EP0098944A2 - Tungsten alloy powder - Google Patents
Tungsten alloy powder Download PDFInfo
- Publication number
- EP0098944A2 EP0098944A2 EP83105070A EP83105070A EP0098944A2 EP 0098944 A2 EP0098944 A2 EP 0098944A2 EP 83105070 A EP83105070 A EP 83105070A EP 83105070 A EP83105070 A EP 83105070A EP 0098944 A2 EP0098944 A2 EP 0098944A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- tungsten
- iron
- particles
- alloy powder
- binder
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000843 powder Substances 0.000 title claims abstract description 40
- 229910001080 W alloy Inorganic materials 0.000 title abstract description 10
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims abstract description 33
- 229910052721 tungsten Inorganic materials 0.000 claims abstract description 33
- 239000010937 tungsten Substances 0.000 claims abstract description 31
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims abstract description 25
- 239000002245 particle Substances 0.000 claims abstract description 25
- 239000011230 binding agent Substances 0.000 claims abstract description 22
- 238000004519 manufacturing process Methods 0.000 claims abstract description 18
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims abstract description 14
- 229910052742 iron Inorganic materials 0.000 claims abstract description 13
- 239000011159 matrix material Substances 0.000 claims abstract description 7
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 7
- 229910017052 cobalt Inorganic materials 0.000 claims abstract description 4
- 239000010941 cobalt Substances 0.000 claims abstract description 4
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims abstract description 4
- 229910020598 Co Fe Inorganic materials 0.000 claims abstract 2
- 229910002519 Co-Fe Inorganic materials 0.000 claims abstract 2
- 239000000243 solution Substances 0.000 claims description 39
- 229910045601 alloy Inorganic materials 0.000 claims description 25
- 239000000956 alloy Substances 0.000 claims description 25
- 229910052751 metal Inorganic materials 0.000 claims description 19
- 239000002184 metal Substances 0.000 claims description 19
- 238000000034 method Methods 0.000 claims description 14
- 238000005245 sintering Methods 0.000 claims description 14
- 150000003839 salts Chemical class 0.000 claims description 12
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 8
- 239000001257 hydrogen Substances 0.000 claims description 8
- 229910052739 hydrogen Inorganic materials 0.000 claims description 8
- 150000002739 metals Chemical class 0.000 claims description 6
- 239000012071 phase Substances 0.000 claims description 6
- 239000000463 material Substances 0.000 claims description 5
- 239000007790 solid phase Substances 0.000 claims description 5
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 claims description 4
- 239000000443 aerosol Substances 0.000 claims description 3
- 238000001704 evaporation Methods 0.000 claims description 3
- 230000008020 evaporation Effects 0.000 claims description 3
- 239000002923 metal particle Substances 0.000 claims description 3
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical class [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 claims description 2
- 150000008064 anhydrides Chemical class 0.000 claims description 2
- 229910052802 copper Inorganic materials 0.000 claims description 2
- 239000010949 copper Substances 0.000 claims description 2
- CMPGARWFYBADJI-UHFFFAOYSA-L tungstic acid Chemical compound O[W](O)(=O)=O CMPGARWFYBADJI-UHFFFAOYSA-L 0.000 claims description 2
- 230000007717 exclusion Effects 0.000 claims 2
- 229910000640 Fe alloy Inorganic materials 0.000 claims 1
- 239000002253 acid Substances 0.000 claims 1
- 150000001412 amines Chemical class 0.000 claims 1
- 239000007864 aqueous solution Substances 0.000 claims 1
- 150000002500 ions Chemical class 0.000 claims 1
- 229910052750 molybdenum Inorganic materials 0.000 claims 1
- 229910052702 rhenium Inorganic materials 0.000 claims 1
- 229910052709 silver Inorganic materials 0.000 claims 1
- 239000010944 silver (metal) Substances 0.000 claims 1
- 239000007791 liquid phase Substances 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- 239000007787 solid Substances 0.000 description 7
- 239000007789 gas Substances 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 5
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 4
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 235000011114 ammonium hydroxide Nutrition 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- 238000000889 atomisation Methods 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 239000011148 porous material Substances 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 238000003825 pressing Methods 0.000 description 3
- 238000001016 Ostwald ripening Methods 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- 238000005452 bending Methods 0.000 description 2
- 230000000536 complexating effect Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- -1 however Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- PBYZMCDFOULPGH-UHFFFAOYSA-N tungstate Chemical compound [O-][W]([O-])(=O)=O PBYZMCDFOULPGH-UHFFFAOYSA-N 0.000 description 2
- 150000003658 tungsten compounds Chemical class 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910003536 H2 WO4 Inorganic materials 0.000 description 1
- 229910052770 Uranium Inorganic materials 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 238000004581 coalescence Methods 0.000 description 1
- QGUAJWGNOXCYJF-UHFFFAOYSA-N cobalt dinitrate hexahydrate Chemical compound O.O.O.O.O.O.[Co+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O QGUAJWGNOXCYJF-UHFFFAOYSA-N 0.000 description 1
- 239000008139 complexing agent Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000004035 construction material Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- XAYGUHUYDMLJJV-UHFFFAOYSA-Z decaazanium;dioxido(dioxo)tungsten;hydron;trioxotungsten Chemical compound [H+].[H+].[NH4+].[NH4+].[NH4+].[NH4+].[NH4+].[NH4+].[NH4+].[NH4+].[NH4+].[NH4+].O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.[O-][W]([O-])(=O)=O.[O-][W]([O-])(=O)=O.[O-][W]([O-])(=O)=O.[O-][W]([O-])(=O)=O.[O-][W]([O-])(=O)=O.[O-][W]([O-])(=O)=O XAYGUHUYDMLJJV-UHFFFAOYSA-Z 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 238000001513 hot isostatic pressing Methods 0.000 description 1
- GXBKELQWVXYOPN-UHFFFAOYSA-N iron tungsten Chemical compound [W][Fe][W] GXBKELQWVXYOPN-UHFFFAOYSA-N 0.000 description 1
- 238000001000 micrograph Methods 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 238000004663 powder metallurgy Methods 0.000 description 1
- 239000010970 precious metal Substances 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000000930 thermomechanical effect Effects 0.000 description 1
- 150000003657 tungsten Chemical class 0.000 description 1
- DNYWZCXLKNTFFI-UHFFFAOYSA-N uranium Chemical compound [U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U] DNYWZCXLKNTFFI-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C1/00—Making non-ferrous alloys
- C22C1/04—Making non-ferrous alloys by powder metallurgy
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C1/00—Making non-ferrous alloys
- C22C1/04—Making non-ferrous alloys by powder metallurgy
- C22C1/045—Alloys based on refractory metals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F9/00—Making metallic powder or suspensions thereof
- B22F9/16—Making metallic powder or suspensions thereof using chemical processes
- B22F9/18—Making metallic powder or suspensions thereof using chemical processes with reduction of metal compounds
- B22F9/20—Making metallic powder or suspensions thereof using chemical processes with reduction of metal compounds starting from solid metal compounds
- B22F9/22—Making metallic powder or suspensions thereof using chemical processes with reduction of metal compounds starting from solid metal compounds using gaseous reductors
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F42—AMMUNITION; BLASTING
- F42B—EXPLOSIVE CHARGES, e.g. FOR BLASTING, FIREWORKS, AMMUNITION
- F42B12/00—Projectiles, missiles or mines characterised by the warhead, the intended effect, or the material
- F42B12/02—Projectiles, missiles or mines characterised by the warhead, the intended effect, or the material characterised by the warhead or the intended effect
- F42B12/04—Projectiles, missiles or mines characterised by the warhead, the intended effect, or the material characterised by the warhead or the intended effect of armour-piercing type
- F42B12/06—Projectiles, missiles or mines characterised by the warhead, the intended effect, or the material characterised by the warhead or the intended effect of armour-piercing type with hard or heavy core; Kinetic energy penetrators
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/12—All metal or with adjacent metals
- Y10T428/12181—Composite powder [e.g., coated, etc.]
Definitions
- the invention relates to a heterogeneous tungsten alloy powder which, in addition to tungsten, contains a binder phase composed of nickel, cobalt and iron, a process for the production of the powder and its use for the production of balancing projectiles.
- High-density materials are required for highly stressed metal parts, especially balancing projectiles.
- uranium and tungsten also meet the requirements for high density.
- the only metal that is traded at high density at a reasonable price is tungsten.
- tungsten is difficult to process because it is very brittle. It is poorly suited as a balancing bullet because it cannot withstand the tensile and compressive loads that occur.
- Balancing bullets are solid cylinders made of metal, the length of which far exceeds the caliber. When a balancing bullet hits an inclined armor plate, the balancing bullet tilts. In the relatively long body, high bending moments occur, which often lead to the bullet breaking and thus to relative ineffectiveness.
- the tungsten grains are always larger than the powder particles in the starting powder.
- the occurrence of a molten phase during the sintering process always results in an additional enlargement of the tungsten grains, which is made possible by the dissolving and redissolving processes between the tungsten and the liquid matrix.
- the phenomenon of the grain enlargement of solid precipitates in contact with liquids is of a fundamental nature and is known as Ostwald ripening.
- Point F lüssigphasengesinterte tungsten alloys s ch typical of a structure of spherical tungsten particles, each in a range of about 10 - present 60 / to large particles, which are embedded in a binder alloy.
- the strength and elongation at break are each limited by the largest particles (here approx. 60 / um). It can often be observed that large grains have grown together. Materials with such a coarse-grained structure do not have sufficient strength and are only slightly deformable.
- the invention has for its object to provide an alloy powder for the production of sintered parts, in particular balancing projectiles, which in addition to high specific weight, high tensile strength (> 1200 N / mm 2 ) and elongation at break (> 20%).
- Embodiments of the invention and manufacturing methods are the subject of subclaims.
- the simultaneous presence of extreme tensile strength with extreme elongation at break is not yet known and identifies the tungsten sintered parts according to the invention as ideal materials for balancing projectiles.
- the material survives both the high pressure and tensile loads when accelerating in the pipe and the high bending moments and compressive forces in the projectile when hitting armor.
- the outstanding properties make the sintered parts according to the invention also suitable for other tasks in science and technology in which the highest demands are placed on strength and toughness.
- F ig. 1 shows the tungsten alloy powder according to the invention in a thousand times magnification. It consists of particles with an approximately spherical shape (large ball to the right of the center of the picture). The diameter is 10 - 50 / um.
- the balls have a sponge-like structure.
- the sponge structure is made up of tungsten grains of about 1 / um diameter, which are covered and held together by a thin coating of binder metal. As a result, the distribution of tungsten and binder metal that is characteristic of the finished workpiece is predetermined.
- the inventive powder is therefore alloyed finished and the B inthe alloy already surrounds the W grains in the form of a coating. So that must be more dense during manufacture Sintered bodies the two processes of forming the binder alloy and coating the tungsten grains can no longer be carried out with the aid of a molten phase.
- the powder can be sintered into dense bodies in the solid phase.
- the sponge structure of the powder particles is loosely structured so that the powder can be compressed to around 50% of the theoretical density of a compact under 3 kbar pressure.
- This high green density in connection with the large specific surface area of the order of 1 m 2 / g enables the pressure-free sealing sintering of the compacts while avoiding liquid phases.
- FIG. 2 shows a micrograph of a sintered alloy according to the invention in a magnification of 600 times.
- FIG. 3 shows an alloy produced according to the prior art, that is to say liquid phase sintered, in a magnification of 600 times.
- the tungsten alloy powder according to the invention is compacted by pressing and then preferably sintered in hydrogen in the solid phase. Even at a sintering temperature of 900 ° C, the sintered density reaches over 95% of the theoretical density. With sinter Temperatures between 1200 0 C and 1300 ° C can be produced non-porous sintered bodies.
- the microstructure of the compacts of the festphasengesinterten Fig. 2 ig, in contrast to the liquid-phase parts of the F. 3 no spherical tungsten grains, but an almost space-filling arrangement of polygonal tungsten grains, between which the matrix metal is distributed in a thin layer. 2 is much more fine-grained than the structure of FIG. 3 achieved by liquid phase sintering.
- the diameter of the tungsten grains is 2-5 ⁇ m and the grain size distribution is very narrow-band.
- a line structure can be achieved (not shown) in which the tungsten grains are deformed by approximately 200%.
- the fine-grained and homogeneous structure is the reason for the superior mechanical properties of the sintered parts produced from the powders according to the invention.
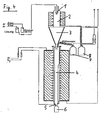
- FIG. 4 shows an arrangement for producing the tungsten powder according to the invention with atomizing nozzle 1, evaporator part 2, separator 3, reducing part 4, hydrogen inlet 5 and discharge member 6, and two containers 7 and 8 for condensate and exhaust gas.
- the powder according to the invention is produced as follows: A common solution of a tungsten salt and the salts of the matrix metals - examples of solution preparation are given below - is sprayed through the atomizer unit 1 and reaches the 800 ° C. evaporator part 2 as an aerosol. Fine particles are formed , which consist of the homogeneously distributed salts (or other compounds) of the alloy components.
- the solid and the gaseous products of the evaporation process are separated in the separator 3. Condensate, and exhaust gas released into the container 7 and 8.
- the reduction part 4 of the solid fraction obtained in separator 3 drops (mostly oxides) a slowly rising flow of hydrogen towards and is reduced at a temperature of 950 ° C to 1200 0 C to the metal.
- the speed of the gas flow can be regulated at the hydrogen inlet 5.
- the finished reduced powder leaves the reduction part 4 through the discharge member 6.
- the salt or oxide production and the subsequent reduction can also be carried out in succession in two separate apparatuses.
- Example 2 a solution of 113.5 g WO 3 in ammonia solution is prepared and filtered through a butterfly filter into a dropping funnel. 39.6 g of Ni (NO 3 ) 2 are placed in a 3-necked flask with two dropping funnels, gas inlet tube, gas outlet with wash bottle and suction tube for the solution. 6H 2 0, 3.6 g FeCl 2 - 4H 2 0 and 2.2 g CoCl 2 weighed out. The first dropping funnel is filled with 450 ml of the filtered WO 3 solution, the second dropping funnel contains 100 ml of semi-concentrated ammonia solution.
- the 3-neck flask and the gas space above the solutions in the dropping funnels are flushed with nitrogen for 20 min. Then the ammonia solution is added dropwise to the salts in the flask with stirring. After the addition has ended, the WO 3 solution from the second dropping funnel is added to the salt solution. Via the suction pipe, the solution can be fed to the metal powder production process described below in the absence of air.
- WO 3 126 g of WO 3 are as triggers e-1 in the example in ammonia solution g. After filtration, the volume is about 900 ml. 393 g of CuSO 4 .5H 2 O are weighed into a 2000 ml beaker and dissolved in 500 ml of water at 50.degree. 500 ml of 33% NH 3 solution are slowly added to the solution. The tungstate and copper solutions are then combined. Avoid standing longer in the cold.
- the hydrogen stron. is 400 standard liters per hour.
- the yield is above 80%.
- the powder contains less than 20 ppm Si0 2 , and depending on the solution preparation between 0 and 900 ppm carbon as well 500 ppm nitrogen.
- the powder particles have a spherical shape. Their average diameter is 20-30 ⁇ m .
- the structure of the particles is spongy.
- the powder of the composition 90% W, 6% Ni, 2% Fe, 2% Co (in percent by weight) has a bulk density of 0.85 g / cm 3 after production. It is compressed by axial or isostatic cold pressing at a pressure of 3 kbar to test specimens, the green density of which is approximately 8.5 g / cm 3 . Due to the sponge-like structure of the powder, which results in good interlocking of the particles after pressing, compacts with high green strength can also be produced without the addition of binders.
- the compact is sintered in dry, flowing hydrogen at 1300 ° C for 4 hours and then degassed in a vacuum of approx. 10 -2 mbar at 1050 ° C for 0.5 hours.
- the resulting sintered body is absolutely non-porous and has a fine-grained sintered structure with W grains of 2 - 5 / um diameter, which are surrounded by a thin skin made of binder alloy.
Abstract
Heterogenes Wolframlegierungspulver zur Herstellung von Sinterteilen mit porenfreiem Gefüge, insbesondere von Wuchtgesch ossen. Das Pulver enthält neben Wolfram eine Binderphase aus Nickel, Kobalt und Eisen und besteht aus 10 - 50 µm grossen Partikeln, wobei die Partikel eine schwammartige Struktur besitzen, die gebildet wird von Wolframkörnern < 1 µm, die ihrerseits umhüllt und zusammengehalten wird von einer dünnen Schicht einer homogenen Ni-Co-Fe-Binderphase (Matrix). Weiterhin werden Herstellungsverfahren angegeben.Heterogeneous tungsten alloy powder for the production of sintered parts with a non-porous structure, in particular balancing projectiles. In addition to tungsten, the powder contains a binder phase made of nickel, cobalt and iron and consists of 10 - 50 µm particles, the particles having a sponge-like structure that is formed by tungsten grains <1 µm, which in turn is enveloped and held together by a thin layer a homogeneous Ni-Co-Fe binder phase (matrix). Manufacturing processes are also specified.
Description
Die Erfindung betrifft ein heterogenes Wolframlegierungspulver, welches neben Wolfram eine Binderphase aus Nickel, Kobalt und Eisen enthält, Verfahren zur Herstellung des Pulvers und seine Verwendung zur Herstellung von Wuchtgeschossen.The invention relates to a heterogeneous tungsten alloy powder which, in addition to tungsten, contains a binder phase composed of nickel, cobalt and iron, a process for the production of the powder and its use for the production of balancing projectiles.
Für hochbeanspruchte Metallteile, insbesondere Wuchtgeschosse, werden Werkstoffe hoher Dichte benötigt. Neben den Edelmetallen Gold und Platin erfüllen noch Uran und Wolfram die Anforderungen nach hoher Dichte. Das einzige Metall, das bei hoher Dichte zu einem vertretbaren Preis gehandelt wird, ist Wolfram. Als reines Metall ist Wolfram jedoch schwierig zu verarbeiten, da es sehr spröde ist. Als Wuchtgeschoss ist es schlecht geeignet, da es die auftretenden Zug- und Druckbelastungen nicht aushält. Wuchtgeschosse sind Vollzylinder aus Metall, deren Länge den Kaliber bei weitem übersteigt. Beim Auftreffen eines Wuchtgeschosses auf eine schrägstehende Panzerplatte verkippt das Wuchtgeschoss. In dem relativ langen Körper treten hohe Biegemomente auf, die häufig zu Bruch des Geschosses und damit zur relativen Wirkungslosigkeit führen.High-density materials are required for highly stressed metal parts, especially balancing projectiles. In addition to the precious metals gold and platinum, uranium and tungsten also meet the requirements for high density. The only metal that is traded at high density at a reasonable price is tungsten. As a pure metal, however, tungsten is difficult to process because it is very brittle. It is poorly suited as a balancing bullet because it cannot withstand the tensile and compressive loads that occur. Balancing bullets are solid cylinders made of metal, the length of which far exceeds the caliber. When a balancing bullet hits an inclined armor plate, the balancing bullet tilts. In the relatively long body, high bending moments occur, which often lead to the bullet breaking and thus to relative ineffectiveness.
Darum kommen für einen Einsatz als Konstruktionswerkstoff für so hoch beanspruchte Bauteile nur Verbundmaterialien in Frage, die das Wolfram in einer duktilen Binderlegierung eingebettet enthalten. Um bei hoher Dichte hohe Festigkeit und Zähigkeit zu erreichen, ist ein Gefügeaufbau erforderlich, der das Wolfram in Form feiner einzelner Partikel enthält, die allseitig von einer sehr dünnen Schicht eines duktilen Bindermetalls umschlossen sind. Das Gefüge darf keine Poren aufweisen. Die mechanischen Eigenschaften (Zugfestigkeit, Bruchdehnung) der Teile sind umso vorteilhafter, je feinkörniger das Gefüge ist.For this reason, only composite materials that contain tungsten embedded in a ductile binder alloy are suitable for use as a construction material for components that are subject to such high stress. In order to achieve high strength and toughness at high density, a structure is required that contains the tungsten in the form of fine individual particles that are enclosed on all sides by a very thin layer of a ductile binder metal. The structure must have no pores. The mechanical properties (tensile strength, elongation at break) of the parts are all the more advantageous the more fine-grained the structure.
Bekannt ist aus F.Eisenkolb, Fortschritte der Pulvermetallurgie, 1963 Band II Seite 439 der Zusatz von in Wolfram löslichen Elementen, wie z.B. Re, die die Duktilität des Wolframs an sich erhöhen. Auf den Seiten 430 bis 433 werden Eigenschaften homogener Wolframlegierungen und die Möglichkeit des Festphasensinterns für homogene Wolframlegierungen angegeben. Homogene Wolframlegierungen eignen sich wegen ihrer geringen Duktilität nicht zur Herstellung von Wuchtgeschossen.It is known from F. Eisenenkolb, progress of powder metallurgy, 1963 volume II page 439 the addition of elements soluble in tungsten, such as e.g. Re, which increase the ductility of the tungsten itself. On pages 430 to 433 properties of homogeneous tungsten alloys and the possibility of solid phase sintering for homogeneous tungsten alloys are given. Homogeneous tungsten alloys are not suitable for the production of balancing bullets because of their low ductility.
Bekannt ist die Herstellung von Bauteilen aus heterogenen Wolframlegierungen durch Flüssigphasensintern. Eine Pulvermischung von Wolframpulver und pulverförmigen Legierungskomponenten wird gepresst und anschliessend gesintert. Um ein porenfreies Gefüge zu erzielen, wird die Technik des Flüssigphasensinterns angewandt. Die Sintertemperatur wird dabei so hoch gewählt, dass die Binderlegierung schmelzflüssig ist. Dabei laufen im wesentlichen drei Prozesse ab:
- 1. Die Binderlegierung bildet sich aus den Pulvern der einzelnen Legierungskomponenten.
- 2. Die schmelzflüssige Binderlegierung umhüllt die Wolframkörner.
- 3. Der Körper verdichtet sich bis zur vollständigen Porenfreiheit.
- 1. The binder alloy is formed from the powders of the individual alloy components.
- 2. The molten binder alloy envelops the tungsten grains.
- 3. The body condenses to complete freedom from pores.
Im gesinterten Zustand sind die Wolframkörner immer grösser als die Pulverpartikel im Ausgangspulver. Das Auftreten einer schmelzflüssigen Phase beim Sinterprozess hat immer eine zusätzliche Vergrösserung der Wolframkörner zur Folge, die durch An- und Umlöseprozesse zwischen Wolfram und flüssiger Matrix ermöglicht wird. Das Phänomen der Kornvergrösserung von festen Ausscheidungen im Kontakt mit Flüssigkeiten ist grundsätzlicher Natur und unter dem Begriff der Ostwaldreifung bekannt.In the sintered state, the tungsten grains are always larger than the powder particles in the starting powder. The occurrence of a molten phase during the sintering process always results in an additional enlargement of the tungsten grains, which is made possible by the dissolving and redissolving processes between the tungsten and the liquid matrix. The phenomenon of the grain enlargement of solid precipitates in contact with liquids is of a fundamental nature and is known as Ostwald ripening.
Flüssigphasengesinterte Wolframlegierungen weisen typi- scherweise ein Gefüge aus kugeligen Wolframteilchen auf, die jeweils in einem Spektrum von etwa 10 - 60 /um großen Partikeln vorliegen, die in einer Binderlegierung eingebettet sind. Die Festigkeit und die Bruchdehnung werden aber jeweils durch die grössten vorhandenen Partikel (hier ca. 60 /um) begrenzt. Häufig ist zu beobachten, dass grosse Körner zusammengewachsen sind. Werkstoffe mit derartig grobkörnigem Gefüge weisen eine nicht ausreichende Festigkeit und nur eine geringe Verformbarkeit auf.Point F lüssigphasengesinterte tungsten alloys s cherweise typical of a structure of spherical tungsten particles, each in a range of about 10 - present 60 / to large particles, which are embedded in a binder alloy. The strength and elongation at break are each limited by the largest particles (here approx. 60 / um). It can often be observed that large grains have grown together. Materials with such a coarse-grained structure do not have sufficient strength and are only slightly deformable.
Auch durch die Wahl feinerer Ausgangspulver sind keine erheblich feineren Gefüge zu erzielen, da die für die Ostwaldreifung verantwortlichen treibenden Kräfte (Verringerung der freien Oberflächenenergie) mit steigender spezifischer Oberfläche der Partikel ansteigen. Auch durch isostatisches Heißpressen ist beim derzeitigen Stand der Technik eine wesentliche Verfeinerung des Gefüges nicht zu erzielen, da dabei ebenfalls eine flüssige Phase benötigt wird, um die Legierungsbildung der Bindermetalle und eine porenfreie Umschliessung der Wolframkörner durch die Binderlegierung zu ermöglichen.The choice of finer starting powders does not result in a significantly finer structure, since the driving forces responsible for the Ostwald ripening (reduction of the free surface energy) increase with increasing specific surface area of the particles. Even with hot isostatic pressing, a substantial refinement of the structure cannot be achieved in the current state of the art, since a liquid phase is also required to enable the binder metals to form alloys and the tungsten grains to be enclosed without pores by the binder alloy.
Der Erfindung liegt die Aufgabe zugrunde, ein Legierungspulver zur Herstellung von Sinterteilen, insbesondere von Wuchtgeschossen, zu schaffen, die neben hohem spezifischen Gewicht eine hohe Zugfestigkeit ( >1200 N/mm2) und Bruchdehnung (> 20 %) aufweisen.The invention has for its object to provide an alloy powder for the production of sintered parts, in particular balancing projectiles, which in addition to high specific weight, high tensile strength (> 1200 N / mm 2 ) and elongation at break (> 20%).
Gelöst wird diese Aufgabe von einem Legierungspulver mit den in Anspruch 1 genannten Merkmalen.This object is achieved by an alloy powder with the features mentioned in claim 1.
Ausführungen der Erfindung und Herstellungsverfahren sind Gegenstände von Unteransprüchen.Embodiments of the invention and manufacturing methods are the subject of subclaims.
Die erfindungsgemässe Legierung weist nach dem Sintern ohne zusätzliche thermomechanische Nachbehandlung folgende Eigenschaften auf:
- Zugfestigkeit > 1200 N/mm2 bei gleichzeitiger Bruchdehnung >25 %. Der Stand der Technik kennt Zugfestigkeiten von 1200 N/mm2 bei lediglich 8 - 10 % Bruchdehnung oder Zugfestigkeiten von 900 N/mm2 bei 25 % Bruchdehnung.
- Tensile strength> 1200 N / mm 2 with simultaneous elongation at break> 25%. The prior art knows tensile strengths of 12 00 N / mm 2 with only 8-10% elongation at break or tensile strengths of 900 N / mm 2 with 25% elongation at break.
Das gleichzeitige Vorliegen von extremer Zugfestigkeit bei extremer Bruchdehnung ist bisher nicht bekannt und weist die erfindungsgemässen Wolfram-Sinterteile als ideale Werkstoffe für Wuchtgeschosse aus. Sowohl die hohen Druck- und Zugbelastungen beim Beschleunigen im Rohr als auch die hohen Biegemomente und Druckkräfte im Geschoss beim Auftreffen auf eine Panzerung werden von dem Material unbeschadet überstanden. Die hervorragenden Eigenschaften prädestinieren die erfindungsgemässen Sinterteile auch für andere Aufgaben in Wissenschaft und Technik, in denen höchste Anforderungen an die Festigkeit und Zähigkeit gestellt werden.The simultaneous presence of extreme tensile strength with extreme elongation at break is not yet known and identifies the tungsten sintered parts according to the invention as ideal materials for balancing projectiles. The material survives both the high pressure and tensile loads when accelerating in the pipe and the high bending moments and compressive forces in the projectile when hitting armor. The outstanding properties make the sintered parts according to the invention also suitable for other tasks in science and technology in which the highest demands are placed on strength and toughness.
Weitere Vorteile, Merkmale und Anwendungen ergeben sich aus den Figuren, die nachfolgend beschrieben werden.Further advantages, features and applications result from the figures which are described below.
Es zeigen:
- Fig. 1 erfindungsgemässe Pulverpartikel,
- Fig. 2 erfindungsgemässes Sintermetall,
- Fig. 3 Sintermetall aus dem Stand der Technik,
- Fig. 4 eine Anordnung zur Herstellung des erfindungsgemässen Legierungspulvers.
- 1 powder particles according to the invention,
- 2 sintered metal according to the invention,
- 3 sintered metal from the prior art,
- Fig. 4 shows an arrangement for producing the alloy powder according to the invention.
Fig. 1 zeigt das erfindungsgemässe Wolframlegierungspulver in tausendfacher Vergrösserung. Es besteht aus Partikeln mit annähernd kugeliger Gestalt (grosse Kugel rechts der Bildmitte). Der Durchmesser beträgt 10 - 50 /um. Die Kugeln weisen eine schwammartige Struktur auf. Die Schwammstruktur wird aufgebaut aus Wolframkörnern von etwa 1 /um Durchmesser, die von einem dünnen Überzug aus Bindermetall bedeckt und zusammengehalten sind. Dadurch ist bereits diejenige Verteilung von Wolfram und Bindermetall vorgegeben, die für das fertige Werkstück kennzeichnend ist. F ig. 1 shows the tungsten alloy powder according to the invention in a thousand times magnification. It consists of particles with an approximately spherical shape (large ball to the right of the center of the picture). The diameter is 10 - 50 / um. The balls have a sponge-like structure. The sponge structure is made up of tungsten grains of about 1 / um diameter, which are covered and held together by a thin coating of binder metal. As a result, the distribution of tungsten and binder metal that is characteristic of the finished workpiece is predetermined.
Im Gegensatz zu den nach dem Stand der Technik verwendeten Pulvermischungen aus W, Ni, Co und Fe-Pulver ist das erfindungsgemässe Pulver also fertig legiert und die Binderlegierung umschliesst bereits die W-Körner in Form eines Überzuges. Damit müssen bei der Herstellung dichter Sinterkörper die beiden Prozesse Bildung der Binderlegierung und Umhüllung der Wolframkörner nicht mehr unter Zuhilfenahme einer schmelzflüssigen Phase durchgeführt werden. Das Pulver kann in der festen Phase zu dichten Körpern versintert werden.In contrast to those used in the prior art powder mixtures of W, Ni, Co and Fe powder, the inventive powder is therefore alloyed finished and the B inthe alloy already surrounds the W grains in the form of a coating. So that must be more dense during manufacture Sintered bodies the two processes of forming the binder alloy and coating the tungsten grains can no longer be carried out with the aid of a molten phase. The powder can be sintered into dense bodies in the solid phase.
Die Schwammstruktur der Pulverpartikel ist locker aufgebaut, so dass das Pulver unter 3 kbar Pressdruck auf etwa 50 % der theoretischen Dichte eines Kompaktums verdichtet werden kann. Diese hohe Gründichte in Verbindung mit der grossen spezifischen Oberfläche von grössenordnungsmässig 1 m2/g ermöglicht das druckfreie Dichtsintern der Presslinge unter Vermeidung von flüssigen Phasen.The sponge structure of the powder particles is loosely structured so that the powder can be compressed to around 50% of the theoretical density of a compact under 3 kbar pressure. This high green density in connection with the large specific surface area of the order of 1 m 2 / g enables the pressure-free sealing sintering of the compacts while avoiding liquid phases.
Fig. 2 zeigt ein Schliffbild einer erfindungsgemässen gesinterten Legierung in 600-facher Vergrösserung.2 shows a micrograph of a sintered alloy according to the invention in a magnification of 600 times.
Fig. 3 zeigt zum Vergleich eine gemäss dem Stand der Technik hergestellte, also flüssigphasengesinterte Legierung in 600-facher Vergrösserung.For comparison, FIG. 3 shows an alloy produced according to the prior art, that is to say liquid phase sintered, in a magnification of 600 times.
Das erfindungsgemässe Wolframlegierungspulver wird durch Pressen kompaktiert und anschliessend vorzugsweise in Wasserstoff in der festen Phase gesintert. Bereits bei einer Sintertemperatur von 900° C erreicht die Sinterdichte über 95 % der theoretischen Dichte. Mit Sintertemperaturen zwischen 12000 C und 1300° C lassen sich porenfreie Sinterkörper herstellen.The tungsten alloy powder according to the invention is compacted by pressing and then preferably sintered in hydrogen in the solid phase. Even at a sintering temperature of 900 ° C, the sintered density reaches over 95% of the theoretical density. With sinter Temperatures between 1200 0 C and 1300 ° C can be produced non-porous sintered bodies.
Das Gefüge der festphasengesinterten Presslinge der Fig. 2 weist im Gegensatz zu den flüssigphasengesinterten Teilen der Fig. 3 keine kugeligen Wolframkörner auf, sondern eine nahezu raumerfüllende Anordnung polygoner Wolframkörner, zwischen denen in dünner Schicht das Matrixmetall verteilt ist. Das Sintergefüge der Fig. 2 ist wesentlich feinkörniger als das durch Flüssigphasensintern erreichte Gefüge der Fig. 3. Wie aus Fig. 2 zu entnehmen ist, beträgt der Durchmesser der Wolframkörner 2 - 5 µm und die Korngrössenverteilung ist sehr schmalbandig. Beim Aufbringen gerichteter Kräfte lässt sich ein zeiliges Gefüge erzielen (nicht gezeigt), in dem die Wolframkörner über etwa 200 % verformt sind. Das feinkörnige und homogene Gefüge ist die Ursache für die überlegenen mechanischen Eigenschaften der aus den erfindungsgemässen Pulvern hergestellten Sinterteile.The microstructure of the compacts of the festphasengesinterten Fig. 2, ig, in contrast to the liquid-phase parts of the F. 3 no spherical tungsten grains, but an almost space-filling arrangement of polygonal tungsten grains, between which the matrix metal is distributed in a thin layer. 2 is much more fine-grained than the structure of FIG. 3 achieved by liquid phase sintering. As can be seen from FIG. 2, the diameter of the tungsten grains is 2-5 μm and the grain size distribution is very narrow-band. When applying directed forces, a line structure can be achieved (not shown) in which the tungsten grains are deformed by approximately 200%. The fine-grained and homogeneous structure is the reason for the superior mechanical properties of the sintered parts produced from the powders according to the invention.
Fig. 4 zeigt eine Anordnung zur Herstellung des erfindungsgemässen Wolframpulvers mit Zerstäuberdüse 1, Verdampferteil 2, Separator 3, Reduktionsteil 4, Wasserstoffeintritt 5 und Austrageorgan 6, sowie zwei Behältern 7 und 8 für Kondensat und Abgas.FIG. 4 shows an arrangement for producing the tungsten powder according to the invention with atomizing nozzle 1, evaporator part 2,
Das erfindungsgemässe Pulver wird wie folgt hergestellt: Eine gemeinsame Lösung eines Wolframsalzes und der Salze der Matrixmetalle - Beispiele zur Lösungsbereitung werden weiter unten angegeben - wird durch die Zerstäubereinheit 1 versprüht und gelangt als Aerosol in den 8000 C heissen Verdampferteil 2. Es entstehen feine Partikel, die aus den homogen ineinander verteilten Salzen (oder anderen Verbindungen) der Legierungskomponenten bestehen.The powder according to the invention is produced as follows: A common solution of a tungsten salt and the salts of the matrix metals - examples of solution preparation are given below - is sprayed through the atomizer unit 1 and reaches the 800 ° C. evaporator part 2 as an aerosol. Fine particles are formed , which consist of the homogeneously distributed salts (or other compounds) of the alloy components.
Im Separator 3 werden die festen und die gasförmigen Produkte des Verdampfungsprozesses getrennt. Kondensat und Abgas gelangen in die Behälter 7 und 8. Im Reduktionsteil 4 fällt der im Separator 3 gewonnene Feststoffanteil (hauptsächlich Oxide) einem langsam aufsteigenden Wasserstoffstrom entgegen und wird bei einer Temperatur von 950° C bis 12000 C zum Metall reduziert. Am Wasserstoffeintritt 5 kann die Geschwindigkeit der Gasströmung geregelt werden. Das fertig reduzierte Pulver verlässt den Reduktionsteil 4 durch das Austrageorgan 6. Die Salz- bzw. Oxidherstellung und die nachfolgende Reduktion können auch nacheinander in zwei getrennten Apparaturen durchgeführt werden.The solid and the gaseous products of the evaporation process are separated in the
Entscheidend für die hohe Sinteraktivität des Pulvers, die allein ein Festphasensintern ermöglicht, sind die Feinheit der Zerstäubung bei der Pulverherstellung, die Konzentration und Zusammensetzung der gemeinsamen Lösung sowie die schonende Reduktion der Salz- bzw. Oxidpartikel, bei der ein Zusammenwachsen der Salz- oder der Metallpartikel vermieden werden muss.Decisive for the high sintering activity of the powder, which is only possible with solid phase sintering, are the fineness of atomization during powder production, the concentration and composition of the common solution and the gentle reduction of the salt or oxide particles, in which a coalescence of the salt or metal particles must be avoided.
Bis zu einer Salzkonzentration, die 600 g aufgelöstem Metall pro Liter Lösung entspricht, ist eine Zerstäubung ausreichend, die ein mittleres Tropfenspektrum von 30 bis 50 µm erzeugt. Die aus der Lösung entstehenden Feststoffpartikel haben eine dem Tropfenspektrum vergleichbare Grössenverteilung. Wichtig ist bereits an dieser Stelle die Schwammstruktur der Feststoffpartikel, die im nachfolgenden Reduktionsschritt kurze Diffusionswege und damit kurze Reaktionszeiten erlaubt. Auf diese Weise ist eine Reduktion der Partikel im freien Fall möglich, bei der ein Zusammenwachsen der Salz- oder Metallpartikel unterbleibt.Up to a salt concentration that corresponds to 600 g of dissolved metal per liter of solution, atomization is sufficient, which produces an average drop spectrum of 30 to 50 µm. The solid particles resulting from the solution have a size distribution comparable to the drop spectrum. The sponge structure of the solid particles is important at this point, which allows short diffusion paths and thus short reaction times in the subsequent reduction step. In this way, a reduction of the particles in free fall is possible, in which the salt or metal particles do not grow together.
Bei den Versuchen hat sich als Lösungsmittel Wasser bewährt. Bei dessen Verwendung fielen die obengenannten Feststoffpartikel als Oxidmischungen an. Als Reduktionsmittel kam in diesen Fällen Wasserstoff zur Anwendung. Die Lösungsbereitung kann auf zwei Wegen erfolgen:
- Entweder man arbeitet in schwach saurem Medium bei einem pH > 3 unter Verwendung von Ammoniummetawolframat als löslicher Wolframverbindung oder man bereitet eine ammoniakalische Lösung von Wolframsäure, deren Anhydrid oder einem ihrer Salze und verhindert die Fällung der Kationen der Matrixmetalle durch Komplexieren entweder mit Ammoniak oder mit den üblichen organischen Komplexbildnern wie z.B. EDTA. Die Verwendung kolloidaler Wolframverbindungen, z.B. in der Form von H2 WO4 aq, WO3 oder Ammoniumparawolframat, führte nach kurzer Zeit zu Störungen bei der Lösungszerstäubung.
- Either one works in a weakly acidic medium at a pH> 3 using ammonium metatungstate as a soluble tungsten compound or one prepares an ammoniacal solution of tungstic acid, its anhydride or a their salts and prevents the precipitation of the cations of the matrix metals by complexing either with ammonia or with the usual organic complexing agents such as EDTA. The use of colloidal tungsten compounds, for example in the form of H 2 WO 4 aq, WO 3 or ammonium paratungstate, led to disturbances in the solution atomization after a short time.
Im Falle eisenhaltiger Lösungen muss beim Komplexieren mit Ammoniak von Salzen des zweiwertigen Eisens ausgegangen und Luftzutritt sorgfältig ausgeschlossen werden. Auch bei der Verwendung von Ammoniummetawolframat stört dreiwertiges Eisen, da es in den üblichen Konzentrationen den pH der Lösung auf Werte um 1 einstellt, so dass nach etwa 1 h Aufbewahrung ein Niederschlag ausfällt, der ein Zerstäuben der Lösung verhindert. Lösungen, die Eisen (II)-Ionen enthalten, bleiben nach dem Filtrieren über Blaubandfilter länger als 24 h bei Raumtemperatur klar.In the case of solutions containing iron, salts of divalent iron must be used when complexing with ammonia and air access must be carefully excluded. Trivalent iron also interferes with the use of ammonium metatungstate, since it adjusts the pH of the solution to values around 1 in the usual concentrations, so that after about 1 hour of storage, precipitation precipitates, which prevents the solution from atomizing. Solutions containing iron (II) ions remain clear for more than 24 hours at room temperature after filtering through blue band filters.
Es folgen Beispiele zur Lösungsherstellung, zur Pulverherstellung und zur Sinterung:The following are examples of solution preparation, powder production and sintering:
In ein 800 ml Becherglas werden 117,3 g W03 eingewogen und mit ca. 300 ml Wasser aufgeschlämmt. Es wird 3 h bei Siedehitze gerührt, bis die Farbe des Bodenkörpers von gelb nach weiß umgeschlagen ist. Nach dem Abkühlen auf Raumtemperatur wird mit 100 ml 33%iger NH3-Lösung versetzt und schwach erwärmt. Nach 30 bis 40 Minuten wird die fast klare Lösung über ein Faltenfilter filtriert.117.3 g of W0 3 are weighed into an 800 ml beaker and suspended with approx. 300 ml of water. The mixture is stirred at boiling heat for 3 h until the color of the base has changed from yellow to white. After cooling to room temperature, 100 ml of 33% NH 3 solution are added and it is weak warms. After 30 to 40 minutes, the almost clear solution is filtered through a pleated filter.
In ein 250 ml Becherglas werden 24,3 g Ni(NO3)2 · 6H20, 6,0 g Co(CH3COO)2· 4H20, 5,06 g Fe(NO3)3 · 9H20 und 45 g EDTA eingewogen und mit 80 ml Wasser aufgerührt. In die Suspension werden langsam 30 - 40 ml 33%ige NH3-Lösung eingetropft, so dass eine dunkelviolette Lösung entsteht, die mit dem Filtrat der Wolframlösung vereinigt wird.In a 250 ml beaker, 24.3 g of Ni (NO 3 ) 2 .6H 2 0, 6.0 g of Co (CH 3 COO) 2 .4H 2 0, 5.06 g of Fe (NO 3 ) 3 .9H 2 0 and 45 g of EDTA are weighed in and stirred with 80 ml of water. 30-40 ml of 33% NH 3 solution are slowly added dropwise to the suspension, so that a dark purple solution is formed, which is combined with the filtrate of the tungsten solution.
Wie im Beispiel 1 wird eine Lösung von 113,5 g WO3 in Ammoniak-Lösung hergestellt und über einen Falterfilter in einen Tropftrichter filtriert. In einen 3-Hals-Kolben mit zwei Tropftrichtern, Gaseinleitungsrohr, Gasaustritt mit Waschflasche und Ansaugrohr für die Lösung werden 39,6 g Ni(NO3)2 . 6H20, 3,6 g FeCl2 - 4H20 und 2,2 g CoCl2 eingewogen. Der erste Tropftrichter ist mit 450 ml der filtrierten WO3-Lösung gefüllt, der zweite Tropftrichter enthält 100 ml halbkonzentrierte Ammoniaklösung.As in Example 1, a solution of 113.5 g WO 3 in ammonia solution is prepared and filtered through a butterfly filter into a dropping funnel. 39.6 g of Ni (NO 3 ) 2 are placed in a 3-necked flask with two dropping funnels, gas inlet tube, gas outlet with wash bottle and suction tube for the solution. 6H 2 0, 3.6 g FeCl 2 - 4H 2 0 and 2.2 g CoCl 2 weighed out. The first dropping funnel is filled with 450 ml of the filtered WO 3 solution, the second dropping funnel contains 100 ml of semi-concentrated ammonia solution.
Der 3-Hals-Kolben und der Gasraum über den Lösungen in den Tropftrichtern wird 20 min mit Stickstoff gespült. Dann wird unter Rühren die Ammoniaklösung zu den Salzen im Kolben getropft. Nach Beendigung der Zugabe wird die WO3-Lösung aus dem zweiten Tropftrichter zur Salzlösung gegeben. über das Ansaugrohr kann die Lösung unter Luftausschluss dem nachstehend beschriebenen Verfahren zur Metallpulverherstellung zugeführt werden.The 3-neck flask and the gas space above the solutions in the dropping funnels are flushed with nitrogen for 20 min. Then the ammonia solution is added dropwise to the salts in the flask with stirring. After the addition has ended, the WO 3 solution from the second dropping funnel is added to the salt solution. Via the suction pipe, the solution can be fed to the metal powder production process described below in the absence of air.
126 g WO3 werden wie im Beispiel 1 in Ammoniaklösung aufge-löst. Nach der Filtration beträgt das Volumen etwa 900 ml. In ein 2000 ml Becherglas werden 393 g CuSO4 · 5H2O eingewogen und bei 50°C in 500 ml Wasser aufgelöst. Zur Lösung werden langsam 500 ml 33%ige NH3-Lösung gegeben. Anschliessend werden die Wolframat- und die Kupferlösung vereinigt. Längeres Stehen in der Kälte ist zu vermeiden.126 g of WO 3 are as triggers e-1 in the example in ammonia solution g. After filtration, the volume is about 900 ml. 393 g of CuSO 4 .5H 2 O are weighed into a 2000 ml beaker and dissolved in 500 ml of water at 50.degree. 500 ml of 33% NH 3 solution are slowly added to the solution. The tungstate and copper solutions are then combined. Avoid standing longer in the cold.
Es werden 800 ml H20 vorgelegt. 485,3 g Ammoniummetawolframat werden unter kräftigem Rühren langsam zugeschüttet. Es wird weitergerührt bis eine klare Lösung entstanden ist. Zur Wolframat-Lösung werden 28,5 g FeCl2 · 4H20 in 500 ml H20 unter starkem Rühren langsam zugegeben. Es ist wichtig, dass die Anwesenheit von dreiwertigem Eisen weitgehend ausgeschlossen wird, da sonst innerhalb kurzer Zeit ein gelb-weißer Niederschlag ausfällt. Zur so bereiteten Eisen-Wolfram-Lösung werden anschliessend 118,9 gNi(NO3)2 · 6H2O und 39,5 g Co(NO3)2 · 6H2O, gelöst im Gesamtvolumen von 500 ml, zugefügt.800 ml of H 2 O are introduced. 485.3 g of ammonium metatungstate are slowly poured in with vigorous stirring. Stirring continues until a clear solution has been found. For tungstate solution 28.5 g of FeCl 2 .4H 2 0 in 500 ml H 2 0 was slowly added with vigorous stirring. It is important that the presence of trivalent iron is largely ruled out, otherwise a yellow-white precipitate will fail within a short time. 118.9 g of Ni (NO 3 ) 2 .6H 2 O and 39.5 g of Co (NO 3 ) 2 .6H 2 O, dissolved in a total volume of 500 ml, are then added to the iron-tungsten solution prepared in this way.
In einem typischen Experiment werden in der oben beschriebenen Anlage 2 1 Lösung pro h versprüht. Der Wasserstoffstron. beträgt 400 Normliter pro h. Die Ausbeute liegt oberhalb 80%. Das Pulver enthält weniger als 20 ppm Si02, undje nach Lösungsherstellung zwischen 0 und 900 ppm Kohlenstoff sowie 500 ppm Stickstoff. Die Pulverpartikel haben Kugelform. Ihr Durchmesser beträgt im Mittel 20 - 30 /um. Die Struktur der Partikel ist schwammartig.In a typical experiment, 2 1 solution per hour are sprayed in the system described above. The hydrogen stron. is 400 standard liters per hour. The yield is above 80%. The powder contains less than 20 ppm Si0 2 , and depending on the solution preparation between 0 and 900 ppm carbon as well 500 ppm nitrogen. The powder particles have a spherical shape. Their average diameter is 20-30 µm . The structure of the particles is spongy.
Das Pulver der Zusammensetzung 90% W, 6% Ni, 2% Fe, 2% Co (in Gewichtsprozenten) besitzt nach der Herstellung eine Schüttdichte von 0,85 g/cm3. Es wird durch axiales oder isostatisches Kaltpressen bei einem Druck von 3 kbar zu Probekörpern verdichtet, deren Gründichte etwa 8,5 g/cm3 beträgt. Aufgrund der schwammartigen Struktur der Pulver, die eine gute Verzahnung der Partikel nach dem Pressen bewirkt, lassen sich auch ohne Zusätze von Bindemitteln Preßlinge mit hoher Grünfestigkeit herstellen.The powder of the composition 90% W, 6% Ni, 2% Fe, 2% Co (in percent by weight) has a bulk density of 0.85 g / cm 3 after production. It is compressed by axial or isostatic cold pressing at a pressure of 3 kbar to test specimens, the green density of which is approximately 8.5 g / cm 3 . Due to the sponge-like structure of the powder, which results in good interlocking of the particles after pressing, compacts with high green strength can also be produced without the addition of binders.
Der Preßling wird in trockenem, strömenden Wasserstoff bei 1300°C 4 Stunden gesintert und anschliessend in Vakuum von ca. 10 -2 mbar bei 1050°C 0,5 Stunden entgast.The compact is sintered in dry, flowing hydrogen at 1300 ° C for 4 hours and then degassed in a vacuum of approx. 10 -2 mbar at 1050 ° C for 0.5 hours.
Der entstandene Sinterkörper ist absolut porenfrei und weist ein feinkörniges Sintergefüge mit W-Körnern von 2 - 5 /um Durchmesser auf, die von einer dünnen Haut aus Binderlegierung umgeben sind.The resulting sintered body is absolutely non-porous and has a fine-grained sintered structure with W grains of 2 - 5 / um diameter, which are surrounded by a thin skin made of binder alloy.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT83105070T ATE25111T1 (en) | 1982-07-16 | 1983-05-21 | TUNGSTEN ALLOY POWDER. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3226648A DE3226648C2 (en) | 1982-07-16 | 1982-07-16 | Heterogeneous tungsten alloy powder |
| DE3226648 | 1982-07-16 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0098944A2 true EP0098944A2 (en) | 1984-01-25 |
| EP0098944A3 EP0098944A3 (en) | 1984-03-28 |
| EP0098944B1 EP0098944B1 (en) | 1987-01-21 |
Family
ID=6168588
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP83105070A Expired EP0098944B1 (en) | 1982-07-16 | 1983-05-21 | Tungsten alloy powder |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US4498395A (en) |
| EP (1) | EP0098944B1 (en) |
| JP (1) | JPS5925950A (en) |
| KR (1) | KR910003572B1 (en) |
| AT (1) | ATE25111T1 (en) |
| DE (2) | DE3226648C2 (en) |
| IL (1) | IL69232A (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0183017A1 (en) * | 1984-10-20 | 1986-06-04 | DORNIER SYSTEM GmbH | Sintering process for prealloyed tungsten powder |
| EP0204909A1 (en) * | 1985-05-29 | 1986-12-17 | Dornier Gmbh | Electrode material for a spar gap assembly |
| EP0304181A1 (en) * | 1987-07-30 | 1989-02-22 | Teledyne Industries, Inc. | High density tungsten-nickel-iron-cobalt alloys having improved hardness, and method for making them |
| EP0326861A1 (en) * | 1988-01-30 | 1989-08-09 | H.C. Starck GmbH & Co. KG | Composite agglomerated metal powder, process for manufacturing it an its use |
| EP0332474A1 (en) * | 1988-03-11 | 1989-09-13 | Camco Drilling Group Limited | Improvements in or relating to cutter assemblies for rotary drill bits |
| FR2659735A1 (en) * | 1990-03-07 | 1991-09-20 | Saint Louis Inst | HYPERSONIC PROJECTILE WITH KINETIC EFFECT AND METHOD FOR THE PRODUCTION THEREOF. |
| DE3637930C1 (en) * | 1985-11-07 | 1992-04-09 | Fraunhofer Ges Forschung | Mfg. composite material for armour piercing ammunition - using alloy powder contg. tungsten@, nickel@, iron@, copper@, titanium@, aluminium@ and/or molybdenum@ |
| FR2672619A1 (en) * | 1985-11-07 | 1992-08-14 | Fraunhofer Ges Forschung | Tungsten-based composite material and process for its preparation |
| EP0384623B1 (en) * | 1989-02-14 | 1994-03-02 | Camco Drilling Group Limited | Improvements in or relating to cutting elements for rotary drill bits |
| FR2784690A1 (en) * | 1998-10-16 | 2000-04-21 | Eurotungstene Poudres | Pre-alloyed tungsten and-or molybdenum and transition metal based powder, useful for sintered parts or cermet binders, comprises micron-size elementary particles |
| CN100363131C (en) * | 2005-12-12 | 2008-01-23 | 北京科技大学 | Method for preparing grain-size-contrdlable superfine crystallite tungsten and tungsten-copper composite material |
| CN104722767A (en) * | 2015-04-16 | 2015-06-24 | 柳州豪祥特科技有限公司 | Tungsten powder preparation method |
| EP3643429A1 (en) * | 2018-10-23 | 2020-04-29 | Bayerische Metallwerke GmbH | Tungsten alloy product and method of preparation and use for a tungsten alloy product |
Families Citing this family (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4970960A (en) * | 1980-11-05 | 1990-11-20 | Feldmann Fritz K | Anti-material projectile |
| DE3715979A1 (en) * | 1985-11-13 | 1988-12-08 | Mtu Muenchen Gmbh | Process for producing dispersion-hardened metal alloys |
| US4897117A (en) * | 1986-03-25 | 1990-01-30 | Teledyne Industries, Inc. | Hardened penetrators |
| DE3634433A1 (en) * | 1986-10-09 | 1988-04-14 | Diehl Gmbh & Co | INSERT FOR HOLLOW LOADS OR Penetrators or balancing bodies for projectiles |
| DE3700805A1 (en) * | 1987-01-14 | 1990-03-08 | Fraunhofer Ges Forschung | Fibre-reinforced composite material based on tungsten/heavy metal |
| DE3705382A1 (en) * | 1987-02-20 | 1988-09-01 | Diehl Gmbh & Co | PENETRATOR AND METHOD FOR THE PRODUCTION THEREOF |
| NL8700935A (en) * | 1987-04-21 | 1988-11-16 | Philips Nv | IMPREGNATED CATHODES WITH A CHECKED POROSITY. |
| FR2615046B1 (en) * | 1987-05-04 | 1992-12-31 | Merlin Gerin | SINTERED COMPOSITE MATERIAL FOR ELECTRICAL CONTACT AND CONTACT PAD USING SAID MATERIAL |
| US5008071A (en) * | 1988-01-04 | 1991-04-16 | Gte Products Corporation | Method for producing improved tungsten nickel iron alloys |
| US4811666A (en) * | 1988-01-04 | 1989-03-14 | Lutfy Eric A | Solid projectiles |
| DE3821474C1 (en) * | 1988-06-25 | 1998-08-27 | Nwm De Kruithoorn Bv | One-piece frangible armour-piercing discarding sabot |
| DE3830111A1 (en) * | 1988-09-05 | 1990-03-15 | Dornier Gmbh | Alloy powder |
| US5088415A (en) * | 1990-10-31 | 1992-02-18 | Safety Shot Limited Partnership | Environmentally improved shot |
| US5189252A (en) * | 1990-10-31 | 1993-02-23 | Safety Shot Limited Partnership | Environmentally improved shot |
| US5913256A (en) | 1993-07-06 | 1999-06-15 | Lockheed Martin Energy Systems, Inc. | Non-lead environmentally safe projectiles and explosive container |
| US5897962A (en) * | 1993-07-16 | 1999-04-27 | Osram Sylvania Inc. | Method of making flowable tungsten/copper composite powder |
| US5821441A (en) * | 1993-10-08 | 1998-10-13 | Sumitomo Electric Industries, Ltd. | Tough and corrosion-resistant tungsten based sintered alloy and method of preparing the same |
| JPH07216478A (en) * | 1994-02-04 | 1995-08-15 | Nippon Yakin Kogyo Co Ltd | Production of tungsten alloy |
| SE504244C2 (en) * | 1994-03-29 | 1996-12-16 | Sandvik Ab | Methods of making composite materials of hard materials in a metal bonding phase |
| MX9700050A (en) * | 1994-07-06 | 1997-12-31 | Lockheed Martin Energy Sys Inc | Non-lead, environmentally safe projectiles and method of making same. |
| US6103392A (en) * | 1994-12-22 | 2000-08-15 | Osram Sylvania Inc. | Tungsten-copper composite powder |
| US5876793A (en) * | 1996-02-21 | 1999-03-02 | Ultramet | Fine powders and method for manufacturing |
| US6607692B2 (en) | 1997-01-30 | 2003-08-19 | Doris Nebel Beal Intervivos Patent Trust | Method of manufacture of a powder-based firearm ammunition projectile employing electrostatic charge |
| US5847313A (en) * | 1997-01-30 | 1998-12-08 | Cove Corporation | Projectile for ammunition cartridge |
| US5789698A (en) * | 1997-01-30 | 1998-08-04 | Cove Corporation | Projectile for ammunition cartridge |
| US6551376B1 (en) | 1997-03-14 | 2003-04-22 | Doris Nebel Beal Inter Vivos Patent Trust | Method for developing and sustaining uniform distribution of a plurality of metal powders of different densities in a mixture of such metal powders |
| KR100217032B1 (en) * | 1997-06-14 | 1999-09-01 | 박호군 | Fabrication method of w-skelton structure for the infiltration of cu melt and composites thereof |
| SE510659C2 (en) * | 1997-10-14 | 1999-06-14 | Sandvik Ab | Process for preparing a cemented carbide comprising coating of particles of the cementitious binder with binder metal |
| CA2334552C (en) * | 2000-02-07 | 2007-04-24 | Halliburton Energy Services, Inc. | High performance powdered metal mixtures for shaped charge liners |
| US7011027B2 (en) * | 2000-05-20 | 2006-03-14 | Baker Hughes, Incorporated | Coated metal particles to enhance oil field shaped charge performance |
| DE10041194A1 (en) * | 2000-08-23 | 2002-03-07 | Starck H C Gmbh | Process for the production of composite components by powder injection molding and suitable composite powder |
| WO2003104742A2 (en) * | 2001-05-15 | 2003-12-18 | Beal Harold F | In-situ formation of cap for ammunition projectile |
| US7243588B2 (en) | 2001-05-15 | 2007-07-17 | Doris Nebel Beal Inter Vivos Patent Trust | Power-based core for ammunition projective |
| US7849695B1 (en) * | 2001-09-17 | 2010-12-14 | Alliant Techsystems Inc. | Rocket thruster comprising load-balanced pintle valve |
| AU2003227884A1 (en) * | 2002-04-25 | 2003-11-10 | The Morgan Crucible Company Plc | Process for manufacturing an alloy material for use in the manufacture of synthetic diamonds |
| US7473432B2 (en) * | 2002-10-11 | 2009-01-06 | Idea Ag | NSAID formulations, based on highly adaptable aggregates, for improved transport through barriers and topical drug delivery |
| US7690312B2 (en) * | 2004-06-02 | 2010-04-06 | Smith Timothy G | Tungsten-iron projectile |
| WO2006083725A2 (en) * | 2005-02-01 | 2006-08-10 | Carlotto John A | Vacuum deposition of coating materials on powders |
| US8196571B2 (en) * | 2006-03-10 | 2012-06-12 | Jenson Martin W | Apparatus, system, and method for launching a granular substance |
| US20090042057A1 (en) * | 2007-08-10 | 2009-02-12 | Springfield Munitions Company, Llc | Metal composite article and method of manufacturing |
| US8522687B2 (en) * | 2007-09-06 | 2013-09-03 | Shaiw-Rong Scott Liu | Kinetic energy penetrator |
| JP5522712B2 (en) * | 2008-08-25 | 2014-06-18 | 公立大学法人兵庫県立大学 | Transition metal-encapsulated tungsten carbide, tungsten carbide-dispersed cemented carbide and method for producing the same |
| JP5522713B2 (en) * | 2008-08-25 | 2014-06-18 | 公立大学法人兵庫県立大学 | Transition metal solid solution tungsten alloy powder and method for producing the same |
| DE102015102763A1 (en) * | 2015-02-26 | 2016-09-01 | Vacuumschmelze Gmbh & Co. Kg | A method of manufacturing a thermoelectric article for a thermoelectric conversion device |
| JP2018035020A (en) | 2016-08-30 | 2018-03-08 | 住友電気工業株式会社 | Aqueous solution composition and method for producing the same, oxide powder and method for producing the same, carbide powder and method for producing the same, and cemented carbide and method for producing the same |
| TWI798480B (en) * | 2016-12-09 | 2023-04-11 | 美商史達克公司 | Fabrication of metallic parts by additive manufacturing and tungsten heavy metal alloy powders therefor |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1054154A (en) * | 1964-10-15 | |||
| GB812848A (en) * | 1956-06-26 | 1959-05-06 | Mallory Metallurg Prod Ltd | Improvements in and relating to high density sintered metal compositions or alloys |
| FR2225980A5 (en) * | 1969-10-28 | 1974-11-08 | Onera (Off Nat Aerospatiale) | |
| US3907546A (en) * | 1974-03-28 | 1975-09-23 | Gte Sylvania Inc | Molybdenum flame spray powder and process |
| US4012230A (en) * | 1975-07-07 | 1977-03-15 | The United States Of America As Represented By The United States Energy Research And Development Administration | Tungsten-nickel-cobalt alloy and method of producing same |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1106965B (en) | 1957-02-12 | 1961-05-18 | Siemens Ag | Process for the production of densely sintered molded bodies from silver composite material |
| DE1100833B (en) | 1958-04-02 | 1961-03-02 | Siemens Ag | High density composite metal for heavy-duty electrical contacts |
| DE1253919B (en) | 1959-01-08 | 1967-11-09 | Renault | Process for the powder metallurgical production of shaped bodies from tungsten and copper |
| GB977249A (en) | 1962-08-09 | 1964-12-02 | Diosgyoeri Gepgyar | Contact pulley for continuously operating softening machines in wire manufacture |
| FR1429965A (en) | 1964-04-21 | 1966-02-25 | English Electric Co Ltd | Contact or electrode for vacuum switches or spark gaps |
| US3382066A (en) | 1965-07-23 | 1968-05-07 | Mallory & Co Inc P R | Method of making tungsten-copper composites |
| FR2155565A5 (en) | 1971-12-09 | 1973-05-18 | Energoinvest Preduzece Za Proj | Tungsten-copper impregnated with copper - by electrolytic impregnation and subsequent heat treatment |
| US4050933A (en) * | 1973-02-21 | 1977-09-27 | Stanadyne, Inc. | Impervious metal object and method of making the same |
| US3973948A (en) | 1973-11-12 | 1976-08-10 | Gte Sylvania Incorporated | Free flowing powder and process for producing it |
| ES199626Y (en) * | 1974-01-19 | 1975-12-16 | Centro De Est. Tec De Mate. Esp. - Inst. Nac. Ind. | SMALL CALIBER PROJECTILE WITH ASYMMETRIC POINT. |
| US4060414A (en) * | 1975-06-06 | 1977-11-29 | Ford Motor Company | Copper coated iron-carbon eutectic alloy powders |
| US4440800A (en) * | 1980-04-24 | 1984-04-03 | Unisearch Limited | Vapor coating of powders |
| JPS56166311A (en) * | 1980-05-26 | 1981-12-21 | Sumitomo Electric Ind Ltd | Manufacture of alloy powder containing molybdenum and tungsten |
-
1982
- 1982-07-16 DE DE3226648A patent/DE3226648C2/en not_active Expired
-
1983
- 1983-05-21 EP EP83105070A patent/EP0098944B1/en not_active Expired
- 1983-05-21 DE DE8383105070T patent/DE3369346D1/en not_active Expired
- 1983-05-21 AT AT83105070T patent/ATE25111T1/en not_active IP Right Cessation
- 1983-07-06 US US06/511,510 patent/US4498395A/en not_active Expired - Fee Related
- 1983-07-15 JP JP58128074A patent/JPS5925950A/en active Granted
- 1983-07-15 KR KR1019830003235A patent/KR910003572B1/en not_active IP Right Cessation
- 1983-07-15 IL IL69232A patent/IL69232A/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB812848A (en) * | 1956-06-26 | 1959-05-06 | Mallory Metallurg Prod Ltd | Improvements in and relating to high density sintered metal compositions or alloys |
| GB1054154A (en) * | 1964-10-15 | |||
| FR2225980A5 (en) * | 1969-10-28 | 1974-11-08 | Onera (Off Nat Aerospatiale) | |
| US3907546A (en) * | 1974-03-28 | 1975-09-23 | Gte Sylvania Inc | Molybdenum flame spray powder and process |
| US4012230A (en) * | 1975-07-07 | 1977-03-15 | The United States Of America As Represented By The United States Energy Research And Development Administration | Tungsten-nickel-cobalt alloy and method of producing same |
Non-Patent Citations (1)
| Title |
|---|
| Winnacker,Küchler,Chemische Technologie, Bd. 6, Metallurgie, 1973, P. 478-481 (Carl Hanser Verlag, München) * |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0183017A1 (en) * | 1984-10-20 | 1986-06-04 | DORNIER SYSTEM GmbH | Sintering process for prealloyed tungsten powder |
| EP0204909A1 (en) * | 1985-05-29 | 1986-12-17 | Dornier Gmbh | Electrode material for a spar gap assembly |
| FR2672619A1 (en) * | 1985-11-07 | 1992-08-14 | Fraunhofer Ges Forschung | Tungsten-based composite material and process for its preparation |
| DE3637930C1 (en) * | 1985-11-07 | 1992-04-09 | Fraunhofer Ges Forschung | Mfg. composite material for armour piercing ammunition - using alloy powder contg. tungsten@, nickel@, iron@, copper@, titanium@, aluminium@ and/or molybdenum@ |
| EP0304181A1 (en) * | 1987-07-30 | 1989-02-22 | Teledyne Industries, Inc. | High density tungsten-nickel-iron-cobalt alloys having improved hardness, and method for making them |
| EP0326861A1 (en) * | 1988-01-30 | 1989-08-09 | H.C. Starck GmbH & Co. KG | Composite agglomerated metal powder, process for manufacturing it an its use |
| US4915733A (en) * | 1988-01-30 | 1990-04-10 | Hermann C. Starck Berlin Gmbh & Co. Kg | Agglomerated metal composite powders |
| EP0332474A1 (en) * | 1988-03-11 | 1989-09-13 | Camco Drilling Group Limited | Improvements in or relating to cutter assemblies for rotary drill bits |
| EP0384623B1 (en) * | 1989-02-14 | 1994-03-02 | Camco Drilling Group Limited | Improvements in or relating to cutting elements for rotary drill bits |
| FR2659735A1 (en) * | 1990-03-07 | 1991-09-20 | Saint Louis Inst | HYPERSONIC PROJECTILE WITH KINETIC EFFECT AND METHOD FOR THE PRODUCTION THEREOF. |
| FR2784690A1 (en) * | 1998-10-16 | 2000-04-21 | Eurotungstene Poudres | Pre-alloyed tungsten and-or molybdenum and transition metal based powder, useful for sintered parts or cermet binders, comprises micron-size elementary particles |
| CN100363131C (en) * | 2005-12-12 | 2008-01-23 | 北京科技大学 | Method for preparing grain-size-contrdlable superfine crystallite tungsten and tungsten-copper composite material |
| CN104722767A (en) * | 2015-04-16 | 2015-06-24 | 柳州豪祥特科技有限公司 | Tungsten powder preparation method |
| EP3643429A1 (en) * | 2018-10-23 | 2020-04-29 | Bayerische Metallwerke GmbH | Tungsten alloy product and method of preparation and use for a tungsten alloy product |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0098944A3 (en) | 1984-03-28 |
| KR840005492A (en) | 1984-11-14 |
| ATE25111T1 (en) | 1987-02-15 |
| DE3226648C2 (en) | 1984-12-06 |
| JPH0224882B2 (en) | 1990-05-31 |
| US4498395A (en) | 1985-02-12 |
| DE3369346D1 (en) | 1987-02-26 |
| IL69232A (en) | 1985-10-31 |
| EP0098944B1 (en) | 1987-01-21 |
| JPS5925950A (en) | 1984-02-10 |
| DE3226648A1 (en) | 1984-01-19 |
| KR910003572B1 (en) | 1991-06-05 |
| IL69232A0 (en) | 1983-11-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0098944B1 (en) | Tungsten alloy powder | |
| DE3802811C2 (en) | ||
| DE2047143C1 (en) | Heat-resistant sintered alloy and process for its production | |
| DE60106149T2 (en) | Hydrogen-absorbing alloy powder and method for producing the same and fuel tank for storing hydrogen | |
| DE60121242T2 (en) | Molybdenum-copper composite powder and its production and processing into a pseudo alloy | |
| EP1242642B1 (en) | method for production of powder mixture or composite powder | |
| EP0956173B1 (en) | Metal powder granulates, method for their production and use of the same | |
| DE102007047523B3 (en) | Process for the production of semi-finished products from NiTi shape memory alloys | |
| EP1042218B1 (en) | Process for preparing nanocrystalline metal hydrides | |
| DE19822663A1 (en) | Sintered metal and alloy powders for powder metallurgical applications and processes for their production and their use | |
| DE69932148T2 (en) | MICRONOON METAL POWDER BASED ON 3D TRANSITION METALS | |
| EP0183017B2 (en) | Sintering process for prealloyed tungsten powder | |
| DE2414909A1 (en) | STEEL POWDER | |
| EP2097549A2 (en) | Metal powder | |
| DE2347375A1 (en) | Process for the production of finely divided spherical nickel powder | |
| DE2200670B2 (en) | ||
| EP2061615A1 (en) | Method for production of composite powders and composite powder | |
| DE3116185A1 (en) | "METAL BINDER FOR COMPRESSING METAL POWDER" | |
| DE2163866C3 (en) | Method for producing a nickel powder having a spherical shape | |
| AT411580B (en) | METHOD FOR THE POWDER METALLURGICAL PRODUCTION OF OBJECTS | |
| EP0223196A2 (en) | Process for manufacturing dispersion-cured metal alloys | |
| DE10301175B4 (en) | Process for the powder metallurgical production of components | |
| DE102018102322A1 (en) | Method for producing a manganese-bismuth alloy | |
| DE3729780A1 (en) | Increasing the penetration of projectile shaped charges - involves using tungsten@-based, and therefore denser, projectile casing to give increased projectile kinetic energy and higher penetration capacity | |
| DE2032862A1 (en) | Process for the preparation of mutual dispersions of carbides and metals or alloys and new products thus obtained |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LI NL SE |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LI NL SE |
|
| 17P | Request for examination filed |
Effective date: 19840829 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE FR GB IT LI NL SE |
|
| REF | Corresponds to: |
Ref document number: 25111 Country of ref document: AT Date of ref document: 19870215 Kind code of ref document: T |
|
| REF | Corresponds to: |
Ref document number: 3369346 Country of ref document: DE Date of ref document: 19870226 |
|
| ET | Fr: translation filed | ||
| ITF | It: translation for a ep patent filed |
Owner name: JACOBACCI & PERANI S.P.A. |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 19890415 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19890525 Year of fee payment: 7 |
|
| ITTA | It: last paid annual fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19900531 Ref country code: CH Effective date: 19900531 Ref country code: BE Effective date: 19900531 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732 |
|
| BERE | Be: lapsed |
Owner name: DORNIER SYSTEM G.M.B.H. Effective date: 19900531 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| EAL | Se: european patent in force in sweden |
Ref document number: 83105070.3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19950420 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 19950512 Year of fee payment: 13 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Effective date: 19960521 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19960522 |
|
| EUG | Se: european patent has lapsed |
Ref document number: 83105070.3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19970531 Year of fee payment: 15 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19981201 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19981201 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19990927 Year of fee payment: 17 Ref country code: DE Payment date: 19990927 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19991011 Year of fee payment: 17 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000521 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20000521 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20010131 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20010301 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |