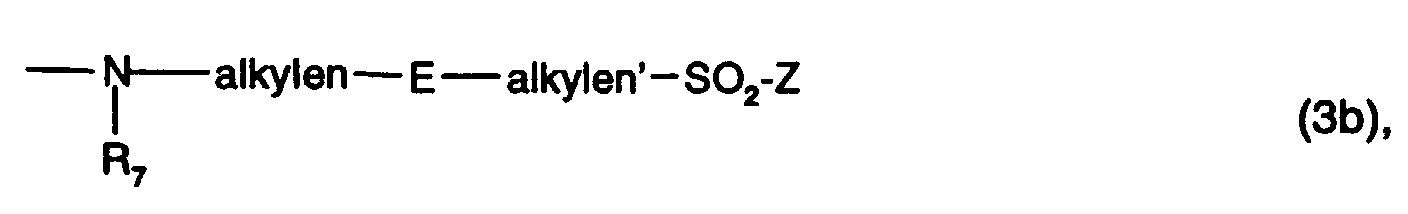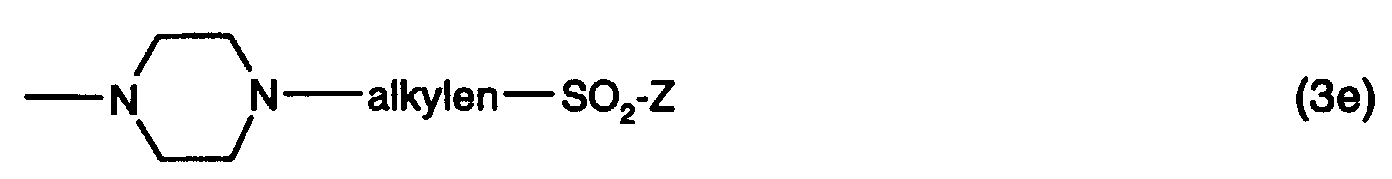EP0886000A2 - Process for ink jet printing textile materials - Google Patents
Process for ink jet printing textile materials Download PDFInfo
- Publication number
- EP0886000A2 EP0886000A2 EP98810525A EP98810525A EP0886000A2 EP 0886000 A2 EP0886000 A2 EP 0886000A2 EP 98810525 A EP98810525 A EP 98810525A EP 98810525 A EP98810525 A EP 98810525A EP 0886000 A2 EP0886000 A2 EP 0886000A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- alkyl
- sulfo
- alkylene
- carboxy
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 0 CC(C=CC=C1)C=C1N=Nc(c(*)n[n]1C2=C*(*)C=CC=C2)c1O Chemical compound CC(C=CC=C1)C=C1N=Nc(c(*)n[n]1C2=C*(*)C=CC=C2)c1O 0.000 description 6
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P5/00—Other features in dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form
- D06P5/30—Ink jet printing
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/38—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using reactive dyes
- D06P1/382—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using reactive dyes reactive group directly attached to heterocyclic group
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/46—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing natural macromolecular substances or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/46—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing natural macromolecular substances or derivatives thereof
- D06P1/48—Derivatives of carbohydrates
- D06P1/50—Derivatives of cellulose
Definitions
- the present invention relates to a method for printing textile fiber materials with reactive dyes according to the inkjet printing process (jet and inkjet processes) as well as the inks used for this process.
- Inkjet printing processes have been in the textile industry for several years applied. These methods make it possible to produce a Eliminate the printing stencil, so that considerable cost and time savings are achieved can be. Especially in the production of sample templates can within reacted to changing needs much less time.
- Corresponding inkjet printing processes should in particular be optimal have application properties. Worth mentioning in this Relationship properties such as viscosity, stability, surface tension and Conductivity of the inks used. There are also increased quality requirements of the prints obtained, e.g. regarding color strength, fiber-dye binding stability as well as wet fastness properties. Of the known methods these requirements are not met in all properties, so there is still a need for there are new processes for textile inkjet printing.
- R 1 , R 2 , R 3 , R 4 and R 5 can be further substituted as alkyl radicals, for example by hydroxy, sulfo, sulfato, cyano or carboxy.
- R 1 , R 2 , R 3 , R 4 and R 5 are preferably, independently of one another, hydrogen or C 1 -C 4 alkyl, in particular hydrogen.
- X 1 is preferably chlorine or in particular fluorine.
- B 1 is preferably a C 2 -C 12 alkylene radical which can be interrupted by 1, 2 or 3 members from the group -NH-, -N (CH 3 ) - or -O- and is unsubstituted or by hydroxyl, sulfo, Sulfato, cyano or carboxy is substituted; or a phenylene radical optionally substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, C 2 -C 4 alkanoylamino, sulfo, halogen or carboxy.
- B 1 is particularly preferably a C 2 -C 12 -alkylene radical which can be interrupted by 1, 2 or 3 members from the group -NH-, -N (CH 3 ) - or in particular -O- and is unsubstituted or by hydroxy, Sulfo, sulfato, cyano or carboxy is substituted.
- the alkylene radical is preferably unsubstituted or substituted by hydroxy, sulfo or sulfato, in particular by hydroxy.
- B 1 is very particularly preferably a C 2 -C 12 alkylene radical, in particular a C 2 -C 6 alkylene radical, which can be interrupted by 1, 2 or 3 members -O- and is unsubstituted or substituted by hydroxy.
- C 2 -C 6 alkylene radicals are of particular interest as bridge members B 1 .
- Alkylene and alkylene ' are independently of each other e.g. a methylene, Ethylene, 1,3-propylene, 1,4-butylene, 1,5-pentylene or 1,6-hexylene radical or their branched isomers.
- Alkylene and alkylene preferably represent a C 2 -C 3 alkylene radical and particularly preferably an ethylene radical.
- Arylene is preferably a 1,3- or 1,4-phenylene radical which is unsubstituted or e.g. by Sulfo, methyl, methoxy or carboxy is substituted.
- the leaving group U 1 is, for example, -Cl, -Br, -F, -OSO 3 H, -SSO 3 H, -OCO-CH 3 , -OPO 3 H 2 , -OCO-C 6 H 5 , - OSO 2 -C 1 -C 4 alkyl or -OSO 2 -N (C 1 -C 4 alkyl) 2 .
- U 1 is preferably a group of the formula -Cl, -OSO 3 H, -SSO 3 H, -OCO-CH 3 , -OCO-C 6 H 5 or -OPO 3 H 2 , in particular -Cl or -OSO 3 H and particularly preferred -OSO 3 H.
- Z is preferably vinyl or a radical of the formula -CH 2 -CH 2 -OSO 3 H, in particular vinyl.
- E preferably represents -NH- and particularly preferably -O-.
- W preferably denotes a group of the formula -NHCO- or in particular -CONH-.
- R is preferably hydrogen or the group -SO 2 -Z, where Z has the meanings and preferences given above. R particularly preferably represents hydrogen.
- R 6 is preferably hydrogen, C 1 -C 4 alkyl or a group -alkylene-SO 2 -Z, in which alkylene and Z each have the meanings given above.
- R 6 is particularly preferably hydrogen or C 1 -C 4 alkyl, in particular hydrogen.
- R 7 is preferably hydrogen or a C 1 -C 4 alkyl radical and particularly preferably hydrogen.
- the number 0 is preferred for t.
- Preferred as fiber-reactive radicals V are those of the formulas (3b) to (3d), in particular that Formula (3b) or (3d) and preferably of formula (3b).
- a 2 is optionally substituted C 1 -C 4 alkyl, phenyl or naphthyl, this may, for example, unsubstituted or substituted by sulfo, sulfato, hydroxyl, carboxyl or phenyl-substituted C 1 -C 4 alkyl; or phenyl or naphthyl which is unsubstituted or substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, carboxy, sulfo or halogen. Phenyl which is unsubstituted or substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, carboxy, sulfo or halogen is preferred.
- a 2 is preferably the residue of a monoazo, polyazo, metal complex azo, anthraquinone, phthalocyanine, formazan or dioxazine chromophore containing at least one sulfo group.
- the radicals A 1 , A 2 and A 3 can contain, as a radical of a monoazo, polyazo, metal complex azo, anthraquinone, phthalocyanine, formazane or dioxazine chromophore on their backbone, the substituents customary in organic dyes.
- substituents in the radicals A 1 , A 2 and A 3 are: Alkyl groups with 1 to 4 carbon atoms, such as methyl, ethyl, propyl, isopropyl or butyl, it being possible for the alkyl radicals to be further substituted, for example by hydroxyl, sulfo or sulfato; Alkoxy groups with 1 to 4 carbon atoms, such as methoxy, ethoxy, propoxy, isopropoxy or butoxy, it being possible for the alkyl radicals to be further substituted, for example by hydroxyl, sulfo or sulfato; phenyl optionally substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy or sulfo; Acylamino groups with 1 to 8 carbon atoms, especially those alkanoylamino groups such as acetylamino or propionylamino; benzoylamino optionally
- a 1 , A 2 and A 3 represent the remainder of a monoazo, polyazo or metal complex azo chromophore, the following are particularly suitable:
- D is the remainder of a diazo component of the benzene or naphthalene series
- M is the remainder of a middle component of the benzene or naphthalene series
- K is the remainder of a coupling component of the benzene, naphthalene, pyrazolone, 6-hydroxypyridone (2) or acetoacetic acid arylamide series
- u are the number 0 or 1, where D, M and K are common substituents in azo dyes, for example C 1 -C 4 -alkyl or C 1 -C 4 -alkoxy, halogen, carboxy, sulfo which is further substituted by hydroxy, sulfo or sulfato , Nitro, cyan, trifluoromethyl, s
- the metal complexes derived from the above dye residues of the formulas (4a) and (4b) are also possible, these being, in particular, dye residues of a 1: 1 copper complex azo dye of the benzene or naphthalene series, in which the copper atom is attached to a metallizable group, such as a hydroxy group, is bound on both sides in the ortho position to the azo bridge. If the chromophore radicals of the formula (4a) or (4b) carry a reactive radical, this preferably corresponds to the formula SO 2 Z given above.
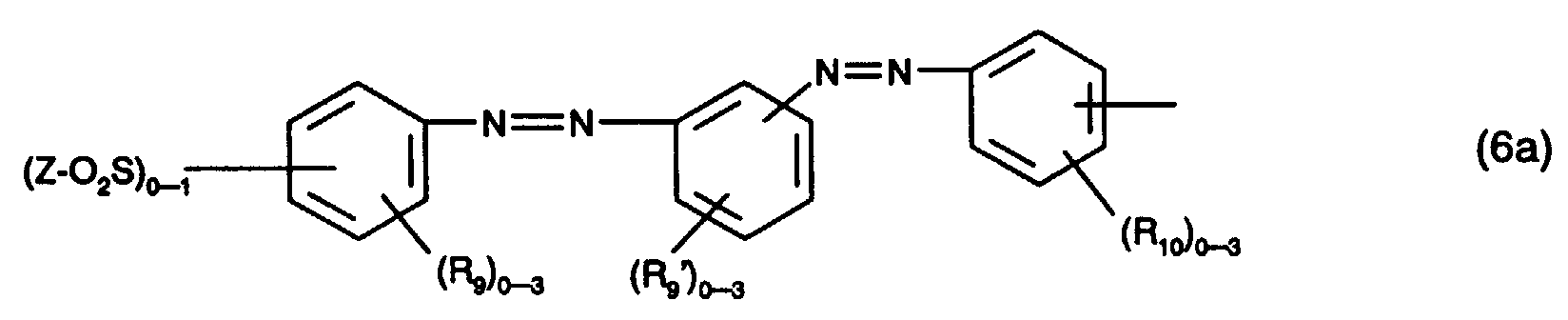
- the radicals of the formulas (4a) and (4b) are preferably those of the formula wherein (R 9 ) 0-3 represents 0 to 3 identical or different substituents from the group C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy and sulfo and Z has the meaning given above, where Z has the meaning given above, wherein (R 10 ) 0-4 for 0 to 4 identical or different substituents from the group halogen, nitro, cyano, trifluoromethyl, sulfamoyl, carbamoyl, C 1 -C 4 alkyl, C 1 -C 4 alkoxy, amino, acetylamino , Ureido, hydroxy, carboxy, sulfomethyl and sulfo and Z has the meaning given above, wherein (R 11 ) 0-3 represents 0 to 3 identical or different substituents from the group C 1 -C 4 alkyl, C 1 -C 4 alk
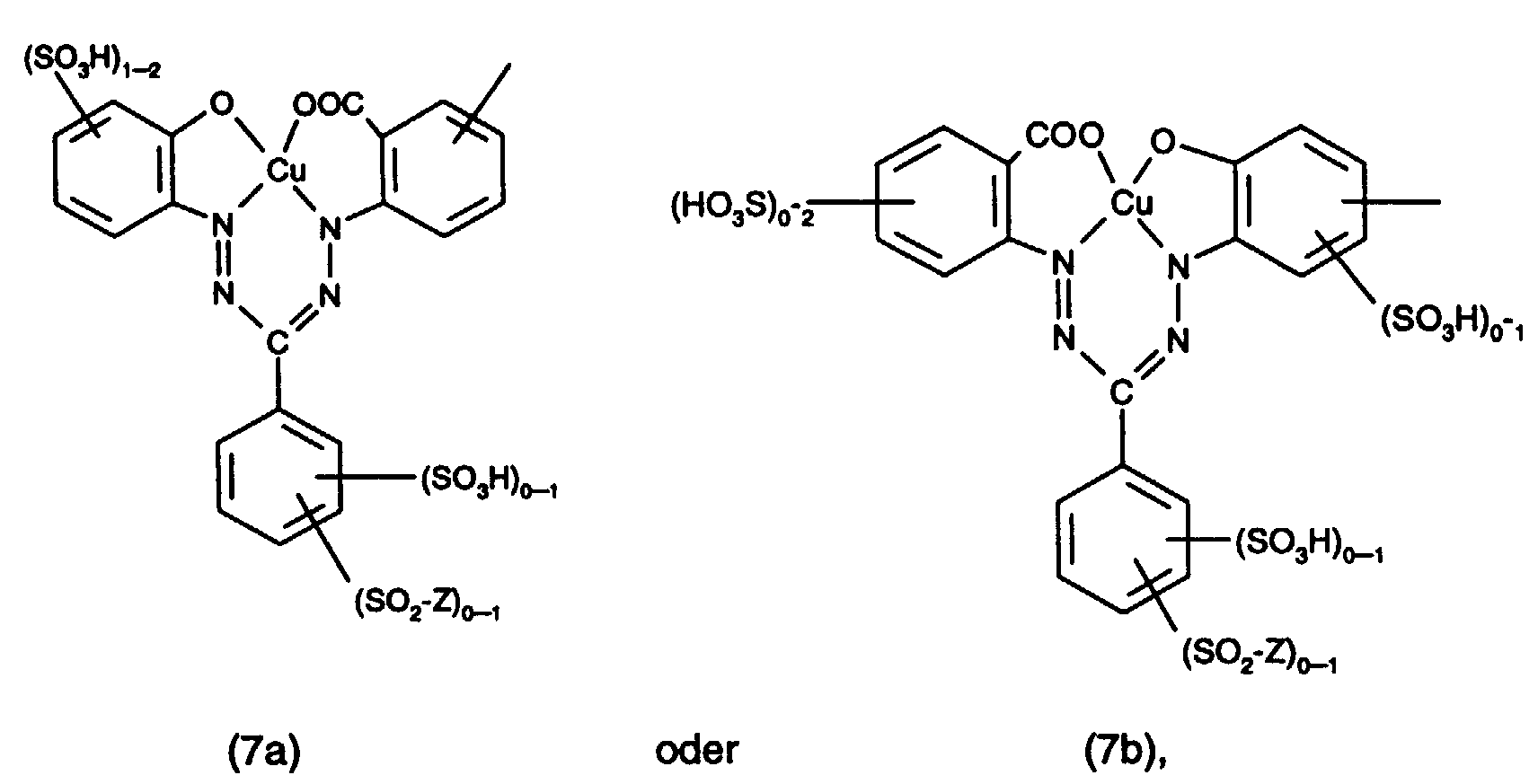
- the rest of a formazan chromophore are preferably those of the formula wherein Z has the meaning given above and the benzene nuclei contain no further substituents or are further substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, C 1 -C 4 alkylsulfonyl, halogen or carboxy.
- the radicals of the formulas (7a) and (7b) preferably contain no further substituents and no —SO 2 —Z radical.
- an anthraquinone chromophore is preferably of the formula wherein G is an unsubstituted or substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy or sulfo phenylene radical or a cyclohexylene, phenylene methylene or C 2 -C 6 alkylene radical.
- the radicals A 1 , A 2 and A 3 are preferably those of the formulas (5a) to (5e), (6a) to (6d), (7a), (7b), (8), (9) or (10), the radicals of the formulas (6a) to (6d), (7a) and (7b) preferably not containing any fiber-reactive radical.
- the radicals of the formulas (5a) to (5e), (7a), (7b) and (10) are particularly interested.
- the radicals A 1 , A 2 and A 3 preferably each contain 1 to 4 sulfo groups and preferably 1 to 3 sulfo groups.
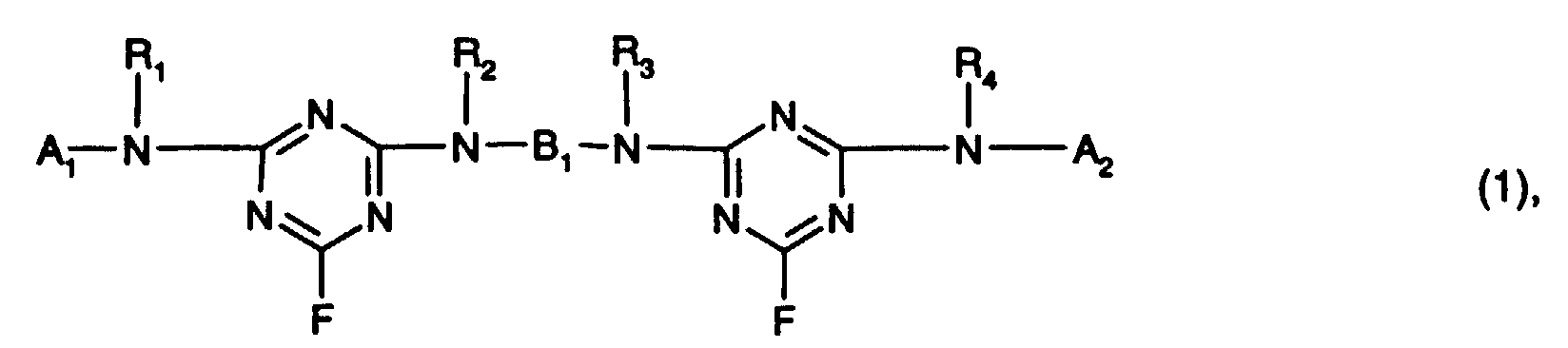
- the reactive dyes of the formulas (1) and (2) each contain at least two fiber reactive groups.
- the reactive dye of formula (1) contains two fiber-reactive ones Triazine residues and the reactive dye of formula (2) a fiber-reactive triazine residue and a fiber-reactive radical V.
- the reactive dyes of the formulas (1) preferably contain and (2) only two fiber reactive groups at a time.
- Reactive dyes of the formula are of particular interest or
- the reactive dyes of the formulas (1) and (2) are known or can be used in analogy to known compounds, e.g. through usual diazotization, Coupling and condensation reactions.
- the reactive dyes of formulas (1) and (2) used in the inks should preferably low in salt, i.e. a total salt content of less than 0.5 wt .-%, based on the weight of the dyes.
- Reactive dyes that due to their manufacture and / or the subsequent addition of coupé agents have higher salt contents, e.g. by membrane separation processes, such as Ultrafiltration, reverse osmosis or dialysis.
- the inks preferably contain exclusively dyes containing sulfo groups, water-soluble reactive dyes, such as those of formulas (1) and (2) above.
- the inks preferably contain a total reactive dye content of the above Formulas (1) and (2) from 5 to 35% by weight, in particular 10 to 35% by weight and preferably 10 to 20% by weight based on the total weight of the ink.
- cellulose ethers e.g. Methyl, ethyl, Hydroxyethyl, methylhydroxyethyl, hydroxypropyl or hydroxypropylmethyl cellulose in Consideration. Methyl cellulose or, in particular, hydroxyethyl cellulose are preferred.
- the Cellulose ethers are usually used in the ink in an amount of 0.01 to 2% by weight, in particular 0.01 to 1 wt .-% and preferably 0.01 to 0.5 wt .-%, based on the Total weight of ink used.
- Alkali alginates and preferably sodium alginate are particularly suitable as alginates Consideration. These are usually contained in the ink in an amount of 0.01 to 2% by weight, in particular 0.01 to 1 wt .-% and preferably 0.01 to 0.5 wt .-%, based on the Total weight of ink used.
- Both the water-soluble, non-ionic cellulose ether used and the Alginates are used as so-called thickeners and allow the setting of one certain viscosity of the ink.
- Inks are preferred which have a viscosity of 1 to 40 mPas, in particular 5 to 40 mPa ⁇ s and preferably 10 to 40 mPa ⁇ s. Inks with a viscosity of 10 to 30 mPa ⁇ s are particularly preferred.
- the inks may also contain buffer substances, such as borax, borate or citrate.
- buffer substances such as borax, borate or citrate.
- borax borate
- citrate sodium borate, sodium tetraborate and sodium citrate. They are used in particular in amounts of 0.1 to 3% by weight, preferably 0.1 to 1% by weight, based on the total weight of the ink, in order to have a pH of, for example, 5 to 9, in particular 6 to 8, adjust.
- a citrate buffer is preferably used in the case of inks containing alginate.
- the inks can e.g. N-methyl-2-pyrrolidone or in particular 1,2-propylene glycol contain. These are usually present in the ink in an amount of 5 to 30% by weight, in particular 5 to 20% by weight and preferably 10 to 20% by weight on the total weight of the ink used.
- the inks can also contain conventional additives such as e.g. foam suppressants or in particular substances that inhibit fungal and / or bacterial growth. This are usually in amounts of 0.01 to 1 wt .-%, based on the total weight the ink used.
- foam suppressants or in particular substances that inhibit fungal and / or bacterial growth. This are usually in amounts of 0.01 to 1 wt .-%, based on the total weight the ink used.
- the method according to the invention for printing textile fiber materials can with performed on known ink jet printers suitable for textile printing will.
- inkjet printing process individual drops of the ink are checked out sprayed onto a substrate using a nozzle.
- the continuous ink jet method is predominantly used for this as well as the drop on demand method.
- the ink-jet method creates the drops continuously, not for printing required drops are drained into a collecting container and recycled.
- Drop on demand method drops are created and printed as desired; i.e. drops are only generated when this is necessary for printing.
- the Generation of the drops can e.g. by means of a piezo inkjet head or by means of thermal Energy (bubble jet). Pressure is preferred for the method according to the invention using the drop-on-demand method, in particular using a piezo inkjet head.
- fiber materials containing hydroxyl groups come as textile fiber materials into consideration.
- cellulose-containing fiber materials which are made up entirely or in part Cellulose exist. Examples are natural fiber materials such as cotton, linen or Hemp and regenerated fiber materials such as Viscose and lyocell. Especially viscose or preferably cotton are preferred.
- the above Fiber materials are preferably in the form of flat textile fabrics, knitted fabrics or webs.
- the aqueous alkaline liquor contains at least one of the usual bases, which in conventional Reactive printing processes are used to fix the reactive dyes.
- the base is e.g. in an amount of 10 to 100 g / l liquor, preferably 10 to 50 g / l Fleet, deployed.
- Sodium bicarbonate is preferred, sodium carbonate or a mixture of water glass and sodium carbonate is used.
- the pH the alkaline liquor is usually 7.5 to 13.5, preferably 8.5 to 12.5.
- the aqueous alkaline liquor can contain other additives, e.g. Hydrotrope, contain.
- Urea is preferably used as the hydrotroping agent, e.g. in a Amount of 25 to 200 g / l liquor, preferably 50 to 150 g / l liquor is used.
- the fiber material is preferably dried after the above pretreatment.
- the fiber material is advantageously dried, preferably at temperatures up to 150 ° C, especially 80 to 120 ° C, and then a heat treatment process subjected to complete the print, or the dye to fix.
- the heat treatment can e.g. through a warm dwell process, a thermal insulation process or preferably be carried out by a steaming process.
- the printed fiber material is e.g. one treatment in one Damper with possibly superheated steam, conveniently at one temperature from 95 to 180 ° C, advantageously in saturated steam.
- the printed fiber material is usually washed with water in the usual way washed out to remove unfixed dye.
- the prints obtainable by the process according to the invention have good prints General realities on; e.g. they have high fiber-dye binding stability both in the acidic and in the alkaline range, good lightfastness, good Wet fastness properties, such as fastness to washing, water, sea water, over-dyeing and perspiration, one good chlorine fastness, rubbing fastness, ironing fastness and pleating fastness as well as sharp contours and high color strength.
- the printing inks used are characterized by good stability and good viscosity properties. So the viscosity remains even during the Preserved high shear forces almost unchanged.
Abstract
Description
Die vorliegende Erfindung betrifft ein Verfahren zum Bedrucken von textilen Fasermaterialien mit Reaktivfarbstoffen nach dem Tintenstrahldruck-Verfahren (Jet- und Ink-Jet-Verfahren) sowie die für dieses Verfahren verwendeten Tinten.The present invention relates to a method for printing textile fiber materials with reactive dyes according to the inkjet printing process (jet and inkjet processes) as well as the inks used for this process.
Tintenstrahldruck-Verfahren werden bereits seit einigen Jahren in der Textilindustrie angewendet. Diese Verfahren ermöglichen es, auf die sonst übliche Herstellung einer Druckschablone zu verzichten, so dass erhebliche Kosten- und Zeiteinsparungen erzielt werden können. Insbesondere bei der Herstellung von Mustervorlagen kann innerhalb deutlich geringerer Zeit auf veränderte Bedürfnisse reagiert werden.Inkjet printing processes have been in the textile industry for several years applied. These methods make it possible to produce a Eliminate the printing stencil, so that considerable cost and time savings are achieved can be. Especially in the production of sample templates can within reacted to changing needs much less time.
Entsprechende Tintenstrahldruck-Verfahren sollten insbesondere optimale anwendungstechnische Eigenschaften aufweisen. Zu erwähnen seien in diesem Zusammenhang Eigenschaften wie die Viskosität, Stabilität, Oberflächenspannung und Leitfähigkeit der verwendeten Tinten. Ferner werden erhöhte Anforderungen an die Qualität der erhaltenen Drucke gestellt, wie z.B. bezüglich Farbstärke, Faser-Farbstoff-Bindungsstabilität sowie Nassechtheitseigenschaften. Von den bekannten Verfahren werden diese Anforderungen nicht in allen Eigenschaften erfüllt, so dass weiterhin ein Bedarf nach neuen Verfahren für den textilen Tintenstrahldruck besteht.Corresponding inkjet printing processes should in particular be optimal have application properties. Worth mentioning in this Relationship properties such as viscosity, stability, surface tension and Conductivity of the inks used. There are also increased quality requirements of the prints obtained, e.g. regarding color strength, fiber-dye binding stability as well as wet fastness properties. Of the known methods these requirements are not met in all properties, so there is still a need for there are new processes for textile inkjet printing.
Gegenstand der Erfindung ist ein Verfahren zum Bedrucken von textilen Fasermaterialien
nach dem Tintenstrahldruck-Verfahren, welches dadurch gekennzeichnet ist, dass man
diese Fasermaterialien mit einer wässrigen Tinte bedruckt, welche
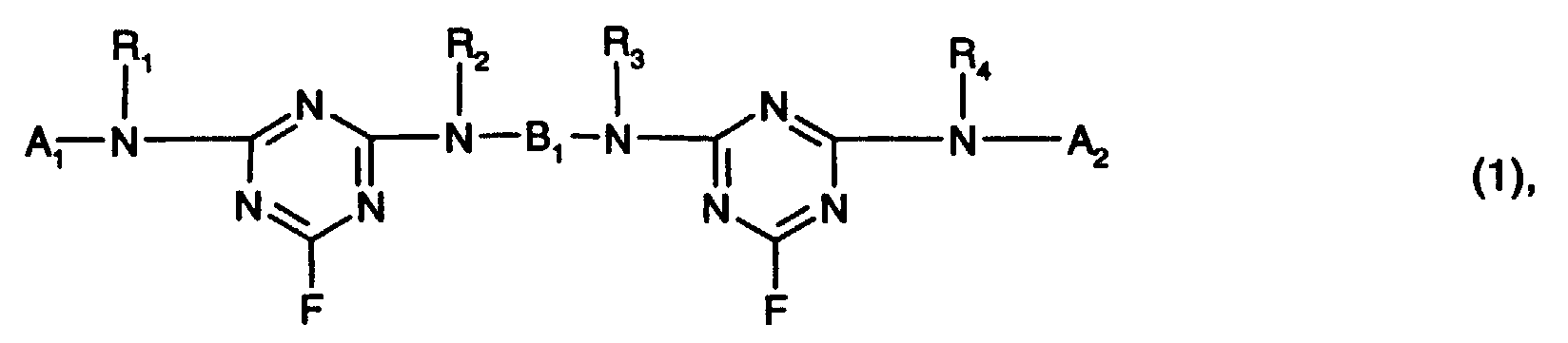
Die Reste R1, R2, R3, R4 und R5 können als Alkylreste weitersubstituiert sein, z.B. durch Hydroxy, Sulfo, Sulfato, Cyano oder Carboxy. Bevorzugt sind R1, R2, R3, R4 und R5 unabhängig voneinander Wasserstoff oder C1-C4-Alkyl, insbesondere Wasserstoff. The radicals R 1 , R 2 , R 3 , R 4 and R 5 can be further substituted as alkyl radicals, for example by hydroxy, sulfo, sulfato, cyano or carboxy. R 1 , R 2 , R 3 , R 4 and R 5 are preferably, independently of one another, hydrogen or C 1 -C 4 alkyl, in particular hydrogen.
X1 ist bevorzugt Chlor oder insbesondere Fluor.X 1 is preferably chlorine or in particular fluorine.
Als organische Brückenglieder B1 kommen z.B. die folgenden in Betracht:
Ferner kommt für den Rest der Formel -N(R2)-B1-N(R3)- auch ein Rest der Formel in Betracht.Furthermore, for the rest of the formula -N (R 2 ) -B 1 -N (R 3 ) - there is also a radical of the formula into consideration.
Bevorzugt ist B1 ein C2-C12-Alkylenrest, welcher durch 1, 2 oder 3 Glieder aus der Gruppe
-NH-, -N(CH3)- oder -O- unterbrochen sein kann und unsubstituiert oder durch Hydroxy,
Sulfo, Sulfato, Cyano oder Carboxy substituiert ist; oder
ein gegebenenfalls durch C1-C4-Alkyl, C1-C4-Alkoxy, C2-C4-Alkanoylamino, Sulfo, Halogen
oder Carboxy substituierter Phenylenrest.B 1 is preferably a C 2 -C 12 alkylene radical which can be interrupted by 1, 2 or 3 members from the group -NH-, -N (CH 3 ) - or -O- and is unsubstituted or by hydroxyl, sulfo, Sulfato, cyano or carboxy is substituted; or
a phenylene radical optionally substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, C 2 -C 4 alkanoylamino, sulfo, halogen or carboxy.
Besonders bevorzugt ist B1 ein C2-C12-Alkylenrest, welcher durch 1, 2 oder 3 Glieder aus der Gruppe -NH-, -N(CH3)- oder insbesondere -O- unterbrochen sein kann und unsubstituiert oder durch Hydroxy, Sulfo, Sulfato, Cyano oder Carboxy substituiert ist. Vorzugsweise ist der Alkylenrest unsubstituiert oder durch Hydroxy, Sulfo oder Sulfato, insbesondere durch Hydroxy, substituiert. B 1 is particularly preferably a C 2 -C 12 -alkylene radical which can be interrupted by 1, 2 or 3 members from the group -NH-, -N (CH 3 ) - or in particular -O- and is unsubstituted or by hydroxy, Sulfo, sulfato, cyano or carboxy is substituted. The alkylene radical is preferably unsubstituted or substituted by hydroxy, sulfo or sulfato, in particular by hydroxy.
Ganz besonders bevorzugt ist B1 ein C2-C12-Alkylenrest, insbesondere ein C2-C6-Alkylenrest, welcher durch 1, 2 oder 3 Glieder -O- unterbrochen sein kann und unsubstituiert oder durch Hydroxy substituiert ist.B 1 is very particularly preferably a C 2 -C 12 alkylene radical, in particular a C 2 -C 6 alkylene radical, which can be interrupted by 1, 2 or 3 members -O- and is unsubstituted or substituted by hydroxy.
Von besonderem Interesse als Brückenglieder B1 sind C2-C6-Alkylenreste.C 2 -C 6 alkylene radicals are of particular interest as bridge members B 1 .
Bei alkylen und alkylen' handelt es sich unabhängig voneinander z.B. um einen Methylen-, Äthylen-, 1,3-Propylen-, 1,4-Butylen-, 1,5-Pentylen- oder 1,6-Hexylenrest oder deren verzweigte Isomere.Alkylene and alkylene 'are independently of each other e.g. a methylene, Ethylene, 1,3-propylene, 1,4-butylene, 1,5-pentylene or 1,6-hexylene radical or their branched isomers.
Bevorzugt stehen alkylen und alkylen für einen C2-C3-Alkylenrest und insbesondere bevorzugt für einen Äthylenrest.Alkylene and alkylene preferably represent a C 2 -C 3 alkylene radical and particularly preferably an ethylene radical.
Arylen ist vorzugsweise ein 1,3- oder 1,4-Phenylenrest, der unsubstituiert oder z.B. durch Sulfo, Methyl, Methoxy oder Carboxy substituiert ist.Arylene is preferably a 1,3- or 1,4-phenylene radical which is unsubstituted or e.g. by Sulfo, methyl, methoxy or carboxy is substituted.
Bei der Abgangsgruppe U1 handelt es sich z.B. um -Cl, -Br, -F, -OSO3H, -SSO3H, -OCO-CH3, -OPO3H2, -OCO-C6H5, -OSO2-C1-C4-Alkyl oder -OSO2-N(C1-C4-Alkyl)2. Bevorzugt ist U1 eine Gruppe der Formel -Cl, -OSO3H, -SSO3H, -OCO-CH3, -OCO-C6H5 oder -OPO3H2, insbesondere -Cl oder -OSO3H und besonders bevorzugt -OSO3H.The leaving group U 1 is, for example, -Cl, -Br, -F, -OSO 3 H, -SSO 3 H, -OCO-CH 3 , -OPO 3 H 2 , -OCO-C 6 H 5 , - OSO 2 -C 1 -C 4 alkyl or -OSO 2 -N (C 1 -C 4 alkyl) 2 . U 1 is preferably a group of the formula -Cl, -OSO 3 H, -SSO 3 H, -OCO-CH 3 , -OCO-C 6 H 5 or -OPO 3 H 2 , in particular -Cl or -OSO 3 H and particularly preferred -OSO 3 H.
Bevorzugt ist Z Vinyl oder ein Rest der Formel -CH2-CH2-OSO3H, insbesondere Vinyl.Z is preferably vinyl or a radical of the formula -CH 2 -CH 2 -OSO 3 H, in particular vinyl.
E steht vorzugsweise für -NH- und insbesondere bevorzugt für -O-.E preferably represents -NH- and particularly preferably -O-.
W bedeutet bevorzugt eine Gruppe der Formel -NHCO- oder insbesondere -CONH-.W preferably denotes a group of the formula -NHCO- or in particular -CONH-.
R bedeutet bevorzugt Wasserstoff oder die Gruppe -SO2-Z, wobei für Z die zuvor angegebenen Bedeutungen und Bevorzugungen gelten. Besonders bevorzugt steht R für Wasserstoff.R is preferably hydrogen or the group -SO 2 -Z, where Z has the meanings and preferences given above. R particularly preferably represents hydrogen.
R6 ist vorzugsweise Wasserstoff, C1-C4-Alkyl oder eine Gruppe -alkylen-SO2-Z, worin alkylen und Z jeweils die zuvor angegebenen Bedeutungen haben. Besonders bevorzugt ist R6 Wasserstoff oder C1-C4-Alkyl, insbesondere Wasserstoff. R 6 is preferably hydrogen, C 1 -C 4 alkyl or a group -alkylene-SO 2 -Z, in which alkylene and Z each have the meanings given above. R 6 is particularly preferably hydrogen or C 1 -C 4 alkyl, in particular hydrogen.
R7 ist vorzugsweise Wasserstoff oder ein C1-C4-Alkylrest und insbesondere bevorzugt Wasserstoff.R 7 is preferably hydrogen or a C 1 -C 4 alkyl radical and particularly preferably hydrogen.
Für t ist die Zahl 0 bevorzugt.The number 0 is preferred for t.
Von besonderem Interesse sind faserreaktive Reste V, worin
R und R6 sind hierbei vorzugsweise Wasserstoff.Of particular interest are fiber-reactive radicals V, in which
R and R 6 are preferably hydrogen.
Bevorzugt als faserreaktive Reste V sind solche der Formeln (3b) bis (3d), insbesondere der Formel (3b) oder (3d) und vorzugsweise der Formel (3b).Preferred as fiber-reactive radicals V are those of the formulas (3b) to (3d), in particular that Formula (3b) or (3d) and preferably of formula (3b).
Steht A2 für gegebenenfalls substituiertes C1-C4-Alkyl, Phenyl oder Naphthyl, so kann dies z.B. unsubstituiertes oder durch Sulfo, Sulfato, Hydroxy, Carboxy oder Phenyl substituiertes C1-C4-Alkyl; oder unsubstituiertes oder durch C1-C4-Alkyl, C1-C4-Alkoxy, Carboxy, Sulfo oder Halogen substituiertes Phenyl oder Naphthyl sein. Bevorzugt ist hierbei unsubstituiertes oder durch C1-C4-Alkyl, C1-C4-Alkoxy, Carboxy, Sulfo oder Halogen substituiertes Phenyl.A 2 is optionally substituted C 1 -C 4 alkyl, phenyl or naphthyl, this may, for example, unsubstituted or substituted by sulfo, sulfato, hydroxyl, carboxyl or phenyl-substituted C 1 -C 4 alkyl; or phenyl or naphthyl which is unsubstituted or substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, carboxy, sulfo or halogen. Phenyl which is unsubstituted or substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, carboxy, sulfo or halogen is preferred.
Bevorzugt ist A2 der Rest eines mindestens eine Sufogruppe enthaltenden Monoazo-, Polyazo-, Metallkomplexazo-, Anthrachinon-, Phthalocyanin-, Formazan- oder Dioxazin-Chromophors.A 2 is preferably the residue of a monoazo, polyazo, metal complex azo, anthraquinone, phthalocyanine, formazan or dioxazine chromophore containing at least one sulfo group.
Die Reste A1, A2 und A3 können als Rest eines Monoazo-, Polyazo-, Metallkomplexazo-, Anthrachinon-, Phthalocyanin-, Formazan- oder Dioxazin-Chromophors an ihrem Grundgerüst die bei organischen Farbstoffen üblichen Substituenten gebunden enthalten. The radicals A 1 , A 2 and A 3 can contain, as a radical of a monoazo, polyazo, metal complex azo, anthraquinone, phthalocyanine, formazane or dioxazine chromophore on their backbone, the substituents customary in organic dyes.
Als Beispiele für Substituenten in den Resten A1, A2 und A3 seien genannt:
Alkylgruppen mit 1 bis 4 Kohlenstoffatomen, wie Methyl, Aethyl, Propyl, Isopropyl oder Butyl,
wobei die Alkylreste z.B. durch Hydroxyl, Sulfo oder Sulfato weitersubstituiert sein können;
Alkoxygruppen mit 1 bis 4 Kohlenstoffatomen, wie Methoxy, Aethoxy, Propoxy, lsopropoxy
oder Butoxy, wobei die Alkylreste z.B. durch Hydroxyl, Sulfo oder Sulfato weitersubstituiert
sein können; gegebenenfalls durch C1-C4-Alkyl, C1-C4-Alkoxy, Halogen, Carboxy oder Sulfo
substituiertes Phenyl; Acylaminogruppen mit 1 bis 8 Kohlenstoffatomen, insbesondere
solche Alkanoylaminogruppen, wie z.B. Acetylamino oder Propionylamino; gegebenenfalls
im Phenylring durch C1-C4-Alkyl, C1-C4-Alkoxy, Halogen oder Sulfo substituiertes
Benzoylamino; gegebenenfalls im Phenylring durch C1-C4-Alkyl, C1-C4-Alkoxy, Halogen oder
Sulfo substituiertes Phenylamino; N,N-Di-β-hydroxyäthylamino; N,N-Di-β-sulfatoäthylamino;
Sulfobenzylamino; N,N-Disulfobenzylamino; Alkoxycarbonyl mit 1 bis 4 Kohlenstoffatomen
im Alkoxyrest, wie Methoxycarbonyl oder Aethoxycarbonyl; Alkylsulfonyl mit 1 bis 4
Kohlenstoffatomen, wie Methylsulfonyl oder Aethylsulfonyl; Trifluormethyl; Nitro; Amino;
Cyano; Halogen, wie Fluor, Chlor oder Brom; Carbamoyl; N-Alkylcarbamoyl mit 1 bis 4
Kohlenstoffatomen im Alkylrest, wie N-Methylcarbamoyl oder N-Aethylcarbamoyl; Sulfamoyl;
N-Mono- oder N,N-Dialkylsulfamoyl mit jeweils 1 bis 4 Kohlenstoffatomen, wie N-Methylsulfamoyl,
N-Aethylsulfamoyl, N-Propylsulfamoyl, N-lsopropylsulfamoyl oder N-Butylsulfamoyl,
wobei die Alkylreste z.B. durch Hydroxy oder Sulfo weitersubstituiert sein
können; N-(β-Hydroxyäthyl)-sulfamoyl; N,N-Di-(β-hydroxyäthyl)-sulfamoyl; gegebenenfalls
durch C1-C4-Alkyl, C1-C4-Alkoxy, Halogen, Carboxy oder Sulfo substituiertes N-Phenylsulfamoyl;
Ureido; Hydroxy; Carboxy; Sulfomethyl oder Sulfo sowie faserreaktive
Reste.Examples of substituents in the radicals A 1 , A 2 and A 3 are:
Alkyl groups with 1 to 4 carbon atoms, such as methyl, ethyl, propyl, isopropyl or butyl, it being possible for the alkyl radicals to be further substituted, for example by hydroxyl, sulfo or sulfato; Alkoxy groups with 1 to 4 carbon atoms, such as methoxy, ethoxy, propoxy, isopropoxy or butoxy, it being possible for the alkyl radicals to be further substituted, for example by hydroxyl, sulfo or sulfato; phenyl optionally substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy or sulfo; Acylamino groups with 1 to 8 carbon atoms, especially those alkanoylamino groups such as acetylamino or propionylamino; benzoylamino optionally substituted in the phenyl ring by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen or sulfo; phenylamino optionally substituted in the phenyl ring by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen or sulfo; N, N-di-β-hydroxyethylamino; N, N-di-β-sulfatoethylamino; Sulfobenzylamino; N, N-disulfobenzylamino; Alkoxycarbonyl having 1 to 4 carbon atoms in the alkoxy radical, such as methoxycarbonyl or ethoxycarbonyl; Alkylsulfonyl having 1 to 4 carbon atoms, such as methylsulfonyl or ethylsulfonyl; Trifluoromethyl; Nitro; Amino; Cyano; Halogen such as fluorine, chlorine or bromine; Carbamoyl; N-alkylcarbamoyl with 1 to 4 carbon atoms in the alkyl radical, such as N-methylcarbamoyl or N-ethylcarbamoyl; Sulfamoyl; N-mono- or N, N-dialkylsulfamoyl each having 1 to 4 carbon atoms, such as N-methylsulfamoyl, N-ethylsulfamoyl, N-propylsulfamoyl, N-isopropylsulfamoyl or N-butylsulfamoyl, it being possible for the alkyl radicals to be further substituted, for example by hydroxyl or sulfo; N- (β-hydroxyethyl) sulfamoyl; N, N-di- (β-hydroxyethyl) sulfamoyl; N-phenylsulfamoyl optionally substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy or sulfo; Ureido; Hydroxy; Carboxy; Sulfomethyl or sulfo and fiber-reactive residues.
Als Beispiele für solche faserreaktiven Reste seien solche der Formel -NH-CO-Y und insbesondere -SO2-Z genannt, wobei für Z die oben angegebenen Bedeutungen und Bevorzugungen gelten und Y ein Rest der Formel -CH(Hal)-CH2-Hal oder -C(Hal)=CH2 und Hal Halogen, insbesondere Chlor oder vorzugsweise Brom, ist.Examples of such fiber-reactive radicals are those of the formula -NH-CO-Y and in particular -SO 2 -Z, where Z has the meanings and preferences given above and Y is a radical of the formula -CH (Hal) -CH 2 - Hal or -C (Hal) = CH 2 and Hal is halogen, in particular chlorine or preferably bromine.
Bedeuten A1, A2 und A3 den Rest eines Monoazo-, Polyazo- oder Metallkomplexazo-Chromophors, kommen insbesondere die folgenden in Betracht:If A 1 , A 2 and A 3 represent the remainder of a monoazo, polyazo or metal complex azo chromophore, the following are particularly suitable:
Chromophorreste eines Mono- oder Disazofarbstoffes der Formel
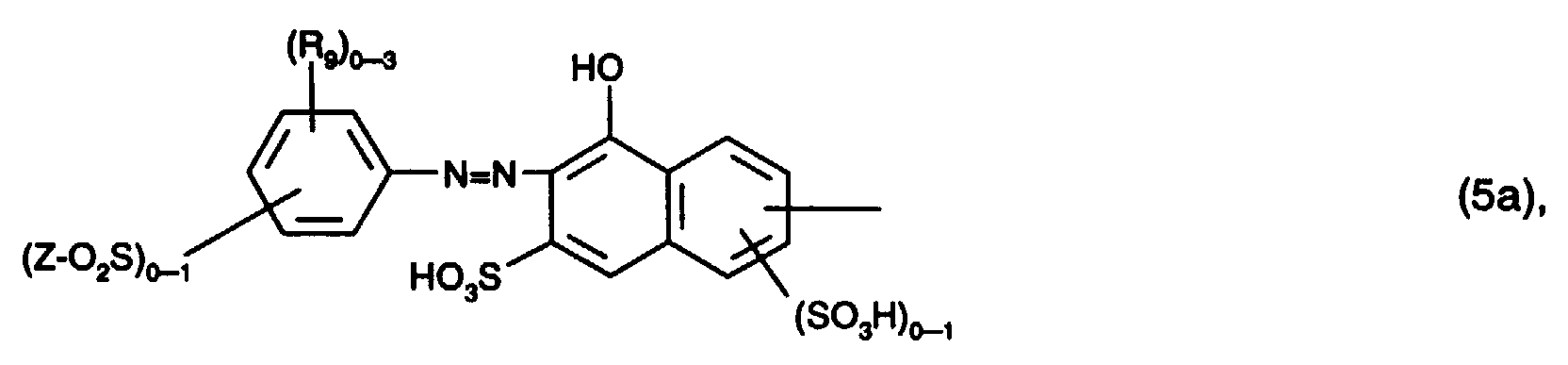
Bei den Resten der Formeln (4a) und (4b) handelt es sich bevorzugt um solche der Formel worin (R9)0-3 für 0 bis 3 gleiche oder verschiedene Substituenten aus der Gruppe C1-C4-Alkyl, C1-C4-Alkoxy, Halogen, Carboxy und Sulfo steht und Z die oben angegebene Bedeutung hat, worin Z die oben angegebene Bedeutung hat, worin (R10)0-4 für 0 bis 4 gleiche oder verschiedene Substituenten aus der Gruppe Halogen, Nitro, Cyan, Trifluormethyl, Sulfamoyl, Carbamoyl, C1-C4-Alkyl, C1-C4-Alkoxy, Amino, Acetylamino, Ureido, Hydroxy, Carboxy, Sulfomethyl und Sulfo steht und Z die oben angegebene Bedeutung hat, worin (R11)0-3 für 0 bis 3 gleiche oder verschiedene Substituenten aus der Gruppe C1-C4-Alkyl, C1-C4-Alkoxy, Halogen, Carboxy und Sulfo steht, worin R12 und R14 unabhängig voneinander Wasserstoff, C1-C4-Alkyl oder Phenyl, und R13 Wasserstoff, Cyano, Carbamoyl oder Sulfomethyl ist, oder oder oder worin (R9)0-3, (R10)0-3, (R11)0-3 und Z jeweils die oben angegebene Bedeutung haben und (R9')0-3 für 0 bis 3 gleiche oder verschiedene Substituenten aus der Gruppe C1-C4-Alkyl, C1-C4-Alkoxy, Halogen, Carboxy oder Sulfo steht. Von besonderem Interesse sind hierbei die Reste der Formeln (5a) bis (5e).The radicals of the formulas (4a) and (4b) are preferably those of the formula wherein (R 9 ) 0-3 represents 0 to 3 identical or different substituents from the group C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy and sulfo and Z has the meaning given above, where Z has the meaning given above, wherein (R 10 ) 0-4 for 0 to 4 identical or different substituents from the group halogen, nitro, cyano, trifluoromethyl, sulfamoyl, carbamoyl, C 1 -C 4 alkyl, C 1 -C 4 alkoxy, amino, acetylamino , Ureido, hydroxy, carboxy, sulfomethyl and sulfo and Z has the meaning given above, wherein (R 11 ) 0-3 represents 0 to 3 identical or different substituents from the group C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy and sulfo, where R 12 and R 14 are independently hydrogen, C 1 -C 4 alkyl or phenyl, and R 13 is hydrogen, cyano, carbamoyl or sulfomethyl, or or or wherein (R 9 ) 0-3 , (R 10 ) 0-3 , (R 11 ) 0-3 and Z each have the meaning given above and (R 9 ') 0-3 for 0 to 3 identical or different substituents the group C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy or sulfo. The residues of the formulas (5a) to (5e) are of particular interest.
Bei dem Rest eines Formazanchromophors handelt es sich bevozugt um solche der Formel worin Z die oben angegebene Bedeutung hat und die Benzolkerne keine weiteren Substituenten enthalten oder durch C1-C4-Alkyl, C1-C4-Alkoxy, C1-C4-Alkylsulfonyl, Halogen oder Carboxy weitersubstituiert sind. Vorzugsweise enthalten die Reste der Formeln (7a) und (7b) keine weiteren Substituenten und keinen Rest -SO2-Z.The rest of a formazan chromophore are preferably those of the formula wherein Z has the meaning given above and the benzene nuclei contain no further substituents or are further substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, C 1 -C 4 alkylsulfonyl, halogen or carboxy. The radicals of the formulas (7a) and (7b) preferably contain no further substituents and no —SO 2 —Z radical.
Bei dem Rest eines Phthalocyaninchromophors handelt es sich bevorzugt um solche der
Formel
Bei dem Rest eines Dioxazinchromophors handelt es sich bevorzugt um solche der Formel
Bei dem Rest eines Anthrachinonchromophors handelt es sich bevorzugt um solche der Formel worin G einen unsubstituierten oder durch C1-C4-Alkyl, C1-C4-Alkoxy, Halogen, Carboxy oder Sulfo substituierten Phenylenrest oder einen Cyclohexylen-, Phenylenmethylen- oder C2-C6-Alkylenrest bedeutet.The rest of an anthraquinone chromophore is preferably of the formula wherein G is an unsubstituted or substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogen, carboxy or sulfo phenylene radical or a cyclohexylene, phenylene methylene or C 2 -C 6 alkylene radical.
Bei den Resten A1, A2 und A3 handelt es sich bevorzugt um solche der Formeln (5a) bis (5e), (6a) bis (6d), (7a), (7b), (8), (9) oder (10), wobei die Reste der Formeln (6a) bis (6d), (7a) und (7b) vorzugsweise keinen faserreaktiven Rest enthalten. Von besonderem Interesse sind die Reste der Formeln (5a) bis (5e), (7a), (7b) und (10), insbesondere die Reste der Formeln (5a), (5b), (5c) und (5e).The radicals A 1 , A 2 and A 3 are preferably those of the formulas (5a) to (5e), (6a) to (6d), (7a), (7b), (8), (9) or (10), the radicals of the formulas (6a) to (6d), (7a) and (7b) preferably not containing any fiber-reactive radical. Of particular interest are the radicals of the formulas (5a) to (5e), (7a), (7b) and (10), in particular the radicals of the formulas (5a), (5b), (5c) and (5e).
Die Reste A1, A2 und A3 enthalten bevorzugt jeweils 1 bis 4 Sulfogruppen und vorzugsweise 1 bis 3 Sulfogruppen.The radicals A 1 , A 2 and A 3 preferably each contain 1 to 4 sulfo groups and preferably 1 to 3 sulfo groups.
Die Reaktivfarbstoffe der Formeln (1) und (2) enthalten jeweils mindestens zwei faserreaktive Gruppen. So enthält der Reaktivfarbstoff der Formel (1) zwei faserreaktive Triazinreste und der Reaktivfarbstoff der Formel (2) einen faserreaktiven Triazinrest und einen faserreaktiven Rest V. Vorzugsweise enthalten die Reaktivfarbstoffe der Formeln (1) und (2) jeweils nur zwei faserreaktive Gruppen.The reactive dyes of the formulas (1) and (2) each contain at least two fiber reactive groups. For example, the reactive dye of formula (1) contains two fiber-reactive ones Triazine residues and the reactive dye of formula (2) a fiber-reactive triazine residue and a fiber-reactive radical V. The reactive dyes of the formulas (1) preferably contain and (2) only two fiber reactive groups at a time.
Von besonderem Interesse sind Reaktivfarbstoffe der Formel oder Reactive dyes of the formula are of particular interest or
Die Reaktivfarbstoffe der Formeln (1) und (2) sind bekannt oder können in Analogie zu bekannten Verbindungen erhalten werden, wie z.B. durch übliche Diazotierungs-, Kupplungs- und Kondensationsreaktionen.The reactive dyes of the formulas (1) and (2) are known or can be used in analogy to known compounds, e.g. through usual diazotization, Coupling and condensation reactions.
Die in den Tinten verwendeten Reaktivfarbstoffe der Formeln (1) und (2) sollten vorzugsweise salzarm sein, d.h. einen Gesamtgehalt an Salzen von weniger als 0,5 Gew.-%, bezogen auf das Gewicht der Farbstoffe, enthalten. Reaktivfarbstoffe, die, bedingt durch ihre Herstellung und/oder die nachträgliche Zugabe von Coupagemitteln grössere Salzgehalte aufweisen, können z.B. durch Membrantrennverfahren, wie Ultrafiltration, Umkehrosmose oder Dialyse, entsalzt werden.The reactive dyes of formulas (1) and (2) used in the inks should preferably low in salt, i.e. a total salt content of less than 0.5 wt .-%, based on the weight of the dyes. Reactive dyes that due to their manufacture and / or the subsequent addition of coupé agents have higher salt contents, e.g. by membrane separation processes, such as Ultrafiltration, reverse osmosis or dialysis.
Vorzugsweise enthalten die Tinten als Farbstoffe ausschliesslich sulfogruppenhaltige, wasserlösliche Reaktivfarbstoffe, wie die der obigen Formeln (1) und (2). The inks preferably contain exclusively dyes containing sulfo groups, water-soluble reactive dyes, such as those of formulas (1) and (2) above.
Die Tinten enthalten bevorzugt einen Gesamtgehalt an Reaktivfarbstoffen der obigen Formeln (1) und (2) von 5 bis 35 Gew.-%, insbesondere 10 bis 35 Gew.-% und vorzugsweise 10 bis 20 Gew.-%, bezogen auf das Gesamtgewicht der Tinte.The inks preferably contain a total reactive dye content of the above Formulas (1) and (2) from 5 to 35% by weight, in particular 10 to 35% by weight and preferably 10 to 20% by weight based on the total weight of the ink.
Als wasserlösliche, nichtionogene Celluloseäther kommen z.B. Methyl-, Aethyl-, Hydroxyäthyl-, Methylhydroxyäthyl-, Hydroxypropyl- oder Hydroxypropylmethylcellulose in Betracht. Bevorzugt sind Methylcellulose oder insbesondere Hydroxyäthylcellulose. Die Celluloseäther werden in der Tinte üblicherweise in einer Menge von 0,01 bis 2 Gew.-%, insbesondere 0,01 bis 1 Gew.-% und vorzugsweise 0,01 bis 0,5 Gew.-%, bezogen auf das Gesamtgewicht der Tinte, verwendet.As water-soluble, non-ionic cellulose ethers e.g. Methyl, ethyl, Hydroxyethyl, methylhydroxyethyl, hydroxypropyl or hydroxypropylmethyl cellulose in Consideration. Methyl cellulose or, in particular, hydroxyethyl cellulose are preferred. The Cellulose ethers are usually used in the ink in an amount of 0.01 to 2% by weight, in particular 0.01 to 1 wt .-% and preferably 0.01 to 0.5 wt .-%, based on the Total weight of ink used.
Als Alginate kommen insbesondere Alkalialginate und vorzugsweise Natriumalginat in Betracht. Diese werden in der Tinte üblicherweise in einer Menge von 0,01 bis 2 Gew.-%, insbesondere 0,01 bis 1 Gew.-% und vorzugsweise 0,01 bis 0,5 Gew.-%, bezogen auf das Gesamtgewicht der Tinte, verwendet.Alkali alginates and preferably sodium alginate are particularly suitable as alginates Consideration. These are usually contained in the ink in an amount of 0.01 to 2% by weight, in particular 0.01 to 1 wt .-% and preferably 0.01 to 0.5 wt .-%, based on the Total weight of ink used.
Sowohl die verwendeten wasserlöslichen, nichtionogenen Celluloseäther als auch die Alginate werden als sogenannte Verdicker verwendet und erlauben die Einstellung einer bestimmten Viskosität der Tinte.Both the water-soluble, non-ionic cellulose ether used and the Alginates are used as so-called thickeners and allow the setting of one certain viscosity of the ink.
Bevorzugt sind Tinten, welche eine Viskosität von 1 bis 40 mPa·s, insbesondere 5 bis 40 mPa·s und vorzugsweise 10 bis 40 mPa·s aufweisen. Tinten mit einer Viskosität von 10 bis 30 mPa·s sind besonders bevorzugt.Inks are preferred which have a viscosity of 1 to 40 mPas, in particular 5 to 40 mPa · s and preferably 10 to 40 mPa · s. Inks with a viscosity of 10 to 30 mPa · s are particularly preferred.
Ferner können die Tinten Puffersubstanzen enthalten, wie z.B. Borax, Borat oder Citrat. Als
Beispiele seien Natriumborat, Natriumtetraborat sowie Natriumcitrat genannt.
Sie werden insbesondere in Mengen von 0,1 bis 3 Gew.-%, vorzugsweise 0,1 bis 1
Gew.-%, bezogen auf das Gesamtgewicht der Tinte, verwendet, um einen pH-Wert von z.B.
5 bis 9, insbesondere 6 bis 8, einzustellen. Vorzugsweise verwendet man im Falle
alginathaltiger Tinten einen Citrat-Puffer.The inks may also contain buffer substances, such as borax, borate or citrate. Examples include sodium borate, sodium tetraborate and sodium citrate.
They are used in particular in amounts of 0.1 to 3% by weight, preferably 0.1 to 1% by weight, based on the total weight of the ink, in order to have a pH of, for example, 5 to 9, in particular 6 to 8, adjust. A citrate buffer is preferably used in the case of inks containing alginate.
Als weitere Zusätze können die Tinten z.B. N-Methyl-2-pyrrolidon oder insbesondere 1,2-Propylenglykol enthalten. Diese werden in der Tinte üblicherweise in einer Menge von 5 bis 30 Gew.-%, insbesondere 5 bis 20 Gew.-% und vorzugsweise 10 bis 20 Gew.-%, bezogen auf das Gesamtgewicht der Tinte, verwendet.As further additives, the inks can e.g. N-methyl-2-pyrrolidone or in particular 1,2-propylene glycol contain. These are usually present in the ink in an amount of 5 to 30% by weight, in particular 5 to 20% by weight and preferably 10 to 20% by weight on the total weight of the ink used.
Ferner können die Tinten noch übliche Zusätze, wie z.B. schaumdämpfende Mittel oder insbesondere das Pilz- und/oder Bakterienwachstum hemmende Stoffe, enthalten. Diese werden üblicherweise in Mengen von 0,01 bis 1 Gew.-%, bezogen auf das Gesamtgewicht der Tinte, verwendet.Furthermore, the inks can also contain conventional additives such as e.g. foam suppressants or in particular substances that inhibit fungal and / or bacterial growth. This are usually in amounts of 0.01 to 1 wt .-%, based on the total weight the ink used.
Das erfindungsgemässe Verfahren zum Bedrucken von textilen Fasermaterialien kann mit an und für sich bekannten für den textilen Druck geeigneten Tintenstrahldruckern ausgeführt werden.The method according to the invention for printing textile fiber materials can with performed on known ink jet printers suitable for textile printing will.
Im Falle des Tintenstrahldruck-Verfahrens werden einzelne Tropfen der Tinte kontrolliert aus einer Düse auf ein Substrat gespritzt. Ueberwiegend werden hierzu die kontinuierliche Ink-Jet-Methode sowie die Drop on demand-Methode verwendet. Im Falle der kontinuierlichen Ink-Jet-Methode werden die Tropfen kontinuierlich erzeugt, wobei nicht für den Druck benötigte Tropfen in einen Auffangbehälter abgeleitet und rezykliert werden. Im Falle der Drop on demand-Methode hingegen werden Tropfen nach Wunsch erzeugt und gedruckt; d.h. es werden nur dann Tropfen erzeugt, wenn dies für den Druck erforderlich ist. Die Erzeugung der Tropfen kann z.B. mittels eines Piezo-Inkjet-Kopfes oder mittels thermischer Energie (Bubble Jet) erfolgen. Bevorzugt ist für das erfindungsgemässe Verfahren der Druck nach der Drop on demand-Methode, insbesondere mittels eines Piezo-Inkjet-Kopfes.In the case of the inkjet printing process, individual drops of the ink are checked out sprayed onto a substrate using a nozzle. The continuous ink jet method is predominantly used for this as well as the drop on demand method. In the case of continuous The ink-jet method creates the drops continuously, not for printing required drops are drained into a collecting container and recycled. In case of Drop on demand method, however, drops are created and printed as desired; i.e. drops are only generated when this is necessary for printing. The Generation of the drops can e.g. by means of a piezo inkjet head or by means of thermal Energy (bubble jet). Pressure is preferred for the method according to the invention using the drop-on-demand method, in particular using a piezo inkjet head.
Als textile Fasermaterialien kommen insbesondere hydroxylgruppenhaltige Fasermaterialien in Betracht. Bevorzugt sind cellulosehaltige Fasermaterialien, die ganz oder teilweise aus Cellulose bestehen. Beispiele sind natürliche Fasermaterialien wie Baumwolle, Leinen oder Hanf und regenerierte Fasermaterialien wie z.B. Viskose sowie Lyocell. Besonders bevorzugt sind hierbei Viskose oder vorzugsweise Baumwolle. Die genannten Fasermaterialien liegen vorzugsweise als flächige textile Gewebe, Gewirke oder Bahnen vor.In particular, fiber materials containing hydroxyl groups come as textile fiber materials into consideration. Preference is given to cellulose-containing fiber materials which are made up entirely or in part Cellulose exist. Examples are natural fiber materials such as cotton, linen or Hemp and regenerated fiber materials such as Viscose and lyocell. Especially viscose or preferably cotton are preferred. The above Fiber materials are preferably in the form of flat textile fabrics, knitted fabrics or webs.
Gemäss einer bevorzugten Ausführungsform der vorliegenden Erfindung erfolgt vor dem Bedrucken eine Vorbehandlung des Fasermaterials, worin man das zu bedruckende Fasermaterial zuerst mit einer wässrigen alkalischen Flotte behandelt und das behandelte Fasermaterial gegebenenfalls trocknet. According to a preferred embodiment of the present invention, before Printing a pretreatment of the fiber material, in which one is to be printed Fiber material first treated with an aqueous alkaline liquor and the treated Fiber material dries if necessary.
Die wässrige alkalische Flotte enthält mindestens eine der üblichen Basen, welche in konventionellen Reaktivdruckverfahren zur Fixierung der Reaktivfarbstoffe eingesetzt werden. Die Base wird z.B. in einer Menge von 10 bis 100 g/l Flotte, vorzugsweise 10 bis 50 g/l Flotte, eingesetzt. Als Base kommen beispielsweise Natriumcarbonat, Natriumhydroxid, Dinatriumphosphat, Trinatriumphosphat, Natriumacetat, Natriumpropionat, Natriumhydrogencarbonat, wässriges Ammoniak oder Alkalispender, wie z.B. Natriumchloracetat oder Natriumformiat in Betracht. Vorzugsweise wird Natriumhydrogencarbonat, Natriumcarbonat oder eine Mischung aus Wasserglas und Natriumcarbonat verwendet. Der pH-Wert der alkalischen Flotte beträgt in der Regel 7,5 bis 13,5, vorzugsweise 8,5 bis 12,5. Die wässrige alkalische Flotte kann ausser den Basen weitere Zusätze, z.B. Hydrotropiermittel, enthalten. Als Hydrotropiermittel wird bevorzugt Harnstoff verwendet, der z.B. in einer Menge von 25 bis 200 g/l Flotte, vorzugsweise 50 bis 150 g/l Flotte eingesetzt wird. Vorzugsweise wird das Fasermaterial nach der obigen Vorbehandlung getrocknet.The aqueous alkaline liquor contains at least one of the usual bases, which in conventional Reactive printing processes are used to fix the reactive dyes. The base is e.g. in an amount of 10 to 100 g / l liquor, preferably 10 to 50 g / l Fleet, deployed. Sodium carbonate, sodium hydroxide, Disodium phosphate, trisodium phosphate, sodium acetate, sodium propionate, sodium hydrogen carbonate, aqueous ammonia or alkali dispensers, e.g. Sodium chloroacetate or sodium formate. Sodium bicarbonate is preferred, sodium carbonate or a mixture of water glass and sodium carbonate is used. The pH the alkaline liquor is usually 7.5 to 13.5, preferably 8.5 to 12.5. The aqueous alkaline liquor can contain other additives, e.g. Hydrotrope, contain. Urea is preferably used as the hydrotroping agent, e.g. in a Amount of 25 to 200 g / l liquor, preferably 50 to 150 g / l liquor is used. The fiber material is preferably dried after the above pretreatment.
Nach dem Bedrucken wird das Fasermaterial vorteilhafterweise getrocknet, vorzugsweise bei Temperaturen bis 150°C, insbesondere 80 bis 120°C, und anschliessend einem Hitzebehandlungsprozess unterworfen, um den Druck zu vervollständigen, bzw. den Farbstoff zu fixieren.After printing, the fiber material is advantageously dried, preferably at temperatures up to 150 ° C, especially 80 to 120 ° C, and then a heat treatment process subjected to complete the print, or the dye to fix.
Die Hitzebehandlung kann z.B. durch ein Warmverweilverfahren, einen Thermosolierprozess oder vorzugsweise durch ein Dämpfverfahren durchgeführt werden.The heat treatment can e.g. through a warm dwell process, a thermal insulation process or preferably be carried out by a steaming process.
Beim Dämpfverfahren wird das bedruckte Fasermaterial z.B. einer Behandlung in einem Dämpfer mit gegebenenfalls überhitztem Dampf, zweckmässigerweise bei einer Temperatur von 95 bis 180° C, vorteilhafterweise im Sattdampf, unterzogen.In the steaming process, the printed fiber material is e.g. one treatment in one Damper with possibly superheated steam, conveniently at one temperature from 95 to 180 ° C, advantageously in saturated steam.
Im Anschluss wird das bedruckte Fasermaterial in der Regel in üblicher Weise mit Wasser ausgewaschen um nichtfixierten Farbstoff zu entfernen.Subsequently, the printed fiber material is usually washed with water in the usual way washed out to remove unfixed dye.
Gegenstand der vorliegenden Erfindung sind ferner wässrige Drucktinten für das
Tintenstrahldruck-Verfahren, welche dadurch gekennzeichnet sind, dass sie
Für die Drucktinten sowie für die Reaktivfarbstoffe der Formeln (1) und (2) gelten hierbei die weiter oben angegebenen Bedeutungen und Bevorzugungen.The same applies here for the printing inks and for the reactive dyes of the formulas (1) and (2) meanings and preferences given above.
Die nach dem erfindungsgemässen Verfahren erhältlichen Drucke weisen gute Allgemeinechtheiten auf; sie besitzen z.B. eine hohe Faser-Farbstoff-Bindungsstabilität sowohl im sauren als auch im alkalischen Bereich, eine gute Lichtechtheit, gute Nassechtheiten, wie Wasch-, Wasser-, Seewasser-, Überfärbe-und Schweissechtheit, eine gute Chlorechtheit, Reibechtheit, Bügelechtheit und Plissierechtheit sowie scharfe Konturen und eine hohe Farbstärke. Die verwendeten Drucktinten zeichnen sich durch gute Stabilität und gute Viskositätseigenschaften aus. So bleibt die Viskosität selbst bei während des Drucks auftretenden hohen Scherkräften nahezu unverändert erhalten.The prints obtainable by the process according to the invention have good prints General realities on; e.g. they have high fiber-dye binding stability both in the acidic and in the alkaline range, good lightfastness, good Wet fastness properties, such as fastness to washing, water, sea water, over-dyeing and perspiration, one good chlorine fastness, rubbing fastness, ironing fastness and pleating fastness as well as sharp contours and high color strength. The printing inks used are characterized by good stability and good viscosity properties. So the viscosity remains even during the Preserved high shear forces almost unchanged.
Die nachfolgenden Beispiele dienen zur Erläuterung der Erfindung. Die Temperaturen sind in Celsiusgraden angegeben, Teile sind Gewichtsteile, die Prozentangaben beziehen sich auf Gewichtsprozente, sofern nicht anders vermerkt. Gewichtsteile stehen zu Volumenteilen im Verhältnis von Kilogramm zu Liter.The following examples serve to explain the invention. The temperatures are stated in degrees Celsius, parts are parts by weight, the percentages refer percentages by weight, unless stated otherwise. Parts by weight relate to parts by volume in the ratio of kilograms to liters.
- 15 Gew.-% des Reaktivfarbstoffs der Formel
- 0,3 Gew.-% Hydroxyaethylcellulose,
- 0,5 Gew.-% Borax und
- 84,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula
- 0.3% by weight of hydroxyethyl cellulose,
- 0.5% by weight of borax and
- 84.2 wt% water
- 15 Gew.-% des Reaktivfarbstoffs der Formel
- 0,3 Gew.-% Hydroxyaethylcellulose,
- 15 Gew.-% 1,2-Propylenglykol
- 0,5 Gew.-% Borax und
- 69,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula
- 0.3% by weight of hydroxyethyl cellulose,
- 15% by weight 1,2-propylene glycol
- 0.5% by weight of borax and
- 69.2% by weight of water
- 15 Gew.-% des Reaktivfarbstoffs der Formel
- 0,3 Gew.-% Hydroxyaethylcellulose,
- 15 Gew.-% N-Methyl-2-pyrrolidon,
- 0,5 Gew.-% Borax und
- 69,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula
- 0.3% by weight of hydroxyethyl cellulose,
- 15% by weight of N-methyl-2-pyrrolidone,
- 0.5% by weight of borax and
- 69.2% by weight of water
- 15 Gew.-% des Reaktivfarbstoffs der Formel
- 0,3 Gew.-% Hydroxyaethylcellulose,
- 0,5 Gew.-% Borax und
- 84,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula
- 0.3% by weight of hydroxyethyl cellulose,
- 0.5% by weight of borax and
- 84.2 wt% water
- 15 Gew.-% des Reaktivfarbstoffs der Formel
- 0,3 Gew.-% Hydroxyaethylcellulose und
- 84,7 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula
- 0.3% by weight of hydroxyethyl cellulose and
- 84.7 wt% water
- 15 Gew.-% des Reaktivfarbstoffs der Formel
- 0,3 Gew.-% Hydroxyaethylcellulose,
- 0,5 Gew.-% Natriumcitrat und
- 84,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula
- 0.3% by weight of hydroxyethyl cellulose,
- 0.5% by weight sodium citrate and
- 84.2 wt% water
- 15 Gew.-% des Reaktivfarbstoffs der Formel (11a),
- 0,3 Gew.-% Natriumalginat,
- 0,5 Gew.-% Natriumcitrat und
- 84,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula (11a),
- 0.3% by weight sodium alginate,
- 0.5% by weight sodium citrate and
- 84.2 wt% water
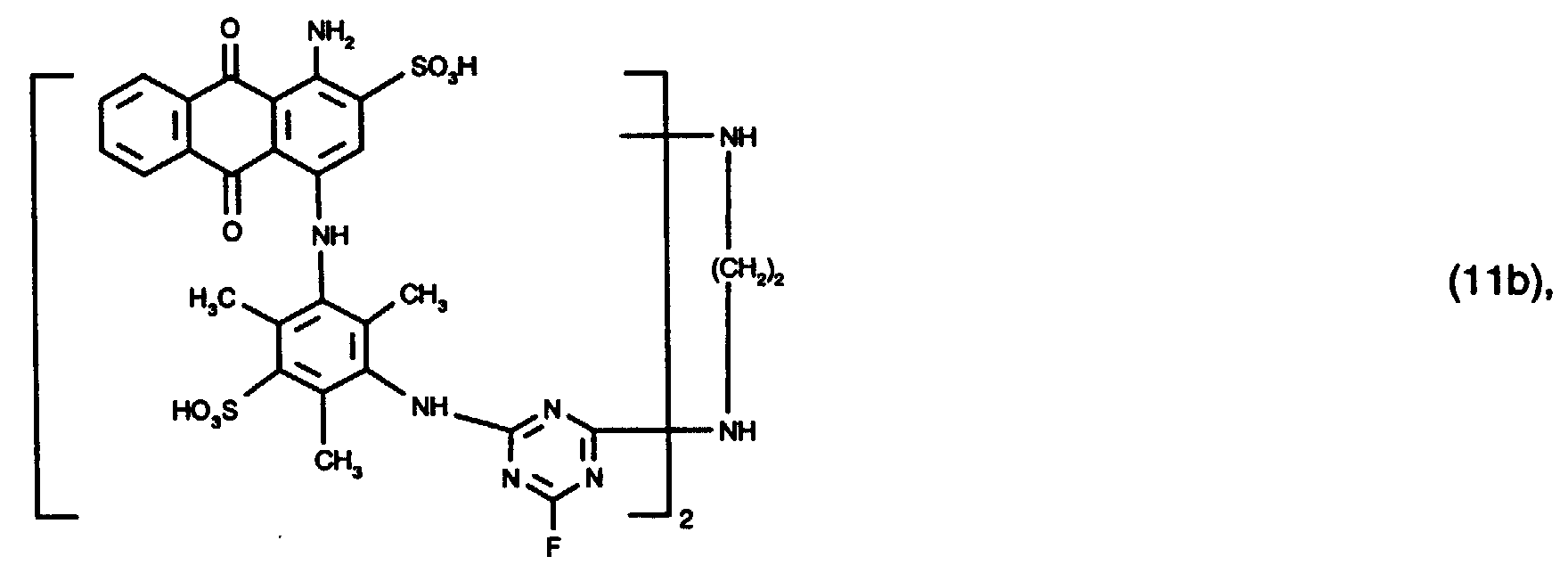
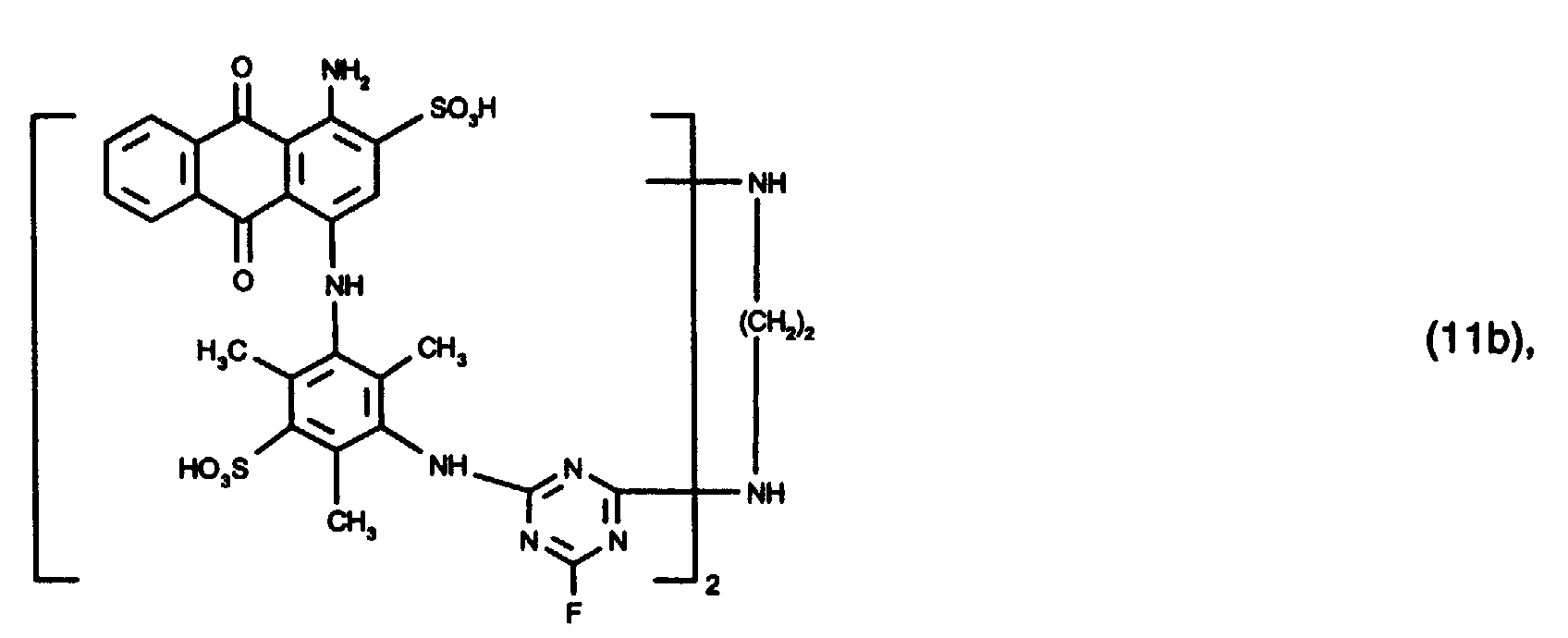
- 15 Gew.-% des Reaktivfarbstoffs der Formel (11b),
- 0,3 Gew.-% Natriumalginat,
- 15 Gew.-% 1,2-Propylenglykol
- 0,5 Gew.-% Natriumcitrat und
- 69,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula (11b),
- 0.3% by weight sodium alginate,
- 15% by weight 1,2-propylene glycol
- 0.5% by weight sodium citrate and
- 69.2% by weight of water
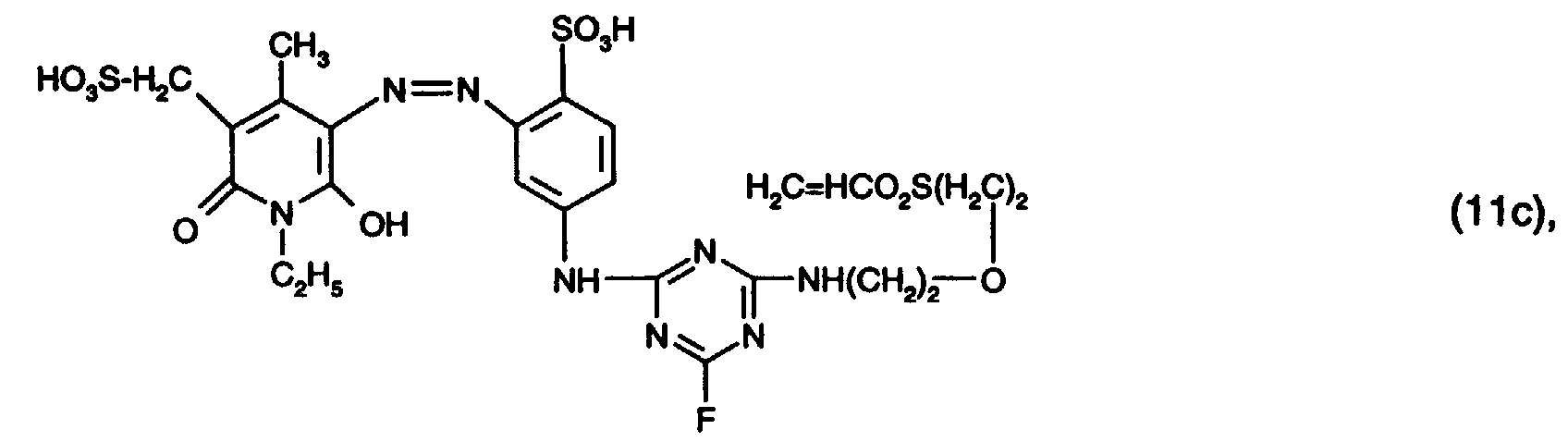
- 15 Gew.-% des Reaktivfarbstoffs der Formel (11c),
- 0,3 Gew.-% Natriumalginat,
- 15 Gew.-% N-Methyl-2-pyrrolidon,
- 0,5 Gew.-% Natriumcitrat und
- 69,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula (11c),
- 0.3% by weight sodium alginate,
- 15% by weight of N-methyl-2-pyrrolidone,
- 0.5% by weight sodium citrate and
- 69.2% by weight of water
- 15 Gew.-% des Reaktivfarbstoffs der Formel (11d),
- 0,3 Gew.-% Natriumalginat,
- 0,5 Gew.-% Natriumcitrat und
- 84,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula (11d),
- 0.3% by weight sodium alginate,
- 0.5% by weight sodium citrate and
- 84.2 wt% water
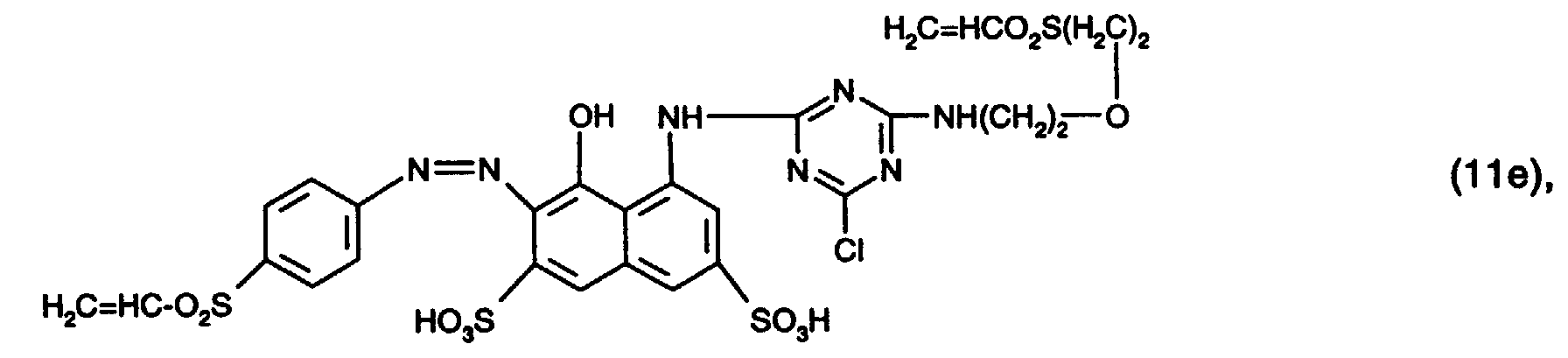
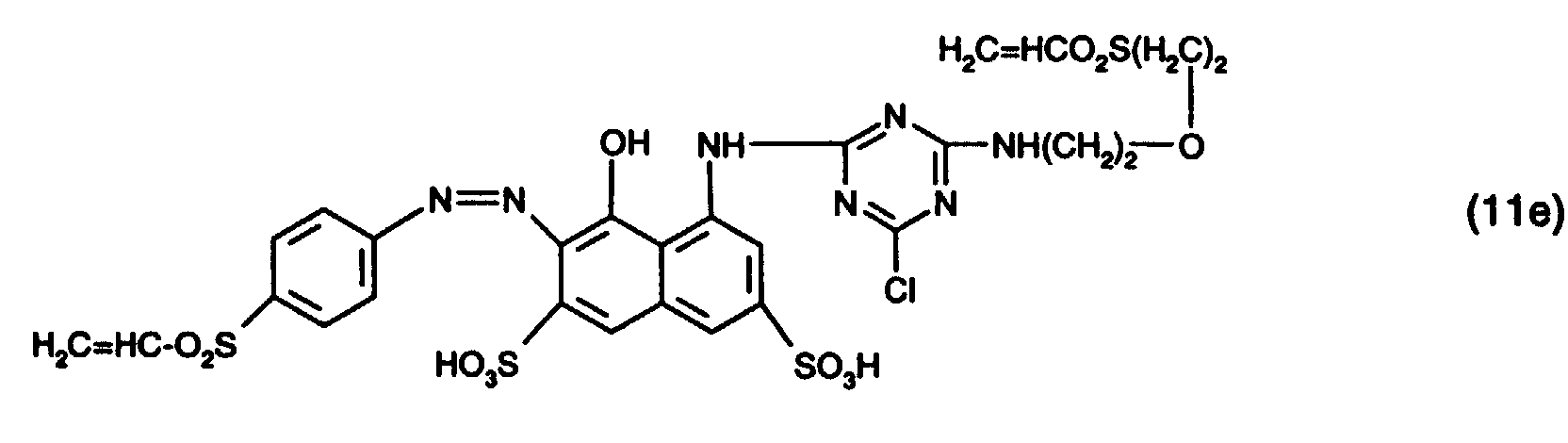
- 15 Gew.-% des Reaktivfarbstoffs der Formel (11e),
- 0,3 Gew.-% Natriumalginat und
- 84,7 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula (11e),
- 0.3% by weight sodium alginate and
- 84.7 wt% water
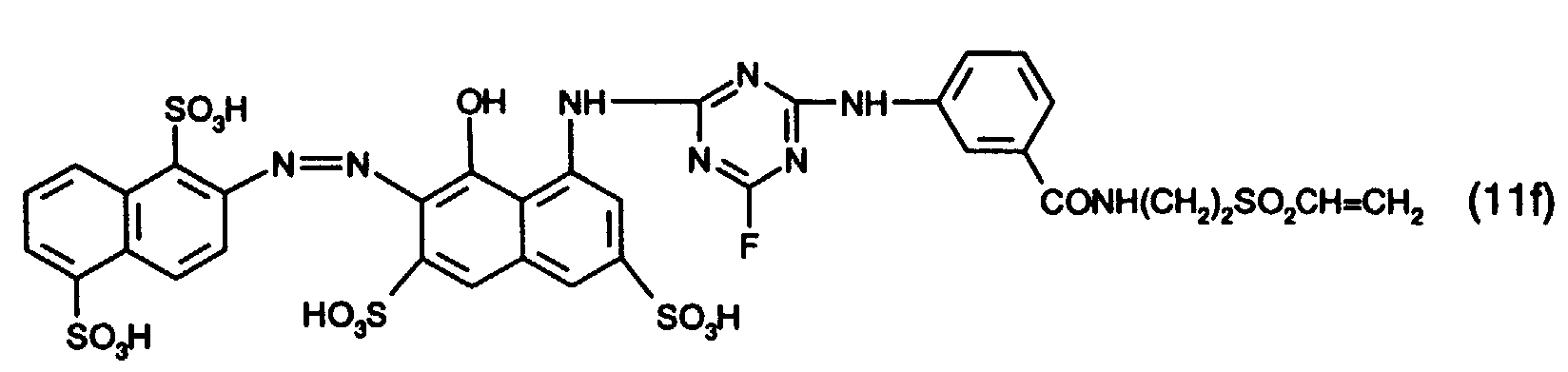
- 15 Gew.-% des Reaktivfarbstoffs der Formel (11f),
- 0,3 Gew.-% Natriumalginat,
- 0,5 Gew.-% Natriumcitrat und
- 84,2 Gew.-% Wasser
- 15% by weight of the reactive dye of the formula (11f),
- 0.3% by weight sodium alginate,
- 0.5% by weight sodium citrate and
- 84.2 wt% water
Claims (15)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH1475/97 | 1997-06-17 | ||
| CH147597 | 1997-06-17 | ||
| CH147597 | 1997-06-17 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0886000A2 true EP0886000A2 (en) | 1998-12-23 |
| EP0886000A3 EP0886000A3 (en) | 2000-02-23 |
| EP0886000B1 EP0886000B1 (en) | 2005-07-20 |
Family
ID=4211360
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98810525A Expired - Lifetime EP0886000B1 (en) | 1997-06-17 | 1998-06-09 | Process for ink jet printing textile materials |
Country Status (13)
| Country | Link |
|---|---|
| US (1) | US5972084A (en) |
| EP (1) | EP0886000B1 (en) |
| JP (1) | JPH1112963A (en) |
| KR (1) | KR100538666B1 (en) |
| CN (1) | CN1140667C (en) |
| BR (1) | BR9802014A (en) |
| DE (1) | DE59812931D1 (en) |
| ES (1) | ES2246065T3 (en) |
| ID (1) | ID20432A (en) |
| PT (1) | PT886000E (en) |
| SG (1) | SG73520A1 (en) |
| TR (1) | TR199801119A2 (en) |
| TW (1) | TW550281B (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000046309A1 (en) * | 1999-02-05 | 2000-08-10 | Ciba Specialty Chemicals Holding Inc. | Black-dyeing inks and their use |
| DE10135042A1 (en) * | 2001-07-11 | 2003-02-06 | Dystar Textilfarben Gmbh & Co | Aqueous ink formulations for the inkjet printing process |
| CN102127333A (en) * | 2011-01-12 | 2011-07-20 | 郑州鸿盛数码科技股份有限公司 | Direct ink-jet printing disperse dye ink and use method thereof |
| CN102153904A (en) * | 2011-01-12 | 2011-08-17 | 郑州鸿盛数码科技股份有限公司 | Ceramic transfer digital printing ink-jet ink and using method thereof |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05161473A (en) * | 1991-12-12 | 1993-06-29 | Tanisake:Kk | Nutritive auxiliary food |
| GB9608488D0 (en) * | 1996-04-25 | 1996-07-03 | Zeneca Ltd | Compositions, processes and uses |
| GB9702354D0 (en) * | 1997-02-05 | 1997-03-26 | Zeneca Ltd | Compounds and their use |
| US6406525B1 (en) | 1998-04-18 | 2002-06-18 | Clariant Finance (Bvi) Limited | Organic compounds |
| GB9916110D0 (en) * | 1999-07-10 | 1999-09-08 | Avecia Ltd | Compounds and inks |
| DE10004954A1 (en) * | 2000-02-04 | 2001-08-16 | Dystar Textilfarben Gmbh & Co | Aqueous printing inks for use by the inkjet printing process, their production and their use on textile fiber materials |
| GB0019464D0 (en) * | 2000-08-09 | 2000-09-27 | Clariant Int Ltd | Organic compounds |
| DE60139949D1 (en) * | 2000-10-10 | 2009-10-29 | Clariant Finance Bvi Ltd | FIBER-REACTIVE COPPER COMPLEXES OF MONOAZO DYES |
| US6869646B2 (en) * | 2001-08-06 | 2005-03-22 | Hewlett-Packard Company, L.P. | Fast drying images and methods for printing on inorganic porous media |
| KR20050116831A (en) * | 2003-03-27 | 2005-12-13 | 시바 스페셜티 케미칼스 홀딩 인크. | Fibre-reactive azo dyes, their preparation and their use |
| TW200512261A (en) * | 2003-07-16 | 2005-04-01 | Nippon Kayaku Kk | Disazocompound and ink composition using same |
| CN102337045B (en) * | 2011-07-13 | 2014-03-26 | 丽源(湖北)科技有限公司 | Blue active dye mixture and preparation and application thereof |
| CN102504583B (en) * | 2011-09-29 | 2014-10-08 | 刘卫斌 | Scarlet azo dye and its preparation and application |
| CN102391683B (en) * | 2011-09-29 | 2014-05-07 | 刘卫斌 | Bright red azo dye mixture and use thereof |
| TWI548704B (en) * | 2015-05-08 | 2016-09-11 | 臺灣永光化學工業股份有限公司 | High fixation ink composition for Digital Textile printing application |
| CN107964258A (en) * | 2017-12-05 | 2018-04-27 | 湖北丽源科技股份有限公司 | A kind of Yellow fluorine-containing reactive dye mixture and its preparation method and application |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4849770A (en) * | 1985-12-13 | 1989-07-18 | Canon Kabushiki Kaisha | Ink for use in ink jet and ink jet printing method using the same |
| US4969951A (en) * | 1985-05-21 | 1990-11-13 | Canon Kabushiki Kaisha | Cloth jet printing method using aqueous ink having hydroxyl or amino-reactive disperse dye |
| GB2252335A (en) * | 1991-01-30 | 1992-08-05 | Ici Plc | Printing process and formulation |
| EP0666362A2 (en) * | 1994-02-08 | 1995-08-09 | Canon Kabushiki Kaisha | Ink-jet printing cloth, and ink-jet printing process and production process of print using the same |
| US5542972A (en) * | 1994-05-20 | 1996-08-06 | Hoechst Ag | Reactive dyes for inkjet printing |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61101574A (en) * | 1984-10-23 | 1986-05-20 | Ricoh Co Ltd | Water based ink |
| JPS60185886A (en) * | 1985-01-29 | 1985-09-21 | キヤノン株式会社 | Printing liquid medium |
| JP2890492B2 (en) * | 1989-07-05 | 1999-05-17 | 三菱化学株式会社 | Recording liquid |
| US5250121A (en) * | 1991-09-26 | 1993-10-05 | Canon Kabushiki Kaisha | Ink-jet textile printing ink and ink-jet textile printing process |
| DE69302039T2 (en) * | 1992-03-06 | 1996-09-12 | Zeneca Ltd | Ink compositions |
| GB9325454D0 (en) * | 1993-01-12 | 1994-02-16 | Zeneca Ltd | Azo compound |
| JP3666895B2 (en) * | 1993-01-13 | 2005-06-29 | キヤノン株式会社 | Ink jet textile printing ink, textile printing method, equipment using such ink, and printed matter obtained |
| US5843217A (en) * | 1996-10-11 | 1998-12-01 | Minolta Co., Ltd. | Ink for ink jet recording |
-
1998
- 1998-06-02 US US09/089,144 patent/US5972084A/en not_active Expired - Lifetime
- 1998-06-09 ES ES98810525T patent/ES2246065T3/en not_active Expired - Lifetime
- 1998-06-09 EP EP98810525A patent/EP0886000B1/en not_active Expired - Lifetime
- 1998-06-09 DE DE59812931T patent/DE59812931D1/en not_active Expired - Fee Related
- 1998-06-09 PT PT98810525T patent/PT886000E/en unknown
- 1998-06-11 ID IDP980855A patent/ID20432A/en unknown
- 1998-06-12 SG SG1998001403A patent/SG73520A1/en unknown
- 1998-06-15 TW TW087109453A patent/TW550281B/en not_active IP Right Cessation
- 1998-06-16 BR BR9802014A patent/BR9802014A/en not_active Application Discontinuation
- 1998-06-16 TR TR1998/01119A patent/TR199801119A2/en unknown
- 1998-06-16 CN CNB981032303A patent/CN1140667C/en not_active Expired - Fee Related
- 1998-06-17 JP JP10169542A patent/JPH1112963A/en active Pending
- 1998-06-17 KR KR1019980022646A patent/KR100538666B1/en not_active IP Right Cessation
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4969951A (en) * | 1985-05-21 | 1990-11-13 | Canon Kabushiki Kaisha | Cloth jet printing method using aqueous ink having hydroxyl or amino-reactive disperse dye |
| US4849770A (en) * | 1985-12-13 | 1989-07-18 | Canon Kabushiki Kaisha | Ink for use in ink jet and ink jet printing method using the same |
| GB2252335A (en) * | 1991-01-30 | 1992-08-05 | Ici Plc | Printing process and formulation |
| EP0666362A2 (en) * | 1994-02-08 | 1995-08-09 | Canon Kabushiki Kaisha | Ink-jet printing cloth, and ink-jet printing process and production process of print using the same |
| US5542972A (en) * | 1994-05-20 | 1996-08-06 | Hoechst Ag | Reactive dyes for inkjet printing |
Non-Patent Citations (2)
| Title |
|---|
| DATABASE WPI Section Ch, Week 198544 Derwent Publications Ltd., London, GB; Class A60, AN 1985-273488 XP002124482 & JP 60 185886 A (CANON KK), 21. September 1985 (1985-09-21) * |
| DATABASE WPI Section Ch, Week 199114 Derwent Publications Ltd., London, GB; Class A88, AN 1991-096910 XP002124603 & JP 03 039372 A (MITSUBISHI KASEI CORP), 20. Februar 1991 (1991-02-20) * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000046309A1 (en) * | 1999-02-05 | 2000-08-10 | Ciba Specialty Chemicals Holding Inc. | Black-dyeing inks and their use |
| DE10135042A1 (en) * | 2001-07-11 | 2003-02-06 | Dystar Textilfarben Gmbh & Co | Aqueous ink formulations for the inkjet printing process |
| CN102127333A (en) * | 2011-01-12 | 2011-07-20 | 郑州鸿盛数码科技股份有限公司 | Direct ink-jet printing disperse dye ink and use method thereof |
| CN102153904A (en) * | 2011-01-12 | 2011-08-17 | 郑州鸿盛数码科技股份有限公司 | Ceramic transfer digital printing ink-jet ink and using method thereof |
| CN102153904B (en) * | 2011-01-12 | 2013-06-19 | 郑州鸿盛数码科技股份有限公司 | Ceramic transfer digital printing ink-jet ink and using method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| KR100538666B1 (en) | 2006-03-28 |
| BR9802014A (en) | 1999-07-20 |
| TR199801119A3 (en) | 1999-10-21 |
| EP0886000A3 (en) | 2000-02-23 |
| PT886000E (en) | 2005-10-31 |
| CN1202551A (en) | 1998-12-23 |
| CN1140667C (en) | 2004-03-03 |
| ID20432A (en) | 1998-12-17 |
| KR19990007044A (en) | 1999-01-25 |
| TR199801119A2 (en) | 1999-10-21 |
| TW550281B (en) | 2003-09-01 |
| EP0886000B1 (en) | 2005-07-20 |
| DE59812931D1 (en) | 2005-08-25 |
| ES2246065T3 (en) | 2006-02-01 |
| US5972084A (en) | 1999-10-26 |
| JPH1112963A (en) | 1999-01-19 |
| SG73520A1 (en) | 2000-06-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0886000B1 (en) | Process for ink jet printing textile materials | |
| EP0885998B1 (en) | Process for ink jet printing textile materials | |
| EP0885999B1 (en) | Process for ink jet printing textile materials | |
| EP0922735B1 (en) | Method for dyeing or printing and new reactive dyes | |
| EP0626429B1 (en) | Fiber-reactive dyes, their preparation and use thereof | |
| EP1086999B1 (en) | Magenta-colouring inks containing copper complexes of azo dyes on the basis of 1-naphthol-di or trisulfonic acids | |
| DE102005047391A1 (en) | Dyes and dye mixtures of fiber-reactive azo dyes, their preparation and their use | |
| DE102008054404A1 (en) | Fiber-reactive azo dyes and dye mixtures, process for their preparation and their use | |
| EP1508596A1 (en) | Mixtures of azo reactive dyes, process for their preparation and the use thereof | |
| EP2035507A2 (en) | Dye mixtures of fibre-reactive azo dyes, production and use thereof | |
| DE102007005795A1 (en) | Mixtures of fiber-reactive azo dyes | |
| DE60008698T2 (en) | BLACK INKING INKS AND THEIR USE | |
| EP0885997A2 (en) | Process for printing fibrous materials | |
| JP2003306627A (en) | Ink composition for ink jet printing of textile product | |
| DE60101051T2 (en) | USE OF A DYE COMPOSITION IN THE INK JET PRINTING PROCESS | |
| DE102009000417A1 (en) | Fiber-reactive azo dyes, process for their preparation and their use | |
| EP1984456A2 (en) | Mixtures of fibre reactive dyes production and use thereof | |
| DE19930858A1 (en) | Fixing prints made by ink-jet printing on fibrous textile, especially cotton or viscose is carried out with ultraviolet, infrared or microwave radiation or dry heat | |
| EP1345994A1 (en) | Black dye mixtures of fibre-reactive azo dyes and the use thereof for dyeing fibre material containing hydroxy and/or carbonamide groups | |
| EP0761768B1 (en) | Reactive dyes, process for their preparation and the use thereof | |
| DE19930866A1 (en) | Ink-jet printing on textile, especially cellulose, e.g. cotton or viscose, with aqueous ink containing reactive dye comprises treatment with fixing alkali only during or after printing | |
| WO2007085574A2 (en) | Water-soluble fiber-reactive dyes, methods for the production thereof, and use thereof | |
| DE19930867A1 (en) | Ink-jet printing, including multicolor printing, on mixed fiber textiles containing polyester and preferably cotton or wool | |
| EP1097971B1 (en) | Reactive dyes, process for their preparation and the use thereof | |
| DE19930995A1 (en) | Ink-jet printing on textile, especially cellulose, e.g. cotton or viscose rayon |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): BE CH DE ES FR GB IT LI PT |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| AKX | Designation fees paid | ||
| 17P | Request for examination filed |
Effective date: 20000529 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): BE CH DE ES FR GB IT LI PT |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE CH DE ES FR GB IT LI PT |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 20050726 |
|
| REF | Corresponds to: |
Ref document number: 59812931 Country of ref document: DE Date of ref document: 20050825 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Effective date: 20050913 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2246065 Country of ref document: ES Kind code of ref document: T3 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20060421 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: HUNTSMAN ADVANCED MATERIALS (SWITZERLAND) GMBH Free format text: CIBA SPECIALTY CHEMICALS HOLDING INC.#KLYBECKSTRASSE 141#4057 BASEL (CH) -TRANSFER TO- HUNTSMAN ADVANCED MATERIALS (SWITZERLAND) GMBH#KLYBECKSTRASSE 200#4057 BASEL (CH) |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20080618 Year of fee payment: 11 Ref country code: CH Payment date: 20080425 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20080630 Year of fee payment: 11 Ref country code: PT Payment date: 20080414 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20080630 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20080628 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20080506 Year of fee payment: 11 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: MM4A Free format text: LAPSE DUE TO NON-PAYMENT OF FEES Effective date: 20091209 |
|
| BERE | Be: lapsed |
Owner name: *CIBA SPECIALTY CHEMICALS HOLDING INC. Effective date: 20090630 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20090609 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20100226 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091209 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090630 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090630 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090630 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20080424 Year of fee payment: 11 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090609 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100101 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090630 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20090610 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090610 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090609 |