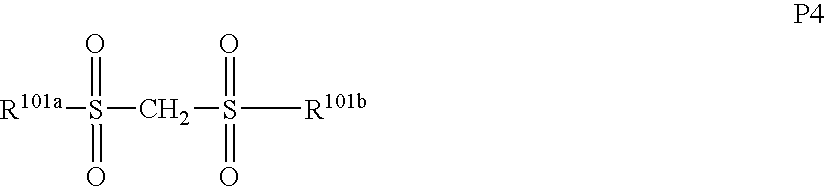US20030198891A1 - Novel ester compounds polymers, resist compositions and patterning process - Google Patents
Novel ester compounds polymers, resist compositions and patterning process Download PDFInfo
- Publication number
- US20030198891A1 US20030198891A1 US10/390,795 US39079503A US2003198891A1 US 20030198891 A1 US20030198891 A1 US 20030198891A1 US 39079503 A US39079503 A US 39079503A US 2003198891 A1 US2003198891 A1 US 2003198891A1
- Authority
- US
- United States
- Prior art keywords
- carbon atoms
- polymer
- groups
- branched
- straight
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 0 C.[1*]C1(C(=O)OC2(C)C=CCCC2)C2CC(C3C4C=CC(C4)C32)C1([2*])[6*] Chemical compound C.[1*]C1(C(=O)OC2(C)C=CCCC2)C2CC(C3C4C=CC(C4)C32)C1([2*])[6*] 0.000 description 26
- HXFIRQHMXJBTRV-UHFFFAOYSA-N CC(C(C)C(O1)=O)C1=O Chemical compound CC(C(C)C(O1)=O)C1=O HXFIRQHMXJBTRV-UHFFFAOYSA-N 0.000 description 2
- NKTCTHZDUUASQR-UHFFFAOYSA-N CC(C)(C)C.CC1(C)C(C)(C)C2(C)C(C)(C)C(C)(C)C1(C)C2(C)C.CC1(C)C=CCCC1.CCC(C)=O Chemical compound CC(C)(C)C.CC1(C)C(C)(C)C2(C)C(C)(C)C(C)(C)C1(C)C2(C)C.CC1(C)C=CCCC1.CCC(C)=O NKTCTHZDUUASQR-UHFFFAOYSA-N 0.000 description 2
- CRMMXYVSCDDAFY-UHFFFAOYSA-N C.CC(=O)OCCCCOC(=O)C1CC2CC1C(C)C2C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 Chemical compound C.CC(=O)OCCCCOC(=O)C1CC2CC1C(C)C2C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 CRMMXYVSCDDAFY-UHFFFAOYSA-N 0.000 description 1
- VIOGZYLVONXPEA-UHFFFAOYSA-N C.CC(=O)OCCCCOC(=O)C1CC2CC1C(C)C2C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 Chemical compound C.CC(=O)OCCCCOC(=O)C1CC2CC1C(C)C2C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 VIOGZYLVONXPEA-UHFFFAOYSA-N 0.000 description 1
- RYRZCUFSYALTGV-UHFFFAOYSA-N C.CC1(C)C=CCCC1 Chemical compound C.CC1(C)C=CCCC1 RYRZCUFSYALTGV-UHFFFAOYSA-N 0.000 description 1
- NWYIJKNBJKMGFM-UHFFFAOYSA-N C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)O)C(C2)C1C.CC1C2CC(C(=O)O)C(C2)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 Chemical compound C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)O)C(C2)C1C.CC1C2CC(C(=O)O)C(C2)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 NWYIJKNBJKMGFM-UHFFFAOYSA-N 0.000 description 1
- IVMPSEVXFRPNQL-UHFFFAOYSA-N C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CC1C2CC(C(=O)OCCO)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 Chemical compound C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CC1C2CC(C(=O)OCCO)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1 IVMPSEVXFRPNQL-UHFFFAOYSA-N 0.000 description 1
- BXKOVYDPRWWWIY-UHFFFAOYSA-N C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1.COCCOCCOC(=O)C1CC2CC1C(C)C2C Chemical compound C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1.COCCOCCOC(=O)C1CC2CC1C(C)C2C BXKOVYDPRWWWIY-UHFFFAOYSA-N 0.000 description 1
- ZHZZQIITSOXBAX-UHFFFAOYSA-N C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OCCO)C(C2)C1C.CCCC(C)CCC(=O)OC(C)(C)C.COC(=O)C1CC2CC1C(C)C2C Chemical compound C.CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OCCO)C(C2)C1C.CCCC(C)CCC(=O)OC(C)(C)C.COC(=O)C1CC2CC1C(C)C2C ZHZZQIITSOXBAX-UHFFFAOYSA-N 0.000 description 1
- SGZBUCRVVUSKOI-UHFFFAOYSA-N C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1.COCCOCCOC(=O)C1CC2CC1C(C)C2C Chemical compound C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CCCC(C)CCC(=O)OC1(C2CCCCC2)CCCC1.COCCOCCOC(=O)C1CC2CC1C(C)C2C SGZBUCRVVUSKOI-UHFFFAOYSA-N 0.000 description 1
- XIYSGWSOAKGGFT-UHFFFAOYSA-N CC(C(=O)O)(C1=CC=C(O)C=C1)C1=CC=C(O)C=C1.CC(C(=O)O)(C1=CC=CC=C1)C1=CC=CC=C1.CC(CCC(=O)O)(C1=CC=C(O)C=C1)C1=CC=C(O)C=C1.CC(CCC(=O)O)(C1=CC=CC=C1)C1=CC=CC=C1.O=C(O)C12CC3CC(CC(C3)C1)C2.O=C(O)C1=CC=CC2=CC=CC=C21.O=C(O)CC12CC3CC(CC(C3)C1)C2.O=C(O)CC1=CC=C(O)C=C1.O=C(O)CC1=CC=CC=C1 Chemical compound CC(C(=O)O)(C1=CC=C(O)C=C1)C1=CC=C(O)C=C1.CC(C(=O)O)(C1=CC=CC=C1)C1=CC=CC=C1.CC(CCC(=O)O)(C1=CC=C(O)C=C1)C1=CC=C(O)C=C1.CC(CCC(=O)O)(C1=CC=CC=C1)C1=CC=CC=C1.O=C(O)C12CC3CC(CC(C3)C1)C2.O=C(O)C1=CC=CC2=CC=CC=C21.O=C(O)CC12CC3CC(CC(C3)C1)C2.O=C(O)CC1=CC=C(O)C=C1.O=C(O)CC1=CC=CC=C1 XIYSGWSOAKGGFT-UHFFFAOYSA-N 0.000 description 1
- LJJJCKVVBLKVCB-UHFFFAOYSA-N CC(C(C)C1C2)C2CC1C(OC(C)(C)C)=O Chemical compound CC(C(C)C1C2)C2CC1C(OC(C)(C)C)=O LJJJCKVVBLKVCB-UHFFFAOYSA-N 0.000 description 1
- LNRZTWZBCPTTHE-UHFFFAOYSA-N CC(C(C)C1C2)C2CC1C(OC1(CCCCC1)C1CCCC1)=O Chemical compound CC(C(C)C1C2)C2CC1C(OC1(CCCCC1)C1CCCC1)=O LNRZTWZBCPTTHE-UHFFFAOYSA-N 0.000 description 1
- SESCSYBWAXMOBX-UHFFFAOYSA-N CC(C)OC1CCCC1.CC(C)OC1CCCCC1.CCC(C)OC.CCCC(C)OC.CCCC(C)OCC.CCCCOCC.CCCOC(C)C.CCCOC(C)CC.CCCOC(C)CCC.CCCOCC.CCOC.CCOC(C)(C)C.CCOC(C)(C)C.CCOC(C)C.CCOC(C)C.CCOC(C)CC.CCOCC.COC(C)(C)C.COC(C)C Chemical compound CC(C)OC1CCCC1.CC(C)OC1CCCCC1.CCC(C)OC.CCCC(C)OC.CCCC(C)OCC.CCCCOCC.CCCOC(C)C.CCCOC(C)CC.CCCOC(C)CCC.CCCOCC.CCOC.CCOC(C)(C)C.CCOC(C)(C)C.CCOC(C)C.CCOC(C)C.CCOC(C)CC.CCOCC.COC(C)(C)C.COC(C)C SESCSYBWAXMOBX-UHFFFAOYSA-N 0.000 description 1
- CJKRPGGHCGFEKB-QOPUZWHJSA-N CC(C)[C@@]1(C)CC2CCC1C2.CC1(C)CC2CC1C1C3CCC(C3)C21.CC1(C)CC2CC1C1CCCC21.CC1(C)CC2CCC1(C)C2(C)C.CC1(C)CC2CCC1C2.CC12CCC(C1)C(C)(C)C2(C)C.CCC(C)[C@@]1(C)CC2CCC1C2.CCCC[C@@]1(C)CC2CCC1C2.CC[C@@]1(C)CC2CC1C1CCCC21.CC[C@@]1(C)CC2CCC1C2 Chemical compound CC(C)[C@@]1(C)CC2CCC1C2.CC1(C)CC2CC1C1C3CCC(C3)C21.CC1(C)CC2CC1C1CCCC21.CC1(C)CC2CCC1(C)C2(C)C.CC1(C)CC2CCC1C2.CC12CCC(C1)C(C)(C)C2(C)C.CCC(C)[C@@]1(C)CC2CCC1C2.CCCC[C@@]1(C)CC2CCC1C2.CC[C@@]1(C)CC2CC1C1CCCC21.CC[C@@]1(C)CC2CCC1C2 CJKRPGGHCGFEKB-QOPUZWHJSA-N 0.000 description 1
- SMEROWZSTRWXGI-UHFFFAOYSA-N CC(CCC(=O)O)C1CCC2C3CCC4CC(O)CCC4(C)C3CCC12C Chemical compound CC(CCC(=O)O)C1CCC2C3CCC4CC(O)CCC4(C)C3CCC12C SMEROWZSTRWXGI-UHFFFAOYSA-N 0.000 description 1
- HPPKHMSHMAHXCC-UHFFFAOYSA-N CC.CC.CC.CC(CC(C1=CC=CC=C1)C1=CC=CC=C1)C1=CC=CC=C1.CC1=CC=C(C(C)(C)C2=CC=C(C)C=C2)C=C1.CC1=CC=C(C2=CC=C(C)C=C2)C=C1.CC1=CC=C(CC2=CC=C(C)C=C2)C=C1.CC1=CC=C(OC2=CC=C(C)C=C2)C=C1.CC1=CC=CC2=CC=CC=C12.CCC(C)(C1=CC=C(C)C=C1)C1=CC=C(C)C=C1.CCCC(C)(C1=CC=C(C)C=C1)C1=CC=C(C)C=C1 Chemical compound CC.CC.CC.CC(CC(C1=CC=CC=C1)C1=CC=CC=C1)C1=CC=CC=C1.CC1=CC=C(C(C)(C)C2=CC=C(C)C=C2)C=C1.CC1=CC=C(C2=CC=C(C)C=C2)C=C1.CC1=CC=C(CC2=CC=C(C)C=C2)C=C1.CC1=CC=C(OC2=CC=C(C)C=C2)C=C1.CC1=CC=CC2=CC=CC=C12.CCC(C)(C1=CC=C(C)C=C1)C1=CC=C(C)C=C1.CCCC(C)(C1=CC=C(C)C=C1)C1=CC=C(C)C=C1 HPPKHMSHMAHXCC-UHFFFAOYSA-N 0.000 description 1
- JCAIJPSRRJQULR-UHFFFAOYSA-N CC.CC1=CC(C(C)CC(C2=CC(C)=C(C)C(C)=C2)C2=CC(C)=C(C)C(C)=C2)=CC(C)=C1C.CC1=CC(C)=C(C2(C)CC(C)(C)C3=CC(C)=C(C)C=C32)C=C1.CC1=CC=C(C)C=C1.CC1=CC=C2CCC(C3=C(C)C=C(C)C=C3)OC2=C1.CCC(C)C1=CC=C(C)C=C1.CCC1=CC=CC(C)=C1 Chemical compound CC.CC1=CC(C(C)CC(C2=CC(C)=C(C)C(C)=C2)C2=CC(C)=C(C)C(C)=C2)=CC(C)=C1C.CC1=CC(C)=C(C2(C)CC(C)(C)C3=CC(C)=C(C)C=C32)C=C1.CC1=CC=C(C)C=C1.CC1=CC=C2CCC(C3=C(C)C=C(C)C=C3)OC2=C1.CCC(C)C1=CC=C(C)C=C1.CCC1=CC=CC(C)=C1 JCAIJPSRRJQULR-UHFFFAOYSA-N 0.000 description 1
- SWMZHLCKZAVGIH-UHFFFAOYSA-N CC.CCC1CC(CC)C(C(=O)OCCOC(C)=O)C1.CCC1CC(CC)C2C3CC(CC3C(=O)OC3(C4CCCCC4)CCCC3)C12 Chemical compound CC.CCC1CC(CC)C(C(=O)OCCOC(C)=O)C1.CCC1CC(CC)C2C3CC(CC3C(=O)OC3(C4CCCCC4)CCCC3)C12 SWMZHLCKZAVGIH-UHFFFAOYSA-N 0.000 description 1
- ILKUZRWHSXAXMF-UHFFFAOYSA-N CC.CCC1CC(CC)C(C(=O)OCCOC)C1.CCC1CC(CC)C2C3CC(CC3C(=O)OC3(C4CCCCC4)CCCC3)C12 Chemical compound CC.CCC1CC(CC)C(C(=O)OCCOC)C1.CCC1CC(CC)C2C3CC(CC3C(=O)OC3(C4CCCCC4)CCCC3)C12 ILKUZRWHSXAXMF-UHFFFAOYSA-N 0.000 description 1
- ALJSBSNAAUSFBW-UHFFFAOYSA-N CC1(C(=O)OC2(C3CCCC3)CCCCC2)CC2C=CC1C2.CC1C2C=CC(C2)C1C(=O)OC1(C2CCCC2)CCCCC1.COC(=O)C1C2C=CC(C2)C1C(=O)OC1(C2CCCC2)CCCCC1.COC(=O)CC1(C(=O)OC2(C3CCCC3)CCCCC2)CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)CCCCC1)C1CC2C=CC1C2 Chemical compound CC1(C(=O)OC2(C3CCCC3)CCCCC2)CC2C=CC1C2.CC1C2C=CC(C2)C1C(=O)OC1(C2CCCC2)CCCCC1.COC(=O)C1C2C=CC(C2)C1C(=O)OC1(C2CCCC2)CCCCC1.COC(=O)CC1(C(=O)OC2(C3CCCC3)CCCCC2)CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)CCCCC1)C1CC2C=CC1C2 ALJSBSNAAUSFBW-UHFFFAOYSA-N 0.000 description 1
- BZSMWNZDMNDACG-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C(C)C2CC1C1C3CC(C(=O)OC4(C5CCCCC5)C=CCC4)C(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CCC1C2 Chemical compound CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C(C)C2CC1C1C3CC(C(=O)OC4(C5CCCCC5)C=CCC4)C(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CCC1C2 BZSMWNZDMNDACG-UHFFFAOYSA-N 0.000 description 1
- BZZQQIJGLDWWRU-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C(C)C2CC1C1C3CC(C(=O)OC4(C5CCCCC5)CCCC4)C(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CCC1C2 Chemical compound CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C(C)C2CC1C1C3CC(C(=O)OC4(C5CCCCC5)CCCC4)C(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CCC1C2 BZZQQIJGLDWWRU-UHFFFAOYSA-N 0.000 description 1
- WYDBUJSVMZPQPE-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CC5CCC4C5)CCCC3)C(C2)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CC5CCC4C5)CCCC3)C(C2)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C WYDBUJSVMZPQPE-UHFFFAOYSA-N 0.000 description 1
- IIFKUEOPEGIEHA-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC(C)(C)C)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC(C)(C)C)C(C2)C1C IIFKUEOPEGIEHA-UHFFFAOYSA-N 0.000 description 1
- VTQNLBTYJPFTCJ-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4C5CC6CCCC4C6C5)CCCC3)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4C5CC6CCCC4C6C5)CCCC3)C(C2)C1C VTQNLBTYJPFTCJ-UHFFFAOYSA-N 0.000 description 1
- XXJRIKBDYFYVFE-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCC4)C=CCCC3)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCC4)C=CCCC3)C(C2)C1C XXJRIKBDYFYVFE-UHFFFAOYSA-N 0.000 description 1
- XEXQLUUALXMCRY-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCC4)CCCCC3)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCC4)CCCCC3)C(C2)C1C XEXQLUUALXMCRY-UHFFFAOYSA-N 0.000 description 1
- AFNQRSRQRJSBRZ-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCC5CCCCC54)CCCC3)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCC5CCCCC54)CCCC3)C(C2)C1C AFNQRSRQRJSBRZ-UHFFFAOYSA-N 0.000 description 1
- JHUSKEMDKWZFHV-UHFFFAOYSA-N CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C Chemical compound CC1C(=O)OC(=O)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C JHUSKEMDKWZFHV-UHFFFAOYSA-N 0.000 description 1
- DNBZYQTXNVKZMQ-UHFFFAOYSA-N CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CC1C2CC(C1C)C1(COC(=O)C1)C2 Chemical compound CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CC1C2CC(C(=O)OC3(C4CCCCC4)CCCC3)C(C2)C1C.CC1C2CC(C1C)C1(COC(=O)C1)C2 DNBZYQTXNVKZMQ-UHFFFAOYSA-N 0.000 description 1
- ZVSYVHOLLLIFFO-UHFFFAOYSA-N CCC1CC(CC)C2C(=O)OCC12.CCC1CC(CC)C2C3CC(CC3C(=O)OC3(C4CCCCC4)CCCC3)C12.O=C1CCCO1 Chemical compound CCC1CC(CC)C2C(=O)OCC12.CCC1CC(CC)C2C3CC(CC3C(=O)OC3(C4CCCCC4)CCCC3)C12.O=C1CCCO1 ZVSYVHOLLLIFFO-UHFFFAOYSA-N 0.000 description 1
- HLPAXFLWACFQKS-UHFFFAOYSA-N CCC1CC2CCCC(C2)C1C1(OC(=O)C2CC3C=CC2C3)CCCC1.O=C(OC1(C2CC3CCC2C3)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC3CCCCC32)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)CCCC1)C1CC2C=CC1C2 Chemical compound CCC1CC2CCCC(C2)C1C1(OC(=O)C2CC3C=CC2C3)CCCC1.O=C(OC1(C2CC3CCC2C3)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC3CCCCC32)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)CCCC1)C1CC2C=CC1C2 HLPAXFLWACFQKS-UHFFFAOYSA-N 0.000 description 1
- LQSQJCHHCWACTG-UHFFFAOYSA-N CCC1CCC(CO)CC1.CCC1COC(=O)O1.CCCCCCCO.CCCCCO.CCCCOCCC.CCCOCCO Chemical compound CCC1CCC(CO)CC1.CCC1COC(=O)O1.CCCCCCCO.CCCCCO.CCCCOCCC.CCCOCCO LQSQJCHHCWACTG-UHFFFAOYSA-N 0.000 description 1
- SKEASRWAJPFADL-AVHMCGKRSA-N CC[C@@]1(OC(=O)C23CC4CC(CC(C4)C2)C3)C[C@H]2CC[C@@H]1C2.O=C(OC1CCCCO1)C1C[C@H]2CC[C@@H]1C2.O=S(=O)([O-])C#C[S+](C#C(F)(F)(F)(F)(F)(F)(F)(F)F)(C1=CC=CC=C1)(C1=CC=CC=C1)C1=CC=CC=C1.[H]C(=O)O[C@H]1C[C@]2([H])[C@@]3(C)CC[C@@H](OC([H])=O)C[C@@]3([H])C[C@@H](OC([H])=O)[C@@]2([H])[C@]2([H])CC[C@H]([C@H](C)CCC(=O)O[C@@]3(CC)C[C@@H]4CC[C@H]3C4)[C@@]12C.[H][C@@]12C[C@H](O)CC[C@]1(C)[C@@]1([H])C[C@H](O)[C@]3(C)[C@@H]([C@H](C)CCC(=O)OC(C)(C)C)CC[C@@]3([H])[C@]1([H])[C@H](O)C2.[O-]C(=S)(OO)[S+](F)(F)(F)(C1=CC=CC=C1)(C1=CC=CC=C1)C1=CC=CC=C1 Chemical compound CC[C@@]1(OC(=O)C23CC4CC(CC(C4)C2)C3)C[C@H]2CC[C@@H]1C2.O=C(OC1CCCCO1)C1C[C@H]2CC[C@@H]1C2.O=S(=O)([O-])C#C[S+](C#C(F)(F)(F)(F)(F)(F)(F)(F)F)(C1=CC=CC=C1)(C1=CC=CC=C1)C1=CC=CC=C1.[H]C(=O)O[C@H]1C[C@]2([H])[C@@]3(C)CC[C@@H](OC([H])=O)C[C@@]3([H])C[C@@H](OC([H])=O)[C@@]2([H])[C@]2([H])CC[C@H]([C@H](C)CCC(=O)O[C@@]3(CC)C[C@@H]4CC[C@H]3C4)[C@@]12C.[H][C@@]12C[C@H](O)CC[C@]1(C)[C@@]1([H])C[C@H](O)[C@]3(C)[C@@H]([C@H](C)CCC(=O)OC(C)(C)C)CC[C@@]3([H])[C@]1([H])[C@H](O)C2.[O-]C(=S)(OO)[S+](F)(F)(F)(C1=CC=CC=C1)(C1=CC=CC=C1)C1=CC=CC=C1 SKEASRWAJPFADL-AVHMCGKRSA-N 0.000 description 1
- GSPNRLCKGGOGJT-TUJRSCDTSA-N O=C(O)C12CC3CC(CC(C3)C1)C2.[H][C@@]12C[C@H](O)CC[C@]1(C)[C@@]1([H])C[C@H](O)[C@]3(C)[C@@H]([C@H](C)CCC(=O)O)CC[C@@]3([H])[C@]1([H])[C@H](O)C2 Chemical compound O=C(O)C12CC3CC(CC(C3)C1)C2.[H][C@@]12C[C@H](O)CC[C@]1(C)[C@@]1([H])C[C@H](O)[C@]3(C)[C@@H]([C@H](C)CCC(=O)O)CC[C@@]3([H])[C@]1([H])[C@H](O)C2 GSPNRLCKGGOGJT-TUJRSCDTSA-N 0.000 description 1
- HSBFMXRBMSIMIG-UHFFFAOYSA-N O=C(OC1(C2CC3CC2C2CCCC32)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CC3CCC2C3)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC2)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC3CCCCC32)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCCC1)C1CC2CC1C1C3C=CC(C3)C21 Chemical compound O=C(OC1(C2CC3CC2C2CCCC32)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CC3CCC2C3)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC2)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC3CCCCC32)CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCCC1)C1CC2CC1C1C3C=CC(C3)C21 HSBFMXRBMSIMIG-UHFFFAOYSA-N 0.000 description 1
- BXCRVWHTRNKJKR-UHFFFAOYSA-N O=C(OC1(C2CCCC2)C=CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC2)CCCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CC1C1C3C=CC(C3)C21 Chemical compound O=C(OC1(C2CCCC2)C=CCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCC2)CCCCC1)C1CC2C=CC1C2.O=C(OC1(C2CCCCC2)C=CCC1)C1CC2CC1C1C3C=CC(C3)C21.O=C(OC1(C2CCCCC2)CCCC1)C1CC2CC1C1C3C=CC(C3)C21 BXCRVWHTRNKJKR-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/013—Esters of alcohols having the esterified hydroxy group bound to a carbon atom of a ring other than a six-membered aromatic ring
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/039—Macromolecular compounds which are photodegradable, e.g. positive electron resists
- G03F7/0392—Macromolecular compounds which are photodegradable, e.g. positive electron resists the macromolecular compound being present in a chemically amplified positive photoresist composition
- G03F7/0397—Macromolecular compounds which are photodegradable, e.g. positive electron resists the macromolecular compound being present in a chemically amplified positive photoresist composition the macromolecular compound having an alicyclic moiety in a side chain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/52—Esters of acyclic unsaturated carboxylic acids having the esterified carboxyl group bound to an acyclic carbon atom
- C07C69/533—Monocarboxylic acid esters having only one carbon-to-carbon double bond
- C07C69/54—Acrylic acid esters; Methacrylic acid esters
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/52—Esters of acyclic unsaturated carboxylic acids having the esterified carboxyl group bound to an acyclic carbon atom
- C07C69/533—Monocarboxylic acid esters having only one carbon-to-carbon double bond
- C07C69/56—Crotonic acid esters; Vinyl acetic acid esters
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/74—Esters of carboxylic acids having an esterified carboxyl group bound to a carbon atom of a ring other than a six-membered aromatic ring
- C07C69/753—Esters of carboxylic acids having an esterified carboxyl group bound to a carbon atom of a ring other than a six-membered aromatic ring of polycyclic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/12—Esters of monohydric alcohols or phenols
- C08F220/16—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms
- C08F220/18—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms with acrylic or methacrylic acids
- C08F220/1811—C10or C11-(Meth)acrylate, e.g. isodecyl (meth)acrylate, isobornyl (meth)acrylate or 2-naphthyl (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F222/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides, or nitriles thereof
- C08F222/04—Anhydrides, e.g. cyclic anhydrides
- C08F222/06—Maleic anhydride
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F232/00—Copolymers of cyclic compounds containing no unsaturated aliphatic radicals in a side chain, and having one or more carbon-to-carbon double bonds in a carbocyclic ring system
- C08F232/08—Copolymers of cyclic compounds containing no unsaturated aliphatic radicals in a side chain, and having one or more carbon-to-carbon double bonds in a carbocyclic ring system having condensed rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F32/00—Homopolymers and copolymers of cyclic compounds having no unsaturated aliphatic radicals in a side chain, and having one or more carbon-to-carbon double bonds in a carbocyclic ring system
- C08F32/08—Homopolymers and copolymers of cyclic compounds having no unsaturated aliphatic radicals in a side chain, and having one or more carbon-to-carbon double bonds in a carbocyclic ring system having two condensed rings
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/039—Macromolecular compounds which are photodegradable, e.g. positive electron resists
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/039—Macromolecular compounds which are photodegradable, e.g. positive electron resists
- G03F7/0392—Macromolecular compounds which are photodegradable, e.g. positive electron resists the macromolecular compound being present in a chemically amplified positive photoresist composition
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/039—Macromolecular compounds which are photodegradable, e.g. positive electron resists
- G03F7/0392—Macromolecular compounds which are photodegradable, e.g. positive electron resists the macromolecular compound being present in a chemically amplified positive photoresist composition
- G03F7/0395—Macromolecular compounds which are photodegradable, e.g. positive electron resists the macromolecular compound being present in a chemically amplified positive photoresist composition the macromolecular compound having a backbone with alicyclic moieties
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/0045—Photosensitive materials with organic non-macromolecular light-sensitive compounds not otherwise provided for, e.g. dissolution inhibitors
Definitions
- the present invention relates to (1) a novel ester compound, (2) a polymer comprising units of the ester compound which is blended as a base resin to formulate a resist composition having a high sensitivity, resolution and etching resistance, and in particular, suitable as micropatterning material for VLSI fabrication, (3) a method for preparing the polymer, (4) a resist composition comprising the polymer, and (5) a patterning process using the resist composition.
- polyhydroxystyrene having a practical level of transmittance and etching resistance is, in fact, a standard base resin.
- polyacrylic or polymethacrylic acid derivatives and polymers comprising aliphatic cyclic compounds in the backbone are under investigation. In either case, the basic concept is that some or all of alkali soluble sites of alkali soluble resin are protected with acid labile or eliminatable groups. The overall performance of resist material is adjusted by a choice from among a variety of acid eliminatable protective groups.
- Exemplary acid eliminatable protective groups include tert-butoxycarbonyl (JP-B 2-27660), tert-butyl (JP-A 62-115440, JP-A 5-80515, and J. Photopolym. Sci. Technol. 7 [3], 507 (1994)), 2-tetrahydropyranyl (JP-A 2-19847, 5-80515 and 5-88367), and 1-ethoxyethyl (JP-A 2-19847 and 4-215661). While it is desired to achieve a finer pattern rule, none of these acid eliminatable protective groups are deemed to exert satisfactory performance.
- tert-butoxycarbonyl and tert-butyl are extremely less reactive with acids so that a substantial quantity of energy radiation must be irradiated to generate a sufficient amount of acid in order to establish a difference in rate of dissolution before and after exposure.
- a photoacid generator of the strong acid type is used, the exposure can be reduced to a relatively low level because reaction can proceed with a small amount of acid generated.
- the deactivation of the generated acid by air-borne basic substances has a relatively large influence, giving rise to such problems as a T-top pattern.
- an object of the present invention is to provide (1) a novel ester compound capable of forming an acid-decomposable polymer, (2) a polymer which is blended as a base resin to formulate a resist composition having a higher sensitivity, resolution and etching resistance than conventional resist compositions, (3) a method for preparing the polymer, (4) a resist composition comprising the polymer as a base resin, and (5) a patterning process using the resist composition.
- a novel ester compound of the following general formula (1) obtained by the method to be described later is useful in preparing an acid-decomposable polymer; that a resist composition comprising as the base resin a novel polymer prepared from the ester compound to a weight average molecular weight of 1,000 to 500,000 has a high sensitivity, resolution and etching resistance; and that this resist composition lends itself to precise micropatterning.
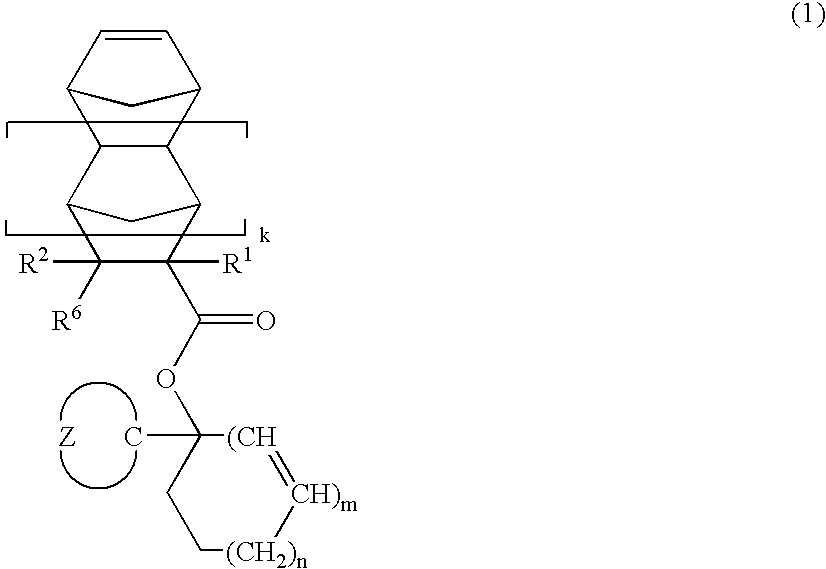
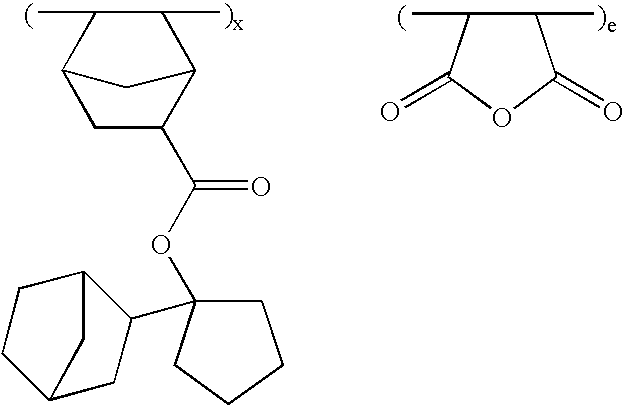
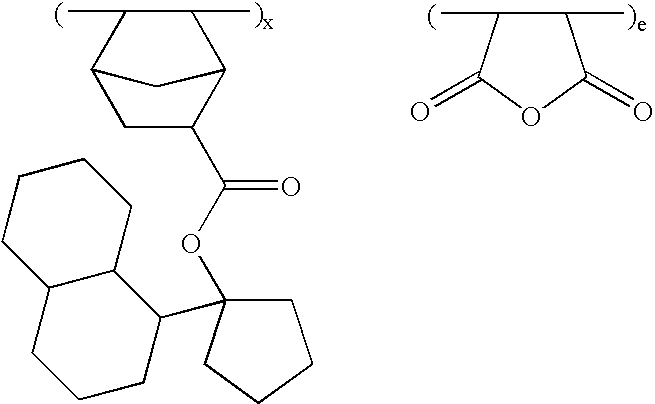
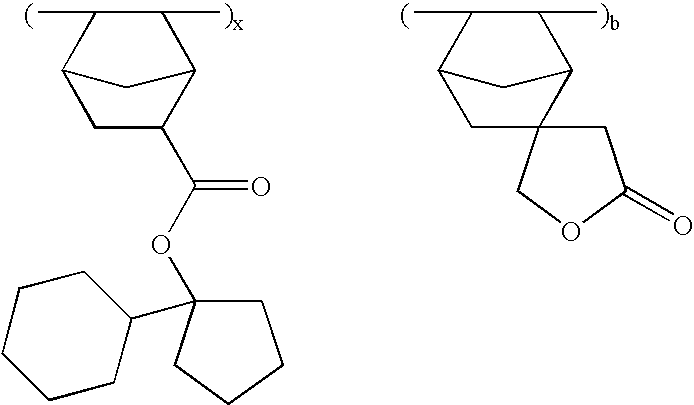
- the invention provides an ester compound of the following general formula (1).
- R 1 is hydrogen, methyl or CH 2 CO 2 R 1
- R 2 is hydrogen, methyl or CO 2 R 3
- R 3 is a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms
- k is 0 or 1
- Z is a divalent hydrocarbon group of 2 to 20 carbon atoms which forms a single ring or bridged ring with the carbon atom and which may contain a hetero atom
- m is 0 or 1
- n is 0, 1, 2 or 3
- 2 m+n is 2 or 3.
- the invention provides a polymer comprising recurring units of the following general formula (1a-1) and/or (1a-2) derived from an ester compound of the general formula (1), and having a weight average molecular weight of 1,000 to 500,000.
- R 1 , R 2 Z, k, m and n are as defined above.
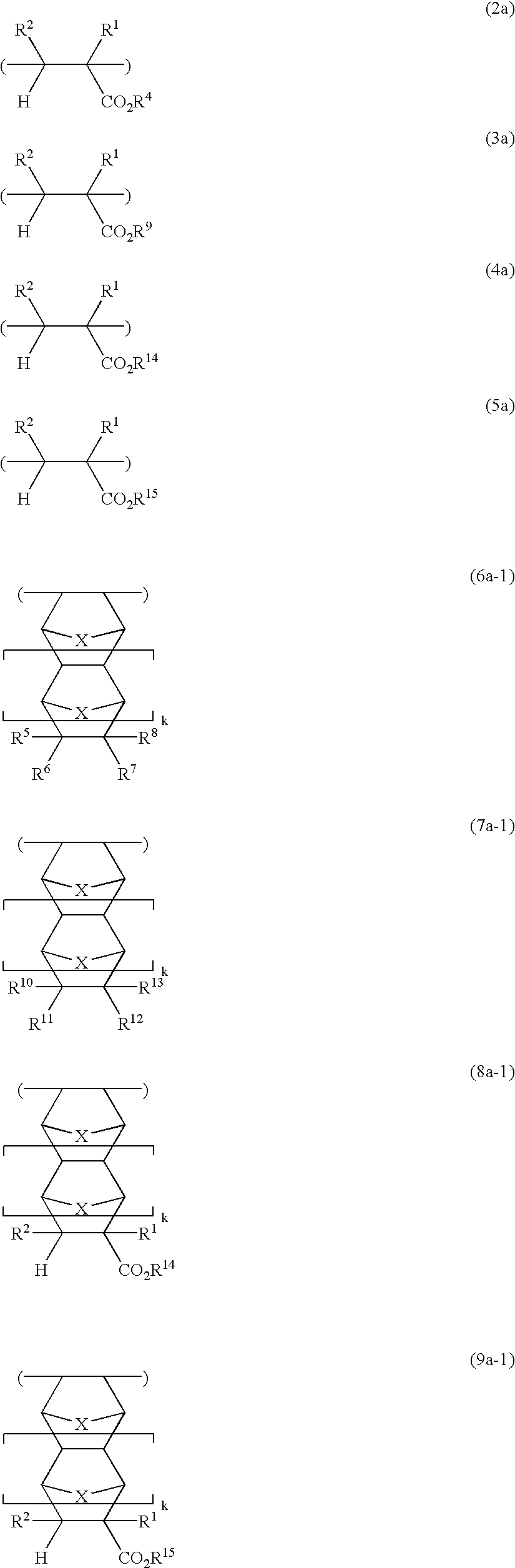
- the polymer further comprises recurring units of at least one of the following formulas (2a) to (10a).
- R 1 , R 2 and k are as defined above;
- R 4 is hydrogen or a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms;
- at least one of R 5 to R 8 is a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms, and the remainders are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, or R 5 to R 8 , taken together, may form a ring, and when they form a ring, at least one of R 5 to R 8 is a carboxyl or hydroxyl-containing divalent hydrocarbon group of 1 to 15 carbon atoms, and the remainders are independently a single bond or a straight, branched or cyclic alkylene group of 1 to 15 carbon atoms;
- R 9 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO 2 — partial structure; at least one of R 10 to R 13
- the invention provides a method for preparing a polymer comprising the step of effecting radical polymerization, anionic polymerization or coordination polymerization between an ester compound of the general formula (1) and another compound having a carbon-to-carbon double bond.
- the invention provides a resist composition comprising the polymer defined above, and more specifically, a resist composition comprising the polymer defined above, a photoacid generator, and an organic solvent.
- the invention provides a process for forming a resist pattern comprising the steps of applying the resist composition defined above onto a substrate to form a coating; heat treating the coating and then exposing it to high-energy radiation or electron beams through a photo mask; and optionally heat treating the exposed coating and developing it with a developer.
- the ester compound of formula (1) and the polymer comprising units of formula (1a-1) and/or (1a-2) employ a cycloalkylcycloalkyl or cycloalkylcycloalkenyl group of the following formula (1b) as the acid-eliminatable protective group, thereby overcoming the drawbacks of the tert-butoxycarbonyl and tert-butyl groups having low reactivity with acid as well as the 2-tetrahydropyranyl and 1-ethoxyethyl groups having too high reactivity with acid and low resistance to alkaline developers.
- Z, m and n are as defined above, the broken line denotes a valence bond.
- the ester compound of formula (1) is decomposed under acidic conditions while generating an olefin compound and a carboxylic acid.
- This mechanism itself is apparently identical with the decomposition of tertiary alkyl esters such as tert-butyl esters although the decomposition rate significantly differs between them.
- carboxylic acid and a tertiary alkyl cation generate first, then the extinction of the cation due to proton release occurs to form an olefin compound, whereby the reaction comes to an end.
- the rate-determining stage of this decomposition reaction is the late stage of cation extinction.
- ester compound of formula (1) provides a high degree of acidolysis surpassing other tertiary alkyl esters.
- the ester compound of formula (1) is basically a tertiary alkyl ester, it, when formulated into a resist composition, does not allow for the infinite progress of decomposition in a duration between exposure and development and film thinning during development as found with 2-tetrahydropyranyl and 1-ethoxyethyl.
- a polymer comprising constituent units derived from the compound is formulated as the base resin, there is obtained a resist composition which possesses a high sensitivity and resolution and has a high dissolution contrast in that the unexposed areas have a very slow rate of dissolution while the areas having undergone an adequate dose of exposure quickly become alkali soluble.
- very high etching resistance is exerted by virtue of the robust aliphatic ring positioned on the backbone of the polymer and a plurality of aliphatic rings contained in the protective group of formula (1b).
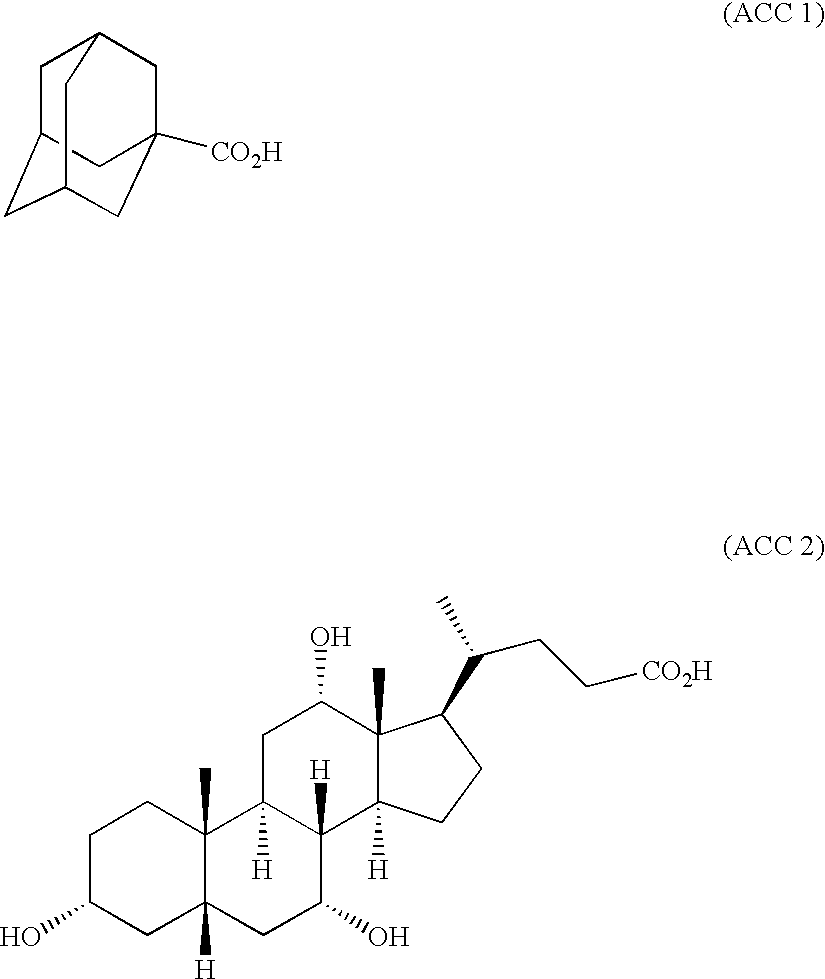
- FIG. 1 is a diagram showing the 1 H-NMR spectrum of the monomer obtained in Synthesis Example 1.
- novel ester compounds according to the first aspect of the invention are of the following general formula (1).
- R 1 is hydrogen, methyl or CH 2 CO 2 R 3 .
- R 2 is hydrogen, methyl or CO 2 R 3 .
- R 3 stands for straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms, such as, for example, methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, tert-amyl, n-pentyl, n-hexyl, cyclopentyl, cyclohexyl, ethylcyclopentyl, butylcyclopentyl, ethylcyclohexyl, butylcyclbhexyl, adamantyl, ethyladamantyl and butyladamantyl.
- Z is a divalent hydrocarbon group of 2 to 20 carbon atoms which forms a single ring or bridged ring with the carbon atom and which may contain a hetero atom, for example, single ringforming hydrocarbon groups such as cyclopropane, cyclobutane, cyclopentane, cyclohexane, cycloheptane, and cyclooctane; bridged ring-forming hydrocarbon groups such as bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane, bicyclo[4.4.0]decane and tricyclo[5.2.1.0 2,6 ]decane; and ring-forming hydrocarbon groups in which some of the hydrogen atoms in the foregoing groups are substituted with alkyl, hydroxy, alkoxy, acyloxy, alkylcarbonyl, hydroxycarbonyl, alkoxycarbonyl and oxo, among others.
- single ringforming hydrocarbon groups such as cyclopropane,
- the letter k is 0 or 1
- m is 0 or 1
- n is 0, 1, 2 or 3
- 2 m+n is equal to 2 or 3.
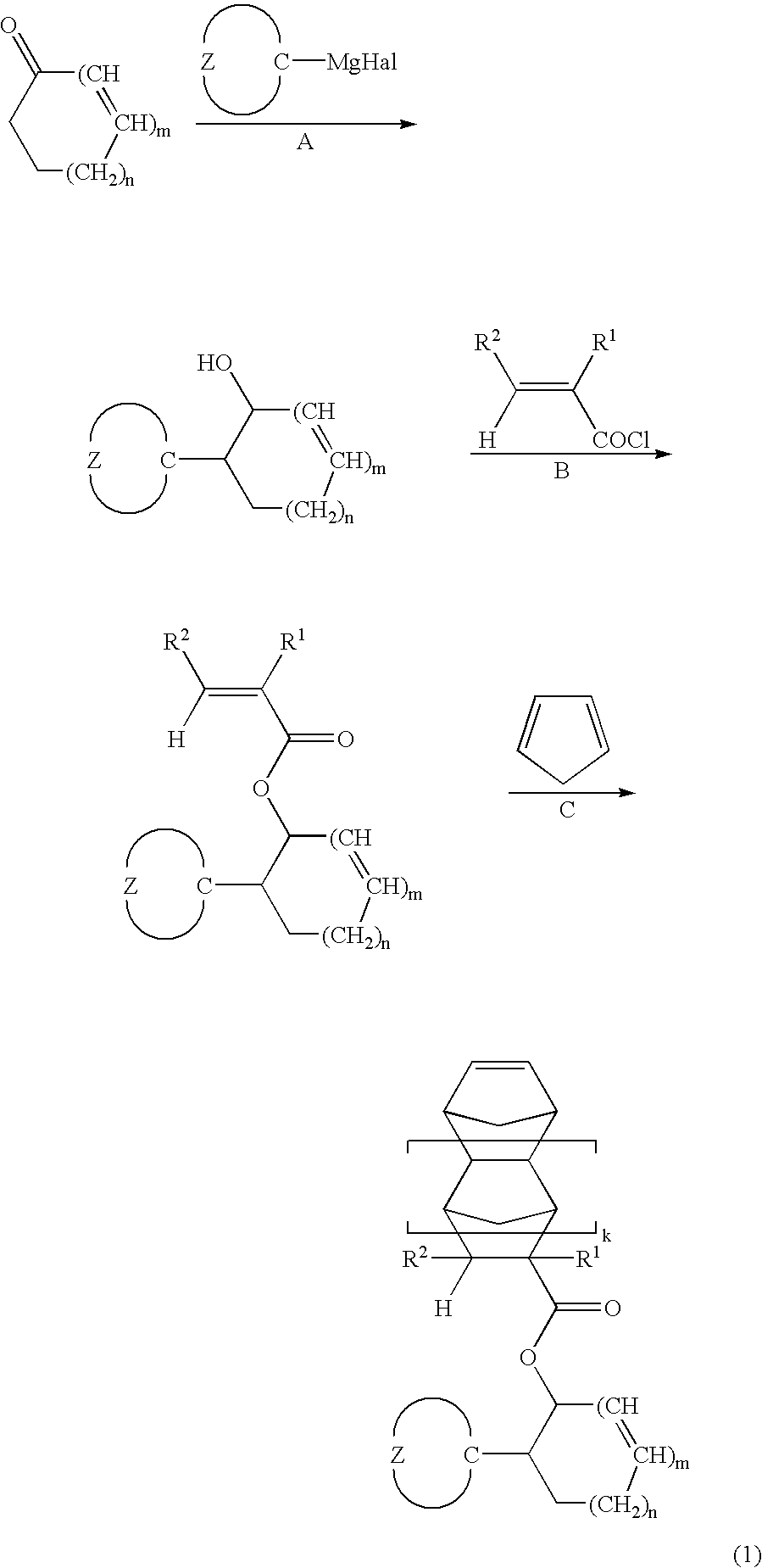
- ester compound of the invention is prepared, for example, by the following process, although the preparation is not limited thereto.
- R 1 , R 2 , Z, k, m and n are as defined above, and Hal is bromine or chlorine.
- Step A is to effect nucleophilic addition reaction with the carbonyl on a cyclic ketone compound to form a tertiary alcohol.
- This stage is typified by Grignard reaction, but not limited thereto. Reaction readily takes place under well-known conditions, preferably by mixing the reactants: cyclic ketone compound, alkyl halide and magnesium in a solvent such as tetrahydrofuran or diethyl ether, while heating or cooling the reaction mixture if desired.
- Step B is to convert the tertiary alcohol to an ester of unsaturated acid. Reaction readily takes place under well-known conditions, preferably by mixing the reactants: tertiary alcohol, unsaturated carboxylic halide (e.g., acrylic chloride or methacrylic chloride) and base (e.g., triethylamine) in a solvent such as methylene chloride, while cooling the reaction mixture if desired.
- unsaturated carboxylic halide e.g., acrylic chloride or methacrylic chloride
- base e.g., triethylamine
- Step C is to construct an alicyclic skeleton through Diels-Alder reaction. Reaction readily takes place under well-known conditions, preferably by mixing the reactants: ester of unsaturated acid and cyclopentadiene in a solventless system or in a solvent such as benzene, optionally in the presence of a catalyst such as boron trifluoride, while heating or cooling the reaction mixture if desired.
- a catalyst such as boron trifluoride
- the invention provides a polymer or high molecular weight compound comprising recurring units of the following general formula-(1a-1) and/or (1a-2) derived from an ester compound of the general formula (1), and having a weight average molecular weight of 1,000 to 500,000, and preferably 5,000 to 100,000.
- R 1 , R 2 , Z, k, m and n are as defined above.
- the polymer of the invention may further comprise recurring units of at least one type selected from recurring units of the following formulas (2a) to (10a) which are derived from monomers of the following general formulas (2) to (10).
- formulae (6a-1) to (9a-2) may also be represented by the following formulae (6a-1-1) to (9a-2-2).
- R 1 and R 2 are as defined above.
- R 4 stands for hydrogen or carboxyl or hydroxyl-containing monovalent hydrocarbon groups of 1 to 15 carbon atoms, for example, carboxyethyl, carboxybutyl, carboxycyclopentyl, carboxycyclohexyl, carboxynorbornyl, carboxyadamantyl,:hydroxyethyl, hydroxybutyl, hydroxycyclopentyl, hydroxycyclohexyl, hydroxynorbornyl, and hydroxyadamantyl.
- At least one of R 5 to R 8 is a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms, and the remaining R's are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms.
- Examples of the carboxyl or hydroxyl-bearing monovalent hydrocarbon group of 1 to 15 carbon atoms include carboxy, carboxymethyl, carboxyethyl, carboxybutyl, hydroxymethyl, hydroxyethyl, hydroxybutyl, 2-carboxyethoxycarbonyl, 4-carboxybutoxycarbonyl, 2:-hydroxyethoxycarbonyl, 4-hydroxybutoxycarbonyl, carboxycyclopentyloxycarbonyl, carboxycyclohexyloxycarbonyl, carboxynorbornyloxycarbonyl, carboxyadamantyloxycarbonyl, hydroxycyclopentyloxycarbonyl, hydroxycyclohexyloxycarbonyl, hydroxynorbornyloxycarbonyl, and hydroxyadamantyloxycarbonyl.
- R 5 to R 8 taken together, may form a ring, and in that event, at least one of R 5 to R 8 is a divalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms.
- Examples of the carboxyl or hydroxyl-bearing divalent hydrocarbon group of 1 to 15 carbon atoms include the groups exemplified as the carboxyl or hydroxyl-bearing monovalent hydrocarbon group, with one hydrogen atom eliminated therefrom.
- Examples of the straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms include the groups exemplified for R 3 , with one hydrogen atom eliminated therefrom.
- R 9 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO 2 — partial structure, for example, 2-oxooxolan-3-yl, 4,4-dimethyl-2-oxooxolan-3-yl, 4-methyl-2-oxooxan-4-yl, 2-oxo-1,3-dioxolan-4-ylmethyl, and 5-methyl-2-oxooxolan-5-yl.
- At least one of R 10 to R 13 is a monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO 2 — partial structure, while the remaining R's are independently hydrogen or straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms.
- Examples of the monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO 2 — partial structure include 2-oxooxolan-3-yloxycarbonyl, 4,4-dimethyl-2-oxooxolan-3-yloxycarbonyl, 4-methyl-2-oxooxan-4-yloxycarbonyl, 2-oxo-1,3-dioxolan-4-ylmethyloxycarbonyl, and 5-methyl-2-oxooxolan-5-yloxycarbonyl.
- Examples of the straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms are the same as exemplified for R 3 .
- R 10 to R 13 taken together, may form a ring, and in that event, at least one of R 10 to R 13 is a divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO 2 — partial structure, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms.
- Examples of the divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO 2 — partial structure include 1-oxo-2-oxapropane-1,3-diyl, 1,3,-dioxo-2-oxapropane-1,3-diyl, 1-oxo-2-oxabutane-1,4-diyl, and 1,3-dioxo-2-oxabutane-1,4-diyl, as well as the groups exemplified as the monovalent hydrocarbon group containing a —CO 2 — partial structure, with one hydrogen atom eliminated therefrom.
- Examples of the straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms include the groups exemplified for R 3 , with one hydrogen atom eliminated therefrom.
- R 14 is a polycyclic hydrocarbon group having 7 to 15 carbon atoms or an alkyl group containing a polycyclic hydrocarbon group, for example, norbornyl, bicyclo[3.3.1]-nonyl, tricyclo[5.2.1.0 2,6 ]decyl, adamantyl, ethyladamantyl, butyladamantyl, norbornylmethyl, and adamantylmethyl.
- R 15 is an acid labile group, and k is 0 or 1.
- X is —CH 2 — or —O—.
- Y is —O— or —(NR 16 )— wherein R 16 is hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atom's, examples of which are as described for R 3 .
- the acid labile groups represented by R 15 may be selected from a variety of such groups.
- Examples of the acid labile group are groups of the following general formulae (L1) to (L4), tertiary alkyl groups of 4 to 20 carbon atoms, preferably 4 to 15 carbon atoms, trialkylsilyl groups in which each alkyl moiety has 1 to 6 carbon atoms, and oxoalkyl groups of 4 to 20 carbon atoms.
- R L01 and R L02 are hydrogen or straight, branched or cyclic alkyl groups of 1 to 18 carbon atoms, preferably 1 to 10 carbon atoms.
- Exemplary alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, cyclopentyl, cyclohexyl, 2-ethylhexyl, and n-octyl.
- R L03 is a monovalent hydrocarbon group of 1 to-18 carbon atoms, preferably 1 to 10 carbon atoms, which may contain a hetero atom such as oxygen, examples of which include unsubstituted straight, branched or cyclic alkyl groups and straight, branched or cyclic alkyl groups in which some hydrogen atoms are replaced by hydroxyl, alkoxy, oxo, amino, alkylamino or the like.
- Illustrative examples are the substituted alkyl groups shown below.
- R L01 and R L02 , R L01 and R L03 , or R L02 and R L03 may form a ring.
- Each of R L01 , R L02 and R L03 is a straight or branched alkylene group of 1 to 18 carbon atoms, preferably 1 to 10 carbon atoms when they form a ring.
- R L04 is a tertiary-alkyl-group of 4 to 20 carbon atoms, preferably 4 to 15 carbon atoms, a trialkylsilyl group in which each alkyl moiety has 1 to 6 carbon atoms, an oxoalkyl group of 4 to 20 carbon atoms, or a group of formula (L1).
- Exemplary tertiary alkyl groups are tert-butyl, tert-amyl, 1,1-diethylpropyl, 1-ethylcyclopentyl, 1-butylcyclopentyl, 1-ethylcyclohexyl, 1-butylcyclohexyl,-1-ethyl-2-cyclopentenyl, 1-ethyl-2-cyclohexenyl, and 2-methyl-2-adamantyl.
- Exemplary trialkylsilyl groups are trimethylsilyl, triethylsilyl, and dimethyl-tert-butylsilyl.
- Exemplary oxoalkyl groups are 3-oxocyclohexyl, 4-methyl-2-oxooxan-4-yl, and 5-methyl-5-oxooxolan-4-yl.
- Letter y is an integer of 0 to 6.
- R L05 is a straight or branched alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms.
- Examples of the straight, branched or cyclic alkyl group include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, tert-amyl, n-pentyl and n-hexyl.
- Exemplary aryl groups are phenyl, methylphenyl, naphthyl, anthryl, phenanthryl, and pyrenyl. Letter m is equal to 0 or 1, n is equal to 0, 1, 2 or 3, and 2 m+n is equal to 2 or 3.
- R L06 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms. Examples of these groups are the same as exemplified for R L05 .
- R L07 to R L16 independently represent hydrogen or monovalent hydrocarbon groups of 1 to 15 carbon atoms which may contain a hetero atom.
- exemplary hydrocarbon groups are straight, branched or cyclic alkyl groups such as methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, tert-amyl, n-pentyl, n-hexyl, n-octyl, n-nonyl, n-decyl, cyclopentyl, cyclohexyl, cyclopentylmethyl, cyclopentylethyl, cyclopentylbutyl, cyclohexylmethyl, cyclohexylethyl and cyclohexylbutyl, and substituted ones of these groups in which some hydrogen atoms are replaced by hydroxyl, alkoxy, carboxy, alkoxycarbonyl
- R L07 to R L16 taken together, form a ring (for example, a pair of R L07 and R L08 , R L07 and R L09 , R L08 and R L10 , R L09 and R L10 , R L11 and R L12 , R L13 and R L14 , or a similar pair form a ring).
- Each of R L07 to R L16 represents a divalent C 1 -C 15 hydrocarbon group which may contain a hetero atom, when they form a ring, examples of which are the ones exemplified above for the monovalent hydrocarbon groups, with one hydrogen atom being eliminated.
- R L07 to R L16 which are attached to adjoining carbon atoms (for example, a pair of R L07 and R L09 , R L09 and R L15 , R L13 and R L15 , or a similar pair) may bond together directly to form a double bond.
- the cyclic ones are, for example, tetrahydrofuran-2-yl, 2-methyltetrahydrofuran-2-yl, tetrahydropyran-2-yl, and 2-methyltetrahydropyran-2-yl.
- Examples of the acid labile groups of formula (L2) include tert-butoxycarbonyl, tert-butoxycarbonylmethyl, tert-amyloxycarbonyl, tert-amyloxycarbonylmethyl, 1,1-diethylpropyloxycarbonyl, 1,1-diethylpropyloxycarbonylmethyl, 1-ethylcyclopentyloxycarbonyl, 1-ethylcyclopentyloxycarbonylmethyl-1-ethyl-2-cyclopentenyloxycarbonyl, 1-ethyl-2-cyclopentenyloxycarbonylmethyl, 1-ethoxyethoxycarbonylmethyl, 2-tetrahydropyranyloxycarbonylmethyl, and 2-tetrahydrofuranyloxycarbonylmethyl groups.
- Examples of the acid labile groups of formula (L3) include 1-methylcyclopentyl, 1-ethylcyclopentyl, 1-n-propylcyclopentyl, 1-n-butylcyclopentyl, 1-sec-butylcyclopentyl, 1-methylcyclohexyl, 1-ethylcyclohexyl, 3-methyl-1-cyclopenten-3-yl, 3-ethyl-1-cyclopenten-3′-yl, 3-methyl-1-cyclohexen-3-yl, and 3-ethyl-1-cyclohexen-3-yl groups.
- the polymer of the invention may be prepared by polymerizing an ester compound of formula (1) or by copolymerizing a first monomer in the form of an ester compound of formula (1) with a second monomer in the form of at least one compound of formulas (2) to (10).
- the polymer can be tailored so that it may exert the preferred performance when blended in resist compositions.
- the polymer of the invention may have copolymerized therewith (iii) another monomer having a carbon-to-carbon double bond other than (i) and (ii).
- Examples of the additional monomer (iii) include substituted acrylic acid esters such as methyl methacrylate, methyl crotonate, dimethyl maleate, and dimethyl itaconate, unsaturated carboxylic acids such as maleic acid, fumaric acid and itaconic acid, substituted or unsubstituted norbornenes such as norbornene and methyl norbornene-5-carboxylate, and unsaturated acid anhydrides such as itaconic anhydride.
- substituted acrylic acid esters such as methyl methacrylate, methyl crotonate, dimethyl maleate, and dimethyl itaconate
- unsaturated carboxylic acids such as maleic acid, fumaric acid and itaconic acid
- substituted or unsubstituted norbornenes such as norbornene and methyl norbornene-5-carboxylate
- unsaturated acid anhydrides such as itaconic anhydride.
- the polymers of the invention may contain
- the polymers of the invention have a weight average molecular weight of 1,000 to 500,000, preferably 3,000 to 100,000. Outside the range, the etching resistance may become extremely low and the resolution may become low because a substantial difference in rate of dissolution before and after exposure is lost.
- a polymer is prepared by effecting radical polymerization, anionic polymerization or coordination polymerization between an ester compound of formula (1) and another compound having a carbon-to-carbon double bond, which is typically selected from the above-described monomers (ii) and (iii).
- reaction conditions include (a) a solvent selected from among hydrocarbons such as benzene, ethers such as tetrahydrofuran, alcohols such as ethanol, and ketones such as methyl isobutyl ketone, (b) a polymerization initiator selected from azo compounds such as 2,2′-azobisisobutyronitrile and peroxides such as benzoyl peroxide and lauroyl peroxide, (c) a temperature of about 0° C. to about 100° C., and (d) a time of about 1 ⁇ 2 hour to about 48 hours. Reaction conditions outside the described range may be employed if desired.
- a solvent selected from among hydrocarbons such as benzene, ethers such as tetrahydrofuran, alcohols such as ethanol, and ketones such as methyl isobutyl ketone
- a polymerization initiator selected from azo compounds such as 2,2′-azobisisobutyronitrile and peroxides such as
- reaction conditions include (a) a solvent selected from among hydrocarbons such as benzene, ethers such as tetrahydrofuran, and liquid ammonia, (b) a polymerization initiator selected from metals such as sodium and potassium, alkyl metals such as n-butyllithium and sec-butyllithium, ketyl, and Grignard reagents, (c) a temperature of about ⁇ 78° C.
- a solvent selected from among hydrocarbons such as benzene, ethers such as tetrahydrofuran, and liquid ammonia
- a polymerization initiator selected from metals such as sodium and potassium, alkyl metals such as n-butyllithium and sec-butyllithium, ketyl, and Grignard reagents
- a stopper selected from among proton-donative compounds such as methanol, halides such as methyl iodide, and electrophilic compounds. Reaction conditions outside the described range may be employed if desired.
- reaction conditions include (a) a solvent selected from among hydrocarbons such as n-heptane and toluene, (b) a catalyst selected from Ziegler-Natta catalysts comprising a transition metal (e.g., titanium) and alkylaluminum, Phillips catalysts of metal oxides having chromium or nickel compounds carried thereon, and olefin-metathesis mixed catalysts as typified by tungsten and rhenium mixed catalysts, (c) a temperature of about 0° C. to about 100° C., and (d) a time of about 1 ⁇ 2 hour to about 48 hours. Reaction conditions outside the described range may be employed if desired.
- a solvent selected from among hydrocarbons such as n-heptane and toluene
- a catalyst selected from Ziegler-Natta catalysts comprising a transition metal (e.g., titanium) and alkylaluminum, Phillips catalysts of metal oxides having chromium or nickel compounds carried thereon,
- the other aspect of the invention provides a resist composition comprising the polymer.
- the resist composition is defined as comprising the polymer, a photoacid generator, and an organic solvent.
- the photoacid generator is a compound capable of generating an acid upon exposure to high energy radiation or electron beams and includes the following:
- R 101a , R 101b , and R 101c independently represent straight, branched or cyclic alkyl, alkenyl, oxoalkyl or oxoalkenyl groups of 1 to 12 carbon atoms, aryl groups of 6 to 20 carbon atoms, or aralkyl or aryloxoalkyl groups of 7 to 12 carbon atoms, wherein some or all of the hydrogen atoms may be replaced by alkoxy or other groups.
- R 101b and R 101c taken together, may form a ring.
- R 101b and R 101c each are alkylene groups of 1 to 6 carbon atoms when they form a ring.
- K ⁇ is a non-nucleophilic counter ion.
- R 101a , R 101b , and R 101c may be the same or different and are illustrated below.
- Exemplary alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, heptyl, octyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclopropylmethyl, 4-methylcyclohexyl, cyclohexylmethyl, norbornyl, and adamantyl.
- Exemplary alkenyl groups include vinyl, allyl, propenyl, butenyl, hexenyl, and cyclohexenyl.
- Exemplary oxoalkyl groups include 2-oxocyclopentyl and 2-oxocyclohexyl as well as 2-oxopropyl, 2-cyclopentyl-2-oxoethyl, 2-cyclohexyl-2-oxoethyl, and 2-(4-methylcyclohexyl)-2-oxoethyl.
- aryl groups include phenyl and naphthyl; alkoxyphenyl groupssuch as p-methoxyphenyl, m-methoxyphenyl, o-methoxyphenyl, ethoxyphenyl, p-tert-butoxyphenyl, and m-tert-butoxyphenyl; alkylphenyl groups such as 2-methylphenyl, 3-methylphenyl, 4-methylphenyl, ethylphenyl, 4-tert-butylphenyl, 4-butylphenyl, and dimethylphenyl; alkylnaphthyl groups such as methylnaphthyl and ethylnaphthyl; alkoxynaphthyl groups such as methoxynaphthyl and ethoxynaphthyl; dialkylnaphthyl groups such as dimethylnaphthyl and diethylnaph,
- Exemplary aralkyl groups include benzyl, phenylethyl, and phenethyl.
- Exemplary aryloxoalkyl groups are 2-aryl-2-oxoethyl groups such as 2-phenyl-2-oxoethyl, 2-(1-naphthyl)-2-oxoethyl, and 2-(2-naphthyl)-2-oxoethyl.
- non-nucleophilic counter ion represented by K ⁇ examples include halide ions such as chloride and bromide ions, fluoroalkylsulfonate ions such as triflate, 1,1,1-trifluoroethanesulfonate, and nonafluorobutanesulfonate arylsulfonate ions such as tosylate, benzenesulfonate, 4-fluorobenzenesulfonate, and 1,2,3,4,5-pentafluorobenzenesulfonate, and alkylsulfonate ions such as mesylate and butanesulfonate.
- halide ions such as chloride and bromide ions
- fluoroalkylsulfonate ions such as triflate, 1,1,1-trifluoroethanesulfonate, and nonafluorobutanesulfonate arylsulfonate ions
- tosylate
- R 102a and R 102b independently represent straight, branched or cyclic alkyl groups of 1 to 8 carbon atoms.
- R 103 represents a straight, branched or cyclic alkylene groups of 1 to 10 carbon atoms.
- R 104a and R 104b independently represent 2-oxoalkyl groups of 3 to 7 carbon atoms.
- K ⁇ is a non-nucleophilic counter ion.
- R 102a and R 102b Illustrative of the groups represented by R 102a and R 102b are methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, heptyl, octyl, cyclopentyl, cyclohexyl, cyclopropylmethyl, 4-methylcyclohexyl, and cyclohexylmethyl.
- R 103 Illustrative of the groups represented by R 103 are methylene, ethylene, propylene, butylene, pentylene, hexylene, heptylene, octylene, nonylene, 1,4-cyclohexylene, 1,2-cyclohexylene, 1,3-cyclopentylene, 1,4-cyclooctylene, and 1,4-cyclohexanedimethylene.
- R 104a and R 104b are 2-oxopropyl, 2-oxocyclopentyl, 2-oxocyclohexyl, and 2-oxocycloheptyl.
- Illustrative examples of the counter ion represented by K ⁇ are the same as exemplified for formulae (P1a-1) and (P1a-2).
- R 105 and R 106 independently represent straight, branched or cyclic alkyl or halogenated alkyl groups of 1 to 12 carbon atoms, aryl or halogenated aryl groups of 6 to 20 carbon atoms, or aralkyl groups of 7 to 12 carbon atoms.
- exemplary alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, heptyl, octyl, amyl, cyclopentyl, cyclohexyl, cycloheptyl, norbornyl, and adamantyl.
- exemplary halogenated alkyl groups include trifluoromethyl, 1,1,1-trifluoroethyl, 1,1,1-trichloroethyl, and nonafluorobutyl.
- Exemplary aryl groups include phenyl; alkoxyphenyl groups such as p-methoxyphenyl, m-methoxyphenyl, o-methoxyphenyl, ethoxyphenyl, p-tert-butoxyphenyl, and m-tert-butoxyphenyl; and alkylphenyl groups such as 2-methyl-phenyl, 3-methylphenyl, 4-methylphenyl, ethylphenyl, 4-tertbutylphenyl, 4-butylphenyl, and dimethylphenyl.
- Exemplary halogenated aryl groups include fluorophenyl, chlorophenyl, and 1,2,3,4,5-pentafluorophenyl.
- Exemplary aralkyl groups include benzyl and phenethyl.
- R 107 , R 108 , and R 109 independently represent straight, branched or cyclic alkyl or halogenated alkyl groups of 1 to 12 carbon atoms, aryl or halogenated aryl groups of 6 to 20 carbon atoms, or aralkyl groups of 7 to 12 carbon atoms. Also, R 108 and R 109 , taken together, may form a ring. R 108 and R 109 each are straight or branched alkylene groups of 1 to 6 carbon atoms when they form a ring.
- Illustrative examples of the alkyl, halogenated alkyl, aryl, halogenated aryl, and aralkyl groups represented by R 107 , R 108 , and R 109 are the same as exemplified for R 105 and R 106 .
- Examples of the alkylene groups represented by R 108 and R 109 include methylene, ethylene, propylene, butylene, and hexylene.
- R 101a and R 101b are as defined above.
- R 110 is an arylene group of 6 to 10 carbon atoms, alkylene group of 1 to 6 carbon atoms, or alkenylene group of 2 to 6 carbon atoms wherein some or all of the hydrogen atoms may be replaced by straight or branched alkyl or alkoxy groups of 1 to 4 carbon atoms, nitro, acetyl, or phenyl groups.
- R 111 is a straight, branched or cyclic alkyl group of 1 to 8,carbon atoms, alkenyl, alkoxyalkyl, phenyl or naphthyl group wherein some or all of the hydrogen atoms may be replaced by alkyl or alkoxy groups of 1 to 4 carbon atoms, phenyl groups (which may have substituted thereon an alkyl or alkoxy of 1 to 4 carbon atoms, nitro, or acetyl group), hetero-aromatic groups of 3 to 5 carbon atoms, or chlorine or fluorine atoms.
- exemplary arylene groups include 1,2-phenylene and 1,8-naphthylene;
- exemplary alkylene groups include methylene, 1,2-ethylene, 1,3-propylene, 1,4-butylene, 1-phenyl-1,2-ethylene, and norbornane-2,3-diyl;
- exemplary alkenylene groups include 1,2-vinylene, 1-phenyl-1,2-vinylene, and 5-norbornene-2,3-diyl.
- exemplary alkyl groups are as exemplified for R 101a to R 101c ;
- exemplary alkenyl groups include vinyl, 1-propenyl, allyl, 1-butenyl, 3-butenyl, isoprenyl, 1-pentenyl, 3-pentenyl, 4-pentenyl, dimethylallyl, 1-hexenyl, 3-hexenyl, 5-hexenyl, 1-heptenyl, 3-heptenyl, 6-heptenyl, and 7-octenyl; and
- exemplary alkoxyalkyl, groups include methoxymethyl, ethoxymethyl, propoxymethyl, butoxymethyl, pentyloxymethyl, hexyloxymethyl, heptyloxymethyl, methoxyethyl, ethoxyethyl, propoxyethyl, butoxyethyl, pentyloxyethyl, hexyloxyethyl, methoxy
- the alkyl groups of 1 to 4 carbon atoms include methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl and tert-butyl;
- the alkoxy groups of 1 to 4,carbon atoms include methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, isobutoxy, and tert-butoxy;
- the phenyl groups which may have substituted thereon an alkyl or alkoxy of 1 to-4 carbon atoms, nitro, or acetyl group include phenyl, tolyl, p-tert-butoxyphenyl, p-acetylphenyl and p-nitrophenyl;
- the hetero-aromatic groups of 3 to 5 carbon atoms include pyridyl and furyl.
- Illustrative examples of the photoacid generator include:
- onium salts such as diphenyliodonium trifluoromethanesulfonate, (p-tert-butoxyphenyl)phenyliodonium trifluoromethanesulfonate, diphenyliodonium p-toluenesulfonate, (p-tert-butoxyphenyl)phenyliodonium p-toluenesulfonate, triphenylsulfonium trifluoromethanesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium trifluoromethanesulfonate, bis(p-tert-butoxyphenyl)phenylsulfonium trifluoromethanesulfonate, tris(p-tert-butoxyphenyl)-sulfonium trifluoromethanesulfonate, triphenylsulfonium p-toluenesulfonate,
- glyoxime derivatives such as bis-o-(p-toluenesulfonyl)- ⁇ -dimethylglyoxime, bis-o-(p-toluenesulfonyl)- ⁇ -diphenylglyoxime, bis-o-(p-toluenesulfonyl)- ⁇ -dicyclohexylgloxime, bis-o-(p-toluenesulfonyl)-2,3-pentanedioneglyoxime, bis-o-(p-toluenesulfonyl)-2-methyl-3,4-pentanedioneglyoxime, bis-o-(n-butanesulfonyl)-a-dimethylglyoxime, bis-o-(n-butanesulfonyl)- ⁇ -diphenylglyoxime, bis-o-(n-butanesulfonyl)- ⁇ -d
- bissulfone derivatives such as bisnaphthylsulfonylmethane, bistrifluoromethylsulfonylmethane, bismethylsulfonylmethane, bisethylsulfonylmethane, bispropylsulfonylmethane, bisisopropylsulfonylmethane, bis-p-toluenesulfonylmethane, and bisbenzenesulfonylmethane;
- ⁇ -ketosulfone derivatives such as 2-cyclohexylcarbonyl-2-(p-toluenesulfonyl)propane and 2-isopropylcarbonyl-2-(p-toluenesulfonyl)propane;
- disulfone derivatives such as diphenyl disulfone and dicyclohexyl disulfone
- nitrobenzyl sulfonate derivatives such as 2,6-dinitrobenzyl p-toluenesulfonate and 2,4-dinitrobenzyl p-toluenesulfonate;
- sulfonic acid ester derivatives such as 1,2,3-tris-(methanesulfonyloxy)benzene, 1,2,3-tris(trifluoromethanesulfonyloxy)benzene, and 1,2,3-tris(p-toluenesulfonyloxy)-benzene; and
- sulfonic acid esters of N-hydroxyimides such as N-hydroxysuccinimide methanesulfonate, N-hydroxysuccinimide trifluoromethanesulfonate, N-hydroxysuccinimide ethanesulfonate, N-hydroxysuccinimide 1-propanesulfonate, N-hydroxysuccinimide 2-propanesulfonate, N-hydroxysuccinimide 1-pentanesulfonate, N-hydroxysuccinimide 1-octanesulfonate, N-hydroxysuccinimide p-toluenesulfonate, N-hydroxysuccinimide p-methoxybenzenesulfonate, N-hydroxysuccinimide 2-chloroethanesulfonate, N-hydroxysuccinimide benzenesulfonate, N-hydroxysuccinimide 2,4,6-trimethylbenzenesulfonate, N-
- Preferred among these photoacid generators are onium salts such as triphenylsulfonium trifluoromethanesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium trifluoromethanesulfonate, tris(p-tert-butoxyphenyl)sulfonium trifluoromethaneesulfonate, triphenylsulfonium p-toluenesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium p-toluenesulfonate, tris(p-tert-butoxyphenyl)sulfonium p-toluenesulfonate, trinaphthylsulfonium trifluoromethanesulfonate, cyclohexylmethyl(2-oxocyclohexyl)-sulfonium trifluoromethanesulfon
- photoacid generators may be used singly or in combinations of two or more thereof.
- Onium salts are effective for improving rectangularity, while diazomethane derivatives and glyoxime derivatives are effective for reducing standing waves.
- the combination of an onium salt with a diazomethane or a glyoxime derivative allows for fine adjustment of the profile.
- the photoacid generator is added in an amount of 0.1 to 15 parts, and especially 0.5 to 8 parts by weight, per 100 parts by weight of the base resin (all parts are by weight, hereinafter). Less than 0.1 part of the photoacid generator would provide a poor sensitivity whereas more than 15 parts of the photoacid generator would adversely affect transparency and resolution.
- the organic solvent used herein may be any organic solvent in which the base resin, photoacid generator, and other components are soluble.
- the organic solvent include ketones such as cyclohexanone and methyl-2-n-amylketone; alcohols such as 3-methoxybutanol, 3-methyl-3-methoxybutanol, 1-methoxy-2-propanol, and 1-ethoxy-2-propanol; ethers such as propylene glycol monomethyl ether, ethylene glycol monomethyl ether, propylene glycol monoethyl ether, ethylene glycol monoethyl ether, propylene glycol dimethyl ether, and diethylene glycol dimethyl ether; and esters such as propylene glycol monomethyl ether acetate, propylene glycol monoethyl ether acetate, ethyl lactate, ethyl pyruvate, butyl acetate, methyl 3-methoxy
- solvents may be used alone or in combinations of two or more thereof.
- organic solvents it is recommended to use diethylene glycol dimethyl ether and 1-ethoxy-2-propanol because the photoacid generator serving as one of the resist components is most soluble therein, propylene glycol monomethyl ether acetate because it is a safe solvent, or a mixture thereof.
- An appropriate amount of the organic solvent used is about 200 to 1,000 parts, especially about 400 to 800 parts by weight per 100 parts by weight of the base resin.
- another polymer other than the inventive polymer comprising recurring units of formula (1a-1) and/or (1a-2) may also be added.
- the other polymers that can be added to the resist composition are, for example, those polymers comprising units of the following formula (R1) or (R2) and having a weight average molecular weight of about 1,000 to about 500,000, especially about 5,000 to about 100,000 although the other polymers are not limited thereto.
- R 001 is hydrogen, methyl or CH 2 Co 2 R 003 .
- R 002 is hydrogen, methyl or CO 2 R 003 .
- R 003 is a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms.
- R 004 is hydrogen or a monovalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group.
- At least one of R 005 to R 008 represents a monovalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group while the remaining R's independently represent hydrogen or a straight, branched or cyclic alkyl group-of 1 to 15 carbon atoms.
- R 005 to R 008 taken together, may form a ring, and in that event, at least one of R 005 to R 008 is a divalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms.
- R 009 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO 2 — partial structure.
- At least one of R 010 to R 013 is a monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO 2 — partial structure, while the remaining R's are independently hydrogen or straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms.
- R 010 to R 013 taken together, may form a ring, and in that event, at least one of R 010 to R 013 is a divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO 2 — partial structure, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms.
- R 014 is a polycyclic hydrocarbon group having 7 to 15 carbon atoms or an alkyl group containing a polycyclic hydrocarbon group.
- R 015 is an acid labile group.
- R 016 is hydrogen or methyl.
- R 017 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms.
- X′, y′ and z′ are integers of 0 to 3, satisfying x′+y′+z′ ⁇ 5 and 1 ⁇ y′+Z′.
- R 1 to R 15 Exemplary groups of, these R's are as exemplified above for R 1 to R 15 .
- inventive polymer comprising recurring units of formula (1a-1) and/or (1a-2)
- the other polymer are preferably blended in a weight ratio from 10:90 to 90:10, more preferably from 20:80 to 80:20. If the blend ratio of the inventive polymer is below this range, the resist composition would become poor in some of the desired properties.
- the properties of the resist composition can be adjusted by properly changing the blend ratio of the inventive polymer.
- the other polymer is not limited to one type and a mixture of two or more other polymers may be added.
- the use of plural polymers allows for easy adjustment of resist properties.
- a dissolution regulator may be added.
- the dissolution regulator is a compound having on the molecule at least two phenolic hydroxyl groups, in which an average of from 0 to 100 mol % of all the hydrogen atoms on the phenolic hydroxyl groups are replaced with acid labile groups or a compound having on the molecule at least one carboxyl group, in which an average of 50 to 100 mol % of all the hydrogen atoms on the carboxyl groups are replaced with acid labile groups, both the compounds having an average molecular weight within a range of 100 to 1,000, and preferably 150 to 800.
- the degree of substitution of the hydrogen atoms on the phenolic hydroxyl groups with acid labile groups is on average at least 0 mol %, and preferably at least 30 mol %, of all the phenolic hydroxyl groups.
- the upper limit is 100 mol %, and preferably 80 mol %.
- the degree of substitution of the hydrogen atoms on the carboxyl groups with acid-labile groups is on average at least 50 mol %, and preferably at least 70 mol %, of all the carboxyl groups, with the upper limit being 100 mol %.
- Preferable examples of such compounds having two or more phenolic hydroxyl groups or compounds having at least one carboxyl group include those of formulas (D1) to (D14) below.
- R 201 and R 202 are each hydrogen or a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms;
- R 203 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or —(R 207 ) h —COOH;
- R 205 is an alkylene of 1 to 10 carbon atoms; an arylene of 6 to 10 carbonatoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom;
- R 206 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or a hydroxyl-substituted phenyl or naphthy
- suitable examples of R 201 and R 202 include hydrogen:, methyl, ethyl, butyl, propyl, ethynyl, and cyclohexyl; suitable examples of R 203 include the same groups as for R 201 and R 202 , as well as —COOH and —CH 2 COOH; suitable examples of R 204 include ethylene, phenylene, carbonyl, sulfonyl, oxygen, and sulfur; suitable examples of R 205 include methylene as well as the same groups as for R 204 ; and suitable examples of R 206 include hydrogen, methyl, ethyl, butyl, propyl, ethynyl, cyclohexyl, and hydroxyl-substituted phenyl or naphthyl.
- Exemplary acid labile groups on the dissolution regulator include groups of the following general formulae (L1) to (L4), tertiary alkyl groups of 4 to 20 carbon atoms, trialkylsilyl groups in which each of the alkyls has 1 to 6 carbon atoms, and oxoalkyl groups of 4 to 20 carbon atoms.
- R L01 and R L02 are each hydrogen or a straight, branched or cyclic-alkyl having 1 to 18,carbon atoms; and R L03 is a monovalent hydrocarbon group of 1 to 18 carbon atoms which may contain a heteroatom (e.g., oxygen).
- a pair of R L01 and R L02 , a pair of R L01 and R L03 , or a pair of R L02 and R L03 . may together form a ring, with the proviso that R L01 , R L02 , and R L03 are each a straight or branched alkylene of 1 to 18 carbon atoms when they form a ring.
- R L14 is a tertiary alkyl group of 4 to 20 carbon atoms, a trialkysilyl group in which each of the alkyls has 1 to 6 carbon atoms, an oxoalkyl group of 4 to 20 carbon atoms, or a group of the formula (L1).
- R L05 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms.
- R L06 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms.
- R L17 to R L16 independently represent hydrogen or monovalent hydrocarbon groups of 1 to 15 carbon atoms which may contain a hetero atom. Alternatively, R L07 to R L16 , taken together, may form a ring. Each of R L07 to R L16 represents a divalent C 1 -C 15 hydrocarbon group which may contain a hetero atom, when they form a ring. Two of R L07 to R L16 which are attached to adjoining carbon atoms may bond together directly to form a double bond.
- Letter y is an integer of 0 to 6.
- Letter m is equal to 0 or 1
- n is equal to 0, 1, 2 or 3
- 2 m+n is equal to 2 or 3.
- the dissolution regulator may be formulated in an amount of 0 to 50 parts, preferably 5 to 50 parts, and more preferably 10 to 30 parts, per 100 parts of the base resin, and may be used singly or as a mixture of two or more thereof. Less than 5 parts of the dissolution regulator may fail to yield an improved resolution, whereas the use of more than 50 parts would lead to slimming of the patterned film, and thus a decline in resolution.
- the dissolution regulator can be synthesized by introducing acid labile groups into a compound having phenolic hydroxyl or carboxyl groups in accordance with an organic chemical formulation.
- a basic compound may be blended.
- a suitable basic compound used herein is a compound capable of suppressing the rate of diffusion when the acid generated by the photoacid generator diffuses within the resist film. The inclusion of this type of basic compound holds down the rate of acid diffusion within the resist film, resulting in better resolution. In addition, it suppresses changes in sensitivity following exposure, thus reducing substrate and environment dependence, as well as improving the exposure latitude and the pattern profile.
- Examples of basic compounds include primary, secondary, and tertiary aliphatic amines, mixed amines, aromatic amines, heterocyclic amines, carboxyl group-bearing nitrogenous compounds, sulfonyl group-bearing nitrogenous compounds, hydroxyl group-bearing nitrogenous compounds, hydroxyphenyl group-bearing nitrogenous compounds, alcoholic nitrogenous compounds, amide derivatives, and imide derivatives.
- Suitable primary aliphatic amines include ammonia, methylamine, ethylamine, n-propylamine, isopropylamine, n-butylamine, iso-butylamine, sec-butylamine, tert-butylamine, pentylamine, tert-amylamine, cyclopentylamine, hexylamine, cyclohexylamine, heptylamine, octylamine, nonylamine, decylamine, dodecylamine, cetylamine, methylenediamine, ethylenediamine, and tetraethylenepentamine.
- Suitable secondary aliphatic amines include dimethylamine, diethylamine, di-n-propylamine, di-iso-propylamine, di-n-butylamine, di-iso-butylamine, di-sec-butylamine, dipentylamine, dicyclopentylamine, dihexylamine, dicyclohexylamine, diheptylamine, dioctylamine, dinonylamine, didecylamine, didodecylamine, dicetylamine, N,N-dimethylmethylenediamine, N,N-dimethylethylenediamine, and N,N-dimethyltetraethylenepentamine.
- Suitable tertiary aliphatic amines include trimethylamine, triethylamine, tri-n-propylamine, tri-iso-propylamine, tri-n-butylamine, tri-iso-butylamine, tri-sec-butylamine, tripentylamine, tricyclopentylamine, trihexylamine, tricyclohexylamine, triheptylamine, trioctylamine, trinonylamine, tridecylamine, tridodecylamine, tricetylamine, N,N,N′,N′-tetramethylmethylenediamine, N,N,N′,N′-tetramethylethylenediamine, and N,N,N′,N′-tetramethyltetraethylenepentamine.
- Examples of suitable mixed amines include dimethylethylamine, methylethylpropylamine, benzylamine, phenethylamine, and benzyldimethylamine.
- suitable aromatic and heterocyclic amines include aniline derivatives (e.g., aniline, N-methylaniline, N-ethylaniline, N-propylaniline, N,N-dimethylaniline, 2-methylaniline, 3-methylaniline, 4-methylaniline, ethylaniline, propylaniline, trimethylaniline, 2-nitroaniline, 3-nitroaniline, 4-nitroaniline, 2,4-dinitroaniline, 2,6-dinitroaniline, 3,5-dinitroaniline, and N,N-dimethyltoluidine), diphenyl(p-tolyl)amine, methyldiphenylamine, triphenylamine, phenylenediamine, naphthylamine, diaminonaphthalene, pyrrol
- suitable carboxyl group-bearing nitrogenous compounds include aminobenzoic acid, indolecarboxylic acid, and amino acid derivatives (e.g. nicotinic acid, alanine, alginine, aspartic acid, glutamic acid, glycine, histidine, isoleucine, glycylleucine, leucine, methionine, phenylalanine, threonine, lysine, 3-aminopyrazine-2-carboxylic acid, and methoxyalanine).
- aminobenzoic acid e.g. nicotinic acid, alanine, alginine, aspartic acid, glutamic acid, glycine, histidine, isoleucine, glycylleucine, leucine, methionine, phenylalanine, threonine, lysine, 3-aminopyrazine-2-carboxylic acid, and meth
- suitable sulfonyl group-bearing nitrogenous compounds include 3-pyridinesulfonic acid and pyridinium p-toluenesulfonate.
- suitable hydroxyl group-bearing nitrogenous compounds, hydroxyphenyl group-bearing nitrogenous compounds, and alcoholic nitrogenous compounds include 2-hydroxypyridine, aminocresol, 2,4-quinolinediol, 3-indolemethanol hydrate, monoethanolamine, diethanolamine, triethanolamine, N-ethyldiethanolamine, N,N-diethylethanolamine, triisopropanolamine, 2,2′-iminodiethanol, 2-aminoethanol, 3-amino-1-propanol, 4-amino-1-butanol, 4-(2-hydroxyethyl)morpholine, 2-(2-hydroxyethyl)pyridine, 1-(2-hydroxyethyl)piperazine, 1-[2-(2-hydroxyethoxy)ethyl]-piperazine, piper
- Suitable amide derivatives include formamide, N-methylformamide, N,N-dimethylformamide, acetamide, N-methylacetamide, N,N-dimethylacetamide, propionamide, and benzamide.
- Suitable imide derivatives include phthalimide, succinimide, and maleimide.
- n is equal to 1, 2 or 3;
- Y is independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 20 carbon atoms which may contain a hydroxyl group or ether; and
- X is independently selected from groups of the following general formulas (X1) to (X3), and two or three X's may bond together to form a ring.
- R 300 , R 302 and R 305 are independently straight or branched alkylene groups of 1 to 4 carbon atoms;
- R 301 and R 304 are independently hydrogen, straight, branched or cyclic alkyl groups of 1 to 20 carbon atoms, which may contain at least one hydroxyl group, ether, ester or lactone ring;
- R 303 is a single bond or a straight or branched alkylene group of 1 to 4 carbon atoms.
- Illustrative examples of the compounds of formula (B1) include tris(2-methoxymethoxyethyl)amine, tris ⁇ 2-(methoxyethoxy)ethyl ⁇ amine, tris ⁇ 2-(2-methoxyethoxymethoxy)ethyl ⁇ amine, tris ⁇ 2-(1-methoxyethoxy)ethyl ⁇ amine, tris ⁇ 2-(1-ethoxyethoxy)ethyl ⁇ amine, tris ⁇ 2—(1-ethoxypropoxy)ethyl ⁇ amine, tris[2- ⁇ 2-(2-hydroxyethoxy)ethoxy ⁇ -ethyl]amine, 4,7,13,16,21,24-hexaoxa-1,10-diazabicyclo-[8.8.8]hexacosane, 4,7,13,18-tetraoxa-1,10-diazabicyclo-[8.5.5]eicosane, 1,4,10,13-tetraoxa
- the basic compound is preferably formulated in an amount of 0.001 to 10 parts, and especially 0.01 to 1 part, per part of the photoacid generator. Less than 0.001 part of the basic compound fails to achieve the desired effects thereof, while the use of more than 10 parts would result in too low a sensitivity and resolution.
- a compound bearing a ⁇ C—COOH group in a molecule may be blended.
- exemplary, non-limiting compounds bearing a ⁇ C—COOH group include one or more compounds selected from Groups I and II below. Including this compound improves the PED stability of the resist and ameliorates edge roughness on nitride film substrates.
- R 408 is hydrogen or methyl;
- R 402 and R 403 are each hydrogen or a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms;
- R 404 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or a —(R 409 ) h —COOR′ group (R′ being hydrogen or —R 409 —COOH);
- R 405 is —(CH 2 ) i — (wherein i is 2 to 10), an arylene of 6 to 10 carbon atoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom;
- R 406 is an alkylene of 1 to 10 carbon atoms, an arylene of 6 to 10 carbon atoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom;
- R 407 is hydrogen, a straight or branched alkyl or alken
- Illustrative, non-limiting examples of the compound bearing a ⁇ C—COOH group include compounds of the general formulas AI-1 to AI-14 and AII-1 to AII-10-below.
- R′′ is hydrogen or a CH 2 COOH group such that the CH 2 COOH group accounts for 10 to 100 mol % of R′′ in each compound, a and K are as defined above.
- the compound bearing a ⁇ C—COOH group within the molecule may be used singly or as combinations of two or more thereof.
- the compound bearing a ⁇ C—COOH group within the molecule is added in an amount ranging from 0 to 5 parts, preferably 0.1 to 5 parts, more preferably 0.1 to 3 parts, further preferably 0.1 to 2 parts, per 100 parts of the base resin. More than 5 parts of the compound can reduce the resolution of the resist composition.
- the resist composition of the invention may additionally include an acetylene alcohol derivative for the purpose of enhancing the shelf stability.
- Preferred acetylene alcohol derivatives are those having the general formula (S1) or (S2) below.
- R 501 , R 502 , R 503 , R 504 and R 505 are each hydrogen or a straight, branched, or cyclic alkyl of 1 to 8 carbon atoms; and X and Y are each 0 or a positive number, satisfying 0 ⁇ X ⁇ 30, 0 ⁇ Y ⁇ 30, and 0 ⁇ X+Y ⁇ 40.
- Preferable examples of the acetylene alcohol derivative include Surfynol 61, Surfynol 82, Surfynol 104, Surfynol 104E, Surfynol 104H, Surfynol 104A, Surfynol TG, Surfynol PC, Surfynol 440, Surfynol 465, and Surfynol 485 from Air Products and Chemicals Inc., and Surfynol E1004 from Nisshin Chemical Industry K.K.
- the acetylene alcohol derivative is preferably added in an amount of 0.01 to 2% by weight, and more preferably 0.02 to 1% by weight, per 100% by weight of the resist composition. Less than 0.01% by weight would be ineffective for improving coating characteristics and shelf stability, whereas more than 2% by weight would result in a resist having a low resolution.
- the resist composition of the invention may include, as an optional ingredient, a surfactant which is commonly used for improving the coating characteristics.
- Optional ingredients may be added in conventional amounts so long as this does not compromise the objects of the invention.
- Nonionic surfactants are preferred, examples of which include perfluoroalkylpolyoxyethylene ethanols, fluorinated alkyl esters, perfluoroalkylamine oxides, perfluoroalkyl EO-addition products, and fluorinated organosiloxane compounds.
- Useful surfactants are commercially available under the trade names Florade FC-430 and FC-431 from Sumitomo 3M K.K., Surflon S-141 and S-145 from Asahi Glass K.K., Unidine DS-401, DS-403 and DS-451 from Daikin Industry K.K., Megaface F-8151 from Dai-Nippon Ink & Chemicals K.K., and X-70-092 and X-70-093 from Shin-Etsu Chemical Co., Ltd.
- Preferred surfactants are Florade FC-430 from Sumitomo 3M K.K. and X-70-093 from Shin-Etsu Chemical Co., Ltd.
- Pattern formation using the resist composition of the invention may be carried out by a known lithographic technique.
- the resist composition is applied onto a substrate such as a silicon wafer by spin coating or the like to form a resist film having a thickness of 0.3 to 2.0 ⁇ m, which is then pre-baked on a hot plate at 60 to 150° C. for 1 to 10 minutes, and preferably at 80 to 130° C. for 1 to 5 minutes.
- a patterning mask having the desired pattern is then placed over the resist film, and the film exposed through the mask to an electron beam or to high-energy radiation such as deep-UV rays, an excimer laser, or x-rays in a dose of about 1 to 200 mJ/cm 2 , and preferably about 10 to 100 mJ/cm 2 , then post-exposure baked (PEB) on a hot plate at 60 to 150° C. for 1 to 5 minutes, and preferably at 80 to 130° C. for 1 to 3 minutes.
- PEB post-exposure baked
- aqueous alkali solution such as a 0.1 to 5% (preferably 2 to 3%) aqueous solution of tetramethylammonium hydroxide (TMAH), this being done by a conventional method such as dipping, puddling, or spraying for a period of 0.1 to 3 minutes, and preferably 0.5 to 2 minutes.
- TMAH tetramethylammonium hydroxide
- the resist composition of the invention is best suited to fine pattern formation with, in particular, deep-UV rays having a wavelength of 248 to 193 nm, an excimer laser, x-rays, or an electron beam.
- the desired pattern may not be obtainable outside the upper and lower limits of the above range.
- the resist composition comprising the polymer as a base resin lends itself to micropatterning with electron beams or deep-UV rays since it is sensitive to high-energy radiation and has excellent sensitivity, resolution, and etching resistance. Especially because of the minimized absorption at the exposure wavelength of an ArF or KrF excimer laser, a finely defined pattern having sidewalls perpendicular to the substrate can easily be formed.
- Ester compounds and polymers containing the same were synthesized according to the following formulation.
- FIG. 1 shows the spectrum of Monomer 1 by 1 H-NMR (CDCl 3 , 270 MHz).
- Polymers 2 to 18 were synthesized by the same procedure as above or a well-known procedure.
- Resist compositions were formulated using inventive polymers and examined for resolution upon KrF excimer laser exposure.
- Resist compositions were prepared by using Polymers 1 to 18 as the base resin, and dissolving the polymer, a photoacid generator (designated as PAG 1 and 2), a dissolution regulator (designated as DRR 1 to 4), a basic compound, and a compound having a ⁇ C—COOH group in the molecule (ACC 1 and 2) in a solvent in accordance with the formulation shown in Tables 1 and 2. These compositions were each filtered through a Teflon filter (pore diameter 0.2 ⁇ m), thereby giving resist solutions.
- the solvents and basic compounds used are as follows. It is noted that the solvents contained 0.01% by weight of surfactant Florade FC-430 (Sumitomo 3M).
- PGMEA propylene glycol methyl ether acetate
- TMMEA trismethoxymethoxyethylamine
- TMEMEA trismethoxyethoxymethoxyethylamine
- resist solutions were spin-coated onto silicon wafers, then baked on a hot plate at 110° C. for 90 seconds to give resist films having a thickness of 0.5 ⁇ m.
- the resist films were exposed using an KrF excimer laser stepper (Nikon Corporation; NA 0.5), then baked (PEB) at 110° C. for 90 seconds, and developed with a solution of 2.38% TMAH in water, thereby giving positive patterns.
- the resulting resist patterns were evaluated as described below. First, the sensitivity (Eth, mJ/cm 2 ) was determined. Next, the optimal dose (Eop, mJ/cm 2 ) was defined as the dose which provides a 1:1 resolution at the top and bottom of a 0.30 ⁇ m line-and-space pattern, and the resolution of the resist under evaluation was defined as the minimum line width ( ⁇ m) of the lines and spaces that separated at this dose. The shape of the resolved resist pattern was examined under a scanning electron microscope.
- resist compositions were formulated and examined for resolution upon KrF excimer laser exposure.
- Resist compositions were prepared as in Example I except that Polymers 19 and 20 shown below were used as the base resin, in accordance with the formulation shown in Table 3.
- Resist compositions were formulated using inventive polymers and examined for resolution upon ArP excimer laser exposure.
- Resist compositions were prepared as in Example I in accordance with the formulation shown in Table 4.
- the resulting resist solutions were spin-coated onto silicon wafers, then baked on a hot plate at 110° C. for 90 seconds to give resist films having a thickness of 0.5 ⁇ m.
- the resist films were exposed using an ArF excimer laser stepper (Nikon Corporation; NA 0.55), then baked (PEB) at 110° C. for 90 seconds, and developed with a solution of 2.38% TMAH in water; thereby giving positive patterns.
- the resulting resist patterns were evaluated as described below. First, the sensitivity (Eth, mJ/cm 2 ) was determined. Next, the, optimal dose (Eop, mJ/cm 2 ) was defined as the dose which provides a 1:1 resolution at the top and bottom of a 0.25 ⁇ m line-and-space pattern, and the resolution of the resist under evaluation was defined as the minimum line width ( ⁇ m) of the lines and spaces that separated at this dose. The shape of the, resolved resist pattern was examined under a scanning electron microscope.
Abstract
R1 is H, methyl or CH2CO2R3, R2 is H, methyl or CO2R3, R3 is C1-C15 alkyl, k is 0 or 1, Z is a divalent C2-C20 hydrocarbon group which forms a single ring or bridged ring with the carbon atom and which may contain a hetero atom, m is 0 or 1, n is 0, 1, 2 or 3, and 2m+n=2 or 3. A resist composition comprising as the base resin a polymer resulting from the ester compound is sensitive to high-energy radiation, has excellent sensitivity, resolution, and etching resistance, and is suited for micropatterning using electron-beams or deep-UV.
Description
- 1. Field of the Invention
- The present invention relates to (1) a novel ester compound, (2) a polymer comprising units of the ester compound which is blended as a base resin to formulate a resist composition having a high sensitivity, resolution and etching resistance, and in particular, suitable as micropatterning material for VLSI fabrication, (3) a method for preparing the polymer, (4) a resist composition comprising the polymer, and (5) a patterning process using the resist composition.
- 2. Prior Art
- While a number of recent efforts are being made to achieve a finer pattern rule in the drive for higher integration and operating speeds in LSI devices, deep-ultraviolet lithography is thought to hold particular promise as the next generation in microfabrication technology. In particular, photolithography using a KrF or ArF excimer laser as the light source is strongly desired to reach the practical level as the micropatterning technique capable of achieving a feature size of 0.3 μm or less.
- For resist materials for use with a KrF excimer lasers, polyhydroxystyrene having a practical level of transmittance and etching resistance is, in fact, a standard base resin. For resist materials for use with ArF excimer lasers, polyacrylic or polymethacrylic acid derivatives and polymers comprising aliphatic cyclic compounds in the backbone are under investigation. In either case, the basic concept is that some or all of alkali soluble sites of alkali soluble resin are protected with acid labile or eliminatable groups. The overall performance of resist material is adjusted by a choice from among a variety of acid eliminatable protective groups.
- Exemplary acid eliminatable protective groups include tert-butoxycarbonyl (JP-B 2-27660), tert-butyl (JP-A 62-115440, JP-A 5-80515, and J. Photopolym. Sci. Technol. 7 [3], 507 (1994)), 2-tetrahydropyranyl (JP-A 2-19847, 5-80515 and 5-88367), and 1-ethoxyethyl (JP-A 2-19847 and 4-215661). While it is desired to achieve a finer pattern rule, none of these acid eliminatable protective groups are deemed to exert satisfactory performance.
- More particularly, tert-butoxycarbonyl and tert-butyl are extremely less reactive with acids so that a substantial quantity of energy radiation must be irradiated to generate a sufficient amount of acid in order to establish a difference in rate of dissolution before and after exposure. If a photoacid generator of the strong acid type is used, the exposure can be reduced to a relatively low level because reaction can proceed with a small amount of acid generated. However, in this event, the deactivation of the generated acid by air-borne basic substances has a relatively large influence, giving rise to such problems as a T-top pattern. On the other hand, 2-tetrahydropyranyl and 1-ethoxyethyl are so reactive with acids that with the acid generated by exposure, elimination reaction may randomly proceed without a need for heat treatment, with the result that substantial dimensional changes occur between exposure and heat treatment/development. Where these groups are used as protective groups for carboxylic acid, they have a low dissolution inhibiting effect to alkali, resulting in a high rate of dissolution in unexposed areas and film thinning during development. If highly substituted polymers are used to avoid such inconvenience, there results an extreme drop of heat resistance. These resins fail to provide a difference in rate of dissolution before and after exposure, resulting in resist materials having a very low resolution.
- Therefore, an object of the present invention is to provide (1) a novel ester compound capable of forming an acid-decomposable polymer, (2) a polymer which is blended as a base resin to formulate a resist composition having a higher sensitivity, resolution and etching resistance than conventional resist compositions, (3) a method for preparing the polymer, (4) a resist composition comprising the polymer as a base resin, and (5) a patterning process using the resist composition.
- The inventor has found that a novel ester compound of the following general formula (1) obtained by the method to be described later is useful in preparing an acid-decomposable polymer; that a resist composition comprising as the base resin a novel polymer prepared from the ester compound to a weight average molecular weight of 1,000 to 500,000 has a high sensitivity, resolution and etching resistance; and that this resist composition lends itself to precise micropatterning.
-
- Herein R 1 is hydrogen, methyl or CH2CO2R1, R2 is hydrogen, methyl or CO2R3, R3 is a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, k is 0 or 1, Z is a divalent hydrocarbon group of 2 to 20 carbon atoms which forms a single ring or bridged ring with the carbon atom and which may contain a hetero atom, m is 0 or 1, n is 0, 1, 2 or 3, and 2 m+n is 2 or 3.
-
- Herein R 1, R2Z, k, m and n are as defined above.
-
- Herein R 1, R2 and k are as defined above; R4 is hydrogen or a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms; at least one of R5 to R8 is a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms, and the remainders are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, or R5 to R8, taken together, may form a ring, and when they form a ring, at least one of R5 to R8 is a carboxyl or hydroxyl-containing divalent hydrocarbon group of 1 to 15 carbon atoms, and the remainders are independently a single bond or a straight, branched or cyclic alkylene group of 1 to 15 carbon atoms; R9 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO2— partial structure; at least one of R10 to R13 is a monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO2— partial structure, and the remainders are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, or R10 to R13, taken together, may form a ring, and when they form a ring, at least one of R10 to R13 is a divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO2— partial structure, and the remainders are independently a single bond or a straight, branched or cyclic alkylene group of 1 to 15 carbon atoms; R14 is a polycyclic hydrocarbon group of 7 to 15 carbon atoms or an alkyl group containing such a polycyclic hydrocarbon group; RS is an acid labile group; X is —CH2— or —O—; and Y is —O— or —(NR16)— wherein R16 is hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms.
- In a third aspect, the invention provides a method for preparing a polymer comprising the step of effecting radical polymerization, anionic polymerization or coordination polymerization between an ester compound of the general formula (1) and another compound having a carbon-to-carbon double bond.
- In a fourth aspect, the invention provides a resist composition comprising the polymer defined above, and more specifically, a resist composition comprising the polymer defined above, a photoacid generator, and an organic solvent.
- In a fifth aspect, the invention provides a process for forming a resist pattern comprising the steps of applying the resist composition defined above onto a substrate to form a coating; heat treating the coating and then exposing it to high-energy radiation or electron beams through a photo mask; and optionally heat treating the exposed coating and developing it with a developer.
- The ester compound of formula (1) and the polymer comprising units of formula (1a-1) and/or (1a-2) employ a cycloalkylcycloalkyl or cycloalkylcycloalkenyl group of the following formula (1b) as the acid-eliminatable protective group, thereby overcoming the drawbacks of the tert-butoxycarbonyl and tert-butyl groups having low reactivity with acid as well as the 2-tetrahydropyranyl and 1-ethoxyethyl groups having too high reactivity with acid and low resistance to alkaline developers.
- Herein, Z, m and n are as defined above, the broken line denotes a valence bond.
- The ester compound of formula (1) is decomposed under acidic conditions while generating an olefin compound and a carboxylic acid. This mechanism itself is apparently identical with the decomposition of tertiary alkyl esters such as tert-butyl esters although the decomposition rate significantly differs between them. In the acidolysis of a tertiary alkyl ester, carboxylic acid and a tertiary alkyl cation generate first, then the extinction of the cation due to proton release occurs to form an olefin compound, whereby the reaction comes to an end. The rate-determining stage of this decomposition reaction is the late stage of cation extinction. The progress of this stage is very rapid in the case of the ester compound of formula (1), probably because the moderated distortion of the ring and the formation of a conjugated system produce a strong thrust. The olefin compound produced has a very low re-reactivity. As a consequence, the ester compound of formula (1) provides a high degree of acidolysis surpassing other tertiary alkyl esters.
- Since the ester compound of formula (1) is basically a tertiary alkyl ester, it, when formulated into a resist composition, does not allow for the infinite progress of decomposition in a duration between exposure and development and film thinning during development as found with 2-tetrahydropyranyl and 1-ethoxyethyl. When a polymer comprising constituent units derived from the compound is formulated as the base resin, there is obtained a resist composition which possesses a high sensitivity and resolution and has a high dissolution contrast in that the unexposed areas have a very slow rate of dissolution while the areas having undergone an adequate dose of exposure quickly become alkali soluble. Additionally, very high etching resistance is exerted by virtue of the robust aliphatic ring positioned on the backbone of the polymer and a plurality of aliphatic rings contained in the protective group of formula (1b).
- The only FIGURE, FIG. 1 is a diagram showing the 1H-NMR spectrum of the monomer obtained in Synthesis Example 1.
- Ester Compound
-
- Herein, R 1 is hydrogen, methyl or CH2CO2R3. R2 is hydrogen, methyl or CO2R3. R3 stands for straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms, such as, for example, methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, tert-amyl, n-pentyl, n-hexyl, cyclopentyl, cyclohexyl, ethylcyclopentyl, butylcyclopentyl, ethylcyclohexyl, butylcyclbhexyl, adamantyl, ethyladamantyl and butyladamantyl.
- Z is a divalent hydrocarbon group of 2 to 20 carbon atoms which forms a single ring or bridged ring with the carbon atom and which may contain a hetero atom, for example, single ringforming hydrocarbon groups such as cyclopropane, cyclobutane, cyclopentane, cyclohexane, cycloheptane, and cyclooctane; bridged ring-forming hydrocarbon groups such as bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane, bicyclo[4.4.0]decane and tricyclo[5.2.1.0 2,6]decane; and ring-forming hydrocarbon groups in which some of the hydrogen atoms in the foregoing groups are substituted with alkyl, hydroxy, alkoxy, acyloxy, alkylcarbonyl, hydroxycarbonyl, alkoxycarbonyl and oxo, among others.
- The letter k is 0 or 1, m is 0 or 1, n is 0, 1, 2 or 3, and 2 m+n is equal to 2 or 3.
-
-
- Herein, R 1, R2, Z, k, m and n are as defined above, and Hal is bromine or chlorine.
- Step A is to effect nucleophilic addition reaction with the carbonyl on a cyclic ketone compound to form a tertiary alcohol. This stage is typified by Grignard reaction, but not limited thereto. Reaction readily takes place under well-known conditions, preferably by mixing the reactants: cyclic ketone compound, alkyl halide and magnesium in a solvent such as tetrahydrofuran or diethyl ether, while heating or cooling the reaction mixture if desired.
- Step B is to convert the tertiary alcohol to an ester of unsaturated acid. Reaction readily takes place under well-known conditions, preferably by mixing the reactants: tertiary alcohol, unsaturated carboxylic halide (e.g., acrylic chloride or methacrylic chloride) and base (e.g., triethylamine) in a solvent such as methylene chloride, while cooling the reaction mixture if desired.
- Step C is to construct an alicyclic skeleton through Diels-Alder reaction. Reaction readily takes place under well-known conditions, preferably by mixing the reactants: ester of unsaturated acid and cyclopentadiene in a solventless system or in a solvent such as benzene, optionally in the presence of a catalyst such as boron trifluoride, while heating or cooling the reaction mixture if desired.
- Polymer
- In the second aspects the invention provides a polymer or high molecular weight compound comprising recurring units of the following general formula-(1a-1) and/or (1a-2) derived from an ester compound of the general formula (1), and having a weight average molecular weight of 1,000 to 500,000, and preferably 5,000 to 100,000.
- Herein R 1, R2, Z, k, m and n are as defined above.
-
-
- Herein R 1 and R2 are as defined above.
- R 4 stands for hydrogen or carboxyl or hydroxyl-containing monovalent hydrocarbon groups of 1 to 15 carbon atoms, for example, carboxyethyl, carboxybutyl, carboxycyclopentyl, carboxycyclohexyl, carboxynorbornyl, carboxyadamantyl,:hydroxyethyl, hydroxybutyl, hydroxycyclopentyl, hydroxycyclohexyl, hydroxynorbornyl, and hydroxyadamantyl.
- At least one of R 5 to R8 is a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms, and the remaining R's are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms. Examples of the carboxyl or hydroxyl-bearing monovalent hydrocarbon group of 1 to 15 carbon atoms include carboxy, carboxymethyl, carboxyethyl, carboxybutyl, hydroxymethyl, hydroxyethyl, hydroxybutyl, 2-carboxyethoxycarbonyl, 4-carboxybutoxycarbonyl, 2:-hydroxyethoxycarbonyl, 4-hydroxybutoxycarbonyl, carboxycyclopentyloxycarbonyl, carboxycyclohexyloxycarbonyl, carboxynorbornyloxycarbonyl, carboxyadamantyloxycarbonyl, hydroxycyclopentyloxycarbonyl, hydroxycyclohexyloxycarbonyl, hydroxynorbornyloxycarbonyl, and hydroxyadamantyloxycarbonyl. Examples of the straight, branched or cyclic alkyl group of 1 to 15 carbon atoms are the same as exemplified for R3. R5 to R8, taken together, may form a ring, and in that event, at least one of R5 to R8 is a divalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms. Examples of the carboxyl or hydroxyl-bearing divalent hydrocarbon group of 1 to 15 carbon atoms include the groups exemplified as the carboxyl or hydroxyl-bearing monovalent hydrocarbon group, with one hydrogen atom eliminated therefrom. Examples of the straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms include the groups exemplified for R3, with one hydrogen atom eliminated therefrom.
- R 9 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO2— partial structure, for example, 2-oxooxolan-3-yl, 4,4-dimethyl-2-oxooxolan-3-yl, 4-methyl-2-oxooxan-4-yl, 2-oxo-1,3-dioxolan-4-ylmethyl, and 5-methyl-2-oxooxolan-5-yl. At least one of R10 to R13 is a monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO2— partial structure, while the remaining R's are independently hydrogen or straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms. Examples of the monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO2— partial structure include 2-oxooxolan-3-yloxycarbonyl, 4,4-dimethyl-2-oxooxolan-3-yloxycarbonyl, 4-methyl-2-oxooxan-4-yloxycarbonyl, 2-oxo-1,3-dioxolan-4-ylmethyloxycarbonyl, and 5-methyl-2-oxooxolan-5-yloxycarbonyl. Examples of the straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms are the same as exemplified for R3. R10 to R13, taken together, may form a ring, and in that event, at least one of R10 to R13 is a divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO2— partial structure, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms. Examples of the divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO2— partial structure include 1-oxo-2-oxapropane-1,3-diyl, 1,3,-dioxo-2-oxapropane-1,3-diyl, 1-oxo-2-oxabutane-1,4-diyl, and 1,3-dioxo-2-oxabutane-1,4-diyl, as well as the groups exemplified as the monovalent hydrocarbon group containing a —CO2— partial structure, with one hydrogen atom eliminated therefrom. Examples of the straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms include the groups exemplified for R3, with one hydrogen atom eliminated therefrom.
- R 14 is a polycyclic hydrocarbon group having 7 to 15 carbon atoms or an alkyl group containing a polycyclic hydrocarbon group, for example, norbornyl, bicyclo[3.3.1]-nonyl, tricyclo[5.2.1.02,6]decyl, adamantyl, ethyladamantyl, butyladamantyl, norbornylmethyl, and adamantylmethyl.
- R 15 is an acid labile group, and k is 0 or 1.
- X is —CH 2— or —O—.
- Y is —O— or —(NR 16)— wherein R16 is hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atom's, examples of which are as described for R3.
- The acid labile groups represented by R 15 may be selected from a variety of such groups. Examples of the acid labile group are groups of the following general formulae (L1) to (L4), tertiary alkyl groups of 4 to 20 carbon atoms, preferably 4 to 15 carbon atoms, trialkylsilyl groups in which each alkyl moiety has 1 to 6 carbon atoms, and oxoalkyl groups of 4 to 20 carbon atoms.
- R L01 and RL02 are hydrogen or straight, branched or cyclic alkyl groups of 1 to 18 carbon atoms, preferably 1 to 10 carbon atoms. Exemplary alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, cyclopentyl, cyclohexyl, 2-ethylhexyl, and n-octyl. RL03 is a monovalent hydrocarbon group of 1 to-18 carbon atoms, preferably 1 to 10 carbon atoms, which may contain a hetero atom such as oxygen, examples of which include unsubstituted straight, branched or cyclic alkyl groups and straight, branched or cyclic alkyl groups in which some hydrogen atoms are replaced by hydroxyl, alkoxy, oxo, amino, alkylamino or the like. Illustrative examples are the substituted alkyl groups shown below.
- A pair of R L01 and RL02, RL01 and RL03, or RL02 and RL03 may form a ring. Each of RL01, RL02 and RL03 is a straight or branched alkylene group of 1 to 18 carbon atoms, preferably 1 to 10 carbon atoms when they form a ring.
- R L04 is a tertiary-alkyl-group of 4 to 20 carbon atoms, preferably 4 to 15 carbon atoms, a trialkylsilyl group in which each alkyl moiety has 1 to 6 carbon atoms, an oxoalkyl group of 4 to 20 carbon atoms, or a group of formula (L1). Exemplary tertiary alkyl groups are tert-butyl, tert-amyl, 1,1-diethylpropyl, 1-ethylcyclopentyl, 1-butylcyclopentyl, 1-ethylcyclohexyl, 1-butylcyclohexyl,-1-ethyl-2-cyclopentenyl, 1-ethyl-2-cyclohexenyl, and 2-methyl-2-adamantyl. Exemplary trialkylsilyl groups are trimethylsilyl, triethylsilyl, and dimethyl-tert-butylsilyl. Exemplary oxoalkyl groups are 3-oxocyclohexyl, 4-methyl-2-oxooxan-4-yl, and 5-methyl-5-oxooxolan-4-yl. Letter y is an integer of 0 to 6.
- R L05 is a straight or branched alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms. Examples of the straight, branched or cyclic alkyl group include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, tert-amyl, n-pentyl and n-hexyl. Exemplary aryl groups are phenyl, methylphenyl, naphthyl, anthryl, phenanthryl, and pyrenyl. Letter m is equal to 0 or 1, n is equal to 0, 1, 2 or 3, and 2 m+n is equal to 2 or 3.
- R L06 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms. Examples of these groups are the same as exemplified for RL05.
- R L07 to RL16 independently represent hydrogen or monovalent hydrocarbon groups of 1 to 15 carbon atoms which may contain a hetero atom. Exemplary hydrocarbon groups are straight, branched or cyclic alkyl groups such as methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, tert-amyl, n-pentyl, n-hexyl, n-octyl, n-nonyl, n-decyl, cyclopentyl, cyclohexyl, cyclopentylmethyl, cyclopentylethyl, cyclopentylbutyl, cyclohexylmethyl, cyclohexylethyl and cyclohexylbutyl, and substituted ones of these groups in which some hydrogen atoms are replaced by hydroxyl, alkoxy, carboxy, alkoxycarbonyl, oxo, amino, alkylamino, cyano, mercapto, alkylthio, sulfo or other groups. Alternatively, RL07 to RL16, taken together, form a ring (for example, a pair of RL07 and RL08, RL07 and RL09, RL08 and RL10, RL09 and RL10, RL11 and RL12, RL13 and RL14, or a similar pair form a ring). Each of RL07 to RL16 represents a divalent C1-C15 hydrocarbon group which may contain a hetero atom, when they form a ring, examples of which are the ones exemplified above for the monovalent hydrocarbon groups, with one hydrogen atom being eliminated. Two of RL07 to RL16 which are attached to adjoining carbon atoms (for example, a pair of RL07 and RL09, RL09 and RL15, RL13 and RL15, or a similar pair) may bond together directly to form a double bond.
-
- Of the acid labile groups of formula (L1), the cyclic ones are, for example, tetrahydrofuran-2-yl, 2-methyltetrahydrofuran-2-yl, tetrahydropyran-2-yl, and 2-methyltetrahydropyran-2-yl.
- Examples of the acid labile groups of formula (L2) include tert-butoxycarbonyl, tert-butoxycarbonylmethyl, tert-amyloxycarbonyl, tert-amyloxycarbonylmethyl, 1,1-diethylpropyloxycarbonyl, 1,1-diethylpropyloxycarbonylmethyl, 1-ethylcyclopentyloxycarbonyl, 1-ethylcyclopentyloxycarbonylmethyl-1-ethyl-2-cyclopentenyloxycarbonyl, 1-ethyl-2-cyclopentenyloxycarbonylmethyl, 1-ethoxyethoxycarbonylmethyl, 2-tetrahydropyranyloxycarbonylmethyl, and 2-tetrahydrofuranyloxycarbonylmethyl groups.
- Examples of the acid labile groups of formula (L3) include 1-methylcyclopentyl, 1-ethylcyclopentyl, 1-n-propylcyclopentyl, 1-n-butylcyclopentyl, 1-sec-butylcyclopentyl, 1-methylcyclohexyl, 1-ethylcyclohexyl, 3-methyl-1-cyclopenten-3-yl, 3-ethyl-1-cyclopenten-3′-yl, 3-methyl-1-cyclohexen-3-yl, and 3-ethyl-1-cyclohexen-3-yl groups.
-
- The polymer of the invention may be prepared by polymerizing an ester compound of formula (1) or by copolymerizing a first monomer in the form of an ester compound of formula (1) with a second monomer in the form of at least one compound of formulas (2) to (10). By properly adjusting the proportion of the respective monomers used in the copolymerization reaction, the polymer can be tailored so that it may exert the preferred performance when blended in resist compositions.
- In addition to (i) the monomer of formula (1) and (ii) the monomer or monomers of formulas (2) to (10), the polymer of the invention may have copolymerized therewith (iii) another monomer having a carbon-to-carbon double bond other than (i) and (ii). Examples of the additional monomer (iii) include substituted acrylic acid esters such as methyl methacrylate, methyl crotonate, dimethyl maleate, and dimethyl itaconate, unsaturated carboxylic acids such as maleic acid, fumaric acid and itaconic acid, substituted or unsubstituted norbornenes such as norbornene and methyl norbornene-5-carboxylate, and unsaturated acid anhydrides such as itaconic anhydride.
- The polymers of the invention may contain
- (I) more than 0 mol % to 100 mol %, preferably 20 to 90 mol %, more preferably 30 to 80 mol % of units of formula-(1a-1) and/or (1a-2) derived from the monomer of formula (0.1),
- (II) 0 mol % to less than 100 mol %, preferably 1 to 95 mol %, more preferably 5 to 90 mol % of units of one or more types of formulae (2a) to (10a) derived from the monomers of formulae (2) to (10), and optionally,
- (III) 0 to 80 mole, preferably 0 to 70 mol %, more preferably 0 to 50 molt of units of one or more types derived from the additional monomers (iii).
- The polymers of the invention have a weight average molecular weight of 1,000 to 500,000, preferably 3,000 to 100,000. Outside the range, the etching resistance may become extremely low and the resolution may become low because a substantial difference in rate of dissolution before and after exposure is lost.
- In the method according to the third aspect of the invention, a polymer is prepared by effecting radical polymerization, anionic polymerization or coordination polymerization between an ester compound of formula (1) and another compound having a carbon-to-carbon double bond, which is typically selected from the above-described monomers (ii) and (iii).
- For radical polymerization, preferred reaction conditions include (a) a solvent selected from among hydrocarbons such as benzene, ethers such as tetrahydrofuran, alcohols such as ethanol, and ketones such as methyl isobutyl ketone, (b) a polymerization initiator selected from azo compounds such as 2,2′-azobisisobutyronitrile and peroxides such as benzoyl peroxide and lauroyl peroxide, (c) a temperature of about 0° C. to about 100° C., and (d) a time of about ½ hour to about 48 hours. Reaction conditions outside the described range may be employed if desired.
- For anionic polymerization, preferred reaction conditions include (a) a solvent selected from among hydrocarbons such as benzene, ethers such as tetrahydrofuran, and liquid ammonia, (b) a polymerization initiator selected from metals such as sodium and potassium, alkyl metals such as n-butyllithium and sec-butyllithium, ketyl, and Grignard reagents, (c) a temperature of about −78° C. to about 0° C., (d) a time of about ½ hour to about 48 hours, and (e) a stopper selected from among proton-donative compounds such as methanol, halides such as methyl iodide, and electrophilic compounds. Reaction conditions outside the described range may be employed if desired.
- For coordination polymerization, preferred reaction conditions include (a) a solvent selected from among hydrocarbons such as n-heptane and toluene, (b) a catalyst selected from Ziegler-Natta catalysts comprising a transition metal (e.g., titanium) and alkylaluminum, Phillips catalysts of metal oxides having chromium or nickel compounds carried thereon, and olefin-metathesis mixed catalysts as typified by tungsten and rhenium mixed catalysts, (c) a temperature of about 0° C. to about 100° C., and (d) a time of about ½ hour to about 48 hours. Reaction conditions outside the described range may be employed if desired.
- Resist Composition
- Since the polymer of the invention is useful as the base polymer of a resist composition, the other aspect of the invention provides a resist composition comprising the polymer. Specifically, the resist composition is defined as comprising the polymer, a photoacid generator, and an organic solvent.
- Photoacid Generator
- The photoacid generator is a compound capable of generating an acid upon exposure to high energy radiation or electron beams and includes the following:
- (i) onium salts of the formula (P1a-1), (P1a-2) or (P1b),
- (ii) diazomethane derivatives of the formula (P2),
- (iii) glyoxime derivatives of the formula (P3),
- (iv) bissulfone derivatives of the formula (P4),
- (v) sulfonic acid esters of N-hydroxyimide compounds of the formula (P5),
- (vi) P-ketosulfonic acid derivatives,
- (vii) disulfone derivatives,
- (viii) nitrobenzylsulfonate derivatives, and
- (ix) sulfonate derivatives.
- These photoacid generators are described in detail.
-
- Herein, R 101a, R101b, and R101c independently represent straight, branched or cyclic alkyl, alkenyl, oxoalkyl or oxoalkenyl groups of 1 to 12 carbon atoms, aryl groups of 6 to 20 carbon atoms, or aralkyl or aryloxoalkyl groups of 7 to 12 carbon atoms, wherein some or all of the hydrogen atoms may be replaced by alkoxy or other groups. Also, R101b and R101c, taken together, may form a ring. R101b and R101c each are alkylene groups of 1 to 6 carbon atoms when they form a ring. K− is a non-nucleophilic counter ion.
- R 101a, R101b, and R101c may be the same or different and are illustrated below. Exemplary alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, heptyl, octyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclopropylmethyl, 4-methylcyclohexyl, cyclohexylmethyl, norbornyl, and adamantyl. Exemplary alkenyl groups include vinyl, allyl, propenyl, butenyl, hexenyl, and cyclohexenyl. Exemplary oxoalkyl groups include 2-oxocyclopentyl and 2-oxocyclohexyl as well as 2-oxopropyl, 2-cyclopentyl-2-oxoethyl, 2-cyclohexyl-2-oxoethyl, and 2-(4-methylcyclohexyl)-2-oxoethyl. Exemplary aryl groups include phenyl and naphthyl; alkoxyphenyl groupssuch as p-methoxyphenyl, m-methoxyphenyl, o-methoxyphenyl, ethoxyphenyl, p-tert-butoxyphenyl, and m-tert-butoxyphenyl; alkylphenyl groups such as 2-methylphenyl, 3-methylphenyl, 4-methylphenyl, ethylphenyl, 4-tert-butylphenyl, 4-butylphenyl, and dimethylphenyl; alkylnaphthyl groups such as methylnaphthyl and ethylnaphthyl; alkoxynaphthyl groups such as methoxynaphthyl and ethoxynaphthyl; dialkylnaphthyl groups such as dimethylnaphthyl and diethylnaphthyl; and dialkoxynaphthyl groups such as dimethoxynaphthyl and diethoxynaphthyl. Exemplary aralkyl groups include benzyl, phenylethyl, and phenethyl. Exemplary aryloxoalkyl groups are 2-aryl-2-oxoethyl groups such as 2-phenyl-2-oxoethyl, 2-(1-naphthyl)-2-oxoethyl, and 2-(2-naphthyl)-2-oxoethyl. Examples of the non-nucleophilic counter ion represented by K− include halide ions such as chloride and bromide ions, fluoroalkylsulfonate ions such as triflate, 1,1,1-trifluoroethanesulfonate, and nonafluorobutanesulfonate arylsulfonate ions such as tosylate, benzenesulfonate, 4-fluorobenzenesulfonate, and 1,2,3,4,5-pentafluorobenzenesulfonate, and alkylsulfonate ions such as mesylate and butanesulfonate.
- Herein, R 102a and R102b independently represent straight, branched or cyclic alkyl groups of 1 to 8 carbon atoms. R103 represents a straight, branched or cyclic alkylene groups of 1 to 10 carbon atoms. R104a and R104b independently represent 2-oxoalkyl groups of 3 to 7 carbon atoms. K− is a non-nucleophilic counter ion.
- Illustrative of the groups represented by R 102a and R102b are methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, heptyl, octyl, cyclopentyl, cyclohexyl, cyclopropylmethyl, 4-methylcyclohexyl, and cyclohexylmethyl. Illustrative of the groups represented by R103 are methylene, ethylene, propylene, butylene, pentylene, hexylene, heptylene, octylene, nonylene, 1,4-cyclohexylene, 1,2-cyclohexylene, 1,3-cyclopentylene, 1,4-cyclooctylene, and 1,4-cyclohexanedimethylene. Illustrative of the: groups represented by R104a and R104b are 2-oxopropyl, 2-oxocyclopentyl, 2-oxocyclohexyl, and 2-oxocycloheptyl. Illustrative examples of the counter ion represented by K− are the same as exemplified for formulae (P1a-1) and (P1a-2).
-
- Herein, R 105 and R106 independently represent straight, branched or cyclic alkyl or halogenated alkyl groups of 1 to 12 carbon atoms, aryl or halogenated aryl groups of 6 to 20 carbon atoms, or aralkyl groups of 7 to 12 carbon atoms.
- Of the groups represented by R 105 and R106, exemplary alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, heptyl, octyl, amyl, cyclopentyl, cyclohexyl, cycloheptyl, norbornyl, and adamantyl. Exemplary halogenated alkyl groups include trifluoromethyl, 1,1,1-trifluoroethyl, 1,1,1-trichloroethyl, and nonafluorobutyl. Exemplary aryl groups include phenyl; alkoxyphenyl groups such as p-methoxyphenyl, m-methoxyphenyl, o-methoxyphenyl, ethoxyphenyl, p-tert-butoxyphenyl, and m-tert-butoxyphenyl; and alkylphenyl groups such as 2-methyl-phenyl, 3-methylphenyl, 4-methylphenyl, ethylphenyl, 4-tertbutylphenyl, 4-butylphenyl, and dimethylphenyl. Exemplary halogenated aryl groups include fluorophenyl, chlorophenyl, and 1,2,3,4,5-pentafluorophenyl. Exemplary aralkyl groups include benzyl and phenethyl.
-
- Herein, R 107, R108, and R109 independently represent straight, branched or cyclic alkyl or halogenated alkyl groups of 1 to 12 carbon atoms, aryl or halogenated aryl groups of 6 to 20 carbon atoms, or aralkyl groups of 7 to 12 carbon atoms. Also, R108 and R109, taken together, may form a ring. R108 and R109 each are straight or branched alkylene groups of 1 to 6 carbon atoms when they form a ring.
- Illustrative examples of the alkyl, halogenated alkyl, aryl, halogenated aryl, and aralkyl groups represented by R 107, R108, and R109 are the same as exemplified for R105 and R106.
- Examples of the alkylene groups represented by R 108 and R109 include methylene, ethylene, propylene, butylene, and hexylene.
-
- Herein, R 101a and R101b are as defined above.
-
- Herein, R 110 is an arylene group of 6 to 10 carbon atoms, alkylene group of 1 to 6 carbon atoms, or alkenylene group of 2 to 6 carbon atoms wherein some or all of the hydrogen atoms may be replaced by straight or branched alkyl or alkoxy groups of 1 to 4 carbon atoms, nitro, acetyl, or phenyl groups. R111 is a straight, branched or cyclic alkyl group of 1 to 8,carbon atoms, alkenyl, alkoxyalkyl, phenyl or naphthyl group wherein some or all of the hydrogen atoms may be replaced by alkyl or alkoxy groups of 1 to 4 carbon atoms, phenyl groups (which may have substituted thereon an alkyl or alkoxy of 1 to 4 carbon atoms, nitro, or acetyl group), hetero-aromatic groups of 3 to 5 carbon atoms, or chlorine or fluorine atoms.
- Of the groups represented by R 110, exemplary arylene groups include 1,2-phenylene and 1,8-naphthylene; exemplary alkylene groups include methylene, 1,2-ethylene, 1,3-propylene, 1,4-butylene, 1-phenyl-1,2-ethylene, and norbornane-2,3-diyl; and exemplary alkenylene groups include 1,2-vinylene, 1-phenyl-1,2-vinylene, and 5-norbornene-2,3-diyl. Of the groups represented by R111, exemplary alkyl groups are as exemplified for R101a to R101c; exemplary alkenyl groups include vinyl, 1-propenyl, allyl, 1-butenyl, 3-butenyl, isoprenyl, 1-pentenyl, 3-pentenyl, 4-pentenyl, dimethylallyl, 1-hexenyl, 3-hexenyl, 5-hexenyl, 1-heptenyl, 3-heptenyl, 6-heptenyl, and 7-octenyl; and exemplary alkoxyalkyl, groups include methoxymethyl, ethoxymethyl, propoxymethyl, butoxymethyl, pentyloxymethyl, hexyloxymethyl, heptyloxymethyl, methoxyethyl, ethoxyethyl, propoxyethyl, butoxyethyl, pentyloxyethyl, hexyloxyethyl, methoxypropyl, ethoxypropyl, propoxypropyl, butoxypropyl, methoxybutyl, ethoxybutyl, propoxybutyl, methoxypentyl, ethoxypentyl, methoxyhexyl, and methoxyheptyl.
- Of the substituents on these groups, the alkyl groups of 1 to 4 carbon atoms include methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl and tert-butyl; the alkoxy groups of 1 to 4,carbon atoms include methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, isobutoxy, and tert-butoxy; the phenyl groups which may have substituted thereon an alkyl or alkoxy of 1 to-4 carbon atoms, nitro, or acetyl group include phenyl, tolyl, p-tert-butoxyphenyl, p-acetylphenyl and p-nitrophenyl; the hetero-aromatic groups of 3 to 5 carbon atoms include pyridyl and furyl.
- Illustrative examples of the photoacid generator include:
- onium salts such as diphenyliodonium trifluoromethanesulfonate, (p-tert-butoxyphenyl)phenyliodonium trifluoromethanesulfonate, diphenyliodonium p-toluenesulfonate, (p-tert-butoxyphenyl)phenyliodonium p-toluenesulfonate, triphenylsulfonium trifluoromethanesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium trifluoromethanesulfonate, bis(p-tert-butoxyphenyl)phenylsulfonium trifluoromethanesulfonate, tris(p-tert-butoxyphenyl)-sulfonium trifluoromethanesulfonate, triphenylsulfonium p-toluenesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium p-toluenesulfonate, bis(p-tert-butoxyphenyl)phenylsulfonium p-toluenesulfonate, tris(p-tert-butoxyphenyl)sulfonium p-toluenesulfonate, triphenylsulfonium nonafluorobutanesulfonate, triphenylsulfonium butanesulfonate, trimethylsulfonium trifluoromethanesulfonate, trimethylsulfonium p-toluenesulfonate, cyclohexylmethyl(2-oxocyclohexyl)sulfonium trifluoromethanesulfonate, cyclohexylmethyl(2-oxocyclohexyl)sulfonium p-toluenesulfonate, dimethylphenylsulfonium trifluoromethanesulfonate, dimethylphenylsulfonium p-toluenesulfonate, dicyclohexylphenylsulfonium trifluoromethanesulfonate, dicyclohexylphenylsulfonium p-toluenesulfonate, trinaphthylsulfonium trifluoromethanesulfonate, cyclohexylmethyl(2-oxocyclohexyl)sulfonium trifluoromethanesulfonate, (2-norbornyl)methyl(2-oxocyclohexyl)sulfonium trifluoromethanesulfonate, ethylenebis-[methyl(2-oxocyclopentyl)sulfonium trifluoromethanesulfonate], and 1,2′-naphthylcarbonylmethyltetrahydrothiophenium triflate; diazomethane derivatives such as bis(benzenesulfonyl)diazomethane, bis(p-toluenesulfonyl)diazomethane, bis(xylenesulfonyl)diazomethane, bis(cyclohexylsulfonyl)diazomethane, bis(cyclopentylsulfonyl)diazomethane bis(n-butylsulfonyl)diazomethane, bis(isobutylsulfonyl)diazomethane, bis(sec-butylsulfonyl)diazomethane, bis(n-propylsulfonyl)diazomethane, bis(isopropylsulfonyl)diazomethane, bis(tert-butylsulfonyl)diazomethane, bis(n-amylsulfonyl)diazomethane, bis(isoamylsulfonyl)diazomethane, bis(sec-amylsulfonyl)diazomethane, bis(tert-amylsulfonyl)diazomethane, 1-cyclohexylsulfonyl-1-(tert-butylsulfonyl)diazomethane, 1-cyclohexylsulfonyl-1-(tert-amylsulfonyl)diazomethane, and 1-tert-amylsulfonyl-1-(tert-butylsuifonyl)diazomethane;
- glyoxime derivatives such as bis-o-(p-toluenesulfonyl)-α-dimethylglyoxime, bis-o-(p-toluenesulfonyl)-α-diphenylglyoxime, bis-o-(p-toluenesulfonyl)-α-dicyclohexylgloxime, bis-o-(p-toluenesulfonyl)-2,3-pentanedioneglyoxime, bis-o-(p-toluenesulfonyl)-2-methyl-3,4-pentanedioneglyoxime, bis-o-(n-butanesulfonyl)-a-dimethylglyoxime, bis-o-(n-butanesulfonyl)-α-diphenylglyoxime, bis-o-(n-butanesulfonyl)-α-dicyclbhexylglyoxime, bis-o-(n-butanesulfonyl)-2,3-pentanedioneglyoxime, bis-o-(n-butanesulfonyl)-2-methyl-3,4-pentanedioneglyoxime, bis-o-(methanesulfonyl)-α-dimethylglyoxime, bis-o-(trifluoromethanesulfonyl)-α-dimethylglyoxime, bis-o-(1,1,1-trifluoroethanesulfonyl)-α-dimethylglyoxime, bis-o-(tert-butanesulfonyl)-α-dimethylglyoxime, bis-o-(perfluorooctanesulfonyl)-α-dimethylglyoxime, bis-o-(cyclohexanesulfonyl)-α-dimethylglyoxime, bis-o-(benzenesulfonyl)-α-dimethylglyoxime, bis-o-(p-fluorobenzenesulfonyl)-α-dimethylglyoxime, bis-o-(p-tert-butylbenzenesulfonyl)-α-dimethylglyoxime, bis-o-(xylenesulfonyl)-α-dimethylglyoxime, and bis-o-(camphorsulfonyl)-α-dimethylglyoxime;
- bissulfone derivatives such as bisnaphthylsulfonylmethane, bistrifluoromethylsulfonylmethane, bismethylsulfonylmethane, bisethylsulfonylmethane, bispropylsulfonylmethane, bisisopropylsulfonylmethane, bis-p-toluenesulfonylmethane, and bisbenzenesulfonylmethane;
- β-ketosulfone derivatives such as 2-cyclohexylcarbonyl-2-(p-toluenesulfonyl)propane and 2-isopropylcarbonyl-2-(p-toluenesulfonyl)propane;
- disulfone derivatives such as diphenyl disulfone and dicyclohexyl disulfone;
- nitrobenzyl sulfonate derivatives such as 2,6-dinitrobenzyl p-toluenesulfonate and 2,4-dinitrobenzyl p-toluenesulfonate;
- sulfonic acid ester derivatives such as 1,2,3-tris-(methanesulfonyloxy)benzene, 1,2,3-tris(trifluoromethanesulfonyloxy)benzene, and 1,2,3-tris(p-toluenesulfonyloxy)-benzene; and
- sulfonic acid esters of N-hydroxyimides such as N-hydroxysuccinimide methanesulfonate, N-hydroxysuccinimide trifluoromethanesulfonate, N-hydroxysuccinimide ethanesulfonate, N-hydroxysuccinimide 1-propanesulfonate, N-hydroxysuccinimide 2-propanesulfonate, N-hydroxysuccinimide 1-pentanesulfonate, N-hydroxysuccinimide 1-octanesulfonate, N-hydroxysuccinimide p-toluenesulfonate, N-hydroxysuccinimide p-methoxybenzenesulfonate, N-hydroxysuccinimide 2-chloroethanesulfonate, N-hydroxysuccinimide benzenesulfonate, N-hydroxysuccinimide 2,4,6-trimethylbenzenesulfonate, N-hydroxysuccinimide 1-naphthalenesulfonate, N-hydroxysuccinimide 2-naphthalenesulfonate, N-hydroxy-2-phenylsuccinimide methanesulfonate, N-hydroxymaleimide methanesulfonate, N-hydroxymaleimide ethanesulfonate, N-hydroxy-2-phenylmaleimide methanesulfonate, N-hydroxyglutarimide methanesulfonate, N-hydroxyglutarimide benzenesulfonate, N-hydroxyphthalimide methanesulfqnate, N-hydroxyphthalimide benzenesulfonate, N-hydroxyphthalimide trifluoromethanesulfonate, N-hydroxyphthalimide p-toluenesulfonate, N-hydroxynaphthalimide methanesulfonate, N-hydroxynaphthalimide benzenesulfonate, N-hydroxy-5-norbornene-2,3-dicarboxyimide methanesulfonate, N-hydroxy-5-norbornene-2,3-dicarboxylmide trifluoromethanesulfonate, and N-hydroxy-5-norbornene-2,3-dicarboxyimide p-toluenesulfonate.
- Preferred among these photoacid generators are onium salts such as triphenylsulfonium trifluoromethanesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium trifluoromethanesulfonate, tris(p-tert-butoxyphenyl)sulfonium trifluoromethaneesulfonate, triphenylsulfonium p-toluenesulfonate, (p-tert-butoxyphenyl)diphenylsulfonium p-toluenesulfonate, tris(p-tert-butoxyphenyl)sulfonium p-toluenesulfonate, trinaphthylsulfonium trifluoromethanesulfonate, cyclohexylmethyl(2-oxocyclohexyl)-sulfonium trifluoromethanesulfonate, (2-norbornyl)methyl(2-oxocylohexyl)sulfonium trifluoromethanesulfonate, and 1,2′-naphthylcarbonylmethyltetrahydrothiophenium triflate; diazomethane derivatives such as bis(benzenesulfonyl)diazomethane, bis(p-toluenesulfonyl)diazomethane, bis(cyclohexylsulfonyl)-diazomethane, bis(n-butylsulfonyl)diazomethane, bis(isobutylsulfonyl)diazomethane, bis(sec-butylsulfonyl)diazomethane, bis(n-propylsulfonyl)diazomethane, bis(isopropylsulfonyl)diazomethane, and bis(tert-butylsulfonyl)diazomethane; glyoxime derivatives such as bis-o-(p-toluenesulfonyl)-α-dimethylglyoxime and bis-6-(n-butanesulfonyl)-α-dimethylglyoxime; bissulfone derivatives such as bisnaphthylsulfonylmethane; and sulfonic acid esters of N-hydroxyimide compounds such as N-hydroxysuccinimide methanesulfonate, N-hydroxysuccinimide trifluoromethanesulfbnate, N-hydroxysuccinimide 1-propanesulfonate, N-hydroxysuccinimide 2-propanesulfonate, N-hydroxysuccinimide 1-pentanesulfonate, N-hydroxysuccinimide p-toluenesulfonate, N-hydroxynaphthalimide methanesulfonate, and N-hydroxynaphthalimide benzenesulfonate.
- These photoacid generators may be used singly or in combinations of two or more thereof. Onium salts are effective for improving rectangularity, while diazomethane derivatives and glyoxime derivatives are effective for reducing standing waves. The combination of an onium salt with a diazomethane or a glyoxime derivative allows for fine adjustment of the profile.
- The photoacid generator is added in an amount of 0.1 to 15 parts, and especially 0.5 to 8 parts by weight, per 100 parts by weight of the base resin (all parts are by weight, hereinafter). Less than 0.1 part of the photoacid generator would provide a poor sensitivity whereas more than 15 parts of the photoacid generator would adversely affect transparency and resolution.
- Organic Solvent
- The organic solvent used herein may be any organic solvent in which the base resin, photoacid generator, and other components are soluble. Illustrative, non-limiting, examples of the organic solvent include ketones such as cyclohexanone and methyl-2-n-amylketone; alcohols such as 3-methoxybutanol, 3-methyl-3-methoxybutanol, 1-methoxy-2-propanol, and 1-ethoxy-2-propanol; ethers such as propylene glycol monomethyl ether, ethylene glycol monomethyl ether, propylene glycol monoethyl ether, ethylene glycol monoethyl ether, propylene glycol dimethyl ether, and diethylene glycol dimethyl ether; and esters such as propylene glycol monomethyl ether acetate, propylene glycol monoethyl ether acetate, ethyl lactate, ethyl pyruvate, butyl acetate, methyl 3-methoxypropionate, ethyl 3-ethoxypropionate, tert-butyl acetate, tert-butyl propionate, and propylene glycol mono-tert-butyl ether acetate. These solvents may be used alone or in combinations of two or more thereof. Of the above organic solvents, it is recommended to use diethylene glycol dimethyl ether and 1-ethoxy-2-propanol because the photoacid generator serving as one of the resist components is most soluble therein, propylene glycol monomethyl ether acetate because it is a safe solvent, or a mixture thereof.
- An appropriate amount of the organic solvent used is about 200 to 1,000 parts, especially about 400 to 800 parts by weight per 100 parts by weight of the base resin.
- Other Polymer
- To the resist composition of the invention, another polymer other than the inventive polymer comprising recurring units of formula (1a-1) and/or (1a-2) may also be added. The other polymers that can be added to the resist composition are, for example, those polymers comprising units of the following formula (R1) or (R2) and having a weight average molecular weight of about 1,000 to about 500,000, especially about 5,000 to about 100,000 although the other polymers are not limited thereto.
- Herein, R 001 is hydrogen, methyl or CH2Co2R003. R002 is hydrogen, methyl or CO2R003. R003 is a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms. R004 is hydrogen or a monovalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group. At least one of R005 to R008 represents a monovalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group while the remaining R's independently represent hydrogen or a straight, branched or cyclic alkyl group-of 1 to 15 carbon atoms. Alternatively, R005 to R008, taken together, may form a ring, and in that event, at least one of R005 to R008 is a divalent hydrocarbon group of 1 to 15 carbon atoms having a carboxyl or hydroxyl group, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms. R009 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO2— partial structure. At least one of R010 to R013 is a monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO2— partial structure, while the remaining R's are independently hydrogen or straight, branched or cyclic alkyl groups of 1 to 15 carbon atoms. R010 to R013, taken together, may form a ring, and in that event, at least one of R010 to R013 is a divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO2— partial structure, while the remaining R's are independently single bonds or straight, branched or cyclic alkylene groups of 1 to 15 carbon atoms. R014 is a polycyclic hydrocarbon group having 7 to 15 carbon atoms or an alkyl group containing a polycyclic hydrocarbon group. R015 is an acid labile group. R016 is hydrogen or methyl. R017 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms. Letter k′ is equal to 0 or 1; a1′, a2′, a3′, b1′, b2′, b3′, c1′, c2′, c3′, d1′, d2′, d3′, and e1′ are numbers from 0 to less than 1, satisfying a1′+a2′+a3′+b1′+b2′+b3′+c1′+c2′+c3′+d1′+d2′+d3′+e′=1; f′, g′, h′, i′, and j′ are numbers from 0 to less than 1, satisfying f′+g′+h′+i′+j′=1. X′, y′ and z′ are integers of 0 to 3, satisfying x′+y′+z′≦5 and 1≦y′+Z′.
- Exemplary groups of, these R's are as exemplified above for R 1 to R15.
- The inventive polymer (comprising recurring units of formula (1a-1) and/or (1a-2)) and the other polymer are preferably blended in a weight ratio from 10:90 to 90:10, more preferably from 20:80 to 80:20. If the blend ratio of the inventive polymer is below this range, the resist composition would become poor in some of the desired properties. The properties of the resist composition can be adjusted by properly changing the blend ratio of the inventive polymer.
- The other polymer is not limited to one type and a mixture of two or more other polymers may be added. The use of plural polymers allows for easy adjustment of resist properties.
- Dissolution Regulator
- To the resist composition, a dissolution regulator may be added. The dissolution regulator is a compound having on the molecule at least two phenolic hydroxyl groups, in which an average of from 0 to 100 mol % of all the hydrogen atoms on the phenolic hydroxyl groups are replaced with acid labile groups or a compound having on the molecule at least one carboxyl group, in which an average of 50 to 100 mol % of all the hydrogen atoms on the carboxyl groups are replaced with acid labile groups, both the compounds having an average molecular weight within a range of 100 to 1,000, and preferably 150 to 800.
- The degree of substitution of the hydrogen atoms on the phenolic hydroxyl groups with acid labile groups is on average at least 0 mol %, and preferably at least 30 mol %, of all the phenolic hydroxyl groups. The upper limit is 100 mol %, and preferably 80 mol %. The degree of substitution of the hydrogen atoms on the carboxyl groups with acid-labile groups is on average at least 50 mol %, and preferably at least 70 mol %, of all the carboxyl groups, with the upper limit being 100 mol %.
-
- In these formulas, R 201 and R202 are each hydrogen or a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms; R203 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or —(R207)h—COOH; R294 is —(CH2)i— (where i=2 to 10), an arylene of 6 to 10 carbon atoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom; R205 is an alkylene of 1 to 10 carbon atoms; an arylene of 6 to 10 carbonatoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom; R206 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or a hydroxyl-substituted phenyl or naphthyl; R207 is a straight or branched alkylene of 1 to 10 carbon atoms; R208 is hydrogen or hydroxyl; the letter j is an integer from 0 to 5; u and h are each 0 or 1; s, t, s′, t′, s″, and t″ are each numbers which satisfy s+t=8, s′+t′=5, and s″+t″=4, and are such that each phenyl skeleton has at least one hydroxyl group; and α is a number such that the compounds of formula (D8) or (D9) have a molecular weight of from 100 to 1,000.
- In the above formulas, suitable examples of R 201 and R202 include hydrogen:, methyl, ethyl, butyl, propyl, ethynyl, and cyclohexyl; suitable examples of R203 include the same groups as for R201 and R202, as well as —COOH and —CH2COOH; suitable examples of R204 include ethylene, phenylene, carbonyl, sulfonyl, oxygen, and sulfur; suitable examples of R205 include methylene as well as the same groups as for R204; and suitable examples of R206 include hydrogen, methyl, ethyl, butyl, propyl, ethynyl, cyclohexyl, and hydroxyl-substituted phenyl or naphthyl.
-
- In these formulas, R L01 and RL02 are each hydrogen or a straight, branched or cyclic-alkyl having 1 to 18,carbon atoms; and RL03 is a monovalent hydrocarbon group of 1 to 18 carbon atoms which may contain a heteroatom (e.g., oxygen). A pair of RL01 and RL02, a pair of RL01 and RL03, or a pair of RL02 and RL03.may together form a ring, with the proviso that RL01, RL02, and RL03 are each a straight or branched alkylene of 1 to 18 carbon atoms when they form a ring. RL14 is a tertiary alkyl group of 4 to 20 carbon atoms, a trialkysilyl group in which each of the alkyls has 1 to 6 carbon atoms, an oxoalkyl group of 4 to 20 carbon atoms, or a group of the formula (L1). RL05 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms. RL06 is a straight, branched or cyclic alkyl group of 1 to 8 carbon atoms or a substituted or unsubstituted aryl group of 6 to 20 carbon atoms. RL17 to RL16 independently represent hydrogen or monovalent hydrocarbon groups of 1 to 15 carbon atoms which may contain a hetero atom. Alternatively, RL07 to RL16, taken together, may form a ring. Each of RL07 to RL16 represents a divalent C1-C15 hydrocarbon group which may contain a hetero atom, when they form a ring. Two of RL07 to RL16 which are attached to adjoining carbon atoms may bond together directly to form a double bond. Letter y is an integer of 0 to 6. Letter m is equal to 0 or 1, n is equal to 0, 1, 2 or 3, and 2 m+n is equal to 2 or 3.
- The dissolution regulator may be formulated in an amount of 0 to 50 parts, preferably 5 to 50 parts, and more preferably 10 to 30 parts, per 100 parts of the base resin, and may be used singly or as a mixture of two or more thereof. Less than 5 parts of the dissolution regulator may fail to yield an improved resolution, whereas the use of more than 50 parts would lead to slimming of the patterned film, and thus a decline in resolution.
- The dissolution regulator can be synthesized by introducing acid labile groups into a compound having phenolic hydroxyl or carboxyl groups in accordance with an organic chemical formulation.
- Basic Compound
- In the resist composition of the invention, a basic compound may be blended. A suitable basic compound used herein is a compound capable of suppressing the rate of diffusion when the acid generated by the photoacid generator diffuses within the resist film. The inclusion of this type of basic compound holds down the rate of acid diffusion within the resist film, resulting in better resolution. In addition, it suppresses changes in sensitivity following exposure, thus reducing substrate and environment dependence, as well as improving the exposure latitude and the pattern profile.
- Examples of basic compounds include primary, secondary, and tertiary aliphatic amines, mixed amines, aromatic amines, heterocyclic amines, carboxyl group-bearing nitrogenous compounds, sulfonyl group-bearing nitrogenous compounds, hydroxyl group-bearing nitrogenous compounds, hydroxyphenyl group-bearing nitrogenous compounds, alcoholic nitrogenous compounds, amide derivatives, and imide derivatives.
- Examples of suitable primary aliphatic amines include ammonia, methylamine, ethylamine, n-propylamine, isopropylamine, n-butylamine, iso-butylamine, sec-butylamine, tert-butylamine, pentylamine, tert-amylamine, cyclopentylamine, hexylamine, cyclohexylamine, heptylamine, octylamine, nonylamine, decylamine, dodecylamine, cetylamine, methylenediamine, ethylenediamine, and tetraethylenepentamine. Examples of suitable secondary aliphatic amines include dimethylamine, diethylamine, di-n-propylamine, di-iso-propylamine, di-n-butylamine, di-iso-butylamine, di-sec-butylamine, dipentylamine, dicyclopentylamine, dihexylamine, dicyclohexylamine, diheptylamine, dioctylamine, dinonylamine, didecylamine, didodecylamine, dicetylamine, N,N-dimethylmethylenediamine, N,N-dimethylethylenediamine, and N,N-dimethyltetraethylenepentamine. Examples of suitable tertiary aliphatic amines include trimethylamine, triethylamine, tri-n-propylamine, tri-iso-propylamine, tri-n-butylamine, tri-iso-butylamine, tri-sec-butylamine, tripentylamine, tricyclopentylamine, trihexylamine, tricyclohexylamine, triheptylamine, trioctylamine, trinonylamine, tridecylamine, tridodecylamine, tricetylamine, N,N,N′,N′-tetramethylmethylenediamine, N,N,N′,N′-tetramethylethylenediamine, and N,N,N′,N′-tetramethyltetraethylenepentamine.
- Examples of suitable mixed amines include dimethylethylamine, methylethylpropylamine, benzylamine, phenethylamine, and benzyldimethylamine. Examples of suitable aromatic and heterocyclic amines include aniline derivatives (e.g., aniline, N-methylaniline, N-ethylaniline, N-propylaniline, N,N-dimethylaniline, 2-methylaniline, 3-methylaniline, 4-methylaniline, ethylaniline, propylaniline, trimethylaniline, 2-nitroaniline, 3-nitroaniline, 4-nitroaniline, 2,4-dinitroaniline, 2,6-dinitroaniline, 3,5-dinitroaniline, and N,N-dimethyltoluidine), diphenyl(p-tolyl)amine, methyldiphenylamine, triphenylamine, phenylenediamine, naphthylamine, diaminonaphthalene, pyrrole derivatives (e.g., pyrrole, 2H-pyrrole, 1-methylpyrrole, 2,4-dimethylpyrrole, 2,5-dimethylpyrrole, and N-methylpyrrole), oxazole derivatives (e.g., oxazole and isooxazole), thiazole derivatives (e.g., thiazole and isothiazole), imidazole derivatives (e.g., imidazole, 4-methylimidazole, and 4-methyl-2-phenylimidazole), pyrazole derivatives, furazan derivatives, pyrroline derivatives (e.g., pyrroline and 2-methyl-1-pyrroline), pyrrolidine derivatives (e.g., pyrrolidine, N-methylpyrrolidine, pyrrolidinone, and N-methylpyrrolidone), imidazoline derivatives, imidazolidine derivatives, pyridine derivatives (e.g., pyridine, methylpyridine, ethylpyridine, propylpyridine, butylpyridine, 4-(1-butylpentyl)pyridine, dimethylpyridine, trimethylpyridine triethylpyridine, phenylpyridine, 3-methyl-2-phenylpyridine, 4-tert-butylpyridine, diphenylpyridine, benzylpyridine, methoxypyridine, butoxypyridine, dimethoxypyridine, 1-methyl-2-pyridine, 4-pyrrolidinopyridine, 1-methyl-4-phenylpyridine, 2-(1-ethylpropyl)pyridine, aminopyridine, and dimethylaminopyridine), pyridazine derivatives, pyrimidine derivatives, pyrazine derivatives, pyrazoline derivatives, pyrazolidine derivatives, piperidine derivatives, piperazine derivatives, morpholine derivatives, indole derivatives, isoindole derivatives, 1H-indazole derivatives, indoline derivatives, quinoline derivatives (e.g., quinoline and 3-quinolinecarbonitrile), isoquinoline derivatives, cinnoline derivatives, quinazoline derivatives, quinoxaline derivatives, phthalazine derivatives, purine derivatives, pteridine derivatives, carbazole derivatives, phenanthridine derivatives, acridine derivatives, phenazine derivatives, 1,10-phenanthroline.derivatives, adenine derivatives, adenosine derivatives, guanine derivatives, guanosine derivatives, uracil derivatives, and uridine derivatives.
- Examples of suitable carboxyl group-bearing nitrogenous compounds include aminobenzoic acid, indolecarboxylic acid, and amino acid derivatives (e.g. nicotinic acid, alanine, alginine, aspartic acid, glutamic acid, glycine, histidine, isoleucine, glycylleucine, leucine, methionine, phenylalanine, threonine, lysine, 3-aminopyrazine-2-carboxylic acid, and methoxyalanine). Examples-of suitable sulfonyl group-bearing nitrogenous compounds include 3-pyridinesulfonic acid and pyridinium p-toluenesulfonate. Examples of suitable hydroxyl group-bearing nitrogenous compounds, hydroxyphenyl group-bearing nitrogenous compounds, and alcoholic nitrogenous compounds include 2-hydroxypyridine, aminocresol, 2,4-quinolinediol, 3-indolemethanol hydrate, monoethanolamine, diethanolamine, triethanolamine, N-ethyldiethanolamine, N,N-diethylethanolamine, triisopropanolamine, 2,2′-iminodiethanol, 2-aminoethanol, 3-amino-1-propanol, 4-amino-1-butanol, 4-(2-hydroxyethyl)morpholine, 2-(2-hydroxyethyl)pyridine, 1-(2-hydroxyethyl)piperazine, 1-[2-(2-hydroxyethoxy)ethyl]-piperazine, piperidine ethanol, 1-(,2-hydroxyethyl)-pyrrolidine, 1-(2-hydroxyethyl)-2-pyrrolidinone, 3-piperidino-1,2-propanediol, 3-pyrrolidino-1,2-propanediol, 8-hydroxyjulolidine, 3-quinuclidinol, 3-tropanol, 1-methyl-2-pyrrolidine ethanol, 1-aziridine ethanol, N-(2-hydroxyethyl)phthalimide, and N-(2-hydroxyethyl)-isonicotinamide. Examples of suitable amide derivatives include formamide, N-methylformamide, N,N-dimethylformamide, acetamide, N-methylacetamide, N,N-dimethylacetamide, propionamide, and benzamide. Suitable imide derivatives include phthalimide, succinimide, and maleimide.
- In addition, basic compounds of the following general formula (B1) may also be included alone or in admixture.
- N(X)n(Y)3-n B1
- In the formula, n is equal to 1, 2 or 3; Y is independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 20 carbon atoms which may contain a hydroxyl group or ether; and X is independently selected from groups of the following general formulas (X1) to (X3), and two or three X's may bond together to form a ring.
- In the formulas, R 300, R302 and R305 are independently straight or branched alkylene groups of 1 to 4 carbon atoms; R301 and R304 are independently hydrogen, straight, branched or cyclic alkyl groups of 1 to 20 carbon atoms, which may contain at least one hydroxyl group, ether, ester or lactone ring; and R303 is a single bond or a straight or branched alkylene group of 1 to 4 carbon atoms.
- Illustrative examples of the compounds of formula (B1) include tris(2-methoxymethoxyethyl)amine, tris{2-(methoxyethoxy)ethyl}amine, tris{2-(2-methoxyethoxymethoxy)ethyl}amine, tris{2-(1-methoxyethoxy)ethyl}amine, tris{2-(1-ethoxyethoxy)ethyl}amine, tris{2—(1-ethoxypropoxy)ethyl}amine, tris[2-{2-(2-hydroxyethoxy)ethoxy}-ethyl]amine, 4,7,13,16,21,24-hexaoxa-1,10-diazabicyclo-[8.8.8]hexacosane, 4,7,13,18-tetraoxa-1,10-diazabicyclo-[8.5.5]eicosane, 1,4,10,13-tetraoxa-7,16-diazabicyclooctadecane, 1-aza-12-crown-4, 1-aza-15-crown-5, 1-aza-18-crown-6, tris(2-formyloxyethyl)amine, tris(2-acetoxyethyl)-amine, tris(2-propionyloxyethyl)amine, tris(2-butyryloxyethyl)amine, tris(2-isobutyryloxyethyl)amine, tris(2-valeryloxyethyl)amine, tris(2-pivaloyloxyethyl)amine, N,N-bis(2-acetoxyethyl)-2-((acetoxyacetoxy)ethylamine, tris(2-methoxycarbonyloxyethyl)amine, tris(2-tert-butoxycarbonyloxyethyl)amine, tris[2-(2-oxopropoxy)ethyl]amine, tris[2-(methoxycarbonylmethyl)oxyethyl]amine, tris[2-(tert-butoxycarbonylmethyloxy)ethyl]amine, tris[2-(cyclohexyloxycarbonylmethyloxy)ethyl]amine, tris(2-methoxycarbonylethyl)amine, tris(2-ethoxycarbonylethyl)amine, N,N-bis(2-hydroxyethyl)-2-(methoxycarbonyl)ethylamine, N,N-bis(2-acetoxyethyl)-2-(methoxycarbonyl)ethylamine, N,N-bis(2-hydroxyethyl)-2-(ethoxycarbonyl)ethylamine, N,N-bis(2-acetoxyethyl)-2-(ethoxycarbonyl)ethylamine, N,N-bis(2-hydroxyethyl)-2-(2-methoxyethoxycarbonyl)ethylamilne, N,N-bis(2-acetoxyethyl)-2-(2-methoxyethoxycarbonyl)ethylamine, N,N-bis(2-hydroxyethyl)-2-(2-hydroxyethoxycarbonyl)ethylamine, N,N-bis(2-acetoxyethyl)-2-(2-acetoxyethoxycarbonyl)ethylamine, N,N-bis(2-hydroxyethyl)-2-[(methoxycarbonyl)methoxycarbonyl]ethylamine, N,N-bis(2-acetoxyethyl)-2-[(methoxycarbonyl)methoxycarbonyl]ethylamine, N,N-bis(2-hydroxyethyl)-2-(2-oxopropoxycarbonyl)ethylamine, N,N-bis(2-acetoxyethyl)-2-(2-oxopropoxycarbonyl)ethylamine, N,N-bis(2-hydroxyethyl)-2-(tetrahydrofurfuryloxycarbonyl)ethylamine, N,N-bis(2-acetoxyethyl)-2-(tetrahydrofurfuryloxycarbonyl)ethylamine, N,N-bis(2-hydroxyethyl)-2-[(2-oxotetrahydrofuran-3-yl)oxycarbonyl]ethylamine, N,N-bis(2-acetoxyethyl)-2-[(2-oxotetrahydrofuran-3-yl)oxycarbonyl]-ethylamine, N,N-bis(2-hydroxyethyl)-2-(4-hydroxybutoxycarbonyl)ethylamine, N,N-bis(2-formyloxyethyl)-2-(4-formyloxybutoxycarbonyl)ethylamine, N,N-bis(2-formyloxyethyl)-2-(2-formyloxyethoxycarbonyl)ethylamine, N,N-bis(2-methoxyethyl)-2-(methoxycarbonyl)ethylamrine, N-(2-hydroxyethyl)-bis[2-(methoxycarbonyl)ethyl]amine, N-(2-acetoxyethyl)-bis[2-(methoxycarbonyl)ethyl]amine, N-(2-hydroxyethyl)-bis[2-(ethoxycarbonyl)ethyl]amine, N-(2-acetoxyethyl)-bis[2-(ethoxycarbonyl)ethyl]amine, N-(3-hydroxy-1-propyl)-bis[2-(methoxycarbonyl)ethyl]amine, N-(3-acetoxy-1-propyl)-bis[2-(methoxycarbonyl)ethyl]amine, N-(2-methoxyethyl)-bis[2-(methoxycarbonyl)ethyl]amine, N-butyl-bis[2-(methoxycarbonyl)ethyl]amine, N-butyl-bis[2-(2-methoxyethoxycarbonyl)ethyl]amine, N-methyl-bis(2-acetoxyethyl)amine, N-ethyl-bis(2-acetoxyethyl)amine, N-methyl-bis(2-pivaloyloxyethyl)amine, N-ethyl-bis[2-(methoxycarbonyloxy)ethyl]amine, N-ethyl-bis[2-(tert-butoxycarbonyloxy)ethyl]amine, tris(methoxycarbonylmethyl)amine, tris(ethoxytarbonylmethyl)amine, N-butyl-bis(methoxycarbonylmethyl)amine, N-hexyl,bis(methoxycarbonylmethyl)amine, and β-(diethylamino)-δ-valerolactone.
- The basic compound is preferably formulated in an amount of 0.001 to 10 parts, and especially 0.01 to 1 part, per part of the photoacid generator. Less than 0.001 part of the basic compound fails to achieve the desired effects thereof, while the use of more than 10 parts would result in too low a sensitivity and resolution.
- Other Components
- In the resist composition, a compound bearing a ≡C—COOH group in a molecule may be blended. Exemplary, non-limiting compounds bearing a ≡C—COOH group include one or more compounds selected from Groups I and II below. Including this compound improves the PED stability of the resist and ameliorates edge roughness on nitride film substrates.
- Group I:
- Compounds in which some or all of the hydrogen atoms on the phenolic hydroxyl groups of the compounds of general formulas (A1) to (A10) below have been replaced with —R 401—COOH (wherein R401 is a straight or branched alkylene of 1 to 10 carbon atoms), and in which the molar ratio C/(C+D) of phenolic hydroxyl groups (C) to ≡C—COOH groups (D) in the molecule is from 0.1 to 1.0.
- In these formulas, R 408 is hydrogen or methyl; R402 and R403 are each hydrogen or a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms; R404 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or a —(R409)h—COOR′ group (R′ being hydrogen or —R409—COOH); R405 is —(CH2)i— (wherein i is 2 to 10), an arylene of 6 to 10 carbon atoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom; R406 is an alkylene of 1 to 10 carbon atoms, an arylene of 6 to 10 carbon atoms, carbonyl, sulfonyl, an oxygen atom, or a sulfur atom; R407 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or a hydroxyl-substituted phenyl or naphthyl; R409 is a straight or branched alkylene of 1 to 10 carbon atoms; R410 is hydrogen, a straight or branched alkyl or alkenyl of 1 to 8 carbon atoms, or a —R411—COOH group; R411 is a straight or branched alkylene of 1 to 10 carbon atoms; the letter j is an integer from 0 to 5; u and h are each 0 or 1; s1, t1, s2, t2, s3, t3, s4, and t4 are each numbers which satisfy s1+t1=8, s2+t2=5, s3+t3=4, and s4+t4=6, and are such that each phenyl skeleton has at least one hydroxyl group; κ is a number such that the compound of formula (A6) may have a weight average molecular weight of 1,000 to 5,000; and λ is a number such that the compound of formula (A7) may have a weight average molecular weight of 1,000 to 10,000.
- Group II:
-
- In these formulas, R 402, R403, and R411 are as defined above; R412 is hydrogen or hydroxyl; s5 and t5 are numbers which satisfy s5≦0, t5≦0, and s5+t5=5; and h′ is equal to 0 or 1.
-
-
- The compound bearing a ≡C—COOH group within the molecule may be used singly or as combinations of two or more thereof.
- The compound bearing a ≡C—COOH group within the molecule is added in an amount ranging from 0 to 5 parts, preferably 0.1 to 5 parts, more preferably 0.1 to 3 parts, further preferably 0.1 to 2 parts, per 100 parts of the base resin. More than 5 parts of the compound can reduce the resolution of the resist composition.
-
- In the formulas, R 501, R502, R503, R504 and R505 are each hydrogen or a straight, branched, or cyclic alkyl of 1 to 8 carbon atoms; and X and Y are each 0 or a positive number, satisfying 0≦X≦30, 0≦Y≦30, and 0≦X+Y≦40.
- Preferable examples of the acetylene alcohol derivative include Surfynol 61, Surfynol 82, Surfynol 104, Surfynol 104E, Surfynol 104H, Surfynol 104A, Surfynol TG, Surfynol PC, Surfynol 440, Surfynol 465, and Surfynol 485 from Air Products and Chemicals Inc., and Surfynol E1004 from Nisshin Chemical Industry K.K.
- The acetylene alcohol derivative is preferably added in an amount of 0.01 to 2% by weight, and more preferably 0.02 to 1% by weight, per 100% by weight of the resist composition. Less than 0.01% by weight would be ineffective for improving coating characteristics and shelf stability, whereas more than 2% by weight would result in a resist having a low resolution.
- The resist composition of the invention may include, as an optional ingredient, a surfactant which is commonly used for improving the coating characteristics. Optional ingredients may be added in conventional amounts so long as this does not compromise the objects of the invention.
- Nonionic surfactants are preferred, examples of which include perfluoroalkylpolyoxyethylene ethanols, fluorinated alkyl esters, perfluoroalkylamine oxides, perfluoroalkyl EO-addition products, and fluorinated organosiloxane compounds. Useful surfactants are commercially available under the trade names Florade FC-430 and FC-431 from Sumitomo 3M K.K., Surflon S-141 and S-145 from Asahi Glass K.K., Unidine DS-401, DS-403 and DS-451 from Daikin Industry K.K., Megaface F-8151 from Dai-Nippon Ink & Chemicals K.K., and X-70-092 and X-70-093 from Shin-Etsu Chemical Co., Ltd. Preferred surfactants are Florade FC-430 from Sumitomo 3M K.K. and X-70-093 from Shin-Etsu Chemical Co., Ltd.
- Pattern formation using the resist composition of the invention may be carried out by a known lithographic technique. For example, the resist composition is applied onto a substrate such as a silicon wafer by spin coating or the like to form a resist film having a thickness of 0.3 to 2.0 μm, which is then pre-baked on a hot plate at 60 to 150° C. for 1 to 10 minutes, and preferably at 80 to 130° C. for 1 to 5 minutes. A patterning mask having the desired pattern is then placed over the resist film, and the film exposed through the mask to an electron beam or to high-energy radiation such as deep-UV rays, an excimer laser, or x-rays in a dose of about 1 to 200 mJ/cm 2, and preferably about 10 to 100 mJ/cm2, then post-exposure baked (PEB) on a hot plate at 60 to 150° C. for 1 to 5 minutes, and preferably at 80 to 130° C. for 1 to 3 minutes. Finally, development is carried out using as the developer an aqueous alkali solution, such as a 0.1 to 5% (preferably 2 to 3%) aqueous solution of tetramethylammonium hydroxide (TMAH), this being done by a conventional method such as dipping, puddling, or spraying for a period of 0.1 to 3 minutes, and preferably 0.5 to 2 minutes. These steps result in the formation of the desired pattern on the substrate. Of the various types of high-energy radiation that may be used, the resist composition of the invention is best suited to fine pattern formation with, in particular, deep-UV rays having a wavelength of 248 to 193 nm, an excimer laser, x-rays, or an electron beam. The desired pattern may not be obtainable outside the upper and lower limits of the above range.
- The resist composition comprising the polymer as a base resin lends itself to micropatterning with electron beams or deep-UV rays since it is sensitive to high-energy radiation and has excellent sensitivity, resolution, and etching resistance. Especially because of the minimized absorption at the exposure wavelength of an ArF or KrF excimer laser, a finely defined pattern having sidewalls perpendicular to the substrate can easily be formed.
- Synthesis Examples and Examples are given below by way of illustration and not by way of limitation.
- Ester compounds and polymers containing the same were synthesized according to the following formulation.
- Synthesis of Monomer 1
- In 500 ml of diethyl ether was dissolved 168.0 g of chlorocyclohexane. Below 60° C., this reaction mixture was added dropwise to 26.4 g of metallic magnesium over one hour. After agitation was continued for 2 hours at room temperature, 84.0 g of cyclopentanone was added dropwise over 45 minutes to the reaction mixture which was kept below 65° C. After agitation was continued for one hour at room temperature, the reaction solution was worked up in a conventional manner. The resulting oily substance was distilled in vacuum, collecting 97.3 g of 1-cyclohexylcyclopentanol. The yield was 57.9%.
- In 1 liter of methylene chloride was dissolved 92.4 g of 1-cyclohexylcyclopentanol. To the solution below −20° C., 74.3 g of acrylic chloride was added dropwise over 10 minutes. To the reaction mixture, 1.0 g of 4-(N,N-dimethylamino)pyridine was added and then, 125.0 g of triethylamine was added dropwise over one hour and below −20° C. This reaction mixture was agitated for 15 hours at room temperature and subjected to conventional post-treatment, obtaining about 130 g of an oily substance. This was used in the subsequent reaction without purification.
- The oily substance obtained in the above step was dissolved in 300 ml of benzene. To this solution below 10° C., 72.6 g of cyclopentadiene was added dropwise over 10 minutes. The reaction mixture was agitated for 17 hours at room temperature, then for one hour at 70° C., and concentrated in vacuum. Vacuum distillation (170° C./0.2 Torr) of the resulting oily substance yielded 106.6 g of 1-cyclohexylcyclopentyl 5-norbornene-2-carboxylate (designated Monomer 1). The yield was 67.3%.
- FIG. 1 shows the spectrum of Monomer 1 by 1H-NMR (CDCl3, 270 MHz).
- Synthesis of Monomers 2 to 8
-
- Synthesis of Polymer 1
- In 1 liter of tetrahydrofuran, 72.0 g of Monomer 1 and 24.5 g of maleic anhydride were dissolved, and 1.8 g of 2,2′-azobisisobutyronitrile added. After agitation was continued for 15 hours at 60° C., the reaction solution was concentrated in vacuum. The residue was dissolved in 400 ml of tetrahydrofuran, which was, in turn, added dropwise to 10 liters of n-hexane. The resulting solids were collected by filtration, washed with 10 liters of n-hexane, and dried in vacuum at 40° C. for 6 hours, obtaining 53.6 g of a polymer, designated Polymer 1. The yield was 55.5%.
- Synthesis of Polymers 2 to 18
- Polymers 2 to 18 were synthesized by the same procedure as above or a well-known procedure.
(Polymer 1) (x = 0.50, e = 0.50, Mw = 15,200) (Polymer 2) (x = 0.50, e = 0.50, Mw = 13,400) (Polymer 3) (x = 0.50, e = 0.50, Mw = 14,100) (Polymer 4) (x = 0.50, e = 0.50, Mw = 11,900) (Polymer 5) (x = 0.50, e = 0.50, Mw = 14,800) (Polymer 6) (x = 0.50, e = 0.50, Mw = 12,700) (Polymer 7) (x = 0.50, e = 0.50, Mw = 18,300) (Polymer 8) (x = 0.50, e = 0.50, Mw = 16,900) (Polymer 9) (x = 0.45, e = 0.50, Mw = 14,500) (Polymer 10) (x = 0.40, e = 0.50, Mw = 15,100) (Polymer 11) (x = 0.40, e = 0.50, Mw = 16,300) (Polymer 12) (x = 0.40, e = 0.50, Mw = 15,700) (Polymer 13) (x = 0.60, b = 0.40, Mw = 20,200) (Polymer 14) (x = 0.60, b = 0.40, Mw = 22,100) (Polymer 15) (x = 0.60, b = 0.40, Mw = 21,500) (Polymer 16) (x = 0.60, b = 0.40, Mw = 31,300) (Polymer 17) (x = 0.60, b = 0.40, Mw = 32,400) (Polymer 18) (x = 0.60, b = 0.40, Mw = 31,700) - Resist compositions were formulated using inventive polymers and examined for resolution upon KrF excimer laser exposure.
- Resist compositions were prepared by using Polymers 1 to 18 as the base resin, and dissolving the polymer, a photoacid generator (designated as PAG 1 and 2), a dissolution regulator (designated as DRR 1 to 4), a basic compound, and a compound having a ≡C—COOH group in the molecule (ACC 1 and 2) in a solvent in accordance with the formulation shown in Tables 1 and 2. These compositions were each filtered through a Teflon filter (pore diameter 0.2 μm), thereby giving resist solutions.
- The solvents and basic compounds used are as follows. It is noted that the solvents contained 0.01% by weight of surfactant Florade FC-430 (Sumitomo 3M).
- PGMEA: propylene glycol methyl ether acetate
- CyHO: cyclohexanone
- TBA: tributylamine
- TEA: triethanolamine
- TMMEA: trismethoxymethoxyethylamine
- TMEMEA: trismethoxyethoxymethoxyethylamine
- These resist solutions were spin-coated onto silicon wafers, then baked on a hot plate at 110° C. for 90 seconds to give resist films having a thickness of 0.5 μm. The resist films were exposed using an KrF excimer laser stepper (Nikon Corporation; NA 0.5), then baked (PEB) at 110° C. for 90 seconds, and developed with a solution of 2.38% TMAH in water, thereby giving positive patterns.
- The resulting resist patterns were evaluated as described below. First, the sensitivity (Eth, mJ/cm 2) was determined. Next, the optimal dose (Eop, mJ/cm2) was defined as the dose which provides a 1:1 resolution at the top and bottom of a 0.30 μm line-and-space pattern, and the resolution of the resist under evaluation was defined as the minimum line width (μm) of the lines and spaces that separated at this dose. The shape of the resolved resist pattern was examined under a scanning electron microscope.
- The composition and test results of the resist materials are shown in Tables 1 and 2.
- For comparison purposes, resist compositions were formulated and examined for resolution upon KrF excimer laser exposure.
-
- These resist materials were similarly evaluated. The composition and test results of these resist materials are shown in Table 3.
TABLE 1 Photoacid Dissolution Basic Optimum Base resin generator regulator compound Solvent dose Resolution Example (pbw) (pbw) (pbw) (pbw) (pbw) (mJ/cm2) (μm) Shape I-1 Polymer 1 PAG 1 TBA PGMEA 24.0 0.22 rectangular (80) (1) (0.078) (480) I-2 Polymer 2 PAG 1 TBA PGMEA 20.0 0.22 rectangular (80) (1) (0.078) (480) I-3 Polymer 3 PAG 1 TBA PGMEA 23.0 0.22 rectangular (80) (1) (0.078) (480) I-4 Polymer 4 PAG 1 TBA PGMEA 21.5 0.22 rectangular (80) (1) (0.078) (480) I-5 Polymer 5 PAG 1 TBA PGMEA 24.5 0.22 rectangular (80) (1) (0.078) (480) I-6 Polymer 6 PAG 1 PBA PGMEA (80) (1) (0.078) (480) 19.5 0.22 rectangular I-7 Polymer 7 PAG 1 TBA PGMEA 25.0 0.22 rectangular (80) (1) (0.078) (480) I-8 Polymer 8 PAG 1 TBA PGMEA (80) (1) (0.078) (480) 22.5 0.22 rectangular I-9 Polymer 9 PAG 1 TBA PGMEA (80) (1) (0.078) (480) 22.0 0.22 rectangular I-10 Polymer 10 PAG 1 TBA PGMEA 25.5 0.22 rectangular (80) (1) (0.078) (480) I-11 Polymer 11 PAG 1 TBA PGMEA 22.5 0.22 rectangular (80) (1) (0.078) (480) I-12 Polymer 12 PAG 1 TBA PGMEA 22.5 0.22 rectangular (80) (1) (0.078) (480) I-13 Polymer 13 PAG 1 TBA CyHO (80) (1) (0.078) (640) 18.0 0.22 rectangular I-14 Polymer 14 PAG 1 TBA CYHO 16.5 0.22 rectangular (80) (1) (0.078) (640) I-15 Polymer 15 PAG 1 TBA CyHO (80) (1) (0.078) (640) 16.0 0.22 rectangular I-16 Polymer 16 PAG 1 TBA CYHO 21.5 0.22 rectangular (80) (1) (0.078) (640) I-17 Polymer 17 PAG 1 TBA CYHO (80) (1) (0.078) (640) 20.5 0.22 rectangular I-18 Polymer 18 PAG 1 TBA CYHO 20.0 0.22 rectangular (80) (1) (0.078) (640) I-19 Polymer 12 PAG 2 TBA PGMEA 23.0 0.22 rectangular (80) (1) (0.078) (480) I-20 Polymer 12 PAG 2 TBA PGMEA 22.0 0.20 rectangular (80) (1) (0.063) (480) -
TABLE 2 Photoacid Dissolution Basic Optimum Base resin generator regulator compound Solvent dose Resolution Example (pbw) (pbw) (pbw) (pbw) (pbw) (mJ/cm2) (μm) Shape I-21 Polymer 12 PAG 2 TMMEA PGMEA 22.0 0.20 rectangular (80) (1) (0.118) (480) I-22 Polymer 12 PAG 2 TMEMEA PGMEA 22.5 0.22 rectangular (80) (1) (0.173) (480) I-23 Polymer 7 PAG 2 DRR I TEA PGMEA 22.0 0.24 rectangular (70) (1) (10) (0.063) (480) I-24 Polymer 7 PAG 2 DRR 2 TEA PGMEA 23.5 0.22 rectangular (70) (1) (10) (0.063) (480) I-25 Polymer 7 PAG 2 DRR 3 TEA PGMEA 26.0 0.22 rectangular (70) (1) (10) (0.063) (480) I-26 Polymer 7 PAG 2 DRR 4 TEA PGMEA 24.5 0.20 rectangular (70) (1) (10) (0.063) (480) I-27 Polymer 7 PAG 2 ACC 1 TEA PGMEA 23.0 0.22 rectangular (80) (1) (4) (0.063) (480) I-28 Polymer 7 PAG 2 ACC 2 TEA PGMEA 24.0 0.22 rectangular (80) (1) (4) (0.063) (480) -
TABLE 3 Compar- Photoacid Dissolution Basic Optimum ative Base resin generator regulator compound Solvent dose Resolution Example (pbw) (pbw) (pbw) (pbw) (pbw) (mJ/cm2) (μm) Shape 1 Polymer 19 PAG 1 TBA PGMEA — — not (80) (1) (0.078) (480) resolved 2 Polymer 20 PAG 1 TBA PGMEA 29.0 0.28 T-top (80) (1) (0.078) (480) 3 Polymer 19 PAG 1 DRR 4 TEA PGMEA 27.0 0.26 forward (70) (1) (10) (0.063) (480) tapered 4 Polymer 20 PAG 1 DRR 4 TEA PGMEA 26.0 0.26 forward (70) (1) (10) (0.063) (480) tapered - It is seen from Tables 1 to 3 that the resist compositions within the scope of the invention have a high sensitivity and resolution upon KrF excimer laser exposure, as compared with prior art compositions.
- Resist compositions were formulated using inventive polymers and examined for resolution upon ArP excimer laser exposure.
- Resist compositions were prepared as in Example I in accordance with the formulation shown in Table 4.
- The resulting resist solutions were spin-coated onto silicon wafers, then baked on a hot plate at 110° C. for 90 seconds to give resist films having a thickness of 0.5 μm. The resist films were exposed using an ArF excimer laser stepper (Nikon Corporation; NA 0.55), then baked (PEB) at 110° C. for 90 seconds, and developed with a solution of 2.38% TMAH in water; thereby giving positive patterns.
- The resulting resist patterns were evaluated as described below. First, the sensitivity (Eth, mJ/cm 2) was determined. Next, the, optimal dose (Eop, mJ/cm2) was defined as the dose which provides a 1:1 resolution at the top and bottom of a 0.25 μm line-and-space pattern, and the resolution of the resist under evaluation was defined as the minimum line width (μm) of the lines and spaces that separated at this dose. The shape of the, resolved resist pattern was examined under a scanning electron microscope.
- The composition and test results of the resist materials are shown in Table 4.
TABLE 4 Photoacid Dissolution Basic Optimum Base resin generator regulator compound Solvent dose Resolution Example (pbw) (pbw) (pbw) (pbw) (pbw) (mJ/cm2) (μm) Shape II-1 Polymer 1 PAG 1 TBA PGMEA 17.0 0.16 rectangular (80) (1) (0.078) (480) II-2 Polymer 1 PAG 1 DRR 4 TEA PGMEA 15.5 0.16 rectangular (70) (1) (10) (0.063) (480) - It is seen from Table 4 that the resist compositions within the scope of the invention have a high sensitivity and resolution upon ArF excimer laser exposure.
- Japanese Patent Application No. 2000-119410 is incorporated herein-by reference.
- Although some preferred embodiments have been described, many modifications and variations may be made thereto in light of the above teachings. It is therefore to be understood that the invention may be practiced otherwise than as specifically described without departing from the scope of the appended claims.
Claims (7)
1. An ester compound of the following general formula (1):
wherein R1 is hydrogen, methyl or CH2CO2R3, R2 is hydrogen, methyl or CO2R3, R3 is a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, k is 0 or 1, Z is a divalent hydrocarbon group of 2 to 20 carbon atoms which forms a single ring or bridged ring with the carbon atom and which may contain a hetero atom, m is 0 or 1, n is 0, 1, 2 or 3, and 2 m+n is 2 or 3.
3. The polymer of claim 2 further comprising recurring units of at least one of the following formulas (2a) to (10a):
wherein R1, R2 and k are as defined above,
R4 is hydrogen or a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms,
at least one of R5 to R8 is a carboxyl or hydroxyl-containing monovalent hydrocarbon group of 1 to 15 carbon atoms, and the remainders are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, or R5 to R8, taken together, may form a ring, and when they form a ring, at least one of R5 to R8 is a carboxyl or hydroxyl-containing divalent hydrocarbon group of 1 to 15 carbon atoms, and the remainders are independently a single bond or a straight, branched or cyclic alkylene group of 1 to 15 carbon atoms,
R9 is a monovalent hydrocarbon group of 3 to 15 carbon atoms containing a —CO2— partial structure,
at least one of R10 to R13 is a monovalent hydrocarbon group of 2 to 15 carbon atoms containing a —CO2— partial structure, and the remainders are independently hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms, or R10 to R13, taken together, may form a ring, and when they form a ring, at least one of R10 to R13 is a divalent hydrocarbon group of 1 to 15 carbon atoms containing a —CO2— partial structure, and the remainders are independently a single bond or a straight, branched or cyclic alkylene group of 1 to 15 carbon atoms,
R14 is a polycyclic hydrocarbon group of 7 to 15 carbon atoms or an alkyl group containing such a polycyclic hydrocarbon group,
R15 is an acid labile group,
X is —CH2— or —O—, and
Y is —O— or —(NR16)— wherein R16 is hydrogen or a straight, branched or cyclic alkyl group of 1 to 15 carbon atoms.
4. A method for preparing a polymer comprising the step of effecting radical polymerization, anionic polymerization or coordination polymerization between an ester compound of the general formula (1) and another compound having a carbon-to-carbon double bond.
5. A resist composition comprising the polymer of claim 2 or 3.
6. A resist composition comprising the polymer of claim 2 or 3, a photoacid generator, and an organic solvent.
7. A process for forming a resist pattern comprising the steps of:
applying the resist composition of claim 5 or 6 onto a substrate to form a coating,
heat treating the coating and then exposing it to high-energy radiation or electron beams through a photo mask, and
optionally heat treating the exposed coating and developing it with a developer.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/390,795 US20030198891A1 (en) | 2000-04-20 | 2003-03-19 | Novel ester compounds polymers, resist compositions and patterning process |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2000-119410 | 2000-04-20 | ||
| JP2000119410 | 2000-04-20 | ||
| US09/837,219 US6596463B2 (en) | 2000-04-20 | 2001-04-19 | Ester compounds, polymers, resist compositions and patterning process |
| US10/390,795 US20030198891A1 (en) | 2000-04-20 | 2003-03-19 | Novel ester compounds polymers, resist compositions and patterning process |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/837,219 Division US6596463B2 (en) | 2000-04-20 | 2001-04-19 | Ester compounds, polymers, resist compositions and patterning process |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20030198891A1 true US20030198891A1 (en) | 2003-10-23 |
Family
ID=18630387
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/837,378 Expired - Lifetime US6586157B2 (en) | 2000-04-20 | 2001-04-19 | Ester compounds, polymers, resist compositions and patterning process |
| US09/837,219 Expired - Lifetime US6596463B2 (en) | 2000-04-20 | 2001-04-19 | Ester compounds, polymers, resist compositions and patterning process |
| US10/390,795 Abandoned US20030198891A1 (en) | 2000-04-20 | 2003-03-19 | Novel ester compounds polymers, resist compositions and patterning process |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/837,378 Expired - Lifetime US6586157B2 (en) | 2000-04-20 | 2001-04-19 | Ester compounds, polymers, resist compositions and patterning process |
| US09/837,219 Expired - Lifetime US6596463B2 (en) | 2000-04-20 | 2001-04-19 | Ester compounds, polymers, resist compositions and patterning process |
Country Status (5)
| Country | Link |
|---|---|
| US (3) | US6586157B2 (en) |
| EP (2) | EP1148045B1 (en) |
| KR (2) | KR100582158B1 (en) |
| DE (2) | DE60101695T2 (en) |
| TW (2) | TW507117B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050282082A1 (en) * | 2004-06-21 | 2005-12-22 | Shin-Etsu Chemical Co., Ltd. | Polymer, resist composition and patterning process |
| CN101950127A (en) * | 2009-05-20 | 2011-01-19 | 罗门哈斯电子材料有限公司 | Novel resins and photoresist compositions comprising same |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100501600B1 (en) * | 1999-11-15 | 2005-07-18 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Polymer, Resist Composition and Patterning Process |
| KR100498440B1 (en) * | 1999-11-23 | 2005-07-01 | 삼성전자주식회사 | Photosensitive polymer having cyclic backbone and resist composition comprising the same |
| US6777157B1 (en) * | 2000-02-26 | 2004-08-17 | Shipley Company, L.L.C. | Copolymers and photoresist compositions comprising same |
| TWI229240B (en) * | 2000-03-06 | 2005-03-11 | Shinetsu Chemical Co | Polymer, resist material, and method for forming pattern |
| TW507117B (en) * | 2000-04-20 | 2002-10-21 | Shinetsu Chemical Co | Novel ester compounds, polymers, resist compositions and patterning process |
| JP3589160B2 (en) * | 2000-07-07 | 2004-11-17 | 日本電気株式会社 | Resist material, chemically amplified resist, and pattern forming method using the same |
| JP4371206B2 (en) * | 2002-09-30 | 2009-11-25 | 信越化学工業株式会社 | Ester compound, polymer compound, resist material, and pattern forming method |
| JP2004219989A (en) * | 2002-12-25 | 2004-08-05 | Tokyo Ohka Kogyo Co Ltd | Positive type resist composition and method of forming resist pattern using the same |
| US6878504B2 (en) * | 2003-05-28 | 2005-04-12 | Everlight Usa, Inc. | Chemically-amplified resist compositions |
| DE102004046405A1 (en) * | 2004-09-24 | 2006-04-06 | Infineon Technologies Ag | New electrically conductive polymer, comprising an acid cleavable group and a condensed aromatic group, useful in photo- and/or electron ray sensitive composition, which is useful for structuring a substrate using photo resisting layer |
| US7476492B2 (en) | 2006-05-26 | 2009-01-13 | International Business Machines Corporation | Low activation energy photoresist composition and process for its use |
| JP4910662B2 (en) * | 2006-11-29 | 2012-04-04 | 信越化学工業株式会社 | Positive resist material and pattern forming method |
| US20110039210A1 (en) | 2009-05-20 | 2011-02-17 | Rohm And Haas Electronic Materials Llc | Novel resins and photoresist compositions comprising same |
| US20130171429A1 (en) * | 2011-12-31 | 2013-07-04 | Rohm And Haas Electronic Materials Llc | Cycloaliphatic monomer, polymer comprising the same, and photoresist composition comprising the polymer |
| US8790861B2 (en) | 2011-12-31 | 2014-07-29 | Rohm And Haas Electronic Materials Llc | Cycloaliphatic monomer, polymer comprising the same, and photoresist composition comprising the polymer |
| JP5772760B2 (en) * | 2012-08-13 | 2015-09-02 | 信越化学工業株式会社 | Positive resist material and pattern forming method using the same |
| JP6904245B2 (en) * | 2017-12-27 | 2021-07-14 | 信越化学工業株式会社 | Photosensitive resin composition, pattern forming method, and manufacturing method of optical semiconductor device |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4491628A (en) * | 1982-08-23 | 1985-01-01 | International Business Machines Corporation | Positive- and negative-working resist compositions with acid generating photoinitiator and polymer with acid labile groups pendant from polymer backbone |
| US4603101A (en) * | 1985-09-27 | 1986-07-29 | General Electric Company | Photoresist compositions containing t-substituted organomethyl vinylaryl ether materials |
| US5069997A (en) * | 1988-05-19 | 1991-12-03 | Keil & Weinkauf | Positive and negative working radiation sensitive mixtures and production of relief patterns |
| US5585219A (en) * | 1991-09-19 | 1996-12-17 | Fujitsu Limited | Resist composition and process for forming resist pattern |
| US6147249A (en) * | 1998-05-11 | 2000-11-14 | Shin-Etsu Chemical Co., Ltd. | Ester compounds, polymers, resist composition and patterning process |
| US20010044071A1 (en) * | 2000-04-20 | 2001-11-22 | Shin-Etsu Chemical Co., Ltd | Novel ester compounds, polymers, resist compositions and patterning process |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2578646B2 (en) | 1988-07-18 | 1997-02-05 | 三洋電機株式会社 | Non-aqueous secondary battery |
| JPH04215661A (en) | 1990-12-14 | 1992-08-06 | Fujitsu Ltd | Formation of resist pattern |
| JPH0588367A (en) | 1991-09-30 | 1993-04-09 | Fujitsu Ltd | Resist composition and formation of resist pattern |
| JP3772249B2 (en) * | 1998-05-11 | 2006-05-10 | 信越化学工業株式会社 | Novel ester compound, polymer compound, resist material, and pattern forming method |
| EP1000924A4 (en) * | 1998-05-25 | 2005-01-05 | Daicel Chem | Acid-sensitive compound and resin composition for photoresist |
| DE19822968A1 (en) * | 1998-05-25 | 1999-12-02 | Rwe Dea Ag | Process for the catalytic hydroformylation of olefins |
| KR20000015014A (en) | 1998-08-26 | 2000-03-15 | 김영환 | New photoresist monomer, polymer, and photoresist compound |
| KR100441734B1 (en) | 1998-11-02 | 2004-08-04 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Novel Ester Compounds, Polymers, Resist Materials and Process for Forming Pattern |
| JP3997382B2 (en) * | 2000-04-28 | 2007-10-24 | 信越化学工業株式会社 | Novel ester compound having alicyclic structure and process for producing the same |
| KR100497091B1 (en) * | 2000-10-02 | 2005-06-27 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Cyclic Acetal Compound, Polymer, Resist Composition and Patterning Process |
-
2001
- 2001-04-19 TW TW090109382A patent/TW507117B/en not_active IP Right Cessation
- 2001-04-19 EP EP01303555A patent/EP1148045B1/en not_active Expired - Lifetime
- 2001-04-19 KR KR1020010021007A patent/KR100582158B1/en active IP Right Grant
- 2001-04-19 US US09/837,378 patent/US6586157B2/en not_active Expired - Lifetime
- 2001-04-19 DE DE60101695T patent/DE60101695T2/en not_active Expired - Lifetime
- 2001-04-19 DE DE60100721T patent/DE60100721T2/en not_active Expired - Lifetime
- 2001-04-19 TW TW090109383A patent/TW520357B/en not_active IP Right Cessation
- 2001-04-19 EP EP01303574A patent/EP1148044B1/en not_active Expired - Lifetime
- 2001-04-19 KR KR1020010021086A patent/KR100582630B1/en active IP Right Grant
- 2001-04-19 US US09/837,219 patent/US6596463B2/en not_active Expired - Lifetime
-
2003
- 2003-03-19 US US10/390,795 patent/US20030198891A1/en not_active Abandoned
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4491628A (en) * | 1982-08-23 | 1985-01-01 | International Business Machines Corporation | Positive- and negative-working resist compositions with acid generating photoinitiator and polymer with acid labile groups pendant from polymer backbone |
| US4603101A (en) * | 1985-09-27 | 1986-07-29 | General Electric Company | Photoresist compositions containing t-substituted organomethyl vinylaryl ether materials |
| US5069997A (en) * | 1988-05-19 | 1991-12-03 | Keil & Weinkauf | Positive and negative working radiation sensitive mixtures and production of relief patterns |
| US5118585A (en) * | 1988-05-19 | 1992-06-02 | Basf Aktiengesellschaft | Positive and negative working radiation sensitive mixtures and production of relief patterns |
| US5585219A (en) * | 1991-09-19 | 1996-12-17 | Fujitsu Limited | Resist composition and process for forming resist pattern |
| US5585222A (en) * | 1991-09-19 | 1996-12-17 | Fujitsu Limited | Resist composition and process for forming resist pattern |
| US6147249A (en) * | 1998-05-11 | 2000-11-14 | Shin-Etsu Chemical Co., Ltd. | Ester compounds, polymers, resist composition and patterning process |
| US20010044071A1 (en) * | 2000-04-20 | 2001-11-22 | Shin-Etsu Chemical Co., Ltd | Novel ester compounds, polymers, resist compositions and patterning process |
| US20020004178A1 (en) * | 2000-04-20 | 2002-01-10 | Shin-Etsu Chemical Co., Ltd. | Novel ester compounds, polymers, resist compositions and patterning process |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050282082A1 (en) * | 2004-06-21 | 2005-12-22 | Shin-Etsu Chemical Co., Ltd. | Polymer, resist composition and patterning process |
| US7598015B2 (en) | 2004-06-21 | 2009-10-06 | Shin-Etsu Chemical Co., Ltd. | Polymer, resist composition and patterning process |
| CN101950127A (en) * | 2009-05-20 | 2011-01-19 | 罗门哈斯电子材料有限公司 | Novel resins and photoresist compositions comprising same |
| EP2293143A3 (en) * | 2009-05-20 | 2011-03-23 | Rohm and Haas Electronic Materials, L.L.C. | Novel resins and photoresist compositions comprising the same |
| TWI468857B (en) * | 2009-05-20 | 2015-01-11 | 羅門哈斯電子材料有限公司 | Novel resins and photoresist compositions comprising same |
Also Published As
| Publication number | Publication date |
|---|---|
| TW520357B (en) | 2003-02-11 |
| EP1148045A1 (en) | 2001-10-24 |
| KR100582630B1 (en) | 2006-05-23 |
| DE60100721T2 (en) | 2004-07-15 |
| DE60101695T2 (en) | 2004-12-09 |
| US20010044071A1 (en) | 2001-11-22 |
| KR20010098725A (en) | 2001-11-08 |
| US6596463B2 (en) | 2003-07-22 |
| TW507117B (en) | 2002-10-21 |
| DE60101695D1 (en) | 2004-02-12 |
| KR20010098739A (en) | 2001-11-08 |
| KR100582158B1 (en) | 2006-05-23 |
| US6586157B2 (en) | 2003-07-01 |
| US20020004178A1 (en) | 2002-01-10 |
| EP1148044A1 (en) | 2001-10-24 |
| DE60100721D1 (en) | 2003-10-16 |
| EP1148044B1 (en) | 2004-01-07 |
| EP1148045B1 (en) | 2003-09-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6596463B2 (en) | Ester compounds, polymers, resist compositions and patterning process | |
| US6667145B1 (en) | Resist composition and patterning process | |
| US6566038B2 (en) | Polymers, resist compositions and patterning process | |
| US6492090B2 (en) | Polymers, resist compositions and patterning process | |
| US6743566B2 (en) | Cyclic acetal compound, polymer, resist composition and patterning process | |
| US6515149B2 (en) | Acetal compound, polymer, resist composition and patterning response | |
| US6399274B1 (en) | Resist composition and patterning process | |
| US6566037B2 (en) | Polymer, resist composition and patterning process | |
| US6531627B2 (en) | Ester compounds, polymers, resist compositions and patterning process | |
| US6509135B2 (en) | Polymer, resist composition and patterning process | |
| US6660448B2 (en) | Polymer, resist composition and patterning process | |
| US6524765B1 (en) | Polymer, resist composition and patterning process | |
| JP3879815B2 (en) | Novel ester compound, polymer compound, resist material, and pattern forming method | |
| US6624335B2 (en) | Ether, polymer, resist composition and patterning process |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |