight of 6000.
- the amine-terminated polyoxyalkylene is a diamine preferably amine-terminated polypropylene glycols. These diamines are represented by the formula
- d is from 1 to about 150, preferably 2 to about 100, more preferably 2 to about 75.
- examples of these amines include Jeffamine® D-230 wherein d is about 2-3; Jeffamine® D-400 wherein d is about 5-6; Jeffamine® D-2000 wherein d is an average of about 33; and Jeffamine® D-4000 wherein d is an average of about 68.
- the diamines are represented by the formula R R 0 R R
- the amine-terminated polyoxyalkylene is a monoamino polyoxyalkylene such as an amino polyoxypropylene-polyoxyethylene-polyoxypropylene, or an amino polyoxypropylene.
- These amines are generally prepared by the reaction of a monohydric alcohol with an epoxide, such as styrene oxide, 1,2-butene oxide, ethylene oxide, propylene oxide and the like, more preferably ethylene oxide, propylene oxide or mixtures thereof. The terminal hydroxyl group is then converted to an amino group.
- amines are represented by the structure: wherein f is 1 to about 150, R 1 is an alkoxy group having 1 to about 18 carbon atoms, and each R is defined above.
- f is l to 100, more preferably about 4 to about 40.
- R is preferably an alkoxy group having from 1 to 12 carbon atoms, more preferably a methoxy group.
- These types of amines are available from Texaco Chemical Company under the tradename Jeffamine. Specific examples of these amines include Jeffamine® M-600; M-1000, M-2005 and M-2070 amines.
- the amine-terminated polyoxyalkylene is a triamine prepared by treating a triol with ethylene oxide, propylene oxide, or mixtures thereof, followed by amination of the terminal hydroxyl group.
- These amines are available commercially from Texaco Chemical Company under the tradename Jeffamine® triamines. Examples of these amines include, Jeffamine® T-403, which is trimethylolpropane treated with about 5-6 moles of propylene oxide, Jeffamine® T-3000, which is glycerine treated with 50 moles of propylene oxide, and Jeffamine® T-5000, which is glycerine treated with 85 moles of propylene oxide.
- the above amine-terminated polyoxyalkylenes are reacted with the above polycarboxylic acylating agents at an equivalent ratio of acylating agent to amine of about (1-4:1), preferably (1-2:1), more preferably (3:2), at 125°C to 250°C, more preferably about 150°C to about 225°C.
- the reaction is usually accomplished within about two to about ten hours, more preferably about two to about six, more preferably about four hours, more preferably between 0.25 to about 2 hours.
- the reaction product (A) of (A-1) a carboxylic acylating agent and (A-2) an amine-terminated polyoxyalkylene or salt of the reaction product may be used in the present invention.
- the salt is a metal salt, ammonium salt or a mixed salt, e.g., ammonium plus metal.
- the ammonium salt of the reaction product (A) may be derived from ammonia or any amine.
- the amine useful in making ammonium salts of amidic acids may be any of the amine-terminated polyoxyalkylenes described above. Further, the amine may be an alkyl monoamine, or a hydroxyamine.
- the alkyl monoamines are primary, secondary or tertiary monoamines.
- the alkyl monoamines generally contain from 1 to about 24 carbon atoms, more preferably 1 to about 12, more preferably 1 to about 6 in each alkyl group.
- Examples of primary monoamines useful in the present invention include methylamine, ethylamine, propyl- amine, butylamine, octylamine, and dodecylamine.
- Examples of secondary monoamines are given above. Tertiary monoamines include trimethylamine, tributylamine, methyldi- ethylamine, ethyldibutylamine
- the amines are hydroxy- amines.
- the hydroxyamines are primary, secondary or tertiary alkanol amines or mixtures thereof.
- Such amines can be represented by the Formulae:
- each R 2 is independently a hydrocarbyl group of one to about eight carbon atoms or hydroxyhydrocarbyl group of two to about eight carbon atoms and R 3 is a divalent hydrocarbyl group of about two to about 18 carbon atoms.
- the group -R 3 -OH in such formulae represents the hydroxyhydrocarbyl group.
- R 3 can be an acyclic, alicyclic or aromatic group.
- R 3 is an acyclic straight or branched alkylene group such as an ethylene, 1,2-propylene, 1,2- butylene or 1,2-octadecylene group, more preferably an ethylene or propylene group, more preferably an ethylene group.
- R 2 groups are present in the same molecule they can be joined by a direct carbon-to-carbon bond or through a heteroatom (e.g., oxygen, nitrogen or sulfur) to form a 5-, 6-, 7- or 8-membered ring structure.
- heterocyclic amines include N-(hydroxyl lower alkyl)-morpholines, -thiomorpholines, -piperidines, -oxazoli- dines, -thiazolidines and the like.
- each R 2 is independently a methyl, ethyl, propyl, butyl, pentyl or hexyl group.
- these hydroxyamines include monoethanol amine, diethanol amine, triethanol amine, diethylethanol amine, ethylethanol amine, etc.
- the hydroxyamines can also be an ether N-(hydroxyhydrocarbyl) amine.
- Such N- (hydroxyhydrocarbyl) amines can be conveniently prepared by reaction of epoxides with afore- described amines and can be represented by the Formulae:
- R 2 may also be a hydroxypoly- (hydrocarbyloxy) group.
- the salts of the amidic acids are formed from hydroxyamines.
- hydroxyamines can be represented by the formula
- R 4 is a hydrocarbyl group having from 8 to about 30 carbon atoms, preferably 8 to about 24, more preferably 10 to about 18 carbon atoms.
- R 4 is preferably an alkyl or alkenyl group, more preferably an alkenyl group.
- R 4 is preferably an octyl, decyl, dodecyl, tridecyl, tetradecyl, hexadecyl, octadecyl, oleyl, tallow or soya.
- g is preferably one to about 100, more preferably 2 to about 50, more preferably 2 to about 20, more preferably 3 to about 10, more preferably about 5.
- each R 3 is as described above.
- each R 3 is independently an ethylene or propylene group.
- hydroxyamines can be prepared by techniques well known in the art, and many such hydroxyamines are commercially available. They may be prepared, for example, by reaction of primary amines containing at least 6 carbon atoms with various amounts of alkylene oxides such as ethylene oxide, propylene oxide, etc.
- the primary amines may be single amines or mixtures of amines such as obtained by the hydrolysis of fatty oils such as tallow oils, sperm oils, coconut oils, etc.
- fatty acid amines containing from about 8 to about 30 carbon atoms include saturated as well as unsaturated aliphatic amines such as octyl amine, decyl amine, lauryl amine, stearyl amine, oleyl amine, myristyl amine, palmityl amine, dodecyl amine, and octadecyl amine.
- the useful hydroxyamines where h in the above formula (V) is zero include 2-hydroxyethylhexylamine, 2-hydroxyethyloctylamine, 2-hydroxyethylpentadecylamine,
- a number of hydroxyamines wherein h is zero are available from the Armak Chemical Division of Akzona, Inc., Chicago, Illinois, under the general trade designation "Ethomeen” and "Propomeen”. Specific examples of such products include “Ethomeen C/15” which is an ethylene oxide condensate of a cocoamine containing about 5 moles of ethylene oxide; “Ethomeen C/20” and “C/25" which also are ethylene oxide condensation products from cocoamine containing about 10 and 15 moles of ethylene oxide respectively; “Ethomeen O/12” which is an ethylene oxide condensation product of oleylamine containing about 2 moles of ethylene oxide per mole of amine.
- Ethomeen S/15 and S/20 which are ethylene oxide condensation products with soyaamine containing about 5 and 10 moles of ethylene oxide per mole of amine respectively; and "Ethomeen T/12, T/15” and “T/25" which are ethylene oxide condensation products of tallowamine containing about 2, 5 and 15 moles of ethylene oxide per mole of amine respectively.
- "Propomeen O/12” is the condensation product of one mole of oleyl amine with 2 moles propylene oxide.
- the salt is formed from Ethomeen C/15 or S/15 or mixtures thereof.
- hydroxyamines where h is one include "Ethoduomeen T/13" and "T/20" which are ethylene oxide condensation products of N-tallow trimethylene diamine containing 3 and 10 moles of ethylene oxide per mole of diamine, respectively.
- the fatty polyamine diamines include mono- or dialkyl, symmetrical or asymmetrical ethylene diamines, propane diamines (1,2, or 1,3), and polyamine analogs of the above. Suitable commercial fatty polyamines are "Duomeen C" (N-coco-1,3-diaminopropane), “Duomeen S” (N-soya-l,3-diaminopropane), “Duomeen T” (N-tallow-1,3- diaminopropane), or “Duomeen O” (N-oleyl-l,3-diaminopro- pane). "Duomeens” are commercially available diamines described in Product Data Bulletin No. 7-10R1 of Armak Chemical Co., Chicago, Illinois. In another embodiment, the secondary amines may be cyclic amines such as piperidine, piperazine, morpholine, etc.
- the metal salt of reaction product (A) may be prepared from an alkali metal, alkaline earth metal or transition metal compound, preferably an alkali metal or an alkaline earth metal compound, more preferably an alkali metal compound.
- metal compounds include sodium, potassium, calcium, magnesium, zinc or aluminum cation, more preferably, a sodium or potassium cation.
- the metal compounds are formed by treating the reaction product with a metal oxide, hydroxide, or halide.
- the metal salt is formed between room temperature and about 120°C, more preferably room temperature to about 80°C.
- reaction products (A) of the present invention relate to reaction products (A) of the present invention. Unless otherwise indicated in the examples and elsewhere in the specification and claims, temperature is in degrees Celsius, parts are parts by weight and pressure is atmospheric.
- Example 1
- a reaction vessel is charged with 3503 parts (0.58 mole) of Jeffamine® ED-6000 (a diamine from Texaco Chemical Co. having an average molecular weight of 6000 and being a primary amine-terminated propylene oxide capped polyoxyethylene) and 497 parts (0.58 mole) of Unidyme® 60 (a trimer acid available commercially from Union Camp Corporation having 75% trimer acid, 25% dimer acid and derived from C 18 monomeric acids).
- the mixture is stirred and heated to 150°-220°C for four hours, while distillate (9.2 milliliters) is removed.
- the residue has a neutralization number of 7.5.
- a reaction vessel is charged with 2400 parts (1.2 moles) of Jeffamine® ED-2001 and 1611 parts (1.89 moles) of Unidyme® 60. The mixture is stirred and heated to 150°- 200°C for 3.7 hours, while distillate (33 milliliters) is removed. The residue has a neutralization number of 42.
- a reaction vessel is charged with 3660 parts
- a reaction vessel is charged with 3000 parts (1 equivalent) of Jeffamine® ED-6000, 412 parts (2 equivalents) of a dimer acid (commercially available from Westvaco Corporation under the tradename Westvaco dimer acid 1550).
- the mixture is heated under nitrogen to 154°C in 40 minutes.
- the reaction is maintained between 150° and 200°C for four hours while 8.5 milliliters of distillate are collected.
- the reaction mixture is cooled to 120°C and the reaction temperature is maintained for 1.5 hours while 1.1 milliliters of distillate is collected.
