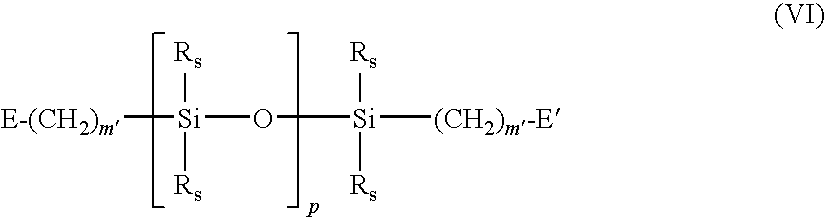US20090145091A1 - Method for treating ophthalmic lenses - Google Patents
Method for treating ophthalmic lenses Download PDFInfo
- Publication number
- US20090145091A1 US20090145091A1 US12/244,386 US24438608A US2009145091A1 US 20090145091 A1 US20090145091 A1 US 20090145091A1 US 24438608 A US24438608 A US 24438608A US 2009145091 A1 US2009145091 A1 US 2009145091A1
- Authority
- US
- United States
- Prior art keywords
- lens
- recess
- solution
- package
- treatment agent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 238000000034 method Methods 0.000 title claims abstract description 36
- 239000000243 solution Substances 0.000 claims abstract description 54
- 239000012756 surface treatment agent Substances 0.000 claims abstract description 24
- 230000001954 sterilising effect Effects 0.000 claims abstract description 10
- 238000007789 sealing Methods 0.000 claims abstract description 8
- 239000007864 aqueous solution Substances 0.000 claims abstract description 6
- 229920001296 polysiloxane Polymers 0.000 claims description 24
- 238000004806 packaging method and process Methods 0.000 claims description 19
- 239000000017 hydrogel Substances 0.000 claims description 14
- 230000000694 effects Effects 0.000 claims description 7
- 229910052739 hydrogen Inorganic materials 0.000 claims description 6
- 239000001257 hydrogen Substances 0.000 claims description 6
- 238000010438 heat treatment Methods 0.000 claims description 4
- 239000000178 monomer Substances 0.000 description 47
- 239000003795 chemical substances by application Substances 0.000 description 29
- -1 cyclic lactams Chemical class 0.000 description 16
- 229920001577 copolymer Polymers 0.000 description 10
- 239000000203 mixture Substances 0.000 description 10
- 125000004432 carbon atom Chemical group C* 0.000 description 9
- 239000000463 material Substances 0.000 description 9
- 239000000126 substance Substances 0.000 description 9
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 5
- 150000003254 radicals Chemical class 0.000 description 5
- 238000004659 sterilization and disinfection Methods 0.000 description 5
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- SEQKRHFRPICQDD-UHFFFAOYSA-N N-tris(hydroxymethyl)methylglycine Chemical compound OCC(CO)(CO)[NH2+]CC([O-])=O SEQKRHFRPICQDD-UHFFFAOYSA-N 0.000 description 4
- 125000000217 alkyl group Chemical group 0.000 description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 4
- 239000011780 sodium chloride Substances 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- LVLANIHJQRZTPY-UHFFFAOYSA-N vinyl carbamate Chemical compound NC(=O)OC=C LVLANIHJQRZTPY-UHFFFAOYSA-N 0.000 description 4
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 3
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- WOBHKFSMXKNTIM-UHFFFAOYSA-N Hydroxyethyl methacrylate Chemical compound CC(=C)C(=O)OCCO WOBHKFSMXKNTIM-UHFFFAOYSA-N 0.000 description 3
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 239000003431 cross linking reagent Substances 0.000 description 3
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 150000002924 oxiranes Chemical class 0.000 description 3
- 230000005855 radiation Effects 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 3
- QRIMLDXJAPZHJE-UHFFFAOYSA-N 2,3-dihydroxypropyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(O)CO QRIMLDXJAPZHJE-UHFFFAOYSA-N 0.000 description 2
- QKPKBBFSFQAMIY-UHFFFAOYSA-N 2-ethenyl-4,4-dimethyl-1,3-oxazol-5-one Chemical compound CC1(C)N=C(C=C)OC1=O QKPKBBFSFQAMIY-UHFFFAOYSA-N 0.000 description 2
- BESKSSIEODQWBP-UHFFFAOYSA-N 3-tris(trimethylsilyloxy)silylpropyl 2-methylprop-2-enoate Chemical group CC(=C)C(=O)OCCC[Si](O[Si](C)(C)C)(O[Si](C)(C)C)O[Si](C)(C)C BESKSSIEODQWBP-UHFFFAOYSA-N 0.000 description 2
- MUWZQYSJSCDUDT-UHFFFAOYSA-N 4,4-dimethyl-2-prop-1-en-2-yl-1,3-oxazol-5-one Chemical compound CC(=C)C1=NC(C)(C)C(=O)O1 MUWZQYSJSCDUDT-UHFFFAOYSA-N 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 2
- DBXNUXBLKRLWFA-UHFFFAOYSA-N N-(2-acetamido)-2-aminoethanesulfonic acid Chemical compound NC(=O)CNCCS(O)(=O)=O DBXNUXBLKRLWFA-UHFFFAOYSA-N 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical compound [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 125000002877 alkyl aryl group Chemical group 0.000 description 2
- 125000005119 alkyl cycloalkyl group Chemical group 0.000 description 2
- 125000000129 anionic group Chemical group 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 150000001732 carboxylic acid derivatives Chemical group 0.000 description 2
- 238000005266 casting Methods 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 239000012632 extractable Substances 0.000 description 2
- VOZRXNHHFUQHIL-UHFFFAOYSA-N glycidyl methacrylate Chemical compound CC(=C)C(=O)OCC1CO1 VOZRXNHHFUQHIL-UHFFFAOYSA-N 0.000 description 2
- 229920001477 hydrophilic polymer Polymers 0.000 description 2
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 2
- 229940088644 n,n-dimethylacrylamide Drugs 0.000 description 2
- YLGYACDQVQQZSW-UHFFFAOYSA-N n,n-dimethylprop-2-enamide Chemical compound CN(C)C(=O)C=C YLGYACDQVQQZSW-UHFFFAOYSA-N 0.000 description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 230000003204 osmotic effect Effects 0.000 description 2
- 229920001983 poloxamer Polymers 0.000 description 2
- 229920001987 poloxamine Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 238000010526 radical polymerization reaction Methods 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- SJHPCNCNNSSLPL-CSKARUKUSA-N (4e)-4-(ethoxymethylidene)-2-phenyl-1,3-oxazol-5-one Chemical compound O1C(=O)C(=C/OCC)\N=C1C1=CC=CC=C1 SJHPCNCNNSSLPL-CSKARUKUSA-N 0.000 description 1
- IEJPPSMHUUQABK-UHFFFAOYSA-N 2,4-diphenyl-4h-1,3-oxazol-5-one Chemical compound O=C1OC(C=2C=CC=CC=2)=NC1C1=CC=CC=C1 IEJPPSMHUUQABK-UHFFFAOYSA-N 0.000 description 1
- NPPNUGUVBUJRAB-UHFFFAOYSA-N 2-[tert-butyl(dimethyl)silyl]oxyethyl ethenyl carbonate Chemical compound CC(C)(C)[Si](C)(C)OCCOC(=O)OC=C NPPNUGUVBUJRAB-UHFFFAOYSA-N 0.000 description 1
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 1
- OMIGHNLMNHATMP-UHFFFAOYSA-N 2-hydroxyethyl prop-2-enoate Chemical compound OCCOC(=O)C=C OMIGHNLMNHATMP-UHFFFAOYSA-N 0.000 description 1
- FBXDMYKUMYUYHT-UHFFFAOYSA-N 4,4-dimethyl-2-prop-1-enyl-1,3-oxazol-5-one Chemical compound CC=CC1=NC(C)(C)C(=O)O1 FBXDMYKUMYUYHT-UHFFFAOYSA-N 0.000 description 1
- UPULOMQHYQDNNT-UHFFFAOYSA-N 5h-1,3-oxazol-2-one Chemical group O=C1OCC=N1 UPULOMQHYQDNNT-UHFFFAOYSA-N 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 206010015946 Eye irritation Diseases 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- 239000007993 MOPS buffer Substances 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 208000002193 Pain Diseases 0.000 description 1
- YGYAWVDWMABLBF-UHFFFAOYSA-N Phosgene Chemical group ClC(Cl)=O YGYAWVDWMABLBF-UHFFFAOYSA-N 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- UZMAPBJVXOGOFT-UHFFFAOYSA-N Syringetin Natural products COC1=C(O)C(OC)=CC(C2=C(C(=O)C3=C(O)C=C(O)C=C3O2)O)=C1 UZMAPBJVXOGOFT-UHFFFAOYSA-N 0.000 description 1
- 239000007997 Tricine buffer Substances 0.000 description 1
- 229920006311 Urethane elastomer Polymers 0.000 description 1
- 150000001253 acrylic acids Chemical class 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 150000003862 amino acid derivatives Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 150000001414 amino alcohols Chemical class 0.000 description 1
- 150000008064 anhydrides Chemical group 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OWMVSZAMULFTJU-UHFFFAOYSA-N bis-tris Chemical compound OCCN(CCO)C(CO)(CO)CO OWMVSZAMULFTJU-UHFFFAOYSA-N 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 125000001951 carbamoylamino group Chemical group C(N)(=O)N* 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000010668 complexation reaction Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- KCFYHBSOLOXZIF-UHFFFAOYSA-N dihydrochrysin Natural products COC1=C(O)C(OC)=CC(C2OC3=CC(O)=CC(O)=C3C(=O)C2)=C1 KCFYHBSOLOXZIF-UHFFFAOYSA-N 0.000 description 1
- 125000005442 diisocyanate group Chemical group 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- KZJNAICCMJTRKF-UHFFFAOYSA-N ethenyl 2-trimethylsilylethyl carbonate Chemical compound C[Si](C)(C)CCOC(=O)OC=C KZJNAICCMJTRKF-UHFFFAOYSA-N 0.000 description 1
- RWEUKWCZWYHIQA-UHFFFAOYSA-N ethenyl 3-trimethylsilylpropyl carbonate Chemical compound C[Si](C)(C)CCCOC(=O)OC=C RWEUKWCZWYHIQA-UHFFFAOYSA-N 0.000 description 1
- NDXTZJDCEOXFOP-UHFFFAOYSA-N ethenyl 3-tris(trimethylsilyloxy)silylpropyl carbonate Chemical compound C[Si](C)(C)O[Si](O[Si](C)(C)C)(O[Si](C)(C)C)CCCOC(=O)OC=C NDXTZJDCEOXFOP-UHFFFAOYSA-N 0.000 description 1
- BHBDVHVTNOYHLK-UHFFFAOYSA-N ethenyl 3-tris(trimethylsilyloxy)silylpropylsulfanylformate Chemical compound C[Si](C)(C)O[Si](O[Si](C)(C)C)(O[Si](C)(C)C)CCCSC(=O)OC=C BHBDVHVTNOYHLK-UHFFFAOYSA-N 0.000 description 1
- ILHMPZFVDISGNP-UHFFFAOYSA-N ethenyl n-[3-tris(trimethylsilyloxy)silylpropyl]carbamate Chemical compound C[Si](C)(C)O[Si](O[Si](C)(C)C)(O[Si](C)(C)C)CCCNC(=O)OC=C ILHMPZFVDISGNP-UHFFFAOYSA-N 0.000 description 1
- KRAZQXAPJAYYJI-UHFFFAOYSA-N ethenyl trimethylsilylmethyl carbonate Chemical compound C[Si](C)(C)COC(=O)OC=C KRAZQXAPJAYYJI-UHFFFAOYSA-N 0.000 description 1
- 229940117927 ethylene oxide Drugs 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 231100000013 eye irritation Toxicity 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 150000002431 hydrogen Chemical group 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 239000000819 hypertonic solution Substances 0.000 description 1
- 229940021223 hypertonic solution Drugs 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- DCUFMVPCXCSVNP-UHFFFAOYSA-N methacrylic anhydride Chemical compound CC(=C)C(=O)OC(=O)C(C)=C DCUFMVPCXCSVNP-UHFFFAOYSA-N 0.000 description 1
- QRWZCJXEAOZAAW-UHFFFAOYSA-N n,n,2-trimethylprop-2-enamide Chemical compound CN(C)C(=O)C(C)=C QRWZCJXEAOZAAW-UHFFFAOYSA-N 0.000 description 1
- ZTUGCJNAJJDKDC-UHFFFAOYSA-N n-(3-hydroxypropyl)-2-methylprop-2-enamide Chemical compound CC(=C)C(=O)NCCCO ZTUGCJNAJJDKDC-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- ARJOQCYCJMAIFR-UHFFFAOYSA-N prop-2-enoyl prop-2-enoate Chemical compound C=CC(=O)OC(=O)C=C ARJOQCYCJMAIFR-UHFFFAOYSA-N 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 1
- 238000004381 surface treatment Methods 0.000 description 1
- 239000012929 tonicity agent Substances 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- UHUUYVZLXJHWDV-UHFFFAOYSA-N trimethyl(methylsilyloxy)silane Chemical compound C[SiH2]O[Si](C)(C)C UHUUYVZLXJHWDV-UHFFFAOYSA-N 0.000 description 1
- 229920001567 vinyl ester resin Polymers 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65B—MACHINES, APPARATUS OR DEVICES FOR, OR METHODS OF, PACKAGING ARTICLES OR MATERIALS; UNPACKING
- B65B25/00—Packaging other articles presenting special problems
- B65B25/008—Packaging other articles presenting special problems packaging of contact lenses
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29D—PRODUCING PARTICULAR ARTICLES FROM PLASTICS OR FROM SUBSTANCES IN A PLASTIC STATE
- B29D11/00—Producing optical elements, e.g. lenses or prisms
- B29D11/00009—Production of simple or compound lenses
- B29D11/00038—Production of contact lenses
- B29D11/00067—Hydrating contact lenses
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D2585/00—Containers, packaging elements or packages specially adapted for particular articles or materials
- B65D2585/54—Containers, packaging elements or packages specially adapted for particular articles or materials for articles of special shape not otherwise provided for
- B65D2585/545—Contact lenses
Definitions
- This invention provides a method for treating ophthalmic lenses in its package with an organic surface treatment agent.
