WO1985000511A1 - Collagen ligament and tendon regeneration method and material - Google Patents
Collagen ligament and tendon regeneration method and material Download PDFInfo
- Publication number
- WO1985000511A1 WO1985000511A1 PCT/US1984/001186 US8401186W WO8500511A1 WO 1985000511 A1 WO1985000511 A1 WO 1985000511A1 US 8401186 W US8401186 W US 8401186W WO 8500511 A1 WO8500511 A1 WO 8500511A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- collagen
- ligament
- piece
- torn
- bone
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/3683—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix subjected to a specific treatment prior to implantation, e.g. decellularising, demineralising, grinding, cellular disruption/non-collagenous protein removal, anti-calcification, crosslinking, supercritical fluid extraction, enzyme treatment
- A61L27/3687—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix subjected to a specific treatment prior to implantation, e.g. decellularising, demineralising, grinding, cellular disruption/non-collagenous protein removal, anti-calcification, crosslinking, supercritical fluid extraction, enzyme treatment characterised by the use of chemical agents in the treatment, e.g. specific enzymes, detergents, capping agents, crosslinkers, anticalcification agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/08—Muscles; Tendons; Ligaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/22—Polypeptides or derivatives thereof, e.g. degradation products
- A61L27/24—Collagen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/3641—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix characterised by the site of application in the body
- A61L27/3645—Connective tissue
- A61L27/3662—Ligaments, tendons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/10—Materials or treatment for tissue regeneration for reconstruction of tendons or ligaments
Definitions
- the invention relates to methods and materials for repairing ligaments and tendons and more particularly to methods using collagen material to repair anterior cruciate ligaments without causing inflammation and avoiding digestion of ligament tissue by synovial fluid.
- a ligament is composed of a number of bundles of collagen fibers, the collagens consisting of complex protein molecules, including strings of amino acids.
- Ligaments and tendons are composed of different kinds of collagens, ligaments having the property that they have very high tensile strength and are very unyielding, whereas tendons stretch more and have more shock absorbing characteristics.
- synovial sheaths which protect the ligaments from synovial fluid which lubricates the knee joints to reduce friction between the surface of the bones forming the joint.
- Synovial fluid has the property that it digests ligament material.
- the anterior cruciate ligament is exposed to synovial fluid if it is torn, and it is this problem that has prevented successful repair of ligaments by simply suturing the torn ligament together.
- Torn ligaments in the knee which are not exposed to synovial fluid, such as posterior cruciate ligaments, are frequently successfully repaired by simply suturing the torn ends together.
- anterior cruciate ligaments have never been successfully repaired because it has not been possible to reconstitute the synovial sheath of a torn anterior cruciate ligament or otherwise protect ligament tissue from digestion by synovial fluid.
- it has been attempted to take fatty tissue and wrap it around a torn anterior cruciate ligament, after suturing it to protect it from synovial fluid. This expedient has failed, and the ligament has dissolved.
- tendon tissue for example, fascia lata tissue from a tough band of tendon near the thigh, and weaving that into the knee, drilling a hole through a portion of the bone forming the joint, tunneling the fascia lata tissue through the drilled hole, and stapling it to the bone.
- tendon tissue for example, fascia lata tissue from a tough band of tendon near the thigh
- this technique works well in that it stabilizes the knee joint.
- the fascia lata tendon tissue gradually stretches, and inevitably fails. Numerous operations have been devised to replace such repairs after they fail or to tighten up the tissue that has been used to replace a torn ligament. More recently, due to the failure of natural collagen materials as ligament replacements for the anterior cruciate ligament, synthetic substitutes have been tried.
- a synthetic ligament referred to as aJenkins ligament has been made in Great Britain beginning in about 1976.
- This ligament consists of a carbon fiber material which is perhaps one-half a millimeter in diamter. Bundles of these fibers are woven together to make the Jenkins ligament, which is then attached to the bone of a knee joint using the above-mentioned drilling technique, tunrieling of the fibers through the drilled hole, and stapling a remote end of the substitute ligament to the bone on the other end of the hole.
- This technique provides a ligament replacement which is initially very strong and provides a high degree of initial stabilization of the joint.
- the fibers usually begin to crack.
- the invention provides a method for treating a skin collagen or a structural collagen of proper density to allow it to function as a scaffolding through which ligament or tendon fibroblasts can propagate, which method achieves a sufficiently high degree of cross-linking of the collagen, avoiding antigenicity of the collagen sufficiently to allow regrowth of a torn synovial sheath of the ligament, by treating the collagen with glutaraldehyde or other similar cross-linking agent that does not leave unbound side radicals that cause inflammation.
- bovine skin collagen, "woven" to produce the needed density is crosslinked by glutaraldehyde.
- This collagen is rolled up as a small coil from a small sheet of the glutaraldehyde-treated collagen material.
- the diameter and length of the collagen coil is selected so that it can be positioned between ends of the torn ligament to be repaired.
- DEXON or PROLENE sutures are provided in a close stitching pattern to attach each end of the collagen coil to the respective torn ends of the ligament.
- as one end of the collagen coil is situated in a drilled out portion of the bone of one member of the knee joint, and peripheral portions of the collagen coil at the level of the surface of that bone member of the joint are peripherally stitched to the periosteum.
- the coiled configuration of the collagen material provides greatly increased surface area which increases structural strength, avoids the weakness due to friability of the collagen material when it becomes wet, and improves the function of the collagen as a scaffold through which ligament fibroblasts can propagate and regenerate themselves.
- Experiments upon dogs and histologic analyses of attempted ligament repairs at different stages of healing have shown that fibroblasts of the torn end of the ligament propagate through the scaffolding structure provided by the woven glutaraldehyde treated bovine skin collagen to form a completely normal appearing regenerated anterior cruciate ligament in only approximately three weeks.
- the regeneration of the synovial sheath is believed to be due to the nonantigenic character of the glutaraldehyde treated, woven bovine skin collagen.
- the structural strength of the glutaraldehyde treated skin collagen has been made sufficiently strong to not only provide a scaffolding structure through which the torn ligament may regenerate, but also allows early de-immobilization of the joint allowing early therapy and avoiding atrophying of the regenerated ligament, cartilage, ligament, and muscle tissue.
- the technique can also be utilized to allow regeneration of tendon tissue.
- Fig. 1 is a section view diagram illustrating a portion of a knee joint, including the cruciate ligaments thereof.
- Fig. 1A is a section diagram similar to Fig. 1, illustrating a torn anterior cruciate ligament.
- Fig. 1B is a cross section of a typical anterior cruciate ligament.
- Fig. 1C is an enlarged view illustrating the torn end portion of a torn ligament and the torn synovial sheath around it.
- Fig. 2A is a schematic magnified diagram showing the basic structure of the woven, treated skin collagen used in accordance with the ligament repair method of the present invention.
- Fig. 3 is a partial perspective view of the scaffolding structure formed by a structural collagen, such as rodent tail collagen.
- Fig. 4 is a diagram illustrating generalized cross-linking of collagen strands.
- Fig. 5 is a perspective view of a coiled configuration of collagen utilized in repair of a torn anterior cruciate ligament in accordance with the present invention.
- Fig. 6 is a section diagram illustrating attachment of a coil collagen configuration to a bone in accordance with the present invention.
- Fig. 7 illustrates an alternate technique for attachment of the collagen of the present invention to the torn bone of a joint using a drill and staple technique.
- Fig. 8A is a section view diagram illustrating use of the coiled collagen mesh of the present invention sutured to repair a partial defect in a ligament.
- Fig. 8B is a section diagram illustrating use of the glutaraldehyde treated woven bovine skin collagen mech of the present invention sutured between frayed ends of a completely torn ligament.
- Fig. 9 is a section view diagram illustrating a completely regenerated anterior cruciate ligament and a regenerated interface thereof with femur and tibia bone portions of a knee joint, and also illustrating a regenerated synovial sheath surrounding the regenerated anterior cruciate ligament.
- Fig. 10 is a photoprint of a dog's knee six weeks after an anterior cruciate ligament had been totally removed, showing a completely regenerated, healthy anterior ligament that, in accordance with the present invention, regrew and replaced the original ligament.
- Fig. 11 is a print of a photomicrograph showing the results of histologic analysis of a regenerated ligament and synovial sheath produced in accordance with the present invention in the anterior cruciate ligament of a dog's knee.
- Fig. 12 is a print of a photomicrograph of a biopsy showing both original, natural anterior cruciate ligament material and ligament material regenerated in accordance with the present invention and a transition zone therebetween in a dog six weeks after implantation of the collagen mesh to repair that ligament in accordance with the invention.
- Fig. 13 is a print of a photograph showing an anterior cruciate ligament/bone interface after twelve weeks in a dog's knee in which the entire original anterior cruciate ligament had been removed and replaced by the collagen mesh of the present invention.
- Fig. 14 is a print of a photomicrograph showing an abrupt transition between normal ligament tissue and MARLEX mesh implanted to repair a human anterior cruciate ligament.
- Fig. 15 is a print of a photomicrograph showing an abrupt transition between normal anterior ligament tissue and DEXON mesh used to repair a human anterior cruciate ligament.
- Fig. 16 is a print of a photomicrograph illustrating rupturing of an inflammation due to carbon fiber mesh used to replace an anterior cruciate ligament in a human knee.
- Fig. 17 is a print of a photomicrograph illustrating the transition between normal ligament tissue which is not exposed to synovial fluid and has been repaired by the prior art technique of suturing or stapling torn ends together.
- Fig. 18 is a print similar to Fig. 17 showing different features thereof. Description of the Invention
- knee joint 1 includes an upper bone 2, called the femur and a lower bone 3, called the tibia.
- the enlarged end of femur 2 is covered by a layer of cartilage 4 which interfaces with a layer of cartilage 5 that is disposed upon the upper end of tibia 3.
- Reference numeral 6 designates the knee cap on the front end of the knee and tendons attached thereto.
- Reference numeral 8 generally designates the cruciate ligaments, which together have the general shape of a cross. Their upper ends are attached to the surfaces of femur 2 and their lower ends are attached to the surface of tibia 3.
- Cruciate ligaments 8 prevent lateral translation of the surface of femur 2 in the direction of arrow 9 relative to tibia 3, and also prevent twisting of tibia 3 relative to femur 2.
- Reference numeral 11 designates the anterior cruciate ligament, the other ligament shown being the posterior cruciate ligament.
- the knee is surrounded by a sac which keeps synovial fluid in the vicinity of the knee, and within that sac is the anterior cruciate ligament 11, including a synovial sheath surrounding anterior cruciate ligament fiber bundles from being digested by synovial fluid.
- anterior cruciate ligament 11 tears of the anterior cruciate ligament 11 occur in the mid portion thereof, as indicated by reference numeral 11A in Fig. 1A. Both the ligament tissue and the synovial sheath surrounding the anterior cruciate ligament are completely torn.
- Fig. 1B a cross section diagram is shown of anterior cruciate ligament 11, which includes a plurality of densely packed bundles 11A of ligament collagen fibers.
- Reference numeral 12 designates the synovial sheath which is disposed about ligament material 11A to protect it from being digested by the above-mentioned synovial fluid.
- Collagen Fleece as a Hemostatic Agent by Silverstein et al., The Journal of Trauma, Volume 21, No. 5, page 388; "Collagen Fibers as a Fleece Hemostastic Agent”, by Silverstein et al., The Journal of Trauma, Volume 20, No. 8, page 688; and "Biological Effects of Residual Glutaraldehyde in Glutaraldehyde-Tanned Collagen Biomaterials", by Speer et al., Journal of Biomedical Materials Research, Volume 14, page 7753, (1980), might sufficiently effectively cross-link woven bovine skin collagen fibers without making the material excessively dense or excessively antigenic to allow that collagen to be used as a ligament repair or replacement medium.
- Fig. 2 shows a print of a photomicrograph of the above described glutaraldehyde-treated woven bovine collagen mesh that is obtained from B. Braun, Inc. before treating it with glutaraldehyde by soaking it in a 10% glutaraldehyde solution to provide an antigenic scaffolding through which fibroblasts can propagate and around which a torn synovial can regenerate.
- the "woven" structure is clearly shown in Fig. 2, wherein reference numeral 111 shows individual fibers of the woven bovine skin collagen. The general orientation of these fibers is in the vertical pattern indicated by the upper arrow.
- the thickness dimension of the illustrated piece of collagen is in the direction of the thickness of the sheet of paper on which Fig. 2 is printed.
- This glutaraldehyde-treated woven bovine skin collagen material was then used to repair torn heel cords of rabbits by suturing a piece of the collagen material between torn ends of these tendons.
- the analyses also showed, very surprisingly, that new synovial sheaths had grown over the repaired ligaments and that fibroblasts of the original ligaments had grown and propagated through the scaffolding provided by the above-mentioned glutaraldehyde-treated woven bovine skin collagen material that had been sutured between the ends of the torn anterior cruciate ligaments.
- the histologic analyses also showed that there were only faint hints of inflammatory reactions in the dogs' knee joints, not enough to prevent new growth of the synovial sheaths, which prevent the synovial fluid from attacking the fibroblasts growing from the torn ends of the anterior cruciate ligaments and prevent them from growing through the collagen scaffolding.
- FIGs. 8A and 8B illustrate how my original experiments were carried out.
- a piece of healthy ligament consisting of approximately a three-fourths, cylinder section of the anterior cruciate ligament 18 was removed, leaving only a bottom section 18C connecting the two ends 18A and 18B together.
- a piece of suitable length glutaraldehyde-treated woven bovine skin collagen material was inserted into the resulting gap in the ligament 18 and sutured peripherally as shown in Fig. 8A.
- the tensile weakness and the high degree of friability of this peice of collagen when it became wet due to blood and other fluids caused considerable difficulty.
- an anterior cruciate ligament of a dog knee was completely severed, as indicated by reference numeral 18 in Fig. 8B, leaving separate ligament sections 18A and 18B with a gap therebetween.
- the glutaraldehyde-treated woven bovine skin collagen coil 16 was positioned between these two torn ligament sections; close stitching of the periphery of each end of the torn ligaments to the opposite ends of the collagen coil 16 was performed.
- the joint was immobilized by a cast for three weeks, and then at various periods from three to fifteen weeks, the animals were sacrificed and the repaired ligaments were subjected to histologic analyses.
- the coiled collagen configuration greatly facilitates the handling and stitching thereof and greatly increases the tensile strength of the repaired ligament immediately and during the initial part of the ligament regrowth process.
- one end of the collagen coil 16 was peripherally stitched to a torn end of an anterior cruciate ligament 19 of a dog's knee by means of sutures 20 after the surface of tibia or femur bone 21 had been drilled or chiseled to produce a void 22 three to five millimeters in depth therein.
- the lower end of the glutaraldehyde-treated woven bovine skin collagen coil was inserted all the way into the hole 22, and at the surface level of the bone 21 peripheral sutures 23 were utilized to attach the collagen coil 16 to the periosteum.
- the joints were immobilized for varying periods of time from three to fifteen weeks, after which the animals were sacrificed and the ligaments and joints were subjected to histologic analyses. These analyses showed that the ligament fibroblasts from ligament fibroblasts from ligament 19 had, within three to four weeks, regrown entirely through the collagen coil scaffolding 16 and had taken route in the bone material. Also, the bone had generated fibroblasts which had propagated toward ligament 19, as will be made clear subsequently.
- Fig. 3 the structure, which closely represents the structure of rodent tail collagen, is very densely woven. It is known to have a very high tensile strength. I believe that if it is properly manipulated and "woven" and then treated with glutaraldehyde, sufficient cross linking may be accomplished, as indicated by reference numerals 30 in Fig. 5, to reduce the antigenicity enough to avoid severe inflammatory reactions; these procedures might be provided so as to result in a density that is low enough to permit propagation of fiberblasts and yet result in a sufficiently high tensile strength that this "structural collagen” may be used in the manner shown in Fig. 7 to replace torn cruciate ligaments without immobiliation of the joint, or at least reducing the amount of time that the joint has to be immobilized to considerably less than three weeks, thereby allowing physical therapy to begin as soon as possible.
- the coil collagen mesh 35 extends through two holes 33 and 34 that have been drilled, respectively, through femur 2 and tibia 3.
- the opposite ends of the coiled collagen mesh 35 are stapled by means of metal staples 36 to the portions of the bones indicated in Fig. 7. If the elongated collagen coil mesh 35 has sufficient tensile strength, the joint might not need to be immobilized, allowing immediate physical therapy. (It should be appreciated that the glutaraldehyde-treated woven bovine skin mesh that I have used up to now in experiments is not nearly as strong as healthy ligament tissue or healthy tendon tissue. Therefore, it has been necessary to immobilize the joints for at least three weeks).
- Fig. 10 is a print of a disarticulated dog's knee taken six weeks after the implantation of a glutaraldehyde-treated woven bovine skin collagen coil which was implanted to completely replace the original anterior cruciate ligament, which had been completely removed. Holes were drilled in both the femur 93A and tibia 93B at the points to which the original removed anterior cruciate ligament had been attached. The holes were drilled down into the cancellous bone tissue designated by reference numeral 21B in Fig.
- Fig. 9 is a diagram of the dog's knee joint and regenerated ligament of Fig. 10.
- Fig. 11 is a print of a photomicrograph of a biopsy of a regenerated anterior ligament connection wherein glutaraldehyde-treated wove bovine skin collagen mesh was sutured to an anterior cruciate ligament in the knee of a dog; the synovial sheath was also completely torn.
- Fig. 11 clearly shows that the torn synovial sheath completely regenerated itself. In this case, the dog was sacrificed after six weeks from the implantation of the collagen coil.
- Reference numeral 86 represents the reconstituted ligament material.
- Reference numeral 87 represents a normal blood vessel therein.
- Reference numeral 88 refers to the regenerated or regrown synovial sheath that grew over the implanted glutaraldehyde-treated woven bovine skin collagen mesh that was implanted. It is to be noted that there is not indication of inflammatory reaction (which would appear as a darkened area) in the synovial sheath or underneath it in the regenerated ligament material 86. Superimposed dotted lines 89 show roughly what an inflammatory action would look like if there had been one. (Such an inflammatory reaction, if present, would cause a densely cellular layer of scar tissue to be formed.)
- Reference numeral 90 designates a clear or pale zone showing the normal collagen bundles that are beginning to form where the collagen coil was originally implanted.
- Fig. 12 shows a print of a micrograph of a biopsy of an anterior cruciate ligament of a dog regenerated in accordance with the present invention.
- the dog was sacrificed six weeks after the collagen coil was sutured to the torn end of the ligament.
- Reference numeral 69 refers to the normal original anterior cruciate ligament material.
- Reference numeral 70 refers to a normal blood vessel therein, which appears as a dark spot in the original ligament material 69.
- Reference numeral 71 designates a dark spot that represents a healthy, normal ligament cell in the original ligament material 69.
- Reference numeral 72 refers, to what appears as a pale zone in the biopsy, and designates normal ligament collagen bundles in the original ligament material 69.
- Reference numeral 73 shows regenerated or regrown ligament material that has propagated through the collagen coil mesh in the dog's ligament during the six weeks after the collagen coil was implanted. There is no evidence in Fig. 12 of the collagen mesh itself, which after six weeks had completely dissolved and disappeared.
- Reference numeral 74 shows a very gradual transition zone between the normal original ligament 69 and the region 73 where the collagen mesh was implanted.
- Reference numeral 75 shows a regrowing blood vessel and the region 73 where the collagen mesh was implanted and the ligament has regenerated.
- Numeral 76 designated dark spots that show regrown ligament cells which are comparable to the ligament cells or fibroblasts 71.
- Reference numeral 77 shows reforming mature collagen bundles that are comparable to the original collagen bundles 72. It is very significant that there are no inflammatory reactions (which would appear as dark areas) in transition zone 74 and in region 73.
- Fig. 13 is a print of a photomicrograph of a biopsy of the junction between collagen mesh that was implanted in a holed drilled to the level of cancellous tissue in bone, wherein the periphery of the collagen mesh coil had been sutured to the periosteum of the bone.
- Reference numeral 80 refers to regenerated ligament material which had propagated from fibroblasts in the cancellous tissue of the bone through the collagen coil such as 16 in Figs. 5 and 6, by the end of a twelve week period in the knee of a dog.
- Reference numeral 81 refers to regenerated ligament cells that appear to be perfectly normal. Note that in this portion of a ligament there are normally very few blood vessels.
- Reference numeral 82 refers to normal bone tissue which has regenerated in the drilled hole in which one end of the collagen coil was implanted.
- Reference numeral 83 refers to regenerated bone cells which appear to be normal in this region. The white spots with dark dots therein represent normal bone cells 83. Note that there are normally very few blood vessels in this part of the bone.
- Reference numeral 84 designates an irregular interface of calcification between regenerated bone and regenerated ligament that also has been regenerated in accordance with the method of the invention.
- This interface region forms as result of a condensation reaction between calcium and other minerals to form what appears in Fig. 13 as a regenerated calcification interface.
- the appearance of regenerated recalcification line 84 appears, after twelve weeks from implantation of the collagen coil, to be perfectly normal. Note that the process, descrihed above with reference to Fig. 13 makes possible the complete regeneration of anterior cruciate ligaments such as 11 in Fig. 9 and 92 in Fig. 10 by regenerating not only ligament tissue, but also a perfect ligamentbone interface to the femur and tibia bones.
- Fig. 14 shows what happens when MARLEX mesh is used in a human body as a ligament replacement.
- Reference numeral 97 designates normal ligament material.
- Reference numeral 98 designates the location of MARLEX mesh.
- Reference numeral 99 designates a very abrupt transition between the normal ligament material and the MARLEX mesh six weeks after implantation of the MARLEX mesh by suturing it to the torn end of an anterior cruciate ligament in a human knee. No gradual transition between the MARLEX and normal ligament material appears, as was the case when glutaraldehyde treated wove bovine skin collagen is implanted in a dog's knee, in which case the torn ligament regenerates itself by growing through the scaffolding provided by the collagen mesh in accordance with the present invention.
- the dark areas such as the ones designated by reference numeral 100, indicate dense, severe, inflammatory reactions that accompany the presence of the MARLEX mesh in the human knee .
- the clear areas indicated by reference numeral 101 show normal collagen bundles in the normal ligament in Fig. 14. There are none of these normal collagen bundles appearing in region 98 with the MARLEX mesh.
- Fig. 15 shows, after six weeks, where woven DEXON material was implanted and sutured to a torn anterior cruciate ligament in a human knee to repair it.
- Reference numeral 104 generally designates the implanted DEXON mesh.
- Reference numeral 105 shows normal ligament material to which the DEXON mesh was sutured.
- Reference numeral 106 shows a very abrupt transition between the normal ligament material 105 and the DEXON material 104.
- Reference numeral 107 shows isolated DEXON synthetic fibers; these fibers clearly have not been absorbed during the six weeks of implantation.
- Reference numeral 108 indicates black areas which represent intense inflammatory reaction that accompanies the presence of DEXON material in the human knee.
- the light areas pointed to by reference numeral 109 show normal collagen fiber bundles in the normal ligament tissue 105; none of this normal collagen fiber bundle tissue is present in the region 104 in which DEXON material is present.
- Fig. 16 is a print of a photomicrograph of a carbon fiber ligament after it has been implanted in a human knee for roughly 18 months.
- Reference numerals 112 show individual carbon fibers which have undergone extensive fracturing and disorientation during the eighteen months.
- Reference numerals 113 designate dark areas that indicate regions of severe cellular inflammation around the individual carbon fibers. Note that in Fig. 16, there is an absence of healthy, well organized collagen bundles of ligament tissue that are present in healthy human anterior cruciate ligaments.
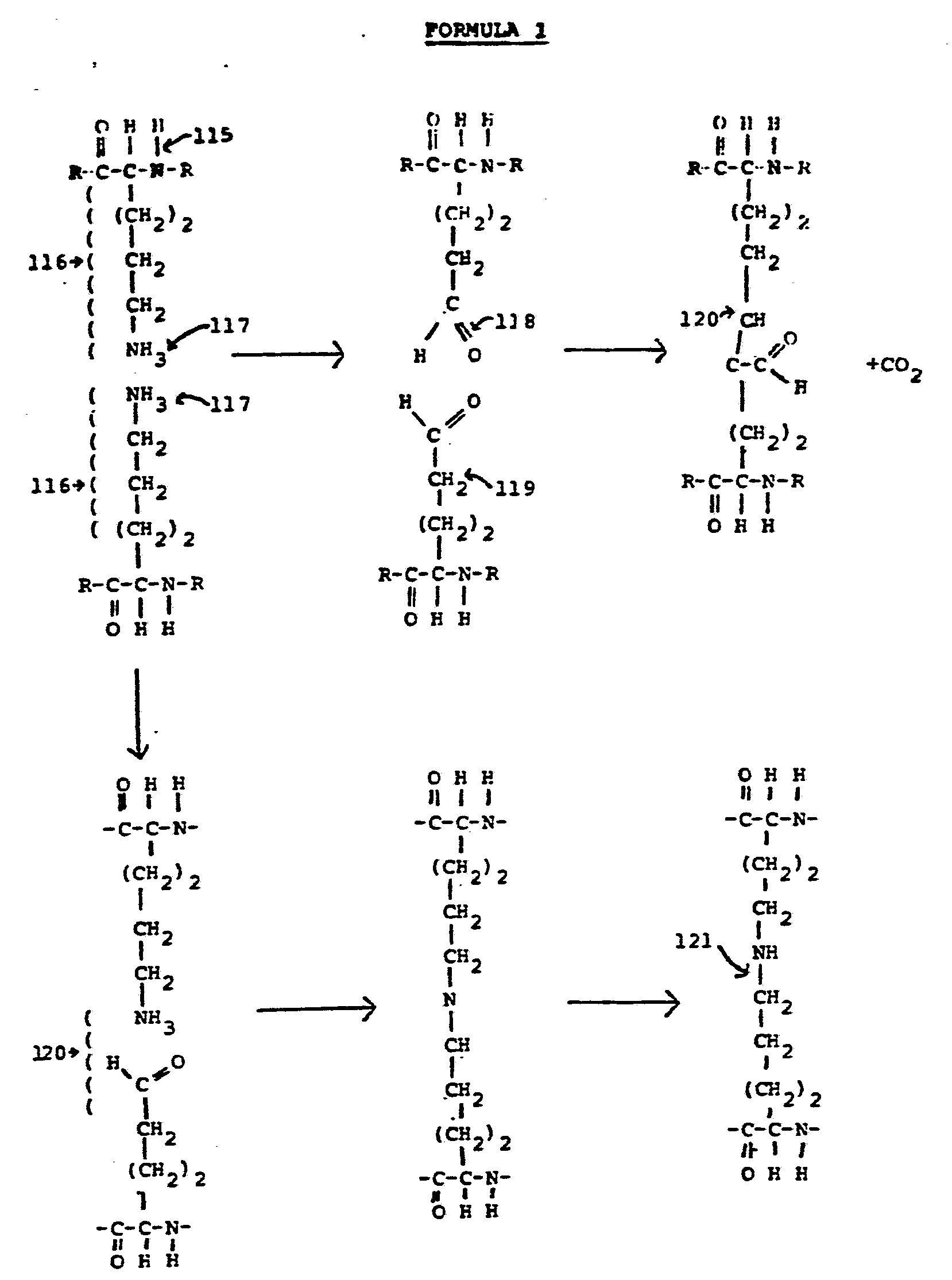
- reference numeral 115 refers to the amino acid chains which form the protein collagen backbone.
- Reference numeral 116 refers to the side chains that extend from the side of the protein molecules.
- the one designated by reference numeral 16 is lysine, an amino acid.
- Reference numeral 117 refers to the amine groups or terminal groups, which are the reactive groups that are desired to be joined in the cross-linking process when the collagen is treated with glutaraldehyde or other similar cross-linking agents, such as other aldehydes.
- the reaction generally designated by the upper half of Formula 1 is a condensation reaction, and the reaction represented by the lower half of Formula 1 is a base shift reaction.
- Formula 1 represents two kinds of cross-linking of the woven bovine skin collagen that occur as a result of being treated with glutaraldehyde.
- the net result of both types of cross-linking is essentially the same, even though there are subtle differences in the chemical bonding that occurs as a result of the two reactions.
- the molecules designated by reference numeral 118 bond with the molecules indicated by reference numeral 119 to produce the condensation reaction.
- R represents the "rest" of the protein chain that is not shown in Formula 1.
- Reference numeral 120 at the bottom of Formula 1 refers to a slightly different base shift chemical reaction that occurs when proteins are cross-linked using glutaraldehyde. This reaction is an oxidation and differs from the condensation reaction shown in the upper part of Formula 1 in that there is a different bond between the two protein chains. But the next effect of each is a stable chemical cross linking bond.
- the qualities of both reactions are that neither leave any reactive side groups or radicals which will be reacted to be human or animal bodies by causing severe inflammatory reactions.
- In effective cross-linking all of the reactive side groups or radicals participate in the cross-linking. This is in contrast to other cross-linking agents, such as chromates, which do result in effective cross-linking, but also leave a salt of the chromate hanging unbonded to the linking molecule; the salt then causes a
- the undesirable reactive groups or radicals can be some other group that a salt of the molecule that has been oxidized by a cross-linking action process and thereby made reactive or antigenic.
- These undesired reactive groups are ones that are not capable of participating in the cross-linking process, however, and are referred to herein simply as radicals. Frequently, they are amines or carboxyl groups.
- radicals are antigenic.
- desirable crosslinking agents are referred to herein as ones that do not have an attached radical that cannot participate in the cross-linking process.
- the radical could be any part of the cross-linking molecule that oxidizes in the cross-linking treatment process and does not have an attached radical that the body will attack to produce an inflammation.
- the Glutaraldehyde-treated woven bovine skin collagen technique of the present invention is expected to be very useful not only in repair of ligaments which are exposed to synovial fluid, but also to ligaments which are not exposed to synovial fluid and also to tendons because even though the tendons and latter mentioned ligaments can be successfully repaired by suturing the torn ends together, it is technically impractical to actually properly suture each one of the torn bundles of fibers together. Therefore, in repairing such ligaments and tendons by suturing, one generally simply sutures or staples the torn ends of the ligament together.
- a biopsy of the resulting repaired ligament, after healing is complete, will show tissue structure that varies considerably from that of normal healthy ligament or tendon tissue in that there is scar tissue which is very disorganized and that the ligament or tendon material in the reapired region is less well cross-linked than is the case for healthy tissue.
- the tensile strength of ligaments or tendons which have been repaired by suturing the torn ends together is not nearly as great as the tensile strength of corresponding healthy ligament or tendon tissue.
- Figs. 17 and 18 which show photomicrographs of biopsys of a torn medial collateral ligament of a human knee, the torn ends of which have been sutured together and allowed to heal for twelve weeks, reference numerals 124 indicates such disorganized, relatively weak scar tissue.
- the techniques and materials described herein provide the great advantage of allowing, for the first time, reliable repair of anterior cruciate ligaments and other ligaments, that are exposed to synovial fluid.
- the techniques and materials described further allow essentially complete regeneration of normal-appearing ligaments from cancellous bone tissue, and also allow, for the first time, regeneration of normal-appearing bone/ ligament calcification interface regions, even if the original ligament has been previously completely removed.
- the described techniques and materials allow, for the first time, regeneration of the synovial sheath which protects some ligaments, such as anterior cruciate ligaments, from digestion by synovial fluid.
- the described techniques and materials also allow, for the first time, repair of tendons and ligaments without the generation of relatively weak, disorganized scar tissue that ordinarily grows where torn ends of ligaments or tendons have been sutured together in accordance with prior techniques. Remobilization of joints much sooner after surgery is made possible by the techniques and materials of the present invention, which is very desirable to prevent atrophying of all related tissue.
Abstract
Description
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US51702083A | 1983-07-25 | 1983-07-25 | |
| US517,020 | 1990-05-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1985000511A1 true WO1985000511A1 (en) | 1985-02-14 |
Family
ID=24058056
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1984/001186 WO1985000511A1 (en) | 1983-07-25 | 1984-07-24 | Collagen ligament and tendon regeneration method and material |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP0152431A4 (en) |
| WO (1) | WO1985000511A1 (en) |
Cited By (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL8701370A (en) * | 1987-06-12 | 1987-08-03 | Stichting Surgical Research Fo | Chamois leather as an adhesive for living tissues. |
| WO1988006872A1 (en) * | 1987-03-09 | 1988-09-22 | Astra Meditec Ab | A resorbable prosthesis |
| WO1988006871A1 (en) * | 1987-03-09 | 1988-09-22 | Astra Meditec Ab | Nerve splicing prosthesis |
| FR2617705A1 (en) * | 1987-07-09 | 1989-01-13 | Lemaire Marcel | ARTIFICIAL LIGAMENTS AND ARTICLES FOR CARRYING OUT THEM |
| EP0306018A1 (en) * | 1987-09-04 | 1989-03-08 | Bio-Products, Inc. | Tendon/ligament substitutes composed of long, parallel, non-antigenic tendon/ligament fibers |
| GB2175507B (en) * | 1985-05-29 | 1989-07-05 | American Hospital Supply Corp | Bone-grafting material |
| US4883486A (en) * | 1988-05-31 | 1989-11-28 | Indu Kapadia | Prosthetic ligament |
| GB2185408B (en) * | 1986-01-16 | 1990-02-14 | Rhode Island Hospital | Neovascularization |
| FR2651994A1 (en) * | 1989-09-19 | 1991-03-22 | Laboureau Jacques Philippe | Prosthetic ligament comprising a network of synthetic or natural fibres impregnated with collagen |
| US5078744A (en) * | 1987-09-04 | 1992-01-07 | Bio-Products, Inc. | Method of using tendon/ligament substitutes composed of long, parallel, non-antigenic tendon/ligament fibers |
| US5320100A (en) * | 1991-09-16 | 1994-06-14 | Atrium Medical Corporation | Implantable prosthetic device having integral patency diagnostic indicia |
| US5370681A (en) * | 1991-09-16 | 1994-12-06 | Atrium Medical Corporation | Polyumenal implantable organ |
| EP0645149A1 (en) * | 1993-09-29 | 1995-03-29 | JOHNSON & JOHNSON MEDICAL, INC. | Absorbable structures for ligament and tendon repair |
| US6017366A (en) * | 1997-04-18 | 2000-01-25 | W. L. Gore & Associates, Inc. | Resorbable interposition arthroplasty implant |
| WO2000078370A1 (en) * | 1999-06-22 | 2000-12-28 | The Brigham And Women's Hospital, Inc. | Biologic replacement for fibrin clot for intra-articular use |
| WO2004045667A1 (en) * | 2002-09-18 | 2004-06-03 | Sdgi Holdings, Inc. | Collagen-based materials and methods for treating synovial joints |
| US6964685B2 (en) | 1999-06-22 | 2005-11-15 | The Brigham And Women's Hospital, Inc. | Biologic replacement for fibrin clot |
| WO2009085769A2 (en) * | 2007-12-20 | 2009-07-09 | Zimmer Orthobiologics, Inc. | Compositions and methods for repair of connective tissue |
| CN103239759A (en) * | 2013-05-03 | 2013-08-14 | 中国人民解放军第三军医大学第三附属医院 | TGF-Beta3 (Transforming Growth Factor Beta3) loaded slow-release tissue engineering synovial sheath |
| US9308242B2 (en) | 2006-09-28 | 2016-04-12 | Children's Medical Center Corporation | Methods and products for tissue repair |
| US9387280B2 (en) | 2008-09-05 | 2016-07-12 | Synovis Orthopedic And Woundcare, Inc. | Device for soft tissue repair or replacement |
| US9757495B2 (en) | 2013-02-01 | 2017-09-12 | Children's Medical Center Corporation | Collagen scaffolds |
| CN107510520A (en) * | 2017-09-21 | 2017-12-26 | 上海松力生物技术有限公司 | Right-angled intersection ligament reproducibility implant and preparation method and application |
| US10786232B2 (en) | 2006-01-25 | 2020-09-29 | The Children's Medical Center Corporation | Methods and procedures for ligament repair |
| US11484578B2 (en) | 2012-02-01 | 2022-11-01 | Children's Medical Center Corporation | Biomaterial for articular cartilage maintenance and treatment of arthritis |
| WO2024005593A1 (en) * | 2022-07-01 | 2024-01-04 | 한양대학교 산학협력단 | Composition for inducing ligamentization of tendon, comprising anterior cruciate ligament-derived stem cells |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4378224A (en) * | 1980-09-19 | 1983-03-29 | Nimni Marcel E | Coating for bioprosthetic device and method of making same |
| US4400833A (en) * | 1981-06-10 | 1983-08-30 | Kurland Kenneth Z | Means and method of implanting bioprosthetics |
| US4458678A (en) * | 1981-10-26 | 1984-07-10 | Massachusetts Institute Of Technology | Cell-seeding procedures involving fibrous lattices |
| US4467478A (en) * | 1982-09-20 | 1984-08-28 | Jurgutis John A | Human ligament replacement |
-
1984
- 1984-07-24 EP EP19840903019 patent/EP0152431A4/en not_active Withdrawn
- 1984-07-24 WO PCT/US1984/001186 patent/WO1985000511A1/en not_active Application Discontinuation
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4378224A (en) * | 1980-09-19 | 1983-03-29 | Nimni Marcel E | Coating for bioprosthetic device and method of making same |
| US4400833A (en) * | 1981-06-10 | 1983-08-30 | Kurland Kenneth Z | Means and method of implanting bioprosthetics |
| US4458678A (en) * | 1981-10-26 | 1984-07-10 | Massachusetts Institute Of Technology | Cell-seeding procedures involving fibrous lattices |
| US4467478A (en) * | 1982-09-20 | 1984-08-28 | Jurgutis John A | Human ligament replacement |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP0152431A4 * |
Cited By (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2175507B (en) * | 1985-05-29 | 1989-07-05 | American Hospital Supply Corp | Bone-grafting material |
| GB2185408B (en) * | 1986-01-16 | 1990-02-14 | Rhode Island Hospital | Neovascularization |
| WO1988006872A1 (en) * | 1987-03-09 | 1988-09-22 | Astra Meditec Ab | A resorbable prosthesis |
| WO1988006871A1 (en) * | 1987-03-09 | 1988-09-22 | Astra Meditec Ab | Nerve splicing prosthesis |
| EP0298533A1 (en) * | 1987-06-12 | 1989-01-11 | Surgical Research Foundation | Sheep dermal collagen as an adhesive medium for living tissues |
| NL8701370A (en) * | 1987-06-12 | 1987-08-03 | Stichting Surgical Research Fo | Chamois leather as an adhesive for living tissues. |
| FR2617705A1 (en) * | 1987-07-09 | 1989-01-13 | Lemaire Marcel | ARTIFICIAL LIGAMENTS AND ARTICLES FOR CARRYING OUT THEM |
| EP0299858A1 (en) * | 1987-07-09 | 1989-01-18 | Cendis Medical Sarl | Artificial ligament and apparatus for its manufacture |
| US5078744A (en) * | 1987-09-04 | 1992-01-07 | Bio-Products, Inc. | Method of using tendon/ligament substitutes composed of long, parallel, non-antigenic tendon/ligament fibers |
| EP0306018A1 (en) * | 1987-09-04 | 1989-03-08 | Bio-Products, Inc. | Tendon/ligament substitutes composed of long, parallel, non-antigenic tendon/ligament fibers |
| US4883486A (en) * | 1988-05-31 | 1989-11-28 | Indu Kapadia | Prosthetic ligament |
| FR2651994A1 (en) * | 1989-09-19 | 1991-03-22 | Laboureau Jacques Philippe | Prosthetic ligament comprising a network of synthetic or natural fibres impregnated with collagen |
| US5320100A (en) * | 1991-09-16 | 1994-06-14 | Atrium Medical Corporation | Implantable prosthetic device having integral patency diagnostic indicia |
| US5370681A (en) * | 1991-09-16 | 1994-12-06 | Atrium Medical Corporation | Polyumenal implantable organ |
| EP0645149A1 (en) * | 1993-09-29 | 1995-03-29 | JOHNSON & JOHNSON MEDICAL, INC. | Absorbable structures for ligament and tendon repair |
| US5514181A (en) * | 1993-09-29 | 1996-05-07 | Johnson & Johnson Medical, Inc. | Absorbable structures for ligament and tendon repair |
| US5595621A (en) * | 1993-09-29 | 1997-01-21 | Johnson & Johnson Medical, Inc. | Method of making absorbable structures for ligament and tendon repair |
| US6017366A (en) * | 1997-04-18 | 2000-01-25 | W. L. Gore & Associates, Inc. | Resorbable interposition arthroplasty implant |
| US6964685B2 (en) | 1999-06-22 | 2005-11-15 | The Brigham And Women's Hospital, Inc. | Biologic replacement for fibrin clot |
| US7838630B2 (en) | 1999-06-22 | 2010-11-23 | Children's Medical Center Corporation | Biologic replacement for fibrin clot |
| WO2000078370A1 (en) * | 1999-06-22 | 2000-12-28 | The Brigham And Women's Hospital, Inc. | Biologic replacement for fibrin clot for intra-articular use |
| WO2004045667A1 (en) * | 2002-09-18 | 2004-06-03 | Sdgi Holdings, Inc. | Collagen-based materials and methods for treating synovial joints |
| US11076846B2 (en) | 2006-01-25 | 2021-08-03 | The Children's Medical Center Corporation | Methods and procedures for ligament repair |
| US11076845B2 (en) | 2006-01-25 | 2021-08-03 | The Children's Medical Center Corporation | Methods and procedures for ligament repair |
| US10786238B2 (en) | 2006-01-25 | 2020-09-29 | The Children's Medical Center Corporation | Methods and procedures for ligament repair |
| US10786239B2 (en) | 2006-01-25 | 2020-09-29 | The Children's Medical Center Corporation | Methods and procedures for ligament repair |
| US10786232B2 (en) | 2006-01-25 | 2020-09-29 | The Children's Medical Center Corporation | Methods and procedures for ligament repair |
| US9308242B2 (en) | 2006-09-28 | 2016-04-12 | Children's Medical Center Corporation | Methods and products for tissue repair |
| US9849213B2 (en) | 2006-09-28 | 2017-12-26 | Children's Medical Center Corporation | Methods and products for tissue repair |
| WO2009085769A3 (en) * | 2007-12-20 | 2010-03-25 | Zimmer Orthobiologics, Inc. | Compositions and methods for repair of connective tissue |
| US9339587B2 (en) | 2007-12-20 | 2016-05-17 | Zimmer Orthobiologics, Inc. | Composition and methods for repair of connective tissue |
| WO2009086313A3 (en) * | 2007-12-20 | 2010-03-25 | Zimmer Orthobiologics, Inc. | Compositions and methods for repair of connective tissue |
| WO2009085769A2 (en) * | 2007-12-20 | 2009-07-09 | Zimmer Orthobiologics, Inc. | Compositions and methods for repair of connective tissue |
| WO2009086313A2 (en) * | 2007-12-20 | 2009-07-09 | Zimmer Orthobiologics, Inc. | Compositions and methods for repair of connective tissue |
| US9387280B2 (en) | 2008-09-05 | 2016-07-12 | Synovis Orthopedic And Woundcare, Inc. | Device for soft tissue repair or replacement |
| US11484578B2 (en) | 2012-02-01 | 2022-11-01 | Children's Medical Center Corporation | Biomaterial for articular cartilage maintenance and treatment of arthritis |
| US10842914B2 (en) | 2013-02-01 | 2020-11-24 | The Children's Medical Center Corporation | Collagen scaffolds |
| US9757495B2 (en) | 2013-02-01 | 2017-09-12 | Children's Medical Center Corporation | Collagen scaffolds |
| US11826489B2 (en) | 2013-02-01 | 2023-11-28 | The Children's Medical Center Corporation | Collagen scaffolds |
| US11839696B2 (en) | 2013-02-01 | 2023-12-12 | The Children's Medical Center Corporation | Collagen scaffolds |
| CN103239759A (en) * | 2013-05-03 | 2013-08-14 | 中国人民解放军第三军医大学第三附属医院 | TGF-Beta3 (Transforming Growth Factor Beta3) loaded slow-release tissue engineering synovial sheath |
| CN107510520A (en) * | 2017-09-21 | 2017-12-26 | 上海松力生物技术有限公司 | Right-angled intersection ligament reproducibility implant and preparation method and application |
| WO2024005593A1 (en) * | 2022-07-01 | 2024-01-04 | 한양대학교 산학협력단 | Composition for inducing ligamentization of tendon, comprising anterior cruciate ligament-derived stem cells |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0152431A1 (en) | 1985-08-28 |
| EP0152431A4 (en) | 1986-11-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1985000511A1 (en) | Collagen ligament and tendon regeneration method and material | |
| US10493180B2 (en) | Device for soft tissue repair or replacement | |
| EP2175800B1 (en) | Composite implant for surgical repair | |
| US4400833A (en) | Means and method of implanting bioprosthetics | |
| US7998204B2 (en) | Composite collagen material and method of forming same | |
| JP4219453B2 (en) | Prosthetic brace with collagen for tissue repair | |
| US4693720A (en) | Device for surgically repairing soft tissues and method for making the same | |
| US4585458A (en) | Means and method of implanting bioprosthetics | |
| US4979956A (en) | Device and method for tendon and ligament repair | |
| US5078744A (en) | Method of using tendon/ligament substitutes composed of long, parallel, non-antigenic tendon/ligament fibers | |
| US5061283A (en) | Method for tendon and ligament repair | |
| US20100249835A1 (en) | Medical device for repair of tissue and method for implantation and fixation | |
| US4920962A (en) | Splint-like element for use in end-to-end nerve suture | |
| JP7034983B2 (en) | Collagen constructs, and how to produce collagen constructs |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP Designated state(s): JP |
|
| AL | Designated countries for regional patents |
Designated state(s): AT DE FR GB SE Kind code of ref document: A1 Designated state(s): AT DE FR GB SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1984903019 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1984903019 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1984903019 Country of ref document: EP |