- the reaction temperature is increased to 200°C while 0.1 milliliter of distillate is removed.
- the residue has a neutralization number of 16.5.
- a reaction vessel is charged with 2225 parts,
- a reaction product is prepared as described in Example 6 using 2600 parts (0.43 mole) of Jeffamine® ED-
- Examples 12-15 are prepared by the procedure described in Example 6, except there is no addition of a 50% aqueous solution of sodium mercaptobenzothiazole.
- the acylated amine-terminated polyoxyalkylenes i.e. reaction product (A) may be used alone or in combination with (B) a surfactant.
- the combination of the acylated amine-terminated polyoxyalkylene and a surfactant gives improved thickening, wetting and emulsifying properties to fluids.
- the combination usually contains from about 5%, preferably from about 15%, more preferably from about 25%, more preferably from about 50% to about 95%, preferably to about 85%, more preferably to about 75% of the acylated amine-terminated polyoxyalkylene.
- the combination usually contains from about 5%, preferably from about 10%, more preferably from about 20% to about 90%, preferably to about 75%, more preferably to about 50% by weight of the surfactant.
- the surfactants include nonionic, cationic and anionic surfactants.
- the surfactants include polyoxyalkylene amines, polyoxyalkylene amides, polyoxyalkylene alcohols, polyoxyalkylene esters, and fatty acid salts or mixtures of these surfactants. Any of the above described polyoxyalkylene amines, including the above-described amine-terminated polyoxyalkylenes and hydroxyamines (Ethomeens, Ethoduomeens, etc.), may be used as surfactants in the present invention.
- the polyoxyalkylene glycols may be polyoxyethylene glycols or polyoxypropylene -glycols.
- Useful polyoxyethylene glycols are available from Union Carbide under the trade name Carbowax® PEG 300, 600, 1000 and 1450.
- the polyoxyalkylene glycols are preferrably polyoxypropylene glycols where the oxypropylene units are at least 80% of the total. The remaining 20% may be ethylene oxide or butylene oxide or other such esters, olefins and the like which may be polarized with polypropylene oxide.
- Useful polyoxypropylene glycols are available from Union Carbide under the trade name NIAX 425; and NIAX 1025.
- Useful polyoxypropylene glycols are available from Dow Chemical and sold by the trade name PPG-1200, and PPG-2000.
- polyoxyalkylene polyols are the liquid polyols available from Wyandotte Chemicals Company under the name PLURONIC Polyols and other similar polyols. These PLURONIC Polyols correspond to the formula
- i, j, and k are integers greater than 1 such that the -CH 2 CH 2 O-groups comprise from about 10% to about 15% by weight of the total number average molecular weight of the glycol, the number average molecular weight of said polyols being from about 2500 to about 4500.
- This type of polyol can be prepared by reacting propylene glycol with propylene oxide and then with ethylene oxide.
- a particularly useful polyoxyalkylene polyol is Pluracol® V-10 which is a polyoxyalkylene derivative of trimethylol propane having a molecular weight of 22,000. This material is available commercially from BASF Corporation, Parsippany, New Jersey, U.S.A.
- the surfactant is an alkyl- terminated polyoxyalkylene.
- alkyl-terminated polyoxyalkylenes are known in the art, and many are available commercially.
- the alkyl-terminated polyoxyalkylenes are produced generally by treating an aliphatic alcohol with an excess of an alkylene oxide such as ethylene oxide or propylene oxide. For example, from about 6 to about 40 moles of ethylene oxide or propylene oxide may be condensed with the aliphatic alcohol.
- the alkyl-terminated polyoxyalkylene polyols useful in the present invention are available commercially under such trade names as "TRITON®” from Rohm & Haas Company, "Carbowax®” and “TERGITOL®” from Union Carbide, "ALFONIC®” from Conoco Chemicals Company, and “NEODOL®” from Shell Chemical Company.
- the TRITON® materials are identified generally as polyethoxylated alcohols or phenols.
- the TERGITOLS® are identified as polyethylene glycol ethers of primary or secondary alcohols; the ALFONIC® materials are identified as ethoxylated linear alcohols which may be represented by the general structural formula
- ALFONIC® ethoxylates characterized by the above formula include ALFONIC® 1012-60 wherein 1 is about 8 to 10 and m is an average of about 5 to 6; ALFONIC® 1214-70 wherein 1 is about 10-12 and m is an average of about 10 to about 11; ALFONIC® 1412-60 wherein 1 is from 10-12 and m is an average of about 7; and
- ALFONIC® 1218-70 wherein 1 is about 10-16 and m is an average of about 10 to about 11.
- the Carbowax® methoxy polyethylene glycols are linear ethoxylated polymer of methanol. Examples of these materials include Carbowax® methoxy polyethylene glycol
- the NEODOL® ethoxylates are ethoxylated alcohols wherein the alcohols are a mixture of alcohols containing from 12 to about 15 carbon atoms, and the alcohols are partially branched chain primary alcohols.
- the ethoxylates are obtained by reacting the alcohols with an excess of ethylene oxide such as from about 3 to about 12 or more moles of ethylene oxide per mole of alcohol.
- NEODOL® ethoxylate 23-6.5 is a partially branched chain alcoholate of 12 to 13 carbon atoms with an average of about 6 to about 7 ethoxy units.
- polystyrene resin Another group of polyols are the commercially available liquid TETRONIC polyols sold by Wyandotte Chemi- cals Corporation. These polyols are represented by the general formula:
- Such hydroxyamines are described in U.S. Patent No. 2,979,528 which is incorporated herein by reference. Those hydroxyamines corresponding to the above formula having a number average molecular weight of up to about 10,000 wherein the ethyleneoxy groups contribute to the total number average molecular weight in the percentage ranges discussed above are preferred. A specific example would be such a hydroxyamine having a number average molecular weight of about 8000 wherein the ethyleneoxy groups account for 7.5%-12% by weight of the total number average molecular weight.
- Such hydroxyamines can be prepared by reacting an alkylene diamine such as ethylene diamine, propylene diamine, hexamethylene diamine etc., with propylene oxide. Then the resulting product is reacted with ethylene oxide.
- the surfactant is a propoxylated hydrazine.
- Propoxylated hydrazines are available commercially under the tradename QxyprufTM. Examples of propoxylated hydrazines include QxyprufTM 6, 12 and 20 which are hydrazine treated with 6, 12 and 20 moles of propylene oxide, respectively.
- the surfactant is a polyoxyalkylated phenol.
- the phenol may be substituted or unsubstituted.
- a preferred polyoxyalkylated phenol is a polyoxyethylate nonylphenol.
- Polyoxyalkylated phenols are availabe commercially from Rohm and Haas Co. under the tradename Triton® and Texaco Chemical Company under the tradename Surfonic®. Examples of polyoxyalkylated phenols include Triton® AG-98, N series, and X series polyoxyethylated nonylphenols.
- the surfactant is a polyoxyalkylene fatty ester.
- Polyoxyalkylene fatty esters may be prepared from any polyoxyalkylene polyol and a fatty acid.
- the polyoxyalkylene polyol is any disclosed herein.
- the fatty acid is preferably the fatty monocarboxylic acid described above.
- Polyoxyalkylene fatty esters are available commercially from Armak Company under the tradename EthofatTM.
- polyoxyalkylene fatty esters include EthofatTM C/15 and C/25, which are coco fatty esters formed using 5 and 15 moles, respectively, of ethylene oxide; EthofatTM O/15 and O/20, which are oleic esters formed using 5 and 10 moles of ethylene oxide; and Ethofat 60/15, 60/20 and 60/25 which are stearic esters formed with 5, 10 and 15 moles of ethylene oxide respectively.
- the surfactant is a polyoxyalkylated fatty amide.
- the fatty amide is polyoxypropylated or polyoxyethylated, more preferably polyoxyethylated.
- fatty acids which may be polyoxyalkylated include oleylamide, stearylamide, tallow- amide, soyaamide, cocoamide, and laurylamide.
- Polyoxyalkylated fatty amides are available commercially from Armak Company under the trade name EthomidTM and Lonza, Inc., under the tradename Unamide®.
- these polyoxyalkylated fatty amides include EthomidTM HT/15 and HT/60, which are hydrogenated tallow acid amides treated with 5 and 50 moles of ethylene oxide respectively; EthomidTM O/15, which is an oleic amide treated with 5 moles of ethylene oxide; Unamide® C-2 and C-5, which are cocamides treated with 2 and 5 moles of ethylene oxide, respectively; and Unamide® L-2 and L-5, which are laur- amides treated with 2 and 5 moles of ethylene oxide, respectively.
- EthomidTM HT/15 and HT/60 which are hydrogenated tallow acid amides treated with 5 and 50 moles of ethylene oxide respectively
- EthomidTM O/15 which is an oleic amide treated with 5 moles of ethylene oxide
- Unamide® C-2 and C-5 which are cocamides treated with 2 and 5 moles of ethylene oxide, respectively
- Unamide® L-2 and L-5 which are laur- amides treated with 2 and 5
- the surfactant is a tall oil, such as distilled tall oil available from Union Camp under the tradename Unitol.
- the surfactant is a nitrogen-containing phosphorus-free reaction product of a carboxylic acylating agent, preferably a succinic acylating agent, having at least one hydrocarbyl group from at least about 12, preferably from about 30, more preferably from about 35 to about 500, preferably to about 300 carbon atoms with at least one N-(hydroxyhydrocarbyl) amine.
- the hydrocarbyl group is derived from a polyalkene having a number average molecular weight from about 500, preferably from about 800 to about 5000, preferably to about 2500, more preferably to about 1200. Number average molecular weight is determined by gel permeation chromatog- raphy.
- the polyalkene is derived from olefins having from about two to about eight carbon atoms, such as ethylene, propylene or butylene, preferably butylene.
- the amine may be a monoamine, polyamine or mixtures thereof.
- the carboxylic acid acylating agents have been described above as well as the hydroxyamines.
- Particularly useful hydroxyhydrocarbyl amines include mono, di and triethanolamine, diethyl ethanolamine, di(3-hydroxypropyl)amine, N-(3-hydroxyl-butyl) amine, N-(4- hydroxyl-butyl)amine, N,N-di(2-hydroxylpropyl) amine, N-(2- hydroxylethyl) morpholine and its thioanalogue, N-(2- hydroxyl-ethyl)cyclohexyl amine, N-3-hydroxylcyclopenthyl amine, O,M-NP-aminophenol, N-(hydroxylethyl) piperazine, N,N'-di(hydroxyethyl)piperazine, and the like.
- Preferred amines are diethylethanolamine and ethanolamine or mixtures thereof.
- the reaction is carried out under ester forming conditions and the product thus formed is an ester/salt.
- the ester/salt is an internal salt, wherein one of the hydroxyl groups of the carboxylic acylating agents becomes ionically bonded to a nitrogen atom within the same group or may be an external salt wherein the ionic salt group is formed with a nitrogen atom which is not part of the same group forming the ester.
- the reaction is carried out at a temperature in the range of about 50°C to about 150°C; but usually at a temperature below a 100°C.
- ester/salts which may be used in the present invention.
- a reaction vessel is charged with 1000 parts of polybutene (number average molecular weight equals 950) substituted succinic anhydride.
- the anhydride is heated to about 90°C over two hours where 209 parts of N,N-diethyl- ethanolamine is added to the vessel.
- the reaction temperature is maintained at 90°C for an additional hour.
- the mixture is cooled to room temperature to provide the desired product. Drilling Fluid
- the reaction products (A) of the present invention are used together with a water based drilling fluid.
- the drilling fluid or mud is generally composed of water, a clay, and a density increasing agent.
- Agents which increase density of drilling muds include galena (PbS), hematite (Fe 2 O 3 ), magnetite (Fe 3 O 4 ), ilmenite (FeOTiO 2 ), barite (BaSO 4 ), siderite (FeCO 3 ), celesite (SrSO 4 ), dolomite (CaCO 3 .MgCO 3 ), and calcite (CaCO 3 ).
- Density increasing agents may also be soluble salts such as sodium chloride, sodium bromide, sodium carbonate, potassium chloride, potassium carbonate, calcium bromide, zinc chloride, and zinc bromide.
- the drilling fluid or mud may also contain commercial clays. These clays include bentonite, attapul- gite, sepiolite, etc. The preferred clay is bentonite.
- the drilling fluid may additionally contain other additives which enhance the lubricating properties of drilling fluids and mud. See, for example, U.S. Patent Nos. 3,214,374 and 4,064,055. These patents are hereby incorporated by reference for their disclosure of drilling fluids and muds, including other additives which may enhance lubricating properties of the fluids and muds.
- the acylated amine terminated polyoxyalkylene may be used alone or in combination with one or more of the above-described surfactants.
- the acylated amine-terminated polyoxyalkylene is generally added to the drilling fluid or mud as a concentrate.
- the concentrates contain at least about 30% by weight of the total composition, preferably at least about 35%.
- the concentrates contain from about 30%, preferably from about 40% to about 80%, preferably to about 70% by weight of acylated amine-terminated polyoxyalkylenes based on the total weight of the concentrate.
- the concentrate may contain only the acylated amine-terminated polyoxyalkylene or may additionally contain one or more of the above- described surfactants.
- the concentrates When used in a drilling fluid, the concentrates are generally added to the drilling fluid at a level of about 0.5% to about 15% by weight concentrate in the drilling fluid. Typically, the concentrates are used from about 0.5% to about 5%, preferably 2% by weight of the drilling fluid.
- the acylated amines are generally present in an amount from about 0.15%, preferably from about 0.25% to about 3%, preferably to about 1% by weight of the drilling fluid.
- a potassium chloride-polymer drilling mud is prepared by mixing 350 parts of water with 12 parts potassium chloride, 8 parts prehydrated bentonite, 1 part starch, 0.5 part of a polyacrylamide (Poly-Plus available from MI Drilling Fluids, Houston, Texas), 0.5 part potas- sium hydroxide, 200 parts of barite and 12 parts of the product of Example 1.
- a polyacrylamide Poly-Plus available from MI Drilling Fluids, Houston, Texas
- potas- sium hydroxide 200 parts of barite and 12 parts of the product of Example 1.
- a sea water/lignosulfonate drilling mud is prepared by mixing 350 parts of water with 20 parts attapulgite, 15 parts sea salt, 3 parts lignosulfonate, 4 parts cellulose (potato starch), 200 parts barite, 0.05 parts defoamer (Sur Tech H-49) and 9 parts of the product of Example 2.
- a fresh water gelled drilling mud is prepared by mixing 350 parts of water, 15 parts prehydrated bentonite, 5 parts drill solids (Rev Dust), 0.5 part cellulose (potato starch), 0.15 part lime, 0.25 part sodium hydroxide, 0.25 part soda ash, 15 parts barite and 9.5 parts of the product of Example 3.
- Example IV A fresh water gelled drilling mud is prepared by mixing 350 parts of water, 15 parts prehydrated bentonite, 5 parts drill solids (Rev Dust), 0.5 part cellulose (potato starch), 0.15 part lime, 0.25 part sodium hydroxide, 0.25 part soda ash, 15 parts barite and 9.5 parts of the product of Example 3.
- a lignosulfonate/fresh water drilling mud is prepared by mixing 350 parts of water, 25 parts prehydrated bentonite, 15 parts drill solids (Rev dust), 0.5 part cellulose (potato starch), 5 parts chrome ligno sulfonate, 2.5 parts lignite, 0.25 part lime, 1 part sodium hydroxide, 0.25 part sodium carbonate, 200 parts barite and 12 parts of the product of Example 4.
- a drilling mud is prepared as described in
- Example I except 18 parts of the product of Example 6 are used in place of 12 parts of the product of Example 1.
- a salt water/ligno sulfonate drilling mud is prepared as described in Example II, except nine parts of the product of Example 9 are used in place of nine parts of the product of Example 2.
- a fresh water gelled drilling mud is prepared as described in Example III, except 15 parts of the product of Example 10 are used in place of 9.5 parts of the product of Example 3.
- a lignosulfonate/fresh water drilling mud is prepared as described in Example IV except 12 parts of the product of Example 11 are used in place of 12 parts of the product of Example 4.
- a salt water/lignosulfonate drilling mud is prepared as described in Example II except 10 parts of the product of Example 14 are used in place of 9 parts of the product of Example 2.
- a lignosulfonate/fresh water drilling mud is prepared as described in Example IV, except 12 parts of the product of Example 15 are used in place of 12 parts of the product of Example 4.
- the acylated amine-terminated polyoxyalkylene may be used alone as a spotting fluid. Such a fluid would be pumped down a well borehole in quantities greater than that required for a lubricant.
- the spotting fluid acts to dislodge previously lodged drill string or pipe.
- the fluids used as spotting fluids are mineral oils and vegetable oils which are toxic to marine life.
- the present acylated amine-terminated polyoxyalkylenes do not adversely affect marine life.
- the acylated amine-terminated polyoxyalkylenes as already described may be used alone or in combination with any one or more of the above-described surfactants.
- the spotting fluids are generally prepared as concentrates
- the concentrates are generally diluted with water at a level of about 0.5% to about 15% concentrate in water.
- the water may be fresh or salt water.
- the concentrate is used from about 1%, preferably from about 3% to about 10%, preferably to about 7%, usually to about 5% by weight concentrate in water.
- Table 3 contains spotting fluid concentrates. Spotting fluids may be prepared from these concentrations by mixing 5% of the concentrate in fresh or salt water.
- the sticking test measures the time necessary for the spotting fluid to unstick a plate.
- Table l are tested using a base mud containing 22 pounds per barrel of bentonite (Aquagel) and 30 pounds per barrel of Martin No. 5 bulk clay which is mixed with water. This mixture is statically aged for 72 hours. Lignosulfonate (Q-broxin) is then added and caustic soda (sodium hydroxide) is added until the pH is about 10.
- Sticking tests are run using a Baroid differential sticking tester apparatus (No. 21150) available from Baroid Co., Houston, Texas U.S.A. The apparatus has been modified by drilling two holes in the cell to allow entrance and exit of spotting fluid.
- the apparatus also uses a bevelled torque plate (Exxon designed torque plate) to establish the height of the filter cake. Modifications of the apparatus are made by Core Laboratories, Lafayette, Louisiana U.S.A. The general procedure involves placing a filter paper and ring attachment in the sticking cell. Approximately 80 milliliters of drilling fluid is poured into the cell. The top of the cell is screwed on and tightened then a yoke attachment is placed on the cell and the desired plate height (cake thickness) is set to 1/64 inch. The cell is pressurized with nitrogen to 500 psi. Filtrate is collected on the filter paper and the plate is allowed to stick. Spotting fluid is added to the cell.
- a bevelled torque plate Exxon designed torque plate
- the cell is inverted and rocked back and forth several times. Any air contained in the cell is bled off. The cell remains inverted after spotting. The time for the cake to unstick is measured. The products are tested at a concentration of 5% by weight in sea water.
- the 96 hour shrimp test is performed on a drilling fluid containing 5% by volume of the spotting fluid.
- the drilling fluid has a pH of 8.46.
- the drilling fluid is thoroughly homogenized for 30 minutes with a high shear mixer.
- the homogenized material is then combined with artificial sea water (salinity equals 20 parts per 1000) in a 1:9 ratio by volume.
- the drilling fluid-sea water mixture characterized by a pH of 7.99, is mixed for five minutes and is allowed to settle for one hour.
- the pH is adjusted with 0.05 milliliter of 6 normal hydrochloric acid to about pH 7.8. Following the settling period, the suspended particulate phase is decanted.
- Mysids (Mysidopsis bahia) used as test organisms are 3-6 days old. The animals are cultured in Core Laboratories Bioassay facilities using brood stock originally purchased from a commercial supplier. The test is conducted at 20°C using artificial sea water (Hawaiian Marine Mix) adjusted to salinity of 20 parts per 1000. Mysids are fed approximately 50 live artemia sp (brine shrimp) per test animal every 24 hours.
- Tests are conducted with five concentrations of suspended particulate phase and a control (of sea water only) with ten Mysids randomly distributed among each concentration. Tests are performed in crystallizing dishes which contain one liter of test solution. Filtered artificial sea water with a salinity of 20 parts per 1000 is used to dilute the suspended particulate phase to test concentrations and as the control solution. A 14 hour light and 10 hour dark photo period is maintained with cool, white flourescent lights. Air is supplied to the test chambers by a commercial aqua culture blower and delivered through glass tubing at a rate of between 50 and 140 cubic centimeters per minute. At a minimum, the number of survivors are determined at 0 and 96 hours. Ninety percent or greater survival occurred in the control exposure.
- the 96 hour reference toxicant test (sodium lauryl sulfate, for this bio essay) is performed according to US EPA protocol.
- the sodium lauryl sulfate is obtained from Aldrich Chemical Co. and is from their lot no. 15K-R- 4G.
- the LC 50 obtained with this most recent sodium lauryl sulfate reference test is 11.5 parts per million sodium lauryl sulfate with 95% confidence level of 10.2 parts per million to 12.9 parts per million.
- the following table contains results of tests performed in the stickometer test and the 96 hour shrimp test.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR9205352A BR9205352A (en) | 1991-07-18 | 1992-07-14 | DRILLING METHOD AND COMPOSITION |
| KR1019930700790A KR930702466A (en) | 1991-07-18 | 1992-07-14 | Well borhaul drilling method and composition used therein |
| NO930976A NO930976D0 (en) | 1991-07-18 | 1993-03-17 | PROCEDURE FOR DRILLING BILLHOLES AND COMPOSITIONS USED THEREOF |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US73227691A | 1991-07-18 | 1991-07-18 | |
| US732,276 | 1991-07-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1993002151A1 true WO1993002151A1 (en) | 1993-02-04 |
Family
ID=24942903
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1992/005879 WO1993002151A1 (en) | 1991-07-18 | 1992-07-14 | Methods of drilling well boreholes and compositions used therein |
Country Status (10)
| Country | Link |
|---|---|
| EP (1) | EP0549770A1 (en) |
| KR (1) | KR930702466A (en) |
| CN (1) | CN1068842A (en) |
| AU (1) | AU647309B2 (en) |
| CA (1) | CA2088480A1 (en) |
| MX (1) | MX9204198A (en) |
| PH (1) | PH30262A (en) |
| TW (1) | TW271448B (en) |
| WO (1) | WO1993002151A1 (en) |
| ZA (1) | ZA925310B (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0561607A2 (en) * | 1992-03-17 | 1993-09-22 | The Lubrizol Corporation | Compositions containing combinations of surfactants and derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same |
| EP0561608A2 (en) * | 1992-03-17 | 1993-09-22 | The Lubrizol Corporation | Compositions containing esters of carboxy-containing interpolymers and methods of using the same |
| EP0561601A2 (en) * | 1992-03-17 | 1993-09-22 | The Lubrizol Corporation | Compositions containing derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same |
| EP0683217A1 (en) * | 1994-05-18 | 1995-11-22 | ARCO Chemical Technology, L.P. | Low-toxicity invert emulsion fluids for well drilling |
| WO2001059028A2 (en) * | 2000-02-11 | 2001-08-16 | M-I L.L.C. | Shale hydration inhibition agent and method of use |
| US6484821B1 (en) * | 2000-11-10 | 2002-11-26 | M-I L.L.C. | Shale hydration inhibition agent and method of use |
| US6609578B2 (en) | 2000-02-11 | 2003-08-26 | Mo M-I Llc | Shale hydration inhibition agent and method of use |
| US6831043B2 (en) | 2002-01-31 | 2004-12-14 | M-I Llc | High performance water based drilling mud and method of use |
| US6857485B2 (en) | 2000-02-11 | 2005-02-22 | M-I Llc | Shale hydration inhibition agent and method of use |
| CN102757773A (en) * | 2011-04-26 | 2012-10-31 | 中国石油化工集团公司 | High-inhibition non-solid phase alkaline completion fluid |
| CN111484835A (en) * | 2020-04-23 | 2020-08-04 | 新疆塔里木油田建设工程有限责任公司 | Water-soluble oil-based drilling fluid lubricant and preparation method thereof |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5599777A (en) * | 1993-10-06 | 1997-02-04 | The Lubrizol Corporation | Methods of using acidizing fluids in wells, and compositions used therein |
| FR2986798B1 (en) * | 2012-02-14 | 2014-01-31 | Rhodia Operations | NOVEL AGENT INHIBITOR FOR SWELLING OF CLAYS, COMPOSITIONS COMPRISING SAID AGENT AND METHODS EMPLOYING SAID AGENT |
| CN102807850B (en) * | 2012-07-30 | 2014-07-23 | 天津中海油服化学有限公司 | Oil-based stuck freeing agent composition for drilling fluid and preparation method for oil-based stuck freeing agent composition |
| CN105441054B (en) * | 2014-08-27 | 2018-09-14 | 中国石油化工股份有限公司 | Surface activator composition and its preparation method and application suitable for low calcium and magnesium oil reservoir |
| CN105368428B (en) * | 2014-08-27 | 2019-12-10 | 中国石油化工股份有限公司 | Anionic-nonionic mixed oil-displacing surfactant and preparation method and application thereof |
| CN107043616B (en) * | 2017-04-20 | 2020-02-07 | 长江大学 | Deepwater white oil flat plate fluid oil base drilling fluid |
| CN110746955B (en) * | 2019-09-26 | 2021-01-12 | 中国石油天然气股份有限公司 | Gemini surfactant for oil displacement, binary composite system, preparation method and application thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3265512A (en) * | 1960-08-04 | 1966-08-09 | Petrolite Corp | Corrosion inhibition |
| US3668129A (en) * | 1970-06-03 | 1972-06-06 | Robert P Willett | Drilling fluid composition and additive therefor |
| US4250042A (en) * | 1979-04-16 | 1981-02-10 | The Lubrizol Corporation | Corrosion inhibition in well-drilling operations using aqueous systems containing ammonium carboxylates |
| WO1990005714A1 (en) * | 1988-11-23 | 1990-05-31 | Esti Kemi A/S | Self-emulsifying ester compounds |
-
1992
- 1992-06-09 TW TW081104472A patent/TW271448B/zh active
- 1992-07-14 KR KR1019930700790A patent/KR930702466A/en not_active Application Discontinuation
- 1992-07-14 AU AU23244/92A patent/AU647309B2/en not_active Ceased
- 1992-07-14 CA CA002088480A patent/CA2088480A1/en not_active Abandoned
- 1992-07-14 WO PCT/US1992/005879 patent/WO1993002151A1/en not_active Application Discontinuation
- 1992-07-14 EP EP92915688A patent/EP0549770A1/en not_active Ceased
- 1992-07-16 ZA ZA925310A patent/ZA925310B/en unknown
- 1992-07-16 CN CN92105790A patent/CN1068842A/en active Pending
- 1992-07-16 MX MX9204198A patent/MX9204198A/en unknown
- 1992-07-17 PH PH44673A patent/PH30262A/en unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3265512A (en) * | 1960-08-04 | 1966-08-09 | Petrolite Corp | Corrosion inhibition |
| US3668129A (en) * | 1970-06-03 | 1972-06-06 | Robert P Willett | Drilling fluid composition and additive therefor |
| US4250042A (en) * | 1979-04-16 | 1981-02-10 | The Lubrizol Corporation | Corrosion inhibition in well-drilling operations using aqueous systems containing ammonium carboxylates |
| WO1990005714A1 (en) * | 1988-11-23 | 1990-05-31 | Esti Kemi A/S | Self-emulsifying ester compounds |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0561607A2 (en) * | 1992-03-17 | 1993-09-22 | The Lubrizol Corporation | Compositions containing combinations of surfactants and derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same |
| EP0561608A2 (en) * | 1992-03-17 | 1993-09-22 | The Lubrizol Corporation | Compositions containing esters of carboxy-containing interpolymers and methods of using the same |
| EP0561601A2 (en) * | 1992-03-17 | 1993-09-22 | The Lubrizol Corporation | Compositions containing derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same |
| EP0561608A3 (en) * | 1992-03-17 | 1993-10-13 | The Lubrizol Corporation | Compositions containing esters of carboxy-containing interpolymers and methods of using the same |
| EP0561607A3 (en) * | 1992-03-17 | 1993-10-13 | The Lubrizol Corporation | Compositions containing combinations of surfactants and derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same |
| EP0561601A3 (en) * | 1992-03-17 | 1993-10-20 | The Lubrizol Corporation | Compositions containing derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same |
| EP0683217A1 (en) * | 1994-05-18 | 1995-11-22 | ARCO Chemical Technology, L.P. | Low-toxicity invert emulsion fluids for well drilling |
| WO2001059028A3 (en) * | 2000-02-11 | 2002-02-07 | Mi Llc | Shale hydration inhibition agent and method of use |
| WO2001059028A2 (en) * | 2000-02-11 | 2001-08-16 | M-I L.L.C. | Shale hydration inhibition agent and method of use |
| US6609578B2 (en) | 2000-02-11 | 2003-08-26 | Mo M-I Llc | Shale hydration inhibition agent and method of use |
| AU2001233113B2 (en) * | 2000-02-11 | 2004-07-08 | M-I L.L.C. | Shale hydration inhibition agent and method of use |
| US6857485B2 (en) | 2000-02-11 | 2005-02-22 | M-I Llc | Shale hydration inhibition agent and method of use |
| US6484821B1 (en) * | 2000-11-10 | 2002-11-26 | M-I L.L.C. | Shale hydration inhibition agent and method of use |
| WO2002038697A3 (en) * | 2000-11-10 | 2003-03-27 | Mi Llc | Shale hydration inhibition agent and method of use |
| US6831043B2 (en) | 2002-01-31 | 2004-12-14 | M-I Llc | High performance water based drilling mud and method of use |
| CN102757773A (en) * | 2011-04-26 | 2012-10-31 | 中国石油化工集团公司 | High-inhibition non-solid phase alkaline completion fluid |
| CN111484835A (en) * | 2020-04-23 | 2020-08-04 | 新疆塔里木油田建设工程有限责任公司 | Water-soluble oil-based drilling fluid lubricant and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2324492A (en) | 1993-02-23 |
| PH30262A (en) | 1997-02-20 |
| CA2088480A1 (en) | 1993-01-19 |
| CN1068842A (en) | 1993-02-10 |
| KR930702466A (en) | 1993-09-09 |
| AU647309B2 (en) | 1994-03-17 |
| EP0549770A1 (en) | 1993-07-07 |
| TW271448B (en) | 1996-03-01 |
| ZA925310B (en) | 1993-04-28 |
| MX9204198A (en) | 1993-07-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5260268A (en) | Methods of drilling well boreholes and compositions used therein | |
| AU647309B2 (en) | Methods of drilling well boreholes and compositions used therein | |
| US5620946A (en) | Compositions containing combinations of surfactants and derivatives of succininc acylating agent or hydroxyaromatic compounds and methods of using the same | |
| US5330662A (en) | Compositions containing combinations of surfactants and derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same | |
| US5593954A (en) | Friction modifier for water-based well drilling fluids and methods of using the same | |
| US8809240B2 (en) | Equivalent circulating density control in deep water drilling | |
| US20120088698A1 (en) | Water in oil emulsion, method for the production thereof | |
| US7786049B2 (en) | Drilling fluids with improved shale inhibition and methods of drilling in subterranean formations | |
| JP4246788B2 (en) | Method for improving ester lubrication performance for well fluids | |
| US5593953A (en) | Friction modifier for oil-based (invert) well drilling fluids and methods of using the same | |
| JPH04503367A (en) | Lipophilic basic amine compounds as additives for invert drilling muds | |
| MXPA01000247A (en) | Oil and oil invert emulsion drilling fluids with improved anti-settling properties. | |
| US5283235A (en) | Compositions containing esters of carboxy-containing interpolymers and methods of using the same | |
| CA2816445A1 (en) | Non-aqueous drilling additive useful to produce a flat temperature-rheology profile | |
| US20170204320A1 (en) | Low Pour-Point Lubricant Additives For Oiled-Based Muds and Synthetic-Based Muds Lubricant Additives for Wellbore or Subterranean Drilling Fluids or Muds | |
| US5599777A (en) | Methods of using acidizing fluids in wells, and compositions used therein | |
| WO2022099399A1 (en) | Deep eutectic solvents | |
| US11879088B2 (en) | Additives to temporarily reduce viscosities in oil-based fluids | |
| US11781056B2 (en) | Stable high solids slurry compositions | |
| AU665619B2 (en) | Compositions containing combinations of surfactants and derivatives of succinic acylating agent or hydroxyaromatic compounds and methods of using the same | |
| US6537951B1 (en) | Fluid and method for releasing tubular elements stuck in a well | |
| WO2022099400A1 (en) | Ternary deep eutectic solvents as drilling fluids | |
| EP0563338A1 (en) | Aqueous functional fluids |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2088480 Country of ref document: CA |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU BR CA FI KR NO RU |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU MC NL SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1992915688 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1992915688 Country of ref document: EP |
|
| WWR | Wipo information: refused in national office |
Ref document number: 1992915688 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1992915688 Country of ref document: EP |