- the surfaces of the lenses may be rendered more biocompatible, for the purpose of reducing or eliminating epithelial cell growth on the lens.
- intraocular lenses are often placed in the eye with an intraocular lens inserter which has surfaces that contact the lens while it is extruded against these surfaces; the lens surfaces may be modified to become more lubricious so as to lower the coefficient of friction for contacting the lens inserted.
- the lens surfaces may be made more wettable by tear film or less resistant to protein and/or lipid deposits from tear film, and more comfortable during wear.
- the lens surfaces have a higher tendency to be hydrophobic with lower wettability, so often a surface treatment is desirable to increase the surface wettability is desired.
- Various methods of changing the surface characteristics of ophthalmic lenses are known that involve attaching a treatment agent to the lens surfaces.
- a conventional manner of packaging ophthalmic lenses, especially contact lenses, is in a so-called blister package.
- Such packages include a recess designed to hold an individual lens, which is typically immersed in a saline packaging solution.
- the packages are then enclosed and sealed with lidstock, the lidstock conventionally being a metallic laminate that can withstand post-packaging heat sterilization conditions.
- the packaging solution may include various agents.
- packaging solutions for blister packages have sometimes been formulated to reduce or eliminate lens folding and sticking. For this reason, polyvinyl alcohol (PVA) has been used in contact lens packaging solutions.
- PVA polyvinyl alcohol
- U.S. Pat. No. 6,440,366 discloses contact lens packaging solutions comprising polyethylene oxide (PEO)/polypropylene oxide (PPO) block copolymers, especially poloxamers or poloxamines.
- Known blister packages include recesses that are concave, recesses that are flat-bottomed, and recesses that are partially concave with a flat bottom.
- a blister package with a flat bottom having four parallel, longitudinal grooves therein which is used to package various contact lenses sold by Bausch & Lomb Incorporated (Rochester, N.Y., USA), and has the general configuration illustrated in FIG. 7 .
- Other specific examples may be found in U.S. Pat. Nos. 5,842,325; 5,722,536; 5,467,868; 2004/0031701; 2004/0004008; 2002/0046958; 6,072,172; 5,143,660; and 6,889,825.
- This invention provides of a method of treating an ophthalmic lens in a package, comprising: placing the lens and an aqueous solution in a recess of package, the solution comprising an organic surface treatment agent that attaches to anterior and posterior surfaces of the lens; and sealing the recess of the package with lidstock and sterilizing the package contents; wherein a bottom of the recess includes grooves.
- This invention recognized that it may be desirable to effect attachment of the treatment agent to the lens surfaces while the lens is contained in a package, for example, to reduce material handling steps and the accompanying costs associated therewith. And if one employs a solution, containing the treatment agent, that is ophthalmically compatible, this solution may serve as the final packaging solution, and the heat sterilization of the package and its contents can serve to effect chemical attachment of the treatment agent to the lens surfaces.
- FIG. 1 is a top perspective view of a first embodiment of a lens blister package suitable for the method of this invention.
- FIG. 2 is a bottom perspective view of the lens blister package of FIG. 1 .
- FIG. 3 is a top plan view of the lens blister package of FIG. 1 .
- FIG. 4 is a cross-sectional view of the lens blister package of FIG. 1 , taken along line A-A of FIG. 3 .
- FIG. 5 is a cross-sectional view of the lens blister package of FIG. 1 , taken along line B-B of FIG. 3 .
- FIG. 6 is a top plan view of a second embodiment of a lens blister package suitable for the method of this invention.
- FIG. 7 is a top plan view of a third embodiment of a lens blister package suitable for the method of this invention.
- This invention provides a method of treating an ophthalmic lens in a package.
- the method comprises: placing the lens and an aqueous solution in a recess of package, the solution comprising an organic surface treatment agent that attaches to anterior and posterior surfaces of the lens; and sealing the recess of the package with lidstock and sterilizing the package contents; wherein a bottom of the recess includes grooves.
- ophthalmic lens means a lens intended for direct contact with ophthalmic tissue, including contact lenses and intraocular lenses.
- contact lenses including contact lenses and intraocular lenses.
- the process is discussed with particular reference to silicone hydrogel contact lenses, a preferred embodiment of this invention, but the invention may be employed for surface treating other polymeric biomedical devices.
- Hydrogels comprise a hydrated, crosslinked polymeric system containing water in an equilibrium state. Accordingly, hydrogels are copolymers prepared from hydrophilic monomers.
- the hydrogel copolymers are generally prepared by polymerizing a mixture containing at least one lens-forming silicone-containing monomer and at least one lens-forming hydrophilic monomer. Either the silicone-containing monomer or the hydrophilic monomer may function as a crosslinking agent (a crosslinking agent being defined as a monomer having multiple polymerizable functionalities), or alternately, a separate crosslinking agent may be employed in the initial monomer mixture from which the hydrogel copolymer is formed.
- a crosslinking agent being defined as a monomer having multiple polymerizable functionalities
- Silicone hydrogels typically have a water content between about 10 to about 80 weight percent.
- Examples of useful lens-forming hydrophilic monomers include: amides such as N,N-dimethylacrylamide and N,N-dimethylmethacrylamide; cyclic lactams such as N-vinyl-2-pyrrolidone; (meth)acrylated alcohols, such as 2-hydroxyethyl methacrylate, 2-hydroxyethyl acrylate and glyceryl methacrylate; (meth)acrylated poly(ethylene glycol)s; (meth)acrylic acids such as methacrylic acid and acrylic acid; and azlactone-containing monomers, such as 2-isopropenyl-4,4-dimethyl-2-oxazolin-5-one and 2-vinyl-4,4-dimethyl-2-oxazolin-5-one.
- amides such as N,N-dimethylacrylamide and N,N-dimethylmethacrylamide
- cyclic lactams such as N-vinyl-2-pyrrolidone
- (meth)” denotes an optional methyl substituent.
- terms such as “(meth)acrylate” denotes either methacrylate or acrylate
- “(meth)acrylic acid” denotes either methacrylic acid or acrylic acid.
- hydrophilic vinyl carbonate or vinyl carbamate monomers disclosed in U.S. Pat. No. 5,070,215
- hydrophilic oxazolone monomers disclosed in U.S. Pat. No. 4,910,277, the disclosures of which are incorporated herein by reference.
- Other suitable hydrophilic monomers will be apparent to one skilled in the art.
- silicone-containing monomers examples include bulky polysiloxanylalkyl (meth)acrylic monomers.
- An example of such monofunctional, bulky polysiloxanylalkyl (meth)acrylic monomers are represented by the following Formula I:
- X denotes —O— or —NR—
- each R 1 independently denotes hydrogen or methyl
- each R 2 independently denotes a lower alkyl radical, phenyl radical or a group represented by
- each R 2 ′ independently denotes a lower alkyl or phenyl radical; and h is 1 to 10.
- One preferred bulky monomer is 3-methacryloxypropyl tris(trimethyl-siloxy)silane or tris(trimethylsiloxy)silylpropyl methacrylate, sometimes referred to as TRIS.
- silicone-containing monomers includes silicone-containing vinyl carbonate or vinyl carbamate monomers such as: 1,3-bis[4-vinyloxycarbonyloxy)but-1-yl]tetramethyldisiloxane; 1,3-bis[4-vinyloxycarbonyloxy)but-1-yl]polydimethylsiloxane; 3-(trimethylsilyl)propyl vinyl carbonate; 3-(vinyloxycarbonylthio)propyl[tris(trimethylsiloxy)silane]; 3-[tris(trimethylsiloxy)silyl]propyl vinyl carbamate; 3-[tris(trimethylsiloxy)silyl]propyl allyl carbamate; 3-[tris(trimethylsiloxy)silyl]propyl vinyl carbonate; t-butyldimethylsiloxyethyl vinyl carbonate; trimethylsilylethyl vinyl carbonate; and trimethylsilylmethyl vinyl carbonate.
- silicone-containing vinyl carbonate or vinyl carbamate monomers are represented by Formula II:
- Y′ denotes —O—, —S— or —NH—
- R Si denotes a silicone-containing organic radical
- R 3 denotes hydrogen or methyl
- d is 1, 2, 3 or 4; and q is 0 or 1.
- Suitable silicone-containing organic radicals R Si include the following:
- p′ is 1 to 6;
- R 5 denotes an alkyl radical or a fluoroalkyl radical having 1 to 6 carbon atoms
- silicone-containing monomers includes polyurethane-polysiloxane macromonomers (also sometimes referred to as prepolymers), which may have hard-soft-hard blocks like traditional urethane elastomers.
- silicone urethane monomers are represented by Formulae IV and V:
- D denotes an alkyl diradical, an alkyl cycloalkyl diradical, a cycloalkyl diradical, an aryl diradical or an alkylaryl diradical having 6 to 30 carbon atoms;
- G denotes an alkyl diradical, a cycloalkyl diradical, an alkyl cycloalkyl diradical, an aryl diradical or an alkylaryl diradical having 1 to 40 carbon atoms and which may contain ether, thio or amine linkages in the main chain;
- a is at least 1;
- A denotes a divalent polymeric radical of Formula VI:
- each R s independently denotes an alkyl or fluoro-substituted alkyl group having 1 to 10 carbon atoms which may contain ether linkages between carbon atoms;
- n′ is at least 1;
- p is a number which provides a moiety weight of 400 to 10,000;
- each of E and E′ independently denotes a polymerizable unsaturated organic radical represented by Formula VII:
- R 6 is hydrogen or methyl
- R 7 is hydrogen, an alkyl radical having 1 to 6 carbon atoms, or a —CO—Y—R 9 radical wherein Y is —O—, —S— or —NH—;
- R 8 is a divalent alkylene radical having 1 to 10 carbon atoms
- R 9 is a alkyl radical having 1 to 12 carbon atoms
- X denotes —CO— or —OCO—
- Z denotes —O— or —NH—
- Ar denotes an aromatic radical having 6 to 30 carbon atoms
- w is 0 to 6; x is 0 or 1; y is 0 or 1; and z is 0 or 1.
- m is at least 1 and is preferably 3 or 4
- a is at least 1 and preferably is 1
- p is a number which provides a moiety weight of 400 to 10,000 and is preferably at least 30
- R 10 is a diradical of a diisocyanate after removal of the isocyanate group, such as the diradical of isophorone diisocyanate
- each E′′ is a group represented by:
- a preferred silicone hydrogel material comprises (based on the initial monomer mixture that is copolymerized to form the hydrogel copolymeric material) 5 to 50 percent, preferably 10 to 25, by weight of one or more silicone macromonomers, 5 to 75 percent, preferably 30 to 60 percent, by weight of one or more polysiloxanylalkyl (meth)acrylic monomers, and 10 to 50 percent, preferably 20 to 40 percent, by weight of a hydrophilic monomer.
- the silicone macromonomer is a poly(organosiloxane) capped with an unsaturated group at two or more ends of the molecule.
- the silane macromonomer is a silicone-containing vinyl carbonate or vinyl carbamate or a polyurethane-polysiloxane having one or more hard-soft-hard blocks and end-capped with a hydrophilic monomer.
- contact lens materials for which the present invention is useful are taught in U.S. Pat. No. 6,891,010 (Kunzler et al.); U.S. Pat. No. 5,908,906 (Kunzler et al.); U.S. Pat. No. 5,714,557 (Kunzler et al.); U.S. Pat. No. 5,710,302 (Kunzler et al.); U.S. Pat. No. 5,708,094 (Lai et al.); U.S. Pat. No. 5,616,757 (Bambury et al.); U.S. Pat. No. 5,610,252 (Bambury et al.); U.S. Pat. No.
- the monomer mixtures may be charged to a mold, and then subjected to heat and/or light radiation, such as UV radiation, to effect curing, or free radical polymerization, of the monomer mixture in the mold.
- heat and/or light radiation such as UV radiation
- the monomer mixtures may be cast in the shape of rods or buttons, which are then lathe cut into a desired shape, for example, into a lens-shaped article.
- extractables include any remaining diluent, unreacted monomers, and oligomers formed from side reactions of the monomers.
- the lens is hydrated, either as part of the extraction process, or in a separate subsequent operation.
- the lens is hydrated to replace the organic solvent with water or aqueous solution. Hydration may be performed while the lens is held in its package, or prior to placing the lens in the package. In any event, ultimately, the lens is contained in the package with an aqueous packaging solution.
- This invention recognized the desirability of effecting attachment of the treatment agent to the lens surfaces while the lens is contained in a package, in contrast to effecting attachment prior to placing the lens in the package. This offers a reduction in material handling steps and the accompanying costs associated therewith. Also, if the solution containing the treatment agent is ophthalmically compatible, this solution may serve as the final packaging solution, and the heat sterilization of the package and its contents can serve to effect chemical attachment of the treatment agent to the lens surfaces.
- attachment of the treatment agent to the lens surface, and like terms, denotes that the treatment agent is substantially adhered to the lens surfaces. Thus, after a single rinsing with water of the lens surfaces with the treatment agent attached thereto, at least 50% of the treatment agent will remain adhered to the lens surfaces.
- the surface treatment agent is attached to the lens surfaces by at least one of covalent bonding, ionic attachment, and hydrogen bonding.
- Covalent bonding denotes that a chemical reaction occurs between the treatment agent and the lens surface, so that covalent bonds are formed therebetween.
- Ionic attachment denotes that the lens surfaces are ionically charged, and the organic surface treatment agent contains moieties with an opposite, ionic charge.
- the lens surfaces may be anionic charged, and the treatment agent may be cationic or zwitterionic, that interacts with the anionic lens surface.
- organic surface treatment agents may be employed, including treatment agents known in the art.
- this invention is applicable for the organic surface treatment agents described in U.S. Pat. No. 7,083,646, the entire disclosure of which is incorporated herein by reference.
- this method involves surface modification of medical devices, particularly ophthalmic lenses, with one or more reactive, hydrophilic polymers as the surface treatment agent.
- the reactive, hydrophilic polymers are copolymers of at least one hydrophilic monomer and at least one monomer that contains reactive chemical functionality.
- the hydrophilic monomers can be aprotic types such as N,N-dimethylacrylamide and N-vinylpyrrolidone, or protic types such as methacrylic acid and 2-hydroxyethyl methacrylate.
- the monomer containing reactive chemical functionality can be an epoxide-containing monomer, such as glycidyl methacrylate.
- the hydrophilic monomer and the monomer containing reactive chemical functionality are copolymerized at a desired molar ratio thereof.
- the hydrophilic monomer serves to render the resultant copolymer hydrophilic.
- the monomer containing reactive chemical functionality provides a reactive group that can react with the lens surface. In other words, this resultant copolymer contains the reactive chemical functionality that can react with complementary functional groups at or near the lens surface, and form covalent bonds therewith.
- this invention is applicable for the organic surface treatment agents described in US 2007/0122540, the entire disclosure of which is incorporated herein by reference.
- this method involves surface modification of medical devices, particularly ophthalmic lenses, where reactive surfactants as the surface treatment agent are covalently bound to the lens surfaces.
- Preferred reactive surfactants are functionalized poloxamers or functionalized poloxamines having reactive functionality that is complimentary to surface functionality of the ophthalmic lens.
- Representative functionalized surfactants are those containing epoxide, methacrylate, or isocyanate functionalities, such as represented below.
- the reactive groups of the treatment agent are matched with the reactive groups on the lens surface.
- the lens surfaces contain carboxylic acid groups
- a glycidyl group can be a reactive group of the surface treatment agent.
- an isocyanate group or carbonyl chloride group can be a reactive group of the surface treatment agent.
- suitable lens-forming monomers providing reactive groups on the lens surface
- suitable lens-forming monomers include those having hydroxy functional groups, such as 2-hydroxyethyl methacrylate, glyceryl methacrylate and 3-hydroxypropyl methacrylamide.
- suitable lens-forming monomers providing the lens surfaces with carboxylic acid reactive groups include methacrylic acid, acrylic acid and N-carboxy- ⁇ -alanine-N-vinyl ester.
- suitable lens-forming monomers providing the lens surface with oxazolinone reactive groups include 2-isopropenyl-4,4-dimethyl-2-oxazolin-5-one, 2-vinyl-4,4-dimethyl-2-oxazolin-5-one, spiro-4′-(2′-isopropenyl-2′-oxazolin-5-one)cyclohexane, spiro-4′-(2′-vinyl-2′-oxazolin-5′-one)cyclohexane and 2-(1-propenyl)-4,4-dimethyl-oxazolin-5-one.
- suitable lens-forming monomers providing the lens surface with anhydride functional groups include methacrylic anhydride, maleic anhydride and acrylic anhydride.
- An example of a suitable lens-forming monomer providing the lens surfaces with epoxide reactive groups includes glycidyl methacrylate.
- the surface treatment agent may be one as employed in the method described in U.S. Pat. No. 6,428,839 (Kunzler et al.), the entire disclosure of which is incorporated herein by reference.
- this method employs poly(acrylic) acid (PAA) surface complexation.
- PAA poly(acrylic) acid
- Hydrogel contact lens copolymers containing polymerized hydrophilic lens-forming monomers having relatively strong proton donating moieties, for example DMA or NVP are treated with water-based solutions containing PAA or PAA co-polymers, acting as wetting agents, to render a lubricious, stable, highly wettable PAA-based surface coating.
- PAA-containing agents may be employed, although generally, coating materials containing carboxylic acid functionality are preferred.
- Surface treatment agents include poly(vinylpyrrolidinone(VP)-co-acrylic acid(AA)), poly(methylvinylether-alt-maleic acid), poly(acrylic acid-graft-ethyleneoxide), poly(AA-co-methacrylic acid), poly(acrylamide-co-AA), poly(AA-co-maleic), and poly(butadiene-maleic acid).
- Particularly preferred polymers are characterized by acid contents of at least about 30 mole percent, preferably at least about 40 mole percent.
- the lens, with its surface in contact with the PAA-containing solution may be heated by autoclaving, or subjected to microwave radiation, to facilitate attachment of the PAA to the lens surface.
- this solution also serves as the final packaging solution, and this solution is ophthalmically compatible. Then, when the package contents are heat sterilized, this heat treatment can serve to effect chemical attachment of the treatment agent to the lens surfaces, if needed. Any excess treatment agent, not attached to the lens surfaces, remains in the final packaging solution.
- this solution is removed with a portion of the organic treatment agent remaining attached to the lens surface. Any excess treatment agent is removed with the solution. If heat treatment is needed to effect chemical attachment of the treatment agent to the lens surfaces, the heat treatment is performed prior to removing this solution from the package recess. Subsequently, after removing this solution, an aqueous packaging solution is added to the recess, followed by sealing the recess with lidstock and sterilizing the package contents.
- the final packaging solution is an aqueous solution, preferably a saline solution. having a pH value within the range of about 6 to about 8, and preferably about 6.5 to about 7.8.
- Suitable buffers may be included, such as: phosphate; borate; citrate; carbonate; tris-(hydroxymethyl)aminomethane (TRIS); bis(2-hydroxyethyl)-imino-tris-(hydroxymethyl)aminoalcohol (bis-tris); zwitterionic buffers such as N-[2-Hydroxy-1,1-bis(hydroxymethyl)ethyl]glycine (Tricine)and N-[2-Hydroxy-1,1-bis(hydroxymethyl)ethyl]glycine, MOPS; N-(Carbamoylmethyl)taurine (ACES); amino acids and amino acid derivatives; and mixtures thereof.
- TriS tris-(hydroxymethyl)aminomethane
- bis-tris bis(2-hydroxyethyl)
- buffers will be used in amounts ranging from about 0.05 to about 2.5 percent by weight, and preferably from about 0.1 to about 1.5 percent by weight of the solution.
- the solutions of the present invention may be adjusted with tonicity agents, to approximate the osmotic pressure of normal lacrimal fluids, which is equivalent to a 0.9 percent solution of sodium chloride or 2.5 percent of glycerol solution.
- the solutions are made substantially isotonic with physiological saline used alone or in combination, otherwise if simply blended with sterile water and made hypotonic or made hypertonic the lenses will lose their desirable optical parameters.
- excess saline may result in the formation of a hypertonic solution, which will cause stinging, and eye irritation.
- Suitable tonicity adjusting agents include, but are not limited to, sodium and potassium chloride, dextrose, calcium and magnesium chloride and the like and mixtures thereof. These agents are typically used individually in amounts ranging from about 0.01 to about 2.5% w/v and preferably from about 0.2 to about 1.5% w/v.
- the tonicity agent will be employed in an amount to provide a final osmotic value of at least about 200 mOsm/kg, preferably from about 200 to about 450 mOsm/kg, more preferably from about 250 to about 400 mOsm/kg, and most preferably from about 280 to about 370 mOsm/kg.
- FIGS. 1 to 7 Representative blister packages that may be employed in the method of this invention are illustrated in FIGS. 1 to 7 .
- FIGS. 1 to 5 illustrate a first embodiment of a blister package.
- Package 1 includes a substrate 3 including recess 2 formed therein, recess 2 sized and configured to hold an individual contact lens 5 therein.
- recess 2 is a concave recess with a flat bottom 6 .
- the anterior surface of contact lens 5 rests on bottom 6 when the package is oriented as in FIG. 4 . This can lead to non-uniform coating of the contact lens, especially this anterior surface of the lens resting on the recess bottom.
- bottom 6 includes three parallel, longitudinal grooves 7 therein. Grooves 7 preferably have a depth of at least 0.2 mm, more preferably at least 0.4 mm.
- contact lens 5 is contained in recess 2 and immersed in solution.
- Recess 2 is then sealed with lidstock, i.e., by sealing the lidstock with heat to raised surface 9 .
- the package and its contents may now be sterilized, preferably by heat sterilization such as autoclaving.
- Recess 2 has a depth greater than a height of the lens, and a sufficiently large volume, that the lens is completely immersed in the final packaging solution.
- FIG. 6 illustrates a second embodiment of a blister package.
- Package 11 includes a substrate 13 including recess 12 formed therein.
- recess 12 is a concave recess, i.e., a rounded bowl shape.
- the bottom includes three interconnected, longitudinal grooves 17 therein.
- FIG. 7 illustrates a third embodiment of a blister package.
- Package 21 includes a substrate 23 including recess 22 formed therein.
- recess 22 has a somewhat oval-shaped flat bottom, and the bottom includes four longitudinal grooves 27 therein.
- the recess is then sealed with lidstock, i.e., by sealing the lidstock with heat.
- the lidstock is sealed to raised surface 9 in FIGS. 1 to 5 .
- the package and its contents may now be sterilized, preferably by heat sterilization such as autoclaving.
- the recess has a depth greater than a height of the lens, and a sufficiently large volume, that the lens is completely immersed in the final packaging solution.
- the recess is filed with at least 0.5 ml of final packaging solution, more preferably at least 1 ml, and most preferably at least 1.2 ml.
Abstract
A method of treating an ophthalmic lens in a package involves: placing the lens and an aqueous solution in a recess of package, the solution including an organic surface treatment agent that attaches to anterior and posterior surfaces of the lens; and sealing the recess of the package with lidstock and sterilizing the package contents. The bottom of the recess includes grooves.
Description
- This application claims the benefit of provisional patent application No. 61/012,856 filed Dec. 11, 2007 which is incorporated by reference herein.
- This invention provides a method for treating ophthalmic lenses in its package with an organic surface treatment agent.
- It is often desired to improve the surface characteristics of an ophthalmic lens. For example, in the case of intraocular lenses, the surfaces of the lenses may be rendered more biocompatible, for the purpose of reducing or eliminating epithelial cell growth on the lens. Also, intraocular lenses are often placed in the eye with an intraocular lens inserter which has surfaces that contact the lens while it is extruded against these surfaces; the lens surfaces may be modified to become more lubricious so as to lower the coefficient of friction for contacting the lens inserted. In the case of contact lenses, the lens surfaces may be made more wettable by tear film or less resistant to protein and/or lipid deposits from tear film, and more comfortable during wear. With respect to silicon-containing lenses, the lens surfaces have a higher tendency to be hydrophobic with lower wettability, so often a surface treatment is desirable to increase the surface wettability is desired. Various methods of changing the surface characteristics of ophthalmic lenses are known that involve attaching a treatment agent to the lens surfaces.
- A conventional manner of packaging ophthalmic lenses, especially contact lenses, is in a so-called blister package. Such packages include a recess designed to hold an individual lens, which is typically immersed in a saline packaging solution. The packages are then enclosed and sealed with lidstock, the lidstock conventionally being a metallic laminate that can withstand post-packaging heat sterilization conditions. The packaging solution may include various agents. As one example, because the lens material may tend to stick to itself and to the lens package, packaging solutions for blister packages have sometimes been formulated to reduce or eliminate lens folding and sticking. For this reason, polyvinyl alcohol (PVA) has been used in contact lens packaging solutions. Additionally, U.S. Pat. No. 6,440,366 discloses contact lens packaging solutions comprising polyethylene oxide (PEO)/polypropylene oxide (PPO) block copolymers, especially poloxamers or poloxamines.
- Various blister packages are known. Known blister packages include recesses that are concave, recesses that are flat-bottomed, and recesses that are partially concave with a flat bottom. One specific example is a blister package with a flat bottom having four parallel, longitudinal grooves therein, which is used to package various contact lenses sold by Bausch & Lomb Incorporated (Rochester, N.Y., USA), and has the general configuration illustrated in
FIG. 7 . Other specific examples may be found in U.S. Pat. Nos. 5,842,325; 5,722,536; 5,467,868; 2004/0031701; 2004/0004008; 2002/0046958; 6,072,172; 5,143,660; and 6,889,825. - This invention provides of a method of treating an ophthalmic lens in a package, comprising: placing the lens and an aqueous solution in a recess of package, the solution comprising an organic surface treatment agent that attaches to anterior and posterior surfaces of the lens; and sealing the recess of the package with lidstock and sterilizing the package contents; wherein a bottom of the recess includes grooves.
- This invention recognized that it may be desirable to effect attachment of the treatment agent to the lens surfaces while the lens is contained in a package, for example, to reduce material handling steps and the accompanying costs associated therewith. And if one employs a solution, containing the treatment agent, that is ophthalmically compatible, this solution may serve as the final packaging solution, and the heat sterilization of the package and its contents can serve to effect chemical attachment of the treatment agent to the lens surfaces.
- However, problems were encountered in that the surfaces of the lens were not uniformly coated with the treatment agent, especially the lens surface in contact with the recess bottom. These problems were overcome by employing packages with recesses including grooves in the bottoms thereof, to ensure better flow of the solution around the lens surfaces.
-
FIG. 1 is a top perspective view of a first embodiment of a lens blister package suitable for the method of this invention. -
FIG. 2 is a bottom perspective view of the lens blister package ofFIG. 1 . -
FIG. 3 is a top plan view of the lens blister package ofFIG. 1 . -
FIG. 4 is a cross-sectional view of the lens blister package ofFIG. 1 , taken along line A-A ofFIG. 3 . -
FIG. 5 is a cross-sectional view of the lens blister package ofFIG. 1 , taken along line B-B ofFIG. 3 . -
FIG. 6 is a top plan view of a second embodiment of a lens blister package suitable for the method of this invention. -
FIG. 7 is a top plan view of a third embodiment of a lens blister package suitable for the method of this invention. - This invention provides a method of treating an ophthalmic lens in a package. The method comprises: placing the lens and an aqueous solution in a recess of package, the solution comprising an organic surface treatment agent that attaches to anterior and posterior surfaces of the lens; and sealing the recess of the package with lidstock and sterilizing the package contents; wherein a bottom of the recess includes grooves.
- The term “ophthalmic lens” means a lens intended for direct contact with ophthalmic tissue, including contact lenses and intraocular lenses. In the following description, the process is discussed with particular reference to silicone hydrogel contact lenses, a preferred embodiment of this invention, but the invention may be employed for surface treating other polymeric biomedical devices.
- Hydrogels comprise a hydrated, crosslinked polymeric system containing water in an equilibrium state. Accordingly, hydrogels are copolymers prepared from hydrophilic monomers. In the case of silicone hydrogels, the hydrogel copolymers are generally prepared by polymerizing a mixture containing at least one lens-forming silicone-containing monomer and at least one lens-forming hydrophilic monomer. Either the silicone-containing monomer or the hydrophilic monomer may function as a crosslinking agent (a crosslinking agent being defined as a monomer having multiple polymerizable functionalities), or alternately, a separate crosslinking agent may be employed in the initial monomer mixture from which the hydrogel copolymer is formed. (As used herein, the term “monomer” or “monomeric” and like terms denote relatively low molecular weight compounds that are polymerizable by free radical polymerization, as well as higher molecular weight compounds also referred to as “prepolymers”, “macromonomers”, and related terms.) Silicone hydrogels typically have a water content between about 10 to about 80 weight percent.
- Examples of useful lens-forming hydrophilic monomers include: amides such as N,N-dimethylacrylamide and N,N-dimethylmethacrylamide; cyclic lactams such as N-vinyl-2-pyrrolidone; (meth)acrylated alcohols, such as 2-hydroxyethyl methacrylate, 2-hydroxyethyl acrylate and glyceryl methacrylate; (meth)acrylated poly(ethylene glycol)s; (meth)acrylic acids such as methacrylic acid and acrylic acid; and azlactone-containing monomers, such as 2-isopropenyl-4,4-dimethyl-2-oxazolin-5-one and 2-vinyl-4,4-dimethyl-2-oxazolin-5-one. (As used herein, the term “(meth)” denotes an optional methyl substituent. Thus, terms such as “(meth)acrylate” denotes either methacrylate or acrylate, and “(meth)acrylic acid” denotes either methacrylic acid or acrylic acid.) Still further examples are the hydrophilic vinyl carbonate or vinyl carbamate monomers disclosed in U.S. Pat. No. 5,070,215, and the hydrophilic oxazolone monomers disclosed in U.S. Pat. No. 4,910,277, the disclosures of which are incorporated herein by reference. Other suitable hydrophilic monomers will be apparent to one skilled in the art.
- Applicable silicone-containing monomeric materials for use in the formation of silicone hydrogels are well known in the art and numerous examples are provided in U.S. Pat. Nos. 4,136,250; 4,153,641; 4,740,533; 5,034,461; 5,070,215; 5,260,000; 5,310,779; and 5,358,995.
- Examples of applicable silicone-containing monomers include bulky polysiloxanylalkyl (meth)acrylic monomers. An example of such monofunctional, bulky polysiloxanylalkyl (meth)acrylic monomers are represented by the following Formula I:
- wherein:
- X denotes —O— or —NR—;
- each R1 independently denotes hydrogen or methyl;
- each R2 independently denotes a lower alkyl radical, phenyl radical or a group represented by
- wherein each R2′ independently denotes a lower alkyl or phenyl radical; and h is 1 to 10. One preferred bulky monomer is 3-methacryloxypropyl tris(trimethyl-siloxy)silane or tris(trimethylsiloxy)silylpropyl methacrylate, sometimes referred to as TRIS.
- Another class of representative silicone-containing monomers includes silicone-containing vinyl carbonate or vinyl carbamate monomers such as: 1,3-bis[4-vinyloxycarbonyloxy)but-1-yl]tetramethyldisiloxane; 1,3-bis[4-vinyloxycarbonyloxy)but-1-yl]polydimethylsiloxane; 3-(trimethylsilyl)propyl vinyl carbonate; 3-(vinyloxycarbonylthio)propyl[tris(trimethylsiloxy)silane]; 3-[tris(trimethylsiloxy)silyl]propyl vinyl carbamate; 3-[tris(trimethylsiloxy)silyl]propyl allyl carbamate; 3-[tris(trimethylsiloxy)silyl]propyl vinyl carbonate; t-butyldimethylsiloxyethyl vinyl carbonate; trimethylsilylethyl vinyl carbonate; and trimethylsilylmethyl vinyl carbonate.
- An example of silicone-containing vinyl carbonate or vinyl carbamate monomers are represented by Formula II:
- wherein
- Y′ denotes —O—, —S— or —NH—;
- RSi denotes a silicone-containing organic radical;
- R3 denotes hydrogen or methyl;
- d is 1, 2, 3 or 4; and q is 0 or 1.
- Suitable silicone-containing organic radicals RSi include the following:
- wherein:
- R4 denotes
- wherein p′ is 1 to 6;
- R5 denotes an alkyl radical or a fluoroalkyl radical having 1 to 6 carbon atoms;
- e is 1 to 200; n′ is 1, 2, 3 or 4; and m′ is 0, 1, 2, 3, 4 or 5.
- An example of a particular species within Formula II is represented by Formula III:
- Another class of silicone-containing monomers includes polyurethane-polysiloxane macromonomers (also sometimes referred to as prepolymers), which may have hard-soft-hard blocks like traditional urethane elastomers. Examples of silicone urethane monomers are represented by Formulae IV and V:
-
E(*D*A*D*G)a*D*A*D*E′; or (IV) -
E(*D*G*D*A)a*D*G*D*E′; (V) - wherein:
- D denotes an alkyl diradical, an alkyl cycloalkyl diradical, a cycloalkyl diradical, an aryl diradical or an alkylaryl diradical having 6 to 30 carbon atoms;
- G denotes an alkyl diradical, a cycloalkyl diradical, an alkyl cycloalkyl diradical, an aryl diradical or an alkylaryl diradical having 1 to 40 carbon atoms and which may contain ether, thio or amine linkages in the main chain;
- * denotes a urethane or ureido linkage;
- a is at least 1;
- A denotes a divalent polymeric radical of Formula VI:
- wherein:
- each Rs independently denotes an alkyl or fluoro-substituted alkyl group having 1 to 10 carbon atoms which may contain ether linkages between carbon atoms;
- m′ is at least 1; and
- p is a number which provides a moiety weight of 400 to 10,000;
- each of E and E′ independently denotes a polymerizable unsaturated organic radical represented by Formula VII:
- wherein:
- R6 is hydrogen or methyl;
- R7 is hydrogen, an alkyl radical having 1 to 6 carbon atoms, or a —CO—Y—R9 radical wherein Y is —O—, —S— or —NH—;
- R8 is a divalent alkylene radical having 1 to 10 carbon atoms;
- R9 is a alkyl radical having 1 to 12 carbon atoms;
- X denotes —CO— or —OCO—;
- Z denotes —O— or —NH—;
- Ar denotes an aromatic radical having 6 to 30 carbon atoms;
- w is 0 to 6; x is 0 or 1; y is 0 or 1; and z is 0 or 1.
- A more specific example of a silicone-containing urethane monomer is represented by Formula (VIII):
- wherein m is at least 1 and is preferably 3 or 4, a is at least 1 and preferably is 1, p is a number which provides a moiety weight of 400 to 10,000 and is preferably at least 30, R10 is a diradical of a diisocyanate after removal of the isocyanate group, such as the diradical of isophorone diisocyanate, and each E″ is a group represented by:
- A preferred silicone hydrogel material comprises (based on the initial monomer mixture that is copolymerized to form the hydrogel copolymeric material) 5 to 50 percent, preferably 10 to 25, by weight of one or more silicone macromonomers, 5 to 75 percent, preferably 30 to 60 percent, by weight of one or more polysiloxanylalkyl (meth)acrylic monomers, and 10 to 50 percent, preferably 20 to 40 percent, by weight of a hydrophilic monomer. In general, the silicone macromonomer is a poly(organosiloxane) capped with an unsaturated group at two or more ends of the molecule. In addition to the end groups in the above structural formulas, U.S. Pat. No. 4,153,641 to Deichert et al. discloses additional unsaturated groups, including acryloxy or methacryloxy. Fumarate-containing materials such as those taught in U.S. Pat. Nos. 5,512,205; 5,449,729; and 5,310,779 to Lai are also useful substrates in accordance with the invention. Preferably, the silane macromonomer is a silicone-containing vinyl carbonate or vinyl carbamate or a polyurethane-polysiloxane having one or more hard-soft-hard blocks and end-capped with a hydrophilic monomer.
- Specific examples of contact lens materials for which the present invention is useful are taught in U.S. Pat. No. 6,891,010 (Kunzler et al.); U.S. Pat. No. 5,908,906 (Kunzler et al.); U.S. Pat. No. 5,714,557 (Kunzler et al.); U.S. Pat. No. 5,710,302 (Kunzler et al.); U.S. Pat. No. 5,708,094 (Lai et al.); U.S. Pat. No. 5,616,757 (Bambury et al.); U.S. Pat. No. 5,610,252 (Bambury et al.); U.S. Pat. No. 5,512,205 (Lai); U.S. Pat. No. 5,449,729 (Lai); U.S. Pat. No. 5,387,662 (Kunzler et al.); U.S. Pat. No. 5,310,779 (Lai); and U.S. Pat. No. 5,260,000 (Nandu et al.), the disclosures of which are incorporated herein by reference.
- Generally, the monomer mixtures may be charged to a mold, and then subjected to heat and/or light radiation, such as UV radiation, to effect curing, or free radical polymerization, of the monomer mixture in the mold. Various processes are known for curing a monomeric mixture in the production of contact lenses or other biomedical devices, including spincasting and static casting. Additionally, the monomer mixtures may be cast in the shape of rods or buttons, which are then lathe cut into a desired shape, for example, into a lens-shaped article.
- Following casting of the lens, the article may be extracted to remove undesired extractables from the device. For example, in the case of contact lenses made from a silicone hydrogel copolymer, extractables include any remaining diluent, unreacted monomers, and oligomers formed from side reactions of the monomers.
- Then, the lens is hydrated, either as part of the extraction process, or in a separate subsequent operation. For example, if an organic solvent is used to extract the lens, then the lens is hydrated to replace the organic solvent with water or aqueous solution. Hydration may be performed while the lens is held in its package, or prior to placing the lens in the package. In any event, ultimately, the lens is contained in the package with an aqueous packaging solution.
- This invention recognized the desirability of effecting attachment of the treatment agent to the lens surfaces while the lens is contained in a package, in contrast to effecting attachment prior to placing the lens in the package. This offers a reduction in material handling steps and the accompanying costs associated therewith. Also, if the solution containing the treatment agent is ophthalmically compatible, this solution may serve as the final packaging solution, and the heat sterilization of the package and its contents can serve to effect chemical attachment of the treatment agent to the lens surfaces.
- As used herein, “attachment” of the treatment agent to the lens surface, and like terms, denotes that the treatment agent is substantially adhered to the lens surfaces. Thus, after a single rinsing with water of the lens surfaces with the treatment agent attached thereto, at least 50% of the treatment agent will remain adhered to the lens surfaces.
- It is preferred that the surface treatment agent is attached to the lens surfaces by at least one of covalent bonding, ionic attachment, and hydrogen bonding. Covalent bonding denotes that a chemical reaction occurs between the treatment agent and the lens surface, so that covalent bonds are formed therebetween. Ionic attachment denotes that the lens surfaces are ionically charged, and the organic surface treatment agent contains moieties with an opposite, ionic charge. As an example the lens surfaces may be anionic charged, and the treatment agent may be cationic or zwitterionic, that interacts with the anionic lens surface.
- A wide variety of organic surface treatment agents may be employed, including treatment agents known in the art.
- As a first example, this invention is applicable for the organic surface treatment agents described in U.S. Pat. No. 7,083,646, the entire disclosure of which is incorporated herein by reference. Generally, this method involves surface modification of medical devices, particularly ophthalmic lenses, with one or more reactive, hydrophilic polymers as the surface treatment agent. The reactive, hydrophilic polymers are copolymers of at least one hydrophilic monomer and at least one monomer that contains reactive chemical functionality. The hydrophilic monomers can be aprotic types such as N,N-dimethylacrylamide and N-vinylpyrrolidone, or protic types such as methacrylic acid and 2-hydroxyethyl methacrylate. The monomer containing reactive chemical functionality can be an epoxide-containing monomer, such as glycidyl methacrylate. The hydrophilic monomer and the monomer containing reactive chemical functionality are copolymerized at a desired molar ratio thereof. The hydrophilic monomer serves to render the resultant copolymer hydrophilic. The monomer containing reactive chemical functionality provides a reactive group that can react with the lens surface. In other words, this resultant copolymer contains the reactive chemical functionality that can react with complementary functional groups at or near the lens surface, and form covalent bonds therewith.
- As a second example, this invention is applicable for the organic surface treatment agents described in US 2007/0122540, the entire disclosure of which is incorporated herein by reference. Generally, this method involves surface modification of medical devices, particularly ophthalmic lenses, where reactive surfactants as the surface treatment agent are covalently bound to the lens surfaces. Preferred reactive surfactants are functionalized poloxamers or functionalized poloxamines having reactive functionality that is complimentary to surface functionality of the ophthalmic lens. Representative functionalized surfactants are those containing epoxide, methacrylate, or isocyanate functionalities, such as represented below.
- For the aforementioned methods where the surface treatment agent is covalently bonded to the lens surface, the reactive groups of the treatment agent are matched with the reactive groups on the lens surface. For example, if the lens surfaces contain carboxylic acid groups, a glycidyl group can be a reactive group of the surface treatment agent. If the lens surfaces contain hydroxy or amino functionality, an isocyanate group or carbonyl chloride group can be a reactive group of the surface treatment agent. A wide variety of suitable combinations of reactive groups will be apparent to those of ordinary skill in the art.
- Examples of suitable lens-forming monomers, providing reactive groups on the lens surface, include those having hydroxy functional groups, such as 2-hydroxyethyl methacrylate, glyceryl methacrylate and 3-hydroxypropyl methacrylamide. Examples of suitable lens-forming monomers providing the lens surfaces with carboxylic acid reactive groups include methacrylic acid, acrylic acid and N-carboxy-β-alanine-N-vinyl ester. Examples of suitable lens-forming monomers providing the lens surface with oxazolinone reactive groups include 2-isopropenyl-4,4-dimethyl-2-oxazolin-5-one, 2-vinyl-4,4-dimethyl-2-oxazolin-5-one, spiro-4′-(2′-isopropenyl-2′-oxazolin-5-one)cyclohexane, spiro-4′-(2′-vinyl-2′-oxazolin-5′-one)cyclohexane and 2-(1-propenyl)-4,4-dimethyl-oxazolin-5-one. Examples of suitable lens-forming monomers providing the lens surface with anhydride functional groups include methacrylic anhydride, maleic anhydride and acrylic anhydride. An example of a suitable lens-forming monomer providing the lens surfaces with epoxide reactive groups includes glycidyl methacrylate.
- As another example, the surface treatment agent may be one as employed in the method described in U.S. Pat. No. 6,428,839 (Kunzler et al.), the entire disclosure of which is incorporated herein by reference. Generally, this method employs poly(acrylic) acid (PAA) surface complexation. Hydrogel contact lens copolymers containing polymerized hydrophilic lens-forming monomers having relatively strong proton donating moieties, for example DMA or NVP, are treated with water-based solutions containing PAA or PAA co-polymers, acting as wetting agents, to render a lubricious, stable, highly wettable PAA-based surface coating. Alternately, other proton-donating wetting agents besides PAA-containing agents may be employed, although generally, coating materials containing carboxylic acid functionality are preferred. Surface treatment agents include poly(vinylpyrrolidinone(VP)-co-acrylic acid(AA)), poly(methylvinylether-alt-maleic acid), poly(acrylic acid-graft-ethyleneoxide), poly(AA-co-methacrylic acid), poly(acrylamide-co-AA), poly(AA-co-maleic), and poly(butadiene-maleic acid). Particularly preferred polymers are characterized by acid contents of at least about 30 mole percent, preferably at least about 40 mole percent. The lens, with its surface in contact with the PAA-containing solution, may be heated by autoclaving, or subjected to microwave radiation, to facilitate attachment of the PAA to the lens surface.
- Other organic surface treatment agents will be apparent to one skilled in the art.
- There are various variations of the sequence of steps employed in the methods of this invention.
- As a first example, after placing the lens and the solution containing the treatment agent in the recess of the package, the recess is sealed with the lidstock without removing the solution. In other words, according to this embodiment, this solution also serves as the final packaging solution, and this solution is ophthalmically compatible. Then, when the package contents are heat sterilized, this heat treatment can serve to effect chemical attachment of the treatment agent to the lens surfaces, if needed. Any excess treatment agent, not attached to the lens surfaces, remains in the final packaging solution.
- As a second example, after placing the lens and the solution containing the treatment agent in the recess of the package, this solution is removed with a portion of the organic treatment agent remaining attached to the lens surface. Any excess treatment agent is removed with the solution. If heat treatment is needed to effect chemical attachment of the treatment agent to the lens surfaces, the heat treatment is performed prior to removing this solution from the package recess. Subsequently, after removing this solution, an aqueous packaging solution is added to the recess, followed by sealing the recess with lidstock and sterilizing the package contents.
- The final packaging solution is an aqueous solution, preferably a saline solution. having a pH value within the range of about 6 to about 8, and preferably about 6.5 to about 7.8. Suitable buffers may be included, such as: phosphate; borate; citrate; carbonate; tris-(hydroxymethyl)aminomethane (TRIS); bis(2-hydroxyethyl)-imino-tris-(hydroxymethyl)aminoalcohol (bis-tris); zwitterionic buffers such as N-[2-Hydroxy-1,1-bis(hydroxymethyl)ethyl]glycine (Tricine)and N-[2-Hydroxy-1,1-bis(hydroxymethyl)ethyl]glycine, MOPS; N-(Carbamoylmethyl)taurine (ACES); amino acids and amino acid derivatives; and mixtures thereof. Generally, buffers will be used in amounts ranging from about 0.05 to about 2.5 percent by weight, and preferably from about 0.1 to about 1.5 percent by weight of the solution. If needed, the solutions of the present invention may be adjusted with tonicity agents, to approximate the osmotic pressure of normal lacrimal fluids, which is equivalent to a 0.9 percent solution of sodium chloride or 2.5 percent of glycerol solution. The solutions are made substantially isotonic with physiological saline used alone or in combination, otherwise if simply blended with sterile water and made hypotonic or made hypertonic the lenses will lose their desirable optical parameters. Correspondingly, excess saline may result in the formation of a hypertonic solution, which will cause stinging, and eye irritation. Examples of suitable tonicity adjusting agents include, but are not limited to, sodium and potassium chloride, dextrose, calcium and magnesium chloride and the like and mixtures thereof. These agents are typically used individually in amounts ranging from about 0.01 to about 2.5% w/v and preferably from about 0.2 to about 1.5% w/v. Preferably, the tonicity agent will be employed in an amount to provide a final osmotic value of at least about 200 mOsm/kg, preferably from about 200 to about 450 mOsm/kg, more preferably from about 250 to about 400 mOsm/kg, and most preferably from about 280 to about 370 mOsm/kg.
- Representative blister packages that may be employed in the method of this invention are illustrated in
FIGS. 1 to 7 . -
FIGS. 1 to 5 illustrate a first embodiment of a blister package.Package 1 includes asubstrate 3 includingrecess 2 formed therein,recess 2 sized and configured to hold anindividual contact lens 5 therein. In the illustrated embodiment,recess 2 is a concave recess with aflat bottom 6. As seen inFIG. 4 , the anterior surface ofcontact lens 5 rests onbottom 6 when the package is oriented as inFIG. 4 . This can lead to non-uniform coating of the contact lens, especially this anterior surface of the lens resting on the recess bottom. These problems were overcome by employing packages with recesses including grooves in the bottoms thereof, to ensure better flow of the solution around the lens surfaces. Accordingly,bottom 6 includes three parallel,longitudinal grooves 7 therein.Grooves 7 preferably have a depth of at least 0.2 mm, more preferably at least 0.4 mm. - After contacting the contact lens with the treatment agent solution,
contact lens 5 is contained inrecess 2 and immersed in solution.Recess 2 is then sealed with lidstock, i.e., by sealing the lidstock with heat to raisedsurface 9. The package and its contents may now be sterilized, preferably by heat sterilization such as autoclaving.Recess 2 has a depth greater than a height of the lens, and a sufficiently large volume, that the lens is completely immersed in the final packaging solution. -
FIG. 6 illustrates a second embodiment of a blister package.Package 11 includes asubstrate 13 includingrecess 12 formed therein. In this illustrated embodiment,recess 12 is a concave recess, i.e., a rounded bowl shape. The bottom includes three interconnected,longitudinal grooves 17 therein. -
FIG. 7 illustrates a third embodiment of a blister package.Package 21 includes asubstrate 23 includingrecess 22 formed therein. In this illustrated embodiment,recess 22 has a somewhat oval-shaped flat bottom, and the bottom includes fourlongitudinal grooves 27 therein. - Once the package recess contains the contact lens immersed in the final packaging solution, the recess is then sealed with lidstock, i.e., by sealing the lidstock with heat. As an example, the lidstock is sealed to raised
surface 9 inFIGS. 1 to 5 . The package and its contents may now be sterilized, preferably by heat sterilization such as autoclaving. The recess has a depth greater than a height of the lens, and a sufficiently large volume, that the lens is completely immersed in the final packaging solution. Preferably, the recess is filed with at least 0.5 ml of final packaging solution, more preferably at least 1 ml, and most preferably at least 1.2 ml. - While there is shown and described herein certain specific structures and compositions and method steps of the present invention, it will be apparent to those skilled in the art that various modifications may be made without departing from the spirit and scope of the underlying inventive concept and that the same is not limited to particular structures herein shown and described except insofar as indicated by the scope of the appended claims.
Claims (16)
1. A method of treating an ophthalmic lens in a package, comprising:
placing the lens and an aqueous solution in a recess of package, the solution comprising an organic surface treatment agent that attaches to anterior and posterior surfaces of the lens; and
sealing the recess of the package with lidstock and sterilizing the package contents;
wherein a bottom of the recess includes grooves.
2. The method of claim 1 , wherein the surface treatment agent attaches to the lens surfaces by at least one of covalent bonding, ionic attachment, and hydrogen bonding.
3. The method of claim 1 , wherein the surface treatment agent attaches to the lens surfaces by covalent bonding.
4. The method of claim 1 , wherein the lens surfaces are ionically charged, and the surface treatment agent has an opposite, ionic charge.
5. The method of claim 1 , wherein the package contents are sterilized by autoclaving, and autoclaving effects attachment of the surface treatment agent to the lens surfaces.
6. The method of claim 1 , wherein attachment of the surface treatment agent is effected by heating the solution while in contact with the lens surfaces in the recess.
7. The method of claim 1 , wherein the anterior surface of the lens contacts a portion of the recess bottom containing the grooves.
8. The method of claim 1 , wherein the grooves have a depth of at least 0.2 mm.
9. The method of claim 8 , wherein the grooves have a depth of at least 0.4 mm.
10. The method of claim 1 , wherein the recess has a depth greater than a height of the lens, and a sufficient volume of solution is placed in the recess so that the lens is completely immersed in the solution.
11. The method of claim 10 , where the volume of the solution is at least 0.5 ml.
12. The method of claim 11 , where the volume of the solution is at least 1 ml.
13. The method of claim 1 , wherein the lens is a hydrogel contact lens.
14. The method of claim 1 , wherein the lens is a silicone hydrogel contact lens.
15. The method of claim 1 , wherein after placing the lens and the solution in the recess of the package, the recess is sealed with the lidstock without removing the solution.
16. The method of claim 1 , wherein after placing the lens and the solution in the recess of the package, the solution is removed with a portion of the surface treatment agent remaining attached to the lens surface, and an aqueous packaging solution is added to the recess prior to sealing the recess with lidstock.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/244,386 US20090145091A1 (en) | 2007-12-11 | 2008-10-02 | Method for treating ophthalmic lenses |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US1285607P | 2007-12-11 | 2007-12-11 | |
| US12/244,386 US20090145091A1 (en) | 2007-12-11 | 2008-10-02 | Method for treating ophthalmic lenses |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20090145091A1 true US20090145091A1 (en) | 2009-06-11 |
Family
ID=40465811
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/244,386 Abandoned US20090145091A1 (en) | 2007-12-11 | 2008-10-02 | Method for treating ophthalmic lenses |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US20090145091A1 (en) |
| WO (1) | WO2009076032A1 (en) |
Cited By (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090145086A1 (en) * | 2007-12-11 | 2009-06-11 | Reynolds Ger M | Method for treating ophthalmic lenses |
| US20110018152A1 (en) * | 2002-06-26 | 2011-01-27 | Peck James M | Contact lens packages |
| US20110062607A1 (en) * | 2009-09-11 | 2011-03-17 | Julie Clements | Methods, devices, and systems for moving wet ophthalmic lenses during their manufacture |
| WO2012016098A1 (en) | 2010-07-30 | 2012-02-02 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| WO2013074535A1 (en) | 2011-11-15 | 2013-05-23 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| US20130341231A1 (en) * | 2010-11-10 | 2013-12-26 | Boehringer Ingelheim Microparts Gmbh | Blister packaging for liquid |
| WO2014093299A1 (en) | 2012-12-11 | 2014-06-19 | Novartis Ag | Method for applying a coating onto a silicone hydrogel lens |
| US20140331602A1 (en) * | 2011-10-18 | 2014-11-13 | Menicon Singapore Pte Ltd. | Systems and methods for multi-stage sealing of contact lens packaging |
| US9005700B2 (en) | 2011-10-12 | 2015-04-14 | Novartis Ag | Method for making UV-absorbing ophthalmic lenses |
| WO2015095157A1 (en) | 2013-12-17 | 2015-06-25 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| US20150223931A1 (en) * | 2013-10-24 | 2015-08-13 | Aaren Scientific, Inc. | Hydrophilic iol packaging system |
| WO2016032926A1 (en) | 2014-08-26 | 2016-03-03 | Novartis Ag | Method for applying stable coating on silicone hydrogel contact lenses |
| US9422447B2 (en) | 2012-06-14 | 2016-08-23 | Novartis Ag | Azetidinium-containing copolymers and uses thereof |
| WO2016145204A1 (en) | 2015-03-11 | 2016-09-15 | University Of Florida Research Foundation, Inc. | Mesh size control of lubrication in gemini hydrogels |
| US20160278915A1 (en) * | 2013-10-24 | 2016-09-29 | Aaren Scientific Inc. | Hydrophilic iol packaging system |
| WO2017037610A1 (en) | 2015-09-04 | 2017-03-09 | Novartis Ag | Method for producing contact lenses with durable lubricious coatings thereon |
| US9655423B1 (en) * | 2016-04-28 | 2017-05-23 | Hon Hai Precision Industry Co., Ltd. | Contact lens package |
| WO2017093834A1 (en) | 2015-12-03 | 2017-06-08 | Novartis Ag | Contact lens packaging solutions |
| WO2017103793A1 (en) | 2015-12-15 | 2017-06-22 | Novartis Ag | Method for applying stable coating on silicone hydrogel contact lenses |
| US9720138B2 (en) | 2014-08-26 | 2017-08-01 | Novartis Ag | Poly(oxazoline-co-ethyleneimine)-epichlorohydrin copolymers and uses thereof |
| WO2018078598A1 (en) | 2016-10-31 | 2018-05-03 | Chnovartis Ag | Method for producing surface coated contact lenses with wearing comfort |
| US20180134475A1 (en) * | 2012-10-18 | 2018-05-17 | Menicon Singapore Pte Ltd. | Systems and Methods for Multi-Stage Sealing of Contact Lens Packaging |
| US10081142B2 (en) | 2015-12-15 | 2018-09-25 | Novartis Ag | Method for producing contact lenses with a lubricious surface |
| WO2018224975A1 (en) | 2017-06-07 | 2018-12-13 | Novartis Ag | Silicone hydrogel contact lenses |
| US10338408B2 (en) | 2012-12-17 | 2019-07-02 | Novartis Ag | Method for making improved UV-absorbing ophthalmic lenses |
| USD880310S1 (en) * | 2018-05-04 | 2020-04-07 | Hope Vision Co., Ltd. | Packaging case for contact lens |
| US10809181B2 (en) | 2017-08-24 | 2020-10-20 | Alcon Inc. | Method and apparatus for determining a coefficient of friction at a test site on a surface of a contact lens |
| US10830923B2 (en) | 2017-12-13 | 2020-11-10 | Alcon Inc. | Method for producing MPS-compatible water gradient contact lenses |
| US10875967B2 (en) | 2017-06-07 | 2020-12-29 | Alcon Inc. | Silicone hydrogel contact lenses |
| WO2021090169A1 (en) | 2019-11-04 | 2021-05-14 | Alcon Inc. | Contact lenses with surfaces having different softness |
| WO2021124099A1 (en) | 2019-12-16 | 2021-06-24 | Alcon Inc. | Wettable silicone hydrogel contact lenses |
| WO2022023966A1 (en) | 2020-07-28 | 2022-02-03 | Alcon Inc. | Contact lenses with softer lens surfaces |
| WO2022224169A1 (en) | 2021-04-22 | 2022-10-27 | Alcon Inc. | Method for applying a coating onto a non-silicone hydrogel lens |
| WO2024038390A1 (en) | 2022-08-17 | 2024-02-22 | Alcon Inc. | A contact lens with a hydrogel coating thereon |
Citations (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4136250A (en) * | 1977-07-20 | 1979-01-23 | Ciba-Geigy Corporation | Polysiloxane hydrogels |
| US4153641A (en) * | 1977-07-25 | 1979-05-08 | Bausch & Lomb Incorporated | Polysiloxane composition and contact lens |
| US4321261A (en) * | 1978-01-05 | 1982-03-23 | Polymer Technology Corporation | Ionic ophthalmic solutions |
| US4740533A (en) * | 1987-07-28 | 1988-04-26 | Ciba-Geigy Corporation | Wettable, flexible, oxygen permeable, substantially non-swellable contact lens containing block copolymer polysiloxane-polyoxyalkylene backbone units, and use thereof |
| US4910277A (en) * | 1988-02-09 | 1990-03-20 | Bambury Ronald E | Hydrophilic oxygen permeable polymers |
| US5034461A (en) * | 1989-06-07 | 1991-07-23 | Bausch & Lomb Incorporated | Novel prepolymers useful in biomedical devices |
| US5070215A (en) * | 1989-05-02 | 1991-12-03 | Bausch & Lomb Incorporated | Novel vinyl carbonate and vinyl carbamate contact lens material monomers |
| US5143660A (en) * | 1988-11-02 | 1992-09-01 | National Research Development Corporation | Method of casting a contact lens |
| US5145643A (en) * | 1990-01-05 | 1992-09-08 | Allergan, Inc. | Nonoxidative ophthalmic compositions and methods for preserving and using same |
| US5192316A (en) * | 1988-02-16 | 1993-03-09 | Allergan, Inc. | Ocular device |
| US5260000A (en) * | 1992-08-03 | 1993-11-09 | Bausch & Lomb Incorporated | Process for making silicone containing hydrogel lenses |
| US5310779A (en) * | 1991-11-05 | 1994-05-10 | Bausch & Lomb Incorporated | UV curable crosslinking agents useful in copolymerization |
| US5358995A (en) * | 1992-05-15 | 1994-10-25 | Bausch & Lomb Incorporated | Surface wettable silicone hydrogels |
| US5387662A (en) * | 1993-02-12 | 1995-02-07 | Bausch & Lomb Incorporated | Fluorosilicone hydrogels |
| US5451237A (en) * | 1993-11-10 | 1995-09-19 | Vehige; Joseph G. | Compositions and methods for inhibiting and reducing lysozyme deposition on hydrophilic contact lenses using biocompatible colored compounds |
| US5467868A (en) * | 1992-12-21 | 1995-11-21 | Johnson & Johnson Vision Products, Inc. | Ophthalmic lens package |
| US5580392A (en) * | 1994-04-05 | 1996-12-03 | Allergan | Contact lens cleaning compositions with particles of variable hardness and processes of use |
| US5616757A (en) * | 1993-04-08 | 1997-04-01 | Bausch & Lomb Incorporated | Organosilicon-containing materials useful for biomedical devices |
| US5708094A (en) * | 1996-12-17 | 1998-01-13 | Bausch & Lomb Incorporated | Polybutadiene-based compositions for contact lenses |
| US5710302A (en) * | 1995-12-07 | 1998-01-20 | Bausch & Lomb Incorporated | Monomeric units useful for reducing the modules of silicone hydrogels |
| US5714557A (en) * | 1995-12-07 | 1998-02-03 | Bausch & Lomb Incorporated | Monomeric units useful for reducing the modulus of low water polymeric silicone compositions |
| US5722536A (en) * | 1996-02-08 | 1998-03-03 | Bausch & Lomb Incorporated | Disposable contact lens package with snap-together feature |
| US5842325A (en) * | 1997-12-11 | 1998-12-01 | Bausch & Lomb Incorporated | Method for labeling packages |
| US5958194A (en) * | 1997-09-18 | 1999-09-28 | Glazier; Alan N. | Gas permeable elastomer contact lens bonded with titanium and/or oxides thereof |
| US6072172A (en) * | 1997-12-22 | 2000-06-06 | Bausch & Lomb Incorporated | Method and apparatus for detecting packages in carton |
| US20020046958A1 (en) * | 2000-09-01 | 2002-04-25 | Lipscomb Lance Kyle | Textured contact lens package |
| US6428839B1 (en) * | 2000-06-02 | 2002-08-06 | Bausch & Lomb Incorporated | Surface treatment of medical device |
| US6440366B1 (en) * | 1997-06-06 | 2002-08-27 | Bausch & Lomb Incorporated | Contact lens packing solutions |
| US20040004008A1 (en) * | 2002-06-26 | 2004-01-08 | Peck James M. | Contact lens packages |
| US6889825B2 (en) * | 2001-07-17 | 2005-05-10 | Menicon Co., Ltd. | Ophthalmic lens storage container |
| US6891010B2 (en) * | 2001-10-29 | 2005-05-10 | Bausch & Lomb Incorporated | Silicone hydrogels based on vinyl carbonate endcapped fluorinated side chain polysiloxanes |
| US20050126940A1 (en) * | 2002-08-12 | 2005-06-16 | Makoto Nakagawa | Hydrating case for moisture-containing lens and hydrating method using it |
| US6958169B2 (en) * | 2002-12-17 | 2005-10-25 | Bausch & Lomb Incorporated | Surface treatment of medical device |
| US7083646B2 (en) * | 2002-06-28 | 2006-08-01 | Bausch & Lomb Incorporated | Surface modification of functional group-containing intraocular lenses |
| US20070122540A1 (en) * | 2005-11-29 | 2007-05-31 | Bausch & Lomb Incorporated | Coatings on ophthalmic lenses |
| US20070149428A1 (en) * | 2005-12-14 | 2007-06-28 | Bausch & Lomb Incorporated | Method of Packaging a Lens |
| US20080110770A1 (en) * | 2006-11-10 | 2008-05-15 | Bausch & Lomb Incorporated | Packaging solutions |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7086526B2 (en) * | 2001-08-17 | 2006-08-08 | Clearlab International Pte Ltd. | Packaging for disposable soft contact lenses |
| AR055909A1 (en) * | 2005-04-15 | 2007-09-12 | Johnson & Johnson Vision Care | METHOD AND APPLIANCES FOR SEALING OPTICAL LENS PACKS |
-
2008
- 2008-10-02 US US12/244,386 patent/US20090145091A1/en not_active Abandoned
- 2008-11-21 WO PCT/US2008/084259 patent/WO2009076032A1/en active Application Filing
Patent Citations (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4136250A (en) * | 1977-07-20 | 1979-01-23 | Ciba-Geigy Corporation | Polysiloxane hydrogels |
| US4153641A (en) * | 1977-07-25 | 1979-05-08 | Bausch & Lomb Incorporated | Polysiloxane composition and contact lens |
| US4321261A (en) * | 1978-01-05 | 1982-03-23 | Polymer Technology Corporation | Ionic ophthalmic solutions |
| US4740533A (en) * | 1987-07-28 | 1988-04-26 | Ciba-Geigy Corporation | Wettable, flexible, oxygen permeable, substantially non-swellable contact lens containing block copolymer polysiloxane-polyoxyalkylene backbone units, and use thereof |
| US4910277A (en) * | 1988-02-09 | 1990-03-20 | Bambury Ronald E | Hydrophilic oxygen permeable polymers |
| US5192316A (en) * | 1988-02-16 | 1993-03-09 | Allergan, Inc. | Ocular device |
| US5143660A (en) * | 1988-11-02 | 1992-09-01 | National Research Development Corporation | Method of casting a contact lens |
| US5070215A (en) * | 1989-05-02 | 1991-12-03 | Bausch & Lomb Incorporated | Novel vinyl carbonate and vinyl carbamate contact lens material monomers |
| US5610252A (en) * | 1989-05-02 | 1997-03-11 | Bausch & Lomb Incorporated | Vinyl carbonate and vinyl carbamate contact lens material monomers |
| US5034461A (en) * | 1989-06-07 | 1991-07-23 | Bausch & Lomb Incorporated | Novel prepolymers useful in biomedical devices |
| US5145643A (en) * | 1990-01-05 | 1992-09-08 | Allergan, Inc. | Nonoxidative ophthalmic compositions and methods for preserving and using same |
| US5310779A (en) * | 1991-11-05 | 1994-05-10 | Bausch & Lomb Incorporated | UV curable crosslinking agents useful in copolymerization |
| US5449729A (en) * | 1991-11-05 | 1995-09-12 | Bausch & Lomb Incorporated | UV curable crosslinking agents useful in copolymerization |
| US5512205A (en) * | 1991-11-05 | 1996-04-30 | Bausch & Lomb Incorporated | UV curable crosslinking agents useful in copolymerization |
| US5358995A (en) * | 1992-05-15 | 1994-10-25 | Bausch & Lomb Incorporated | Surface wettable silicone hydrogels |
| US5260000A (en) * | 1992-08-03 | 1993-11-09 | Bausch & Lomb Incorporated | Process for making silicone containing hydrogel lenses |
| US5467868A (en) * | 1992-12-21 | 1995-11-21 | Johnson & Johnson Vision Products, Inc. | Ophthalmic lens package |
| US5387662A (en) * | 1993-02-12 | 1995-02-07 | Bausch & Lomb Incorporated | Fluorosilicone hydrogels |
| US5616757A (en) * | 1993-04-08 | 1997-04-01 | Bausch & Lomb Incorporated | Organosilicon-containing materials useful for biomedical devices |
| US5451237A (en) * | 1993-11-10 | 1995-09-19 | Vehige; Joseph G. | Compositions and methods for inhibiting and reducing lysozyme deposition on hydrophilic contact lenses using biocompatible colored compounds |
| US5580392A (en) * | 1994-04-05 | 1996-12-03 | Allergan | Contact lens cleaning compositions with particles of variable hardness and processes of use |
| US5710302A (en) * | 1995-12-07 | 1998-01-20 | Bausch & Lomb Incorporated | Monomeric units useful for reducing the modules of silicone hydrogels |
| US5714557A (en) * | 1995-12-07 | 1998-02-03 | Bausch & Lomb Incorporated | Monomeric units useful for reducing the modulus of low water polymeric silicone compositions |
| US5908906A (en) * | 1995-12-07 | 1999-06-01 | Bausch & Lomb Incorporated | Monomeric units useful for reducing the modulus of silicone hydrogels |
| US5722536A (en) * | 1996-02-08 | 1998-03-03 | Bausch & Lomb Incorporated | Disposable contact lens package with snap-together feature |
| US5708094A (en) * | 1996-12-17 | 1998-01-13 | Bausch & Lomb Incorporated | Polybutadiene-based compositions for contact lenses |
| US6440366B1 (en) * | 1997-06-06 | 2002-08-27 | Bausch & Lomb Incorporated | Contact lens packing solutions |
| US20030109390A1 (en) * | 1997-06-06 | 2003-06-12 | Bausch & Lomb Incorporated | Contact lens packing solutions |
| US5958194A (en) * | 1997-09-18 | 1999-09-28 | Glazier; Alan N. | Gas permeable elastomer contact lens bonded with titanium and/or oxides thereof |
| US5842325A (en) * | 1997-12-11 | 1998-12-01 | Bausch & Lomb Incorporated | Method for labeling packages |
| US6072172A (en) * | 1997-12-22 | 2000-06-06 | Bausch & Lomb Incorporated | Method and apparatus for detecting packages in carton |
| US6428839B1 (en) * | 2000-06-02 | 2002-08-06 | Bausch & Lomb Incorporated | Surface treatment of medical device |
| US20020046958A1 (en) * | 2000-09-01 | 2002-04-25 | Lipscomb Lance Kyle | Textured contact lens package |
| US6889825B2 (en) * | 2001-07-17 | 2005-05-10 | Menicon Co., Ltd. | Ophthalmic lens storage container |
| US6891010B2 (en) * | 2001-10-29 | 2005-05-10 | Bausch & Lomb Incorporated | Silicone hydrogels based on vinyl carbonate endcapped fluorinated side chain polysiloxanes |
| US20040004008A1 (en) * | 2002-06-26 | 2004-01-08 | Peck James M. | Contact lens packages |
| US20040031701A1 (en) * | 2002-06-26 | 2004-02-19 | Peck James M. | Contact lens packages |
| US7083646B2 (en) * | 2002-06-28 | 2006-08-01 | Bausch & Lomb Incorporated | Surface modification of functional group-containing intraocular lenses |
| US20050126940A1 (en) * | 2002-08-12 | 2005-06-16 | Makoto Nakagawa | Hydrating case for moisture-containing lens and hydrating method using it |
| US6958169B2 (en) * | 2002-12-17 | 2005-10-25 | Bausch & Lomb Incorporated | Surface treatment of medical device |
| US20070122540A1 (en) * | 2005-11-29 | 2007-05-31 | Bausch & Lomb Incorporated | Coatings on ophthalmic lenses |
| US20070149428A1 (en) * | 2005-12-14 | 2007-06-28 | Bausch & Lomb Incorporated | Method of Packaging a Lens |
| US20080110770A1 (en) * | 2006-11-10 | 2008-05-15 | Bausch & Lomb Incorporated | Packaging solutions |
Cited By (69)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110018152A1 (en) * | 2002-06-26 | 2011-01-27 | Peck James M | Contact lens packages |
| US9585450B2 (en) | 2002-06-26 | 2017-03-07 | Johnson & Johnson Vision Care, Inc. | Contact lens packages |
| US20090145086A1 (en) * | 2007-12-11 | 2009-06-11 | Reynolds Ger M | Method for treating ophthalmic lenses |
| US9296160B2 (en) * | 2009-09-11 | 2016-03-29 | Coopervision International Holding Company, Lp | Method for moving wet ophthalmic lenses during their manufacture |
| US20110062607A1 (en) * | 2009-09-11 | 2011-03-17 | Julie Clements | Methods, devices, and systems for moving wet ophthalmic lenses during their manufacture |
| US10953615B2 (en) | 2009-09-11 | 2021-03-23 | Coopervision International Limited | Methods and devices for moving wet ophthalmic lenses during their manufacture |
| US9816009B2 (en) | 2010-07-30 | 2017-11-14 | Novartis Ag | Silicone hydrogel lenses with water-rich surfaces |
| US9738813B2 (en) | 2010-07-30 | 2017-08-22 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| US8529057B2 (en) | 2010-07-30 | 2013-09-10 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| US8480227B2 (en) | 2010-07-30 | 2013-07-09 | Novartis Ag | Silicone hydrogel lenses with water-rich surfaces |
| US8939577B2 (en) | 2010-07-30 | 2015-01-27 | Novartis Ag | Silicone hydrogel lenses with water-rich surfaces |
| US8944592B2 (en) | 2010-07-30 | 2015-02-03 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9507173B2 (en) | 2010-07-30 | 2016-11-29 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9411171B2 (en) | 2010-07-30 | 2016-08-09 | Novartis Ag | Silicone hydrogel lenses with water-rich surfaces |
| WO2012016098A1 (en) | 2010-07-30 | 2012-02-02 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9239409B2 (en) | 2010-07-30 | 2016-01-19 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9244200B2 (en) | 2010-07-30 | 2016-01-26 | Novartis Ag | Silicone hydrogel lenses with water-rich surfaces |
| US10781340B2 (en) | 2010-07-30 | 2020-09-22 | Alcon Inc. | Silicone hydrogel lenses with water-rich surfaces |
| US20130341231A1 (en) * | 2010-11-10 | 2013-12-26 | Boehringer Ingelheim Microparts Gmbh | Blister packaging for liquid |
| US9005700B2 (en) | 2011-10-12 | 2015-04-14 | Novartis Ag | Method for making UV-absorbing ophthalmic lenses |
| US11077969B2 (en) * | 2011-10-18 | 2021-08-03 | Menicon Singapore Pte Ltd. | Systems and methods for multi-stage sealing of contact lens packaging |
| US20140331602A1 (en) * | 2011-10-18 | 2014-11-13 | Menicon Singapore Pte Ltd. | Systems and methods for multi-stage sealing of contact lens packaging |
| US9505184B2 (en) | 2011-11-15 | 2016-11-29 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| WO2013074535A1 (en) | 2011-11-15 | 2013-05-23 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9422447B2 (en) | 2012-06-14 | 2016-08-23 | Novartis Ag | Azetidinium-containing copolymers and uses thereof |
| US10100219B2 (en) | 2012-06-14 | 2018-10-16 | Novartis Ag | Azetidinium-containing copolymers and uses thereof |
| US20180134475A1 (en) * | 2012-10-18 | 2018-05-17 | Menicon Singapore Pte Ltd. | Systems and Methods for Multi-Stage Sealing of Contact Lens Packaging |
| US9575332B2 (en) | 2012-12-11 | 2017-02-21 | Novartis Ag | Method for applying a coating onto a silicone hydrogel lens |
| WO2014093299A1 (en) | 2012-12-11 | 2014-06-19 | Novartis Ag | Method for applying a coating onto a silicone hydrogel lens |
| US10338408B2 (en) | 2012-12-17 | 2019-07-02 | Novartis Ag | Method for making improved UV-absorbing ophthalmic lenses |
| US9795474B2 (en) * | 2013-10-24 | 2017-10-24 | Carl Zeiss Meditec Ag | Hydrophilic IOL packaging system |
| US10299914B2 (en) * | 2013-10-24 | 2019-05-28 | Carl Zeiss Meditec Production, LLC | Hydrophilic IOL packaging system |
| US20150223931A1 (en) * | 2013-10-24 | 2015-08-13 | Aaren Scientific, Inc. | Hydrophilic iol packaging system |
| US20160278915A1 (en) * | 2013-10-24 | 2016-09-29 | Aaren Scientific Inc. | Hydrophilic iol packaging system |
| WO2015095157A1 (en) | 2013-12-17 | 2015-06-25 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9708087B2 (en) | 2013-12-17 | 2017-07-18 | Novartis Ag | Silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9720138B2 (en) | 2014-08-26 | 2017-08-01 | Novartis Ag | Poly(oxazoline-co-ethyleneimine)-epichlorohydrin copolymers and uses thereof |
| US11002884B2 (en) | 2014-08-26 | 2021-05-11 | Alcon Inc. | Method for applying stable coating on silicone hydrogel contact lenses |
| WO2016032926A1 (en) | 2014-08-26 | 2016-03-03 | Novartis Ag | Method for applying stable coating on silicone hydrogel contact lenses |
| US10723842B2 (en) | 2015-03-11 | 2020-07-28 | University Of Florida Research Foundation, Inc. | Mesh size control of lubrication in gemini hydrogels |
| WO2016145204A1 (en) | 2015-03-11 | 2016-09-15 | University Of Florida Research Foundation, Inc. | Mesh size control of lubrication in gemini hydrogels |
| US9810812B2 (en) | 2015-09-04 | 2017-11-07 | Novartis Ag | Method for producing contact lenses with durable lubricious coatings thereon |
| WO2017037610A1 (en) | 2015-09-04 | 2017-03-09 | Novartis Ag | Method for producing contact lenses with durable lubricious coatings thereon |
| WO2017093834A1 (en) | 2015-12-03 | 2017-06-08 | Novartis Ag | Contact lens packaging solutions |
| US9829723B2 (en) | 2015-12-03 | 2017-11-28 | Novartis Ag | Contact lens packaging solutions |
| EP3543004A1 (en) | 2015-12-03 | 2019-09-25 | Novartis AG | Contact lens packaging solutions |
| US10081142B2 (en) | 2015-12-15 | 2018-09-25 | Novartis Ag | Method for producing contact lenses with a lubricious surface |
| WO2017103793A1 (en) | 2015-12-15 | 2017-06-22 | Novartis Ag | Method for applying stable coating on silicone hydrogel contact lenses |
| US10449740B2 (en) | 2015-12-15 | 2019-10-22 | Novartis Ag | Method for applying stable coating on silicone hydrogel contact lenses |
| US9655423B1 (en) * | 2016-04-28 | 2017-05-23 | Hon Hai Precision Industry Co., Ltd. | Contact lens package |
| US10718960B2 (en) | 2016-10-31 | 2020-07-21 | Alcon Inc. | Method for producing contact lenses with wearing comfort |
| WO2018078598A1 (en) | 2016-10-31 | 2018-05-03 | Chnovartis Ag | Method for producing surface coated contact lenses with wearing comfort |
| WO2018224975A1 (en) | 2017-06-07 | 2018-12-13 | Novartis Ag | Silicone hydrogel contact lenses |
| EP4235274A2 (en) | 2017-06-07 | 2023-08-30 | Alcon Inc. | Silicone hydrogel contact lenses |
| US10866344B2 (en) | 2017-06-07 | 2020-12-15 | Alcon Inc. | Silicone hydrogel contact lenses |
| US10875967B2 (en) | 2017-06-07 | 2020-12-29 | Alcon Inc. | Silicone hydrogel contact lenses |
| EP3928967A1 (en) | 2017-06-07 | 2021-12-29 | Alcon Inc. | Silicone hydrogel contact lenses |
| US10809181B2 (en) | 2017-08-24 | 2020-10-20 | Alcon Inc. | Method and apparatus for determining a coefficient of friction at a test site on a surface of a contact lens |
| US10830923B2 (en) | 2017-12-13 | 2020-11-10 | Alcon Inc. | Method for producing MPS-compatible water gradient contact lenses |
| US11029446B2 (en) | 2017-12-13 | 2021-06-08 | Alcon Inc. | Method for producing MPS-compatible water gradient contact lenses |
| US11029447B2 (en) | 2017-12-13 | 2021-06-08 | Alcon Inc. | Method for producing MPS-compatible water gradient contact lenses |
| US11256003B2 (en) | 2017-12-13 | 2022-02-22 | Alcon Inc. | Weekly and monthly disposable water gradient contact lenses |
| USD880310S1 (en) * | 2018-05-04 | 2020-04-07 | Hope Vision Co., Ltd. | Packaging case for contact lens |
| WO2021090169A1 (en) | 2019-11-04 | 2021-05-14 | Alcon Inc. | Contact lenses with surfaces having different softness |
| WO2021124099A1 (en) | 2019-12-16 | 2021-06-24 | Alcon Inc. | Wettable silicone hydrogel contact lenses |
| US11513257B2 (en) | 2019-12-16 | 2022-11-29 | Alcon Inc. | Wettable silicone hydrogel contact lenses |
| WO2022023966A1 (en) | 2020-07-28 | 2022-02-03 | Alcon Inc. | Contact lenses with softer lens surfaces |
| WO2022224169A1 (en) | 2021-04-22 | 2022-10-27 | Alcon Inc. | Method for applying a coating onto a non-silicone hydrogel lens |
| WO2024038390A1 (en) | 2022-08-17 | 2024-02-22 | Alcon Inc. | A contact lens with a hydrogel coating thereon |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2009076032A1 (en) | 2009-06-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20090145091A1 (en) | Method for treating ophthalmic lenses | |
| US20090145086A1 (en) | Method for treating ophthalmic lenses | |
| US9910190B2 (en) | Poly(oxazoline-co-ethyleneimine)-epichlorohydrin copolymers and uses thereof | |
| JP4712265B2 (en) | contact lens | |
| US9527617B2 (en) | Packaging solutions | |
| JP5689418B2 (en) | Ionic silicone hydrogels with improved hydrolytic stability | |
| US7837934B2 (en) | Packaging solutions | |
| US7919136B2 (en) | Surface treatment of biomedical devices | |
| JP6165714B2 (en) | Medical device having uniform charge density and method of manufacturing the same | |
| TWI720509B (en) | Silicone hydrogel contact lenses | |
| ES2372010T3 (en) | PACKING SOLUTIONS. | |
| US20070149428A1 (en) | Method of Packaging a Lens | |
| US20130031873A1 (en) | Wettable Silicone Hydrogel Contact Lenses And Related Compositions And Methods | |
| US20100149482A1 (en) | Contact lens | |
| US20090108479A1 (en) | Method for Making Biomedical Devices | |
| US20140329859A1 (en) | Ionic silicone hydrogels comprising pharmaceutical and/or neuticeutical components and having improved hydrolytic stability | |
| US20090142485A1 (en) | Process for Making Biomedical Devices | |
| US20080151181A1 (en) | Coatings and Solutions for Contact Lenses | |
| US20100168356A1 (en) | Biomedical Devices | |
| US20090173643A1 (en) | Packaging Solutions | |
| WO2009094368A1 (en) | Contact lens | |
| US7884141B2 (en) | Biomedical devices | |
| EP4165446B1 (en) | Imidazolium zwitterion polymerizable compounds and ophthalmic devices incorporating them | |
| US20210388142A1 (en) | Amino acid-based polymerizable compounds and ophthalmic devices prepared therefrom | |
| US20090156745A1 (en) | Surface modified biomedical devices |